Summary
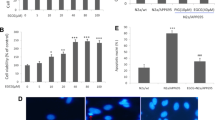
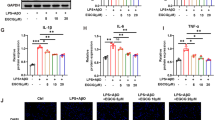
Alzheimer’s disease (AD) shows cognitive impairments in clinic, which is multifactorial with different etiopathogenic mechanisms such as Aβ deposition, neuroinflammation and neuronal dystrophy involved. Therefore, multi-targets drugs with neuroprotective, anti-amyloidogenic and anti-inflammatory properties will be effective in AD treatment. Epigallocatechin-3-gallate (EGCG) possesses a broad spectrum of pharmacological activities in the prevention and treatment of multiple neurodegenerative diseases. In the present study, we showed that oral administration of EGCG (50 mg/kg) for 4 months significantly attenuated the cognitive deficits in APP/PS1 transgenic mice, which served as AD model. Moreover, EGCG induced an improvement in dendritic integrity and expression levels of synaptic proteins in the brain of APP/PS1 mice. And EGCG exerted obvious anti-inflammatory effects, which was manifested by alleviating microglia activation, decreasing pro-inflammatory cytokine (IL-1β) and increasing anti-inflammatory cytokines (IL-10, IL-13). Furthermore, β-amyloid (Aβ) plaques were markedly reduced in the hippocampus of 6-month old APP/PS1 mice after EGCG treatment. In conclusion, these findings indicate that EGCG improves AD-like cognitive impairments through neuroprotective, anti-amyloidogenic and anti-inflammatory effects, thus is a promising therapeutic candidate for AD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Iqbal K, Grundke-Iqbal I, Zaidi T, et al. Defective brain microtubule assembly in Alzheimer’s disease. Lancet, 1986,2(8504):421–426
Bjorklund G, Aaseth J, Dadar M, et al. Molecular Targets in Alzheimer’s Disease. Mol Neurobiol, 2019,56(10):7032–7044
Long JM, Holtzman DM. Alzheimer Disease: An Update on Pathobiology and Treatment Strategies. Cell, 2019,179(2):312–339
Vassar R, Kovacs DM, Yan R, et al. The beta-secretase enzyme BACE in health and Alzheimer’s disease: regulation, cell biology, function, and therapeutic potential. J Neurosci, 2009,29(41):12 787–12 794
Yan R, Vassar R. Targeting the beta secretase BACE1 for Alzheimer’s disease therapy. Lancet Neurol, 2014,13(3):319–329
De Strooper B, Vassar R, Golde T. The secretases: enzymes with therapeutic potential in Alzheimer disease. Nat Rev Neurol, 2010,6(2):99–107
Iqbal K, Alonso Adel C, Grundke-Iqbal I. Cytosolic abnormally hyperphosphorylated tau but not paired helical filaments sequester normal MAPs and inhibit microtubule assembly. J Alzheimers Dis, 2008,14(4):365–370
Iqbal K, Grundke-Iqbal I. Ubiquitination and abnormal phosphorylation of paired helical filaments in Alzheimer’s disease. Mol Neurobiol, 1991,5(2–4):399–410
Iqbal K, Grundke-Iqbal I. Alzheimer neurofibrillary degeneration: significance, etiopathogenesis, therapeutics and prevention. J Cell Mol Med, 2008,12(1):38–55
Alonso AC, Zaidi T, Grundke-Iqbal I, et al. Role of abnormally phosphorylated tau in the breakdown of microtubules in Alzheimer disease. Proc Natl Acad Sci USA, 1994,91(12):5562–5566
Hoover BR, Reed MN, Su J, et al. Tau Mislocalization to Dendritic Spines Mediates Synaptic Dysfunction Independently of Neurodegeneration. Neuron, 2010,68(6):1067–1081
Domingues C, da Cruz ESOAB, Henriques AG. Impact of Cytokines and Chemokines on Alzheimer’s Disease Neuropathological Hallmarks. Curr Alzheimer Res, 2017,14(8):870–882
McGeer PL, McGeer EG. The amyloid cascade-inflammatory hypothesis of Alzheimer disease: implications for therapy. Acta Neuropathol, 2013,126(4):479–497
Han QQ, Shen TT, Wang F, et al. Preventive and Therapeutic Potential of Vitamin C in Mental Disorders. Curr Med Sci, 2018,38(1):1–10
Agostinho P, Cunha RA, Oliveira C. Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer’s disease. Curr Pharm Des, 2010,16(25):2766–1678
Dzamba D, Harantova L, Butenko O, et al. Glial Cells — The Key Elements of Alzheimer’s Disease. Curr Alzheimer Res, 2016,13(8):894–911
Guerriero F, Sgarlata C, Francis M, et al. Neuroinflammation, immune system and Alzheimer disease: searching for the missing link. Aging Clin Exp Res, 2017,29(5):821–831
Meda L, Cassatella MA, Szendrei GI, et al. Activation of microglial cells by beta-amyloid protein and interferon-gamma. Nature, 1995,374(6523):647–650
Meraz-Rios MA, Toral-Rios D, Franco-Bocanegra D, et al. Inflammatory process in Alzheimer’s Disease. Front Integr Neurosci, 2013,7:59
Morales I, Guzman-Martinez L, Cerda-Troncoso C, et al. Neuroinflammation in the pathogenesis of Alzheimer’s disease. A rational framework for the search of novel therapeutic approaches. Front Cell Neurosci, 2014,8:112
Canter RG, Penney J, Tsai LH. The road to restoring neural circuits for the treatment of Alzheimer’s disease. Nature, 2016,539(7628):187–196
Querfurth HW, LaFerla FM. Alzheimer’s disease. N Engl J Med, 2010,362(4):329–344
Guo S, Bezard E, Zhao B. Protective effect of green tea polyphenols on the SH-SY5Y cells against 6-OHDA induced apoptosis through ROS-NO pathway. Free Radical Biol Med, 2005,39(5):682–695
Nanjo F, Goto K, Seto R, et al. Scavenging effects of tea catechins and their derivatives on 1,1-diphenyl-2-picrylhydrazyl radical. Free Radical Biol Med, 1996,21(6):895–902
Singh R, Akhtar N, Haqqi TM. Green tea polyphenol epigallocatechin-3-gallate: inflammation and arthritis. Life Sci, 2010,86(25–26):907–918
Unno K, Takabayashi F, Kishido T, et al. Suppressive effect of green tea catechins on morphologic and functional regression of the brain in aged mice with accelerated senescence (SAMP10). Exp Gerontol, 2004,39(7):1027–1034
Unno K, Takabayashi F, Yoshida H, et al. Daily consumption of green tea catechin delays memory regression in aged mice. Biogerontology, 2007,8(2):89–95
Rezai-Zadeh K, Arendash GW, Hou H, et al. Green tea epigallocatechin-3-gallate (EGCG) reduces beta-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res, 2008,1214:177–187
Rezai-Zadeh K, Shytle D, Sun N, et al. Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. J Neurosci, 2005,25(38):8807–8814
Vorhees CV, Williams MT. Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc, 2006,1(2):848–858
Ding Y, Qiao AM, Wang ZQ, et al. Retinoic Acid Attenuates beta-Amyloid Deposition and Rescues Memory Deficits in an Alzheimer’s Disease Transgenic Mouse Model. J Neurosci, 2008,28(45):11 622–11 634
Cao DF, Lu HL, Lewis TL, et al. Intake of sucrose-sweetened water induces insulin resistance and exacerbates memory deficits and amyloidosis in a transgenic mouse model of Alzheimer disease. J Biol Chem, 2007,282(50):36 275–36 282
Kurt MA, Davies DC, Kidd M, et al. Hyperphosphorylated tau and paired helical filament-like structures in the brains of mice carrying mutant amyloid precursor protein and mutant presenilin-1 transgenes. Neurobiol Dis, 2003,14(1):89–97
Arab H, Mahjoub S, Hajian-Tilaki K, et al. The effect of green tea consumption on oxidative stress markers and cognitive function in patients with Alzheimer’s disease: A prospective intervention study. Caspian J Intern Med, 2016,7(3):188–194
Chan DKY, Mellick GD, et al. Genetic and environmental risk factors and their interactions for Parkinson’s disease in a Chinese population. J Clin Neurosci, 2003,10(3):313–315
Kuriyama S, Hozawa A, Ohmori K, et al. Green tea consumption and cognitive function: a cross-sectional study from the Tsurugaya Project 1. Am J Clin Nutr, 2006,83(2):355–361
Ritchie K, Lovestone S. The dementias. Lancet, 2002,360(9347):1759–1766
Grinberg LN, Newmark H, Kitrossky N, et al. Protective effects of tea polyphenols against oxidative damage to red blood cells. Biochem Pharmacol, 1997,54(9):973–978
van Acker SA, van den Berg DJ, Tromp MN, et al. Structural aspects of antioxidant activity of flavonoids. Free Radical Biol Med, 1996,20(3):331–342
Okello EJ, Leylabi R, McDougall GJ. Inhibition of acetylcholinesterase by green and white tea and their simulated intestinal metabolites. Food Funct, 2012,3(6):651–661
Qin XY, Cheng Y, Yu LC. Potential protection of green tea polyphenols against intracellular amyloid beta-induced toxicity on primary cultured prefrontal cortical neurons of rats. Neurosci Lett, 2012,513(2):170–173
Singh NA, Mandal AK, Khan ZA. Potential neuroprotective properties of epigallocatechin-3-gallate (EGCG). Nutrition J, 2016,15(1):60
Haque AM, Hashimoto M, Katakura M, et al. Long-term administration of green tea catechins improves spatial cognition learning ability in rats. J Nutr, 2006,136(4):1043–1047
Erba D, Riso P, Bordoni A, et al. Effectiveness of moderate green tea consumption on antioxidative status and plasma lipid profile in humans. J Nutr Biochem, 2005,16(3):144–149
Panza VSP, Wazlawik E, Schuetz GR, et al. Consumption of green tea favorably affects oxidative stress markers in weight-trained men. Nutrition, 2008,24(5):433–442
Sartor L, Pezzato E, Garbisa S. (−)Epigallocatechin-3-gallate inhibits leukocyte elastase: potential of the phyto-factor in hindering inflammation, emphysema, and invasion. J Leukocyte Biol, 2002,71(1):73–79
Dona M, Dell’Aica I, Calabrese F, et al. Neutrophil, restraint by green tea: Inhibition of inflammation, associated angiogenesis, and pulmonary fibrosis. J Immunol, 2003,170(8):4335–4341
San Yeoh B, Olvera RA, Singh V, et al. Epigallocatechin-3-Gallate Inhibition of Myeloperoxidase and Its Counter-Regulation by Dietary Iron and Lipocalin 2 in Murine Model of Gut Inflammation. Am J Pathol, 2016,186(4):912–926
Ban JY, Jeon SY, Bae K, et al. Catechin and epicatechin from Smilacis chinae rhizome protect cultured rat cortical neurons against amyloid beta protein (25–35)-induced neurotoxicity through inhibition of cytosolic calcium elevation. Life Sci, 2006,79(24):2251–2259
Koh SH, Lee SM, Kim HY, et al. The effect of epigallocatechin gallate on suppressing disease progression of ALS model mice. Neurosci Lett, 2006,395(2):103–107
Reznichenko L, Amit T, Youdim MB, et al. Green tea polyphenol (−)-epigallocatechin-3-gallate induces neurorescue of long-term serum-deprived PC12 cells and promotes neurite outgrowth. J Neurochem, 2005,93(5):1157–1167
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors have no conflict of interest.
This work was supported in part by the National Natural Science Foundation of China (No. 31800851).
Rights and permissions
About this article
Cite this article
Bao, J., Liu, W., Zhou, Hy. et al. Epigallocatechin-3-gallate Alleviates Cognitive Deficits in APP/PS1 Mice. CURR MED SCI 40, 18–27 (2020). https://doi.org/10.1007/s11596-020-2142-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2142-z