Abstract
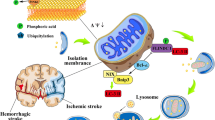
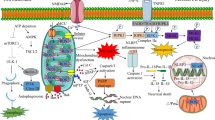
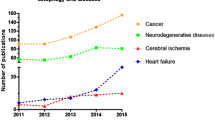
Ischemic stroke is a serious cerebrovascular disease with high morbidity and mortality. As a result of ischemia-reperfusion, a cascade of pathophysiological responses is triggered by the imbalance in metabolic supply and demand, resulting in cell loss. These cellular injuries follow various molecular mechanisms solely or in combination with this disorder. Mitochondria play a driving role in the pathophysiological processes of ischemic stroke. Once ischemic stroke occurs, damaged cells would respond to such stress through mitophagy. Mitophagy is known as a conservatively selective autophagy, contributing to the removal of excessive protein aggregates and damaged intracellular components, as well as aging mitochondria. Moderate mitophagy may exert neuroprotection against stroke. Several pathways associated with the mitochondrial network collectively contribute to recovering the homeostasis of the neurovascular unit. However, excessive mitophagy would also promote ischemia-reperfusion injury. Therefore, mitophagy is a double-edged sword, which suggests that maximizing the benefits of mitophagy is one of the direction of future efforts. This review emphasized the role of mitophagy in ischemic stroke, and highlighted the crosstalk between mitophagy and apoptosis/necroptosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Benjamin EJ, Muntner P, Alonso A, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation, 2019, 139(10):e56–e528
Lakhan SE, Kirchgessner A, Hofer M. Inflammatory mechanisms in ischemic stroke: therapeutic approaches. J Transl Med, 2009,7:97
Barthels D, Das H. Current advances in ischemic stroke research and therapies. Biochimi Biophys Acta Mol Basis Dis, 2020,1866(4):165260
National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med, 1995,333(24):1581–1587
Goyal M, Demchuk AM, Menon BK, et al. Randomized Assessment of Rapid Endovascular Treatment of Ischemic Stroke. N Engl J Med, 2015,372(11):1019–1030
Chen W, Huang JJ, Hu YQ, et al. Mitochondrial Transfer as a Therapeutic Strategy Against Ischemic Stroke. Transl Stroke Res, 2020,11(6):1214–1228
Mayevsky A, Kutai-Asis H, Tolmasov M. Mitochondrial function and brain Metabolic Score (BMS) in ischemic Stroke: Evaluation of “neuroprotectants” safety and efficacy. Mitochondrion, 2020,50:170–194
Hu HZ, Feng XB, Shao ZW, et al. Application and Prospect of Mixed Reality Technology in Medical Field. Curr Med Sci, 2019,39(1):1–6
Lip GYH, Nieuwlaat R, Pisters R, et al. Refining Clinical Risk Stratification for Predicting Stroke and Thromboembolism in Atrial Fibrillation Using a Novel Risk Factor-Based Approach The Euro Heart Survey on Atrial Fibrillation. Chest, 2010,137(2):263–272
Hacke W, Kaste M, Bluhmki E, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med, 2008,359(13):1317–1329
Rabinstein AA. Update on Treatment of Acute Ischemic Stroke. Continuum (Minneap Minn), 2020,26(2):268–286
Zhang D, Zou X, Sy C, et al. Thrombolysis and reperfusion: advanced understanding of early management strategies in acute ischemic stroke. Neurol Res, 2014,36(5):391–396
Cagalinec M, Safiulina D, Liiv M, et al. Principles of the mitochondrial fusion and fission cycle in neurons. J Cell Sci, 2013,126(Pt 10):2187–2197
Chan PH. Mitochondrial dysfunction and oxidative stress as determinants of cell death/survival in stroke. Ann N Y Acad Sci, 2005,1042:203–209
Correa F, Soto V, Zazueta C. Mitochondrial permeability transition relevance for apoptotic triggering in the post-ischemic heart. Int J Biochem Cell Biol, 2007,39(4):787–798
Carinci M, Vezzani B, Patergnani S, et al. Different Roles of Mitochondria in Cell Death and Inflammation: Focusing on Mitochondrial Quality Control in Ischemic Stroke and Reperfusion. Biomedicines, 2021,9(2):169
Wang L, Qi H, Tang Y, et al. Post-translational Modifications of Key Machinery in the Control of Mitophagy. Trends Biochem Sci, 2020,45(1):58–75
Li X, Huang L, Lan J, et al. Molecular mechanisms of mitophagy and its roles in neurodegenerative diseases. Pharmacol Res, 2021,163:105240
Bingol B, Sheng M. Mechanisms of mitophagy: PINK1, Parkin, USP30 and beyond. Free Radic Biol Med, 2016,100:210–222
Lu W, Sun J, Yoon JS, et al. Mitochondrial Protein PGAM5 Regulates Mitophagic Protection against Cell Necroptosis. PLoS One, 2016,11(1):e0147792
Shen L, Gan Q, Yang Y, et al. Mitophagy in Cerebral Ischemia and Ischemia/Reperfusion Injury. Front Aging Neurosci, 2021,13:687246
Kapadia M, De Snoo ML, Kalia LV, et al. Regulation of Parkin-dependent mitophagy by Bcl-2-associated athanogene (BAG) family members. Neural Regen Res, 2021,16(4):684–685
Filippov MA, Tatarnikova OG, Pozdnyakova NV, et al. Inflammation/bioenergetics-associated neurodegenerative pathologies and concomitant diseases: a role of mitochondria targeted catalase and xanthophylls. Neural Regen Res, 2021,16(2):223–233
Park HA, Broman K, Jonas EA. Oxidative stress battles neuronal Bcl-xL in a fight to the death. Neural Regen Res, 2021,16(1):12–15
Ott M, Robertson JD, Gogvadze V, et al. Cytochrome c release from mitochondria proceeds by a two-step process. Proc Natl Acad Sci USA, 2002,99(3):1259–1263
Baechler BL, Bloemberg D, Quadrilatero J. Mitophagy regulates mitochondrial network signaling, oxidative stress, and apoptosis during myoblast differentiation. Autophagy, 2019,15(9):1606–1619
Cen X, Chen Y, Xu X, et al. Pharmacological targeting of MCL-1 promotes mitophagy and improves disease pathologies in an Alzheimer’s disease mouse model. Nat Commun, 2020,11(1):5731
Kang L, Liu S, Li J, et al. Parkin and Nrf2 prevent oxidative stress-induced apoptosis in intervertebral endplate chondrocytes via inducing mitophagy and anti-oxidant defenses. Life Sci, 2020,243:117244
Erustes AG, D’Eletto M, Guarache GC, et al. Overexpression of alpha-synuclein inhibits mitoch-ondrial Ca(2+) trafficking between the endoplasmic reticulum and mitochondria through MAMs by altering the GRP75-IP3R interaction. J Neurosci Res, 2021,99(11):2932–2947
Yoo SM, Jung YK. A Molecular Approach to Mitophagy and Mitochondrial Dynamics. Mol Cells, 2018,41(1):18–26
Roberts RF, Tang MY, Fon EA, et al. Defending the mitochondria: The pathways of mitophagy and mitochondrial-derived vesicles. Int J Biochem Cell Biol, 2016,79:427–436
Narendra D, Tanaka A, Suen DF, et al. Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol, 2008,183(5):795–803
Narendra DP, Jin SM, Tanaka A, et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol, 2010,8(1):e1000298
Lazarou M, Jin SM, Kane LA, et al. Role of PINK1 binding to the TOM complex and alternate intracellular membranes in recruitment and activation of the E3 ligase Parkin. Dev Cell, 2012,22(2):320–333
Deas E, Plun-Favreau H, Gandhi S, et al. PINK1 cleavage at position A103 by the mitochondrial protease PARL. Hum Mol Genet, 2011,20(5):867–879
Sekine S. PINK1 import regulation at a crossroad of mitochondrial fate: the molecular mechanisms of PINK1 import. J Biochem, 2020,167(3):217–224
Okatsu K, Oka T, Iguchi M, et al. PINK1 autophosphorylation upon membrane potential dissipation is essential for Parkin recruitment to damaged mitochondria. Nat Commun, 2012,3:1016
Zhang T, Liu Q, Gao W, et al. The multifaceted regulation of mitophagy by endogenous metabolites. Autophagy, 2021,1–24
Ni HM, Williams JA, Ding WX. Mitochondrial dynamics and mitochondrial quality control. Redox Biol, 2015,4:6–13
Yamada T, Dawson TM, Yanagawa T, et al. SQSTM1/p62 promotes mitochondrial ubiquitination independently of PINK1 and PRKN/parkin in mitophagy. Autophagy, 2019,15(11):2012–2018
Yamano K, Matsuda N, Tanaka K. The ubiquitin signal and autophagy: an orchestrated dance leading to mitochondrial degradation. EMBO Rep, 2016, 17(3):300–316
Yamano K, Queliconi BB, Koyano F, et al. Site-specific Interaction Mapping of Phosphorylated Ubiquitin to Uncover Parkin Activation. J Biol Chem, 2015,290(42):25199–25211
Poole AC, Thomas RE, Yu S, et al. The mitochondrial fusion-promoting factor mitofusin is a substrate of the PINK1/parkin pathway. PLoS One, 2010,5(4):e10054
Wang X, Winter D, Ashrafi G, et al. PINK1 and Parkin target Miro for phosphorylation and degradation to arrest mitochondrial motility. Cell, 2011,147(4):893–906
Sarraf SA, Raman M, Guarani-Pereira V, et al. Landscape of the PARKIN-dependent ubiquitylome in response to mitochondrial depolarization. Nature, 2013,496(7445):372–376
Koyano F, Yamano K, Kosako H, et al. Parkin recruitment to impaired mitochondria for nonselective ubiquitylation is facilitated by MITOL. J Biol Chem, 2019,294(26):10300–10314
Matsuda N, Yamano K. Two sides of a coin: Physiological significance and molecular mechanisms for damage-induced mitochondrial localization of PINK1 and Parkin. Neurosci Res, 2020,159:16–24
Hamacher-Brady A, Brady NR. Mitophagy programs: mechanisms and physiological implications of mitochondrial targeting by autophagy. Cell Mol Life Sci, 2016,73(4):775–795
Chen G, Cizeau J, Vande Velde C, et al. Nix and Nip3 form a subfamily of pro-apoptotic mitochondrial proteins. J Biol Chem, 1999,274(1):7–10
Bellot G, Garcia-Medina R, Gounon P, et al. Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol Cell Biol, 2009,29(10):2570–2581
Choi GE, Lee HJ, Chae CW, et al. BNIP3L/NIX-mediated mitophagy protects against glucocorticoid-induced synapse defects. Nat Commun, 2021,12(1):487
Lei L, Yang S, Lu X, et al. Research Progress on the Mechanism of Mitochondrial Autophagy in Cerebral Stroke. Front Aging Neurosci, 2021,13:698601
Kuang Y, Ma K, Zhou C, et al. Structural basis for the phosphorylation of FUNDC1 LIR as a molecular switch of mitophagy. Autophagy, 2016,12(12):2363–2373
Cai Y, Yang E, Yao X, et al. FUNDC1-dependent mitophagy induced by tPA protects neurons against cerebral ischemia-reperfusion injury. Redox Biol, 2021, 38:101792
Liu H, Zang C, Yuan F, et al. The role of FUNDC1 in mitophagy, mitochondrial dynamics and human diseases. Biochem Pharmacol, 2022,197:114891
Liu L, Feng D, Chen G, et al. Mitochondrial outermembrane protein FUNDC1 mediates hypoxia-induced mitophagy in mammalian cells. Nat Cell Biol, 2012,14(2):177–185
Chen G, Han Z, Feng D, et al. A regulatory signaling loop comprising the PGAM5 phosphatase and CK2 controls receptor-mediated mitophagy. Mol Cell, 2014,54(3):362–377
Sugo M, Kimura H, Arasaki K, et al. Syntaxin 17 regulates the localization and function of PGAM5 in mitochondrial division and mitophagy. EMBO J, 2018,37(21):e98899
Xin Y, Zhang X, Li J, et al. New Insights Into the Role of Mitochondria Quality Control in Ischemic Heart Disease. Front Cardiovasc Med, 2021,8:774619
Nan J, Zhu W, Rahman MS, et al. Molecular regulation of mitochondrial dynamics in cardiac disease. Biochim Biophys Acta Mol Cell Res, 2017,1864(7):1260–1273
Anzell AR, Fogo GM, Gurm Z, et al. Mitochondrial fission and mitophagy are independent mechanisms regulating ischemia/reperfusion injury in primary neurons. Cell Death Dis, 2021,12(5):475
Jia J, Jin H, Nan D, et al. New insights into targeting mitochondria in ischemic injury. Apoptosis, 2021,26(3–4):163–183
Zuo W, Zhang S, Xia CY, et al. Mitochondria autophagy is induced after hypoxic/ischemic stress in a Drp1 dependent manner: the role of inhibition of Drp1 in ischemic brain damage. Neuropharmacology, 2014,86:103–115
Zhou BH, Wei SS, Jia LS, et al. Drp1/Mff signaling pathway is involved in fluoride-induced abnormal fission of hepatocyte mitochondria in mice. Sci Total Environ, 2020,725:138192
Luan Y, Ren KD, Luan Y, et al. Mitochondrial Dynamics: Pathogenesis and Therapeutic Targets of Vascular Diseases. Front Cardiovasc Med, 2021,8:770574
Wu P, Li Y, Zhu S, et al. Mdivi-1 Alleviates Early Brain Injury After Experimental Subarachnoid Hemorrhage in Rats, Possibly via Inhibition of Drp1-Activated Mitochondrial Fission and Oxidative Stress. Neurochem Res, 2017,42(5):1449–1458
Smith G, Gallo G. To mdivi-1 or not to mdivi-1: Is that the question? Dev Neurobiol, 2017,77(11):1260–1268
Flippo KH, Gnanasekaran A, Perkins GA, et al. AKAP1 Protects from Cerebral Ischemic Stroke by Inhibiting Drp1-Dependent Mitochondrial Fission. J Neurosci, 2018,38(38):8233–8242
Lai Y, Lin P, Chen M, et al. Restoration of L-OPA1 alleviates acute ischemic stroke injury in rats via inhibiting neuronal apoptosis and preserving mitochondrial function. Redox Biol, 2020,34:101503
Zeng KW, Wang JK, Wang LC, et al. Small molecule induces mitochondrial fusion for neuroprotection via targeting CK2 without affecting its conventional kinase activity. Signal Transduct Target Ther, 2021,6(1):71
Puri R, Cheng XT, Lin MY, et al. Mul1 restrains Parkin-mediated mitophagy in mature neurons by maintaining ER-mitochondrial contacts. Nat Commun, 2019,10(1):3645
Ham PB 3rd, Raju R. Mitochondrial function in hypoxic ischemic injury and influence of aging. Prog Neurobiol, 2017,157:92–116
Sun T, Ding W, Xu T, et al. Parkin Regulates Programmed Necrosis and Myocardial Ischemia/Reperfusion Injury by Targeting Cyclophilin-D. Antioxid Redox Signal, 2019,31(16):1177–1193
Ning XJ, Yan X, Wang YF, et al. Parkin deficiency elevates hepatic ischemia/reperfusion injury accompanying decreased mitochondrial autophagy, increased apoptosis, impaired DNA damage repair and altered cell cycle distribution. Mol Med Rep, 2018,18(6):5663–5668
Wang H, Chen S, Zhang Y, et al. Electroacupuncture ameliorates neuronal injury by Pink1/Parkin-mediated mitophagy clearance in cerebral ischemia-reperfusion. Nitric Oxide, 2019,91:23–34
Chen JL, Wang XX, Chen L, et al. A sphingosine kinase 2-mimicking TAT-peptide protects neurons against ischemia-reperfusion injury by activating BNIP3-mediated mitophagy. Neuropharmacology, 2020,181: 108326
Yepes M. The Plasminogen Activation System Promotes Neurorepair in the Ischemic Brain. Curr Drug Targets, 2019,20(9):953–959
Jeanneret V, Yepes M. Tissue-type plasminogen activator is a homeostatic regulator of synaptic function in the central nervous system. Neural Regen Res, 2017, 12(3):362–365
Thiebaut AM, Gauberti M, Ali C, et al. The role of plasminogen activators in stroke treatment: fibrinolysis and beyond. Lancet Neurol, 2018,17(12):1121–1132
Feng J, Chen X, Guan B, et al. Inhibition of Peroxynitrite-Induced Mitophagy Activation Attenuates Cerebral Ischemia-Reperfusion Injury. Mol Neurobiol, 2018,55(8):6369–6386
Hang Y, He Y, Wu, et al. Rehmapicroside ameliorates cerebral ischemia-reperfusion injury via attenuating peroxynitrite-mediated mitophagy activation. Free Radic Biol Med, 2020,160:526–539
Shao Z, Dou S, Zhu J, et al. The Role of Mitophagy in Ischemic Stroke. Front Neurol, 2020,11:608610
Guan R, Zou W, Dai X, et al. Mitophagy, a potential therapeutic target for stroke. J Biomed Sci, 2018,25(1):87
Cain K, Bratton SB, Langlais C, et al. Apaf-1 oligomerizes into biologically active approximately 700-kDa and inactive approximately 1.4-MDa apoptosome complexes. J Biol Chem, 2000,275(9):6067–6070
Bratton SB, Walker G, Srinivasula SM, et al. Recruitment, activation and retention of caspases-9 and -3 by Apaf-1 apoptosome and associated XIAP complexes. EMBO J, 2001,20(5):998–1009
Hu XM, Li ZX, Zhang DY, et al. A systematic summary of survival and death signalling during the life of hair follicle stem cells. Stem Cell Res Ther, 2021,12(1):453
Hu XM, Li ZX, Lin RH, et al. Guidelines for Regulated Cell Death Assays: A Systematic Summary, A Categorical Comparison, A Prospective. Front Cell Dev Biol, 2021, 9:634690
Yang L, Ma YM, Shen XL, et al. The Involvement of Mitochondrial Biogenesis in Selenium Reduced Hyperglycemia-Aggravated Cerebral Ischemia Injury. Neurochem Res, 2020,45(8):1888–1901
Dong G, Xu N, Wang M, et al. Anthocyanin Extract from Purple Sweet Potato Exacerbate Mitophagy to Ameliorate Pyroptosis in Klebsiella pneumoniae Infection. Int J Mol Sci, 2021,22(21):11422
Hong T, Zhou Y, Peng L, et al. Knocking down peroxiredoxin 6 aggravates cerebral ischemia-reperfusion injury by enhancing mitophagy. Neuroscience, 2021, 482:30–42
Li Y, Hu K, Liang M, et al. Stilbene glycoside upregulates SIRT3/AMPK to promotes neuronal mitochondrial autophagy and inhibit apoptosis in ischemic stroke. Adv Clin Exp Med, 2021,30(2):139–146
He M, Zhang T, Fan Y, et al. Deletion of mitochondrial uncoupling protein 2 exacerbates mitophagy and cell apoptosis after cerebral ischemia and reperfusion injury in mice. Int J Med Sci, 2020,17(17):2869–2878
Ma H, Guo X, Cui S, et al. Dephosphorylation of AMP-activated kinase exacerbates ischemia/reperfusion-induced acute kidney injury via mitochondrial dysfunction. Kidney Int, 2022,101(2):315–330
Wu Q, Liu J, Mao Z, et al. Ligustilide attenuates ischemic stroke injury by promoting Drp1-mediated mitochondrial fission via activation of AMPK. Phytomedicine, 2021,95:153884
Geisler S, Holmstrom KM, Skujat D, et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat Cell Biol, 2010,12(2):119–131
Yao GY, Zhu Q, Xia J, et al. Ischemic postconditioning confers cerebroprotection by stabilizing VDACs after brain ischemia. Cell Death Dis, 2018,9(10):1033
Shi RY, Zhu SH, Li V, et al. BNIP3 interacting with LC3 triggers excessive mitophagy in delayed neuronal death in stroke. CNS Neurosci Ther, 2014,20(12):1045–1055
Lim SY, Hausenloy DJ, Arjun S, et al. Mitochondrial cyclophilin-D as a potential therapeutic target for post-myocardial infarction heart failure. J Cell Mol Med, 2011,15(11):2443–2451
Xu K, Lu C, Ren X, et al. Overexpression of HIF-1alpha enhances the protective effect of mitophagy on steroid-induced osteocytes apoptosis. Environ Toxicol, 2021,36(11):2123–2137
Shang L, Ding W, Li N, et al. The effects and regulatory mechanism of RIP3 on RGC-5 necroptosis following elevated hydrostatic pressure. Acta Bioch Bioph Sin (Shanghai), 2017,49(2):128–137
Guo LM, Wang Z, Li SP, et al. RIP3/MLKL-mediated neuronal necroptosis induced by methamphetamine at 39°C. Neural Regen Res, 2020,15(5):865–874
Wang Z, Guo LM, Zhou HK, et al. Using drugs to target necroptosis: dual roles in disease therapy. Histol Histopathol, 2018,33(8):773–789
Yan WT, Lu S, Yang YD, et al. Research trends, hot spots and prospects for necroptosis in the field of neuroscience. Neural Regen Res, 2021,16(8):1628–1637
Zhang Q, Wan XX, Hu XM, et al. Targeting Programmed Cell Death to Improve Stem Cell Therapy: Implications for Treating Diabetes and Diabetes-Related Diseases. Front Cell Dev Biol, 2021,9:809656
Yan WT, Yang YD, Hu XM, et al. Do pyroptosis, apoptosis, and necroptosis (PANoptosis) exist in cerebral ischemia? Evidence from cell and rodent studies. Neural Regen Res, 2022,17(8):1761–1768
Horvath C, Young M, Jarabicova I, et al. Inhibition of Cardiac RIP3 Mitigates Early Reperfusion Injury and Calcium-Induced Mitochondrial Swelling without Altering Necroptotic Signalling. Int J Mol Sci, 2021, 22(15):7983
Zhang T, Zhang Y, Cui M, et al. CaMKII is a RIP3 substrate mediating ischemia- and oxidative stress- induced myocardial necroptosis. Nat Med, 2016, 22(2):175–182
She L, Tu H, Zhang YZ, et al. Inhibition of Phosphoglycerate Mutase 5 Reduces Necroptosis in Rat Hearts Following Ischemia/Reperfusion Through Suppression of Dynamin-Related Protein 1. Cardiovasc Drugs Ther, 2019,33(1):13–23
Zhu P, Hu S, Jin Q, et al. Ripk3 promotes ER stress-induced necroptosis in cardiac IR injury: A mechanism involving calcium overload/XO/ROS/mPTP pathway. Redox Biol, 2018,16:157–168
Zhu P, Wan K, Yin M, et al. RIPK3 Induces Cardiomyocyte Necroptosis via Inhibition of AMPK-Parkin-Mitophagy in Cardiac Remodelling after Myocardial Infarction. Oxid Med Cell Longev, 2021, 2021:6635955
Berkhemer OA, Fransen PS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med, 2015,372(1):11–20
Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med, 2015,372(24):2296–2306
Baker TS, Robeny J, Cruz D, et al. Stimulating the Facial Nerve to Treat Ischemic Stroke: A Systematic Review. Front Neurol, 2021,12:753182
Xie Q, Zhang L, Xie L, et al. Z-ligustilide: A review of its pharmacokinetics and pharmacology. Phytother Res, 2020,34(8):1966–1991
Mondal NK, Behera J, Kelly KE, et al. Tetrahydrocurcumin epigenetically mitigates mitochondrial dysfunction in brain vasculature during ischemic stroke. Neurochem Int, 2019,122:120–138
Di Y, He YL, Zhao T, et al. Methylene Blue Reduces Acute Cerebral Ischemic Injury via the Induction of Mitophagy. Mol Med, 2015,21(1):420–429
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflict of interest.
Author Kun XIONG is a member of the Young Editorial Board for Current Medical Science. The paper was handled by the other editor and has undergone rigorous peer review process. Author Kun XIONG was not involved in the journal’s review of, or decision related to, this manuscript.
Additional information
This work was supported by grants from the National Natural Science Foundation of China (No. 81971891, No. 82172196, No. 81772134 and No. 81571939), Key Laboratory of Emergency and Trauma (Hainan Medical University) of Ministry of Education (No. KLET-202108), and the College Students’ Innovation and Entrepreneurship Project (No. S20210026020013).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, Yd., Li, Zx., Hu, Xm. et al. Insight into Crosstalk Between Mitophagy and Apoptosis/Necroptosis: Mechanisms and Clinical Applications in Ischemic Stroke. CURR MED SCI 42, 237–248 (2022). https://doi.org/10.1007/s11596-022-2579-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2579-3