Abstract
Background
The aim of this study was to compare the long-term quality of life (QoL) in patients after Sweet, Ivor-Lewis, or Mckeown esophagectomy.
Methods
Esophageal cancer patients after Sweet, Ivor-Lewis, or Mckeown esophagectomy from 2010 to 2012 were included. QoL was assessed according to the European Organization for Research and Treatment of Cancer general questionnaire: QLQ-C30 and esophagus-specific questionnaire: QLQ-OES18.
Results
A total of 126 qualified patients who have been alive for more than 3 years without tumor recurrence were divided into three groups: the Sweet group (n = 40), Ivor-Lewis group (n = 38), and Mckeown group (n = 48). Among these three groups, the QLQ-C30 mean scores of global health status, functional and symptom scales, and general QoL were similar. The symptom scales of QLQ-OSE18 showed that patients who had a Mckeown operation experienced more problem of eating (P = 0.029), choking when swallowing (P = 0.010) and coughing (P = 0.016), while patients undergoing Sweet operation complained more symptom of reflux (P = 0.003) and pain (P = 0.000).
Conclusions
All three types of esophagectomy provided a generally good long-term QoL. However, patients in Sweet and Mckeown group tend to suffer from a higher symptomatic burden as compared to Ivor-Lewis approach.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgery remains the cornerstone of the multimodality treatment for advanced esophageal cancer.1 Various surgical approaches for esophageal cancer are currently being used, which can be divided into two groups: intrathoracic anastomosis group (Ivor-Lewis and Sweet) and cervical anastomosis group (transhiatal and Mckeown), according to the site of anastomosis.2 Still, there are no definitive guidelines for choosing a specific surgical approach for esophageal cancer.3 Ivor-Lewis and Mckeown have been gradually accepted as the standard and route procedure for esophageal cancer, while the approach of Sweet is debated, mainly for its limited extent of lymphadenectomy, especially for the dissection of the right upper mediastinal lymph nodes (RUM-LND). However, our previous study has investigated the efficiency of Sweet procedure with the RUM-LND resection.4 The result demonstrated a comparable overall survival between groups of Ivor-Lewis and Sweet. Hence, Sweet procedure maybe still worthwhile in modern esophagectomy.5
Quality of life (QoL) plays an essential role postoperatively, which was seen as a prognostic value in patients with esophageal cancer.6,7 Different types of esophagectomy might have different impact on patients’ QoL, which include the functional and symptomatic effects, and these impacts were correlated with patients’ both short-term and long-term QoL.8,9,10 Although the effect of surgical approaches on QoL in esophageal cancer patients has been studied by several studies, most studies either have followed their cohorts for 2 years or less or, in case of longer follow-up, usually have not controlled for wide variations in elapsed time since surgery within studied groups.11,12,13,14 The aim of this study was to evaluate the long-term (> 3 years) QoL of patients after Ivor-Lewis, Sweet, and Mckeown esophagectomy without tumor recurrence using an available cancer-specific questionnaire with tumor-specific module.
Materials and Methods
Patients
From May 2010 to May 2012, a total of 432 patients who had undergone esophagectomy were investigated. This study was approved by the ethics committee of West China Hospital, and informed consents were obtained from all included patients. The pathologic stage of esophageal cancer was diagnosed according to the American Joint Committee on Cancer/Union for International Cancer Control, 7th edition.15 Inclusion criteria for this study were as follows: (1) patients were diagnosed with esophageal squamous cell carcinoma; (2) esophagectomy with subtotal lymphadenectomy was performed via Ivor-Lewis, Mckeown or Sweet approach; (3) gastric conduit was used for esophageal replacement and posterior mediastinal route was used for reconstruction. The exclusion criteria used are as follows: (1) patients who lost to follow-up; (2) patients who have palliative surgery and R1 or R2 resection; (3) patients who died within 2 years; (4) patients who have tumor recurrence.
QoL Assessment
The qualified patients’ health-related QoL was assessed according to the European Organization for Research and Treatment of Cancer (EORTC) QLQ-C30 (version 3.0)16 and (esophagus-specific) QLQ-OES18 developed by Blazeby.17,18 Structured questionnaires were used to collect data from patients via face-to-face, telephone, or mail interview. EORTC QLQ-30 is a worldwide and cancer-specific questionnaire used for evaluating the QoL of patients with malignant diseases, which incorporates five functional scales (physical, role, cognitive, emotional, and social), three symptom scales (fatigue, pain, and nausea and vomiting), a global health status/QoL scale, and a number of single items assessing additional symptoms commonly reported by cancer patients (dyspnea, loss of appetite, insomnia, constipation, and diarrhea) and perceived financial impact of the disease. Each scale refers to several questions while each single item corresponds to single question, and all items’ responses have been classified into four levels which represent “Not at all,” “A little,” “Quite a bit,” and “Very much” in order corresponding to 1–4, consequently except for the global health status which includes 7-point items ranging from “very poor” to “excellent”. The QLQ-OES18 consists of four scales (dysphagia, eating, reflux, and pain) and six single items: swallowing saliva, choking when swallowing, dry mouth, taste problems, coughing, and speech problems, which is more concerned about eating problems. These items’ responses have the same categories as EORTC QLQ-C30 with four levels ranging from “Not at all” to “Very much.” All the responses to the scales or single item are then scored from 0 to 100 according to the EORTC QLQ-C30 scoring manual,16 a high score for a functional scale represents a high level of functioning, and a high score for the global health status represents a high QoL, but a high score for symptom scale represents a high level of symptomatology.
The calculated results of QoL were compared among different surgical approaches. EORTC QLQ-C30 and QLQ-OSE18 values of each surgical approach group were also compared with the available reference healthy population (RHP) published by Scott19 and Derogar20, respectively.
Statistical Analysis
The clinicopathologic features among different surgical approach groups were analyzed by the Fisher exact test and chi-square test. The mean values with standard deviation (SD) were used to present the data of QoL and the outcomes of QoL comparison among different groups were achieved by the analysis of variance (ANOVA). At the same time, when comparing the mean score of each item to RHP, Student’s t test was applied. The statistical significance was regarded as the probability value < 0.05. Statistical analyses were conducted by IBM® SPSS® Statistics Version 21.0.
Results
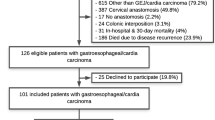
A total of 354 esophageal cancer patients were included in the analyses. Of these, 135 patients (38.1%) were already deceased or suffered recurrences, and 54 patients (15.2%) were lost to follow-up at the time of analysis (April 2016). Of the remaining 165 patients, 39 patients refused to respond, and 126 patients were eligible for completing questionnaires and finally included in our study. The specific study population selection is exhibited in Fig. 1.
After that, the patients were divided into three groups: the Sweet group (open left-thoracic esophagectomy with thoracic anastomosis), 40 patients (31.7%); the Ivor-Lewis group (abdominal and right-thoracic esophagectomy with thoracic anastomosis), 38 patients (30.2%); the Mckeown group (abdominal, right-thoracic and cervical esophagectomy with cervical anastomosis.), 48 patients (38.1%). The clinicopathologic features among the three groups are shown in Table 1.
QLQ-C30
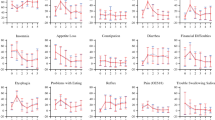
In about 3 to 5 years after curative-intent esophagectomy for esophageal cancer, the three patient groups stratified by surgical approaches shared virtually comparable mean scores of global health/QOL, general symptom scales, and function scales. The comparisons of QLQ-C30 questionnaire among three groups are shown in Table 2. We also compared the mean scores of each group with that of the reference healthy population (RHP) available from EORTC.20,21 The result showed that social function deficits irrespective of type of surgical approaches were observed when contrasted to the RHP (P = 0.040 in the Sweet group, P = 0.001 in the Ivor-Lewis group, P = 0.049 in the Mckeown group) (Fig. 2). Role and emotional and cognitive function, however, were similar with that of the RHP. For general symptom scales, a higher burden of appetite loss, diarrhea, nausea, and vomiting was found in each group when compared to RHP, indicating that the digestive symptoms were the most striking complaint among these patients.
QLQ-OSE18
From the analysis of variance, the symptom scales of QLQ-OSE18 showed that patients who had a Mckeown operation experienced more problem of eating (P = 0.029), choking when swallowing (P = 0.010), and coughing (P = 0.016), while patients undergoing the Sweet operation complained more symptom of reflux (P = 0.003) and pain (P = 0.000) (Table 3). Then, the comparisons of the mean scores of each group with that of the RHP were carried out (Fig. 3). Similarly, eating problem was observed in every group when contrasted to RHP (P = 0.001 in the Ivor-Lewis group, P = 0.000 in both the Sweet group and Mckeown group). More complaints of reflux (P = 0.000), pain (P = 0.009), and trouble talking (P = 0.036) in the Sweet group, more choking when swallowing (P = 0.000), swallowing saliva (P = 0.038) and trouble with taste (P = 0.006) in the Mckeown group, and more choking when swallowing (P = 0.015) in the Ivor-Lewis were reported as compared with that of RHP. Of these symptoms, choking when swallowing was the most striking complaint with the maximal difference between the mean scores of each group and that of the RHP, followed by the reflux and eating with trouble.
Discussion
To date, there is still a significant debate regarding the best surgical approaches for esophagectomy. In addition to the survival after operation, long-term QoL represents the other important issues for patients.8,10 Our study provided further insight into the long-term quality of life in patients with esophageal cancer according to surgical approaches. Outcomes from the questionnaire of QLQ-C30 showed that neither was there a significant difference of mean sores in global health status nor in general QoL among three groups. Meanwhile, none of the groups showed any significant difference in global quality of life, role, and emotional or cognitive function as compared to RHP. Several reasons may explain the above outcomes. On the one hand, QoL would drop immediately following operation because of surgical trauma, which recovers during the postoperative rehabilitation period. Even the long-term QoL would get close to that of RHP but never reach to the preoperative level, which was confirmed by Egberts et al.22 On the other hand, during the recovery period, patients adapt to the situation of coexisting with esophageal cancer and do not act out of fear, anger, and unacceptability, which allowed achieving a new set of knowledge based on the self-knowledge of each patient which contributed for an improved overall QoL.7,21,22
For the symptom scales in the QLQ-C30, the result showed that most patients suffered from almost the same symptoms as most of researches had ever reported: diarrhea, appetite loss, nausea and vomiting, and function decrease.7,13,14 While advances in surgical technique for esophagectomy, the surgical trauma continues to be appreciably higher than other similarly complex operations such as pancreatectomy, gastrectomy, and hepatectomy.23,24,25 Not surprisingly, the considerably anatomical and physiological changes by this surgery will lead to long-lasting symptoms.
The EORTC QLQ-OSE18 questionnaire was confirmed as the only suitable measure in accompany with the QLQ-C30 to assess QoL of patients with esophageal cancer.13,14,22,26 In our study, reflux is the most striking complaint in the Sweet group, while choking when swallowing in the Mckeown group. The reasons for reflux are the removal of esophagogastric junction, which destroys its anti-reflux barrier, and the difference in pressure change also contributes to the reflux. In addition, lower anastomotic location has been demonstrated with higher risk of reflux.27 The Sweet operation was preferentially performed in patients with tumor in lower thoracic esophagus in our study, and its esophagogastric anastomosis usually completed at a relatively lower level. Hence, the site of anastomosis may play a non-neglectable role in the striking burden of reflux in the Sweet group. And longer gastric conduit blood supply, tension of anastomosis, and the extrinsic compression are also some of the possible explanations.28,29
Choking when swallowing caused the greatest impact on patients in the Mckeown group from our studies; however, there is no difference found in dysphagia among the three groups. As compared with intrathoracic esophagogastric anastomoses, the cervical esophagogastric anastomoses has been reported with a relative higher risk of hematoma in the neck and recurrent laryngeal nerve palsy duo to cervical esophageal mobilization30 and a predisposition of benign stricture formation duo to a high incidence of anastomotic leakage.31 Thus, the abovementioned potential factors may increase the complaint of choking when swallowing and ultimately worsens global quality of life. The explaination of the comparable dysphagia is that the removal of the cancer-related stenosis in esophagus contributed to the improvement in dysphagia after operation and patients complained less about dysphagia and gradually adapted to the new eating conditions though the swallowing function was not the same as that before the operation. Egberts et al.22 once prospectively assessed the QoL of 105 patients who underwent esophagectomy for esophageal cancer by means of EORTC QLQ-C30 and QLQ-OSE18 and reported an improvement in dysphagia even 3 months after discharge from hospital.
Pain after operation is another main complaint. From our study, the Sweet group showed the highest mean score of pain. The first probable explanation is the difference of surgical procedures. Although the Sweet procedure requires only one incision through left chest, mobilization of the stomach is done with manual manipulation through the cut-opened hiatus and diaphragm and a larger incision for thoracotomy is inevitable, and the bigger surgical wound contributes to more pain in patients even 2 years after operation. However, more than half of patients who accepted the Ivor-Lewis or Mckeown procedure underwent through thoracoscopy and laparoscopy, and these minimally invasive surgeries are correlated to less postoperative pain for patients. Different location of anastomosis was also associated with pain among esophageal cancer patients. Both Wormald et al.21 and Blazeby et al.10 had reported that scores for pain were significantly higher among the group of patients with intrathoracic anastomosis compared to those with cervical anastomosis. The results were also confirmed by the observations in a longitudinal study by Egberts et al.22 who compared the QoL of patients in cervical and intrathoracic anastomosis groups separately.
Several limitations to this study should be mentioned, such as its retrospective work with the potential for selection bias. Another limitation of this study was that we did not evaluate the postoperative QoL at multiple time points and therefore cannot comment the dynamic effect of different surgical approaches. Several studies have demonstrated the benefit in QoL among esophageal cancer patients who experience minimally invasive surgery.32 Thus, the variable surgical technique among the three groups would potentially introduce bias in the comparison of QoL across surgical approaches. However, the subgroup analysis by stratifying these patients based on surgical technique was confined, since the high risk of type II error caused by the relatively small number of patients receiving MIE and the different types of minimally invasive procedures (total or hybrid).
Conclusion
Our study highlighted that, although the three surgical approaches (the Ivor-Lewis, Mckeown, or Sweet) shared a comparable general health-related QoL for esophageal cancer patients in about 3 to 5 years after curative-intent esophagectomy, a higher burden of digestive disorder were associated with the Sweet and Mckeown operation. Meanwhile, further studies on researching the impact of the surgical approaches on patients’ postoperative QoL are needed.
Reference
Lagergren J, Smyth E, Cunningham D, and Lagergren P. Oesophageal cancer. Lancet 2017; 390: 2383–2396.
Huang HT, Wang F, Shen L, Xia CQ, Lu CX, and Zhong CJ. Clinical Outcome of Middle Thoracic Esophageal Cancer with Intrathoracic or Cervical Anastomosis. Thorac Cardiovasc Surg 2015; 63: 328–34.
Noordman BJ, Wijnhoven BP, and van Lanschot JJ. Optimal surgical approach for esophageal cancer in the era of minimally invasive esophagectomy and neoadjuvant therapy. Dis Esophagus 2016; 29: 773–779.
Wang ZQ, Wang WP, Yuan Y, Hu Y, Peng J, Wang YC, et al. Left thoracotomy for middle or lower thoracic esophageal carcinoma: still Sweet enough?. J Thorac Dis 2016; 8: 3187–3196.
Ma J, Zhan C, Wang L, Jiang W, Zhang Y, Shi Y, et al. The sweet approach is still worthwhile in modern esophagectomy. Ann Thorac Surg 2014; 97: 1728–33.
Deschamps C, Nichols FC, 3rd, Cassivi SD, Allen MS, and Pairolero PC. Long-term function and quality of life after esophageal resection for cancer and Barrett's. Surg Clin North Am 2005; 85: 649–56, xi.
Courrech Staal EF, van Sandick JW, van Tinteren H, Cats A, and Aaronson NK. Health-related quality of life in long-term esophageal cancer survivors after potentially curative treatment. J Thorac Cardiovasc Surg 2010; 140: 777–83.
McLarty AJ, Deschamps C, Trastek VF, Allen MS, Pairolero PC, and Harmsen WS. Esophageal resection for cancer of the esophagus: long-term function and quality of life. Ann Thorac Surg 1997; 63: 1568–72.
Schmidt CE, Bestmann B, Kuchler T, Schmid A, and Kremer B. Quality of life associated with surgery for esophageal cancer: differences between collar and intrathoracic anastomoses. World J Surg 2004; 28: 355–60.
Blazeby JM, Farndon JR, Donovan J, and Alderson D. A prospective longitudinal study examining the quality of life of patients with esophageal carcinoma. Cancer 2000; 88: 1781–7.
Viklund P, Wengstrom Y, Rouvelas I, Lindblad M, and Lagergren J. Quality of life and persisting symptoms after oesophageal cancer surgery. Eur J Cancer 2006; 42: 1407–14.
Avery KN, Metcalfe C, Barham CP, Alderson D, Falk SJ, and Blazeby JM. Quality of life during potentially curative treatment for locally advanced oesophageal cancer. Br J Surg 2007; 94: 1369–76.
Donohoe CL, McGillycuddy E, and Reynolds JV. Long-term health-related quality of life for disease-free esophageal cancer patients. World J Surg 2011; 35: 1853–60.
Gutschow CA, Holscher AH, Leers J, Fuchs H, Bludau M, Prenzel KL, et al. Health-related quality of life after Ivor Lewis esophagectomy. Langenbecks Arch Surg 2013; 398: 231–7.
Edge SB and Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 2010; 17: 1471–4.
Fayers P, Aaronson NK, Bjordal K, Grønvold M, Curran D, and Bottomley A. EORTC QLQ-C30 Scoring Manual (3rd edition). 2001;
Blazeby JM, Alderson D, Winstone K, Steyn R, Hammerlid E, Arraras J, et al. Development of an EORTC questionnaire module to be used in quality of life assessment for patients with oesophageal cancer. The EORTC Quality of Life Study Group. Eur J Cancer 1996; 32A: 1912–7.
Blazeby JM, Conroy T, Hammerlid E, Fayers P, Sezer O, Koller M, et al. Clinical and psychometric validation of an EORTC questionnaire module, the EORTC QLQ-OES18, to assess quality of life in patients with oesophageal cancer. Eur J Cancer 2003; 39: 1384–94.
Scott NW, Fayers P, Aaronson NK, Bottomley A, de Graeff A, Groenvold M, et al. EORTC QLQ-C30 reference values manual. 2008;
Derogar M and Lagergren P. Health-related quality of life among 5-year survivors of esophageal cancer surgery: a prospective population-based study. J Clin Oncol 2012; 30: 413–8.
Wormald JC, Bennett J, van Leuven M, and Lewis MP. Does the site of anastomosis for esophagectomy affect long-term quality of life?. Dis Esophagus 2016; 29: 93–8.
Egberts JH, Schniewind B, Bestmann B, Schafmayer C, Egberts F, Faendrich F, et al. Impact of the site of anastomosis after oncologic esophagectomy on quality of life--a prospective, longitudinal outcome study. Ann Surg Oncol 2008; 15: 566–75.
Atkins BZ, Shah AS, Hutcheson KA, Mangum JH, Pappas TN, Harpole DH, Jr., et al. Reducing hospital morbidity and mortality following esophagectomy. Ann Thorac Surg 2004; 78: 1170–6; discussion 1170–6.
Raymond DP, Seder CW, Wright CD, Magee MJ, Kosinski AS, Cassivi SD, et al. Predictors of Major Morbidity or Mortality After Resection for Esophageal Cancer: A Society of Thoracic Surgeons General Thoracic Surgery Database Risk Adjustment Model. Ann Thorac Surg 2016; 102: 207–14.
Low DE, Alderson D, Cecconello I, Chang AC, Darling GE, D’Journo XB, et al. International Consensus on Standardization of Data Collection for Complications Associated With Esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann Surg 2015; 262: 286–94.
Daster S, Soysal SD, Stoll L, Peterli R, von Flue M, and Ackermann C. Long-term quality of life after Ivor Lewis esophagectomy for esophageal cancer. World J Surg 2014; 38: 2345–51.
Pennathur A, Zhang J, Chen H, and Luketich JD. The “best operation” for esophageal cancer?. Ann Thorac Surg 2010; 89: S2163–7.
Yamamoto S, Makuuchi H, Shimada H, Chino O, Nishi T, Kise Y, et al. Clinical analysis of reflux esophagitis following esophagectomy with gastric tube reconstruction. J Gastroenterol 2007; 42: 342–5.
Yajima K, Kosugi S, Kanda T, Matsuki A, and Hatakeyama K. Risk factors of reflux esophagitis in the cervical remnant following esophagectomy with gastric tube reconstruction. World J Surg 2009; 33: 284–9.
Sato Y, Kosugi S, Aizawa N, Ishikawa T, Kano Y, Ichikawa H, et al. Risk Factors and Clinical Outcomes of Recurrent Laryngeal Nerve Paralysis After Esophagectomy for Thoracic Esophageal Carcinoma. World J Surg 2016; 40: 129–36.
van Heijl M, Gooszen JA, Fockens P, Busch OR, van Lanschot JJ, and van Berge Henegouwen MI. Risk factors for development of benign cervical strictures after esophagectomy. Ann Surg 2010; 251: 1064–9.
Pennathur A LR, Villa Sanchez M, Schuchert MJ, Awais O, Ward J, Gooding WE, Landreneau RJ, Dhupar R, Sarkaria I, Christie NA, Luketich JD. Prospective Study of Quality of Life after Esophagectomy with a Focus on Minimally Invasive Esophagectomy. American Association for Thoracic Surgery Annual Meeting 2018; San Diego, CA. April 2018
Author information
Authors and Affiliations
Contributions
(I) Conception and design: Long-Qi Chen, Yu-Shang Yang, and Yong Yuan;
(II) Administrative support: Wei-Peng Hu and Yu-Shang Yang;
(III) Provision of study materials or patients: Qi-Xin Shang and Yu-Shang Yang;
(IV) Collection and assembly of data: Yu-Shang Yang, Xiao-Ying Wu, and Qi-Xin Shang;
(V) Data analysis and interpretation: Qi-Xin Shang, Wei-Peng Hu, and Yong Yuan;
(VI) Manuscript writing: All authors;
(VII) Final approval of manuscript: All authors.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yang, YS., Shang, QX., Yuan, Y. et al. Comparison of Long-term Quality of Life in Patients with Esophageal Cancer after Ivor-Lewis, Mckeown, or Sweet Esophagectomy. J Gastrointest Surg 23, 225–231 (2019). https://doi.org/10.1007/s11605-018-3999-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-018-3999-z