Abstract
A new fungal generic name Hymenochaetopsis is proposed to replace Pseudochaete, which is a nomen illegitimum according to Art. 53.1 in the International Code of Nomenclature for algae, fungi, and plants. Twelve new combinations are proposed for species in genera Hymenochaete and Pseudochaete. Hymenochaetopsis laricicola is described and illustrated as a new species from Northeast China based on morphological and molecular evidence. It is characterized by effused-reflexed to pileate basidiocarps, presence of a cortex and hyphal layer, relative small hymenial setae, short cylindrical basidiospores, and exclusive growth on Larix. Hymenochaetopsis gigasetosa, which was previously reported from its type locality only, is newly recorded in China. An identification key is provided for the 13 known species of Hymenochaetopsis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pseudochaete T. Wagner & M. Fisch. was segregated from Hymenochaete Lév. as a monotypic genus with Auricularia tabacina as the type species (Wagner & Fischer 2002). Recently, ten species, either newly described or combined from Hymenochaete, Cyclomyces Kunze ex Fr., and Hydnochaete Bres. were added to the genus (He & Dai 2012; He & Li 2013; Parmasto et al. 2014) based on morphological and molecular data. However, Pseudochaete has already been used as a generic name for algae since 1903 (West & West 1903; Guiry & Guiry 2015), which makes it a nom. illegit. for fungi according to Art. 53.1 in the International Code of Nomenclature for algae, fungi, and plants (ICN, IAPT: http://www.iapt-taxon.org). Therefore, a new fungal generic name is needed to accommodate the fungal species under the name of Pseudochaete.
Several specimens collected on Larix from Northeastern China were identified by the senior author as Hymenochaete lenta, which was reported from Costa Rica in South America (He & Li 2011). Those specimens were recently restudied and compared with a H. lenta specimen collected from Venezuela. The results show that the Chinese species is not H. lenta, but represents an undescribed taxon. In addition, two specimens collected from Yunnan Province, southwestern China, are identified as Hymenochaete gigasetosa, which was previously reported from its type locality in India only.
In this paper: (1) a new generic name Hymenochaetopsis and twelve new combinations are proposed; (2) Hymenochaetopsis laricicola sp. nov. and H. gigasetosa new to China are illustrated and described; and (3) an emended description of Hymenochaetopsis and an identification key to all the 13 known species are provided.
Materials and methods
Morphological studies.—Voucher specimens are deposited in the herbarium of Beijing Forestry University (BJFC). Microscopic procedures follow Dai (2010). In the text the following abbreviations are used: L = mean spore length, W = mean spore width, Q = variation in the L/W ratios between the specimens studied, n = the number of spores measured from given number of specimens, KOH = potassium hydroxide, IKI– = inamyloid and nondextrinoid, CB– = acyanophilous. Color designations are from Kornerup and Wanscher (1978). Herbarium codes follow Index Herbariorum (Thiers 2015).
DNA extraction and sequencing.—A CTAB plant genome rapid extraction kit-DN14 (Aidlab Biotechnologies Co., Ltd) was employed for DNA extraction and PCR amplification from dried specimens according to the manufacturer’s instructions. Approximately 600 base pairs of the ITS rDNA were amplified with primers ITS5 and ITS4 (White et al. 1990), using the following procedure: initial denaturation at 95 °C for 3 min, followed by 34 cycles of 94 °C for 40 s, 58 °C for 45 s and 72 °C for 1 min, and a final extension of 72 °C for 10 min. DNA sequencing was performed at Beijing Genomics Institute, and the newly generated sequences were deposited in the GenBank database.
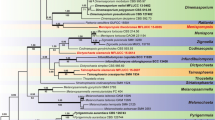
Phylogenetic analysis.—The molecular phylogeny of Hymenochaetopsis was inferred from the analysis of 15 ITS sequences of 12 species (Table 1). The generic type of Hymenochaete, H. rubiginosa (Dicks.) Lév. was selected as the outgroup. The sequences were aligned using the ClustalX 1.83 (Chenna et al. 2003). Alignments were optimized manually in BioEdit 7.0.5.3 (Hall 1999).
Maximum parsimony analysis was performed using PAUP* 4.0b10 (Swofford 2002). Gaps in the alignments were treated as missing data. Trees were generated using 100 replicates of random stepwise addition of sequence and tree-bisection reconnection (TBR) branch-swapping algorithm, with all characters given equal weight. Branch supports for all parsimony analysis were estimated by performing 1000 bootstrap replicates (Felsenstein 1985) with a heuristic search of ten random-addition replicates for each bootstrap replicate. The tree length (TL), consistency indices (CI), retention indices (RI), rescaled consistency indices (RC), and homoplasy index (HI) were calculated for each tree generated. Trees were figured in Treeview 1.6.6 (Page 1996).
Phylogeny results
The ITS dataset includes 16 sequences representing 12 ingroup and one outgroup species. Five sequences were newly obtained (Table 1). The dataset has an aligned length of 667 characters with 104 of them parsimony informative. One parsimony tree is a yield from the analysis (TL = 388, CI = 0.804, RI = 0.757, RC = 0.609, HI = 0.196, Fig. 1). The analyses show that species of Hymenochaetopsis are well separated from the outgroup, and H. laricicola appears as a distinct lineage with strong support (bootstrap value = 100 %). Hymenochaetopsis gigasetosa is closely related to H. latesetosa.
Taxonomy
Hymenochaetopsis S.H. He & Jiao Yang, nom. nov.
MycoBank: MB 814943
Typus: Auricularia tabacina Sowerby, Brit. Fung., pl. 25, 1797.
Synonym: Pseudochaete T. Wagner & M. Fisch., Mycol. Progr. 1: 100, 2002 (nom. illegit.); non Pseudochaete W. West & G.S. West, Journal of Botany, London 41: 37, 1903 (Algae).
Etymology: Hymenochaetopsis (Lat.) refers to the morphological resemblence to Hymenochaete.
Basidiocarps annual, resupinate, effused-reflexed or pileate, thin, membranous, coriaceous or corky. Hymenophore smooth, hydnoid, semi-lamellate or lamellate, more or less cracked with age, usually brown colored, turning black in KOH. Hyphal system monomitic or subdimitic; generative hyphae simple-septate; setal hyphae present in some species. Setae usually abundant, reddish-brown, subulate, more or less encrusted. Basidia clavate to subcylindrical, with four sterigmata, simple-septate at the base. Basidiospores narrowly cylindrical to allantoid, hyaline, thin-walled, smooth, IKI–, CB–. Causing a white rot on angiospermous or gymnospermous wood.
Hymenochaetopsis laricicola S.H. He & Jiao Yang, sp. nov. (Figs. 2c and 3)
MycoBank: MB 814944
Type.—CHINA. Heilongjiang Prov., Ning’an County, Jingbohu Forestry Park, on fallen branch of Larix sp., 5.IX.2013, Y.C. Dai 13458 (holotype, BJFC!).
Etymology.—laricicola (Lat.) refers to the growth on Larix.
Fruitbody.—Basidiocarps annual, broadly effused-reflexed or pileate, closely adnate, coriaceous or corky, more or less brittle when dry. Pilei confluent, imbricate, dimidiate, semicircular or fan-shaped, projecting up to 1.5 cm, up to 0.5 mm at the base; pileal surface radiately fibrillose, silky, becoming glabrous with age, concentrically zonate and sulcate, light brown (6D6–8), brown (7E4–5), grey (6E1) or brownish grey (6E2); margin thin, entire, curving down when dry, concolorous with the pileal suface. Hymenophore smooth, usually densely cracked with age, with numerous crevices, brown (7E5–8), brownish grey (7 F2), reddish brown (8E4–6) or dark brown (7 F5–8); margin thinning out, fimbriate, lighter colored or concolorous with hymenophore surface.
Hyphal structure.—Hyphal system monomitic; generative hyphae without clamp connections; tissue darkening but otherwise unchanged in KOH.
Subiculum.—Tomentum, cortex and hyphal layer present. Cortex composed of strongly agglutinated hyphae, 30–40 μm thick. Generative hyphae in hyphal layer hyaline or yellowish-brown, regularly arranged or more or less interwoven, thin- to thick-walled with a wide lumen, rarely branched, 2–4.5 μm in diam.
Stratified hymenium.—Hyphae in this layer similar to those in subiculum, yellowish-brown, agglutinated, interwoven, 2–4 μm in diam. Hymenium composed of setae, basidia, and basidioles. Hymenial setae abundant, subulate, reddish-brown, thick-walled, sometimes slightly encrusted with crystals in the tips, with bluntly acute or obtuse tip, (20) 25–50 (60) × (6) 7–10 (15) μm, projecting up to 30 μm above the hymenium. Setae in subhymenium (setal hyphae) frequent, more or less parallel along the substrate with tips bending into the hymenium, up to 120 × 12 μm. Cystidia and hyphidia absent. Basidia clavate to subcylindrical, hyaline, with 4-sterigmata, and simple-septate at the base, 12–25 × 3–5 μm; walls thickening toward the base; basidioles numerous, similar to basidia but shorter.
Spores.—Basidiospores narrowly and shortly cylindrical, hyaline, thin-walled, smooth, IKI–, CB–, 4–6 (7) × 1.5–2 μm, L = 5 μm, W = 1.7 μm, Q = 2.8–3.2 (n = 90/3).
Habitat.—Growing on the bark of dead or fallen trees of Larix.
Additional specimens examined.—Hymenochaetopsis laricicola: CHINA. Heilongjiang Prov., Huma County, Nanwenghe Nat. Res., on fallen trunk of Larix gmelinii, 27.VIII.2014, Y.C. Dai 14607; Jilin Prov., Antu County, Changbaishan Nat. Res., on bark of Larix, VII.2012, S.H. Wu 1207–122; Neimenggu Autonomous Region, Genhe County, Greater Higgnan Mountains Nat. Res., on dead tree of Larix gmelinii, 28.VIII.2009, Y.C. Dai 11046 (ITS: JQ279616); Arxan County, Arxan National Forest Park, on fallen trunk of Larix gmelinii, 20.X.2015, S.H. He 3109.
Hymenochaetopsis lenta: Venezuela: Estado Bolivar, Las Nieves, on dead hardwood, 12.VI.1995, L. Ryvarden 37887 (O!).
Hymenochaetopsis corrugata (Fr.) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814946
Basionym: Thelephora corrugata Fr., Observ. Mycol. 1: 154, 1815
Synonyms: Hymenochaete corrugata (Fr.) Lév., Ann. Sci. Nat., Bot., III 5: 152, 1846; Pseudochaete corrugata (Fr.) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis gigasetosa (Parmasto) S.H. He & Jiao Yang, comb. nov. (Figs. 2b and 4)
MycoBank: MB 814947
Basionym: Hymenochaete gigasetosa Parmasto, Folia Cryptog. Estonica, Fasc. 40: 41, 2003
Fruitbody.—Basidiocarps annual, resupinate, closely adnate, coriaceous, first as small patches, later confluent up to 20 cm, 100–300 μm thick in cross-section. Hymenophore smooth, brownish orange (6C5–8) or light brown (6D5–8), not cracked with age; margin thinning out, distinct, fimbriate, white (6D1) when juvenile, becoming indistinct, concolorous with hymenophore surface with age.
Hyphal structure.—Hyphal system monomitic; generative hyphae without clamp connections; tissue darkening but otherwise unchanged in KOH.
Subiculum.—Tomentum absent. Cortex absent or sometimes a thin dark line present, 5–20 μm in diam., composed of strongly agglutinated hyphae. Hyphal layer present, usually indistinct with thickening setal layer. Generative hyphae hyaline to yellowish brown, thin- to thick-walled with a wide lumen, moderately septate and branched, loosely interwoven, 2–5 μm in diam. Setal hyphae (embedded setae) frequently present when juvenile, more or less parallel along substrate, longer and narrower than hymenial setae, usually bending downwards and penetrating out of the hymenium to represent setae.
Stratified hymenium.—Hyphae in this layer similar to those in subiculum, yellowish brown, thick-walled, agglutinated. Setal layer composed of 1–3 rows of overlapping setae. Setae scattered or rare, originating from hymenium or subiculum, setae from hymenium broadly subulate, vertical to hymenium, usually swollen at the base, shorter and wider than those from subiculum; setae from subiculum usually with a setal hyphae-like base, bent downwards and penetrating out the hymenium; both types of setae reddish brown, with blunt or acute tips, slightly encrusted with crystals in the upper part, projecting up to 100 μm above the hymenium, (70) 80–190 (200) × (8) 9–24 (26) μm. Cystidia absent. Hyphidia present, numerous or sometimes scattered, yellowish brown, usually sinuous, not bifurcated or branched, thick-walled, with round or acute tip, sometimes curved or coiled at the tips. Basidia clavate, with four sterigmata, and simple-septate at the base, 16–20 × 3–4 μm; walls usually thickening at basal part; basidioles similar to basidia but slightly smaller.
Spores.—Basidiospores narrowly cylindrical to slightly allantoid, hyaline, thin-walled, smooth, IKI–, CB–, 6–8 × 1.5–2 μm, L = 6.9 μm, W = 1.9 μm, Q = 3.6–3.8 (n = 60/2).
Type of rot.—Causing a white fibrose rot of wood.
Specimens examined.—CHINA. Yunnan Prov.: Baoshan, Gaoligongshan Nat. Res., Baihualing, elevation ca. 1600 m, on fallen angiosperm twig, 28.X.2012, S.H. He 1442 & 1461. INDIA. Uppar Pradesh, Mundali, Chakrata Forest Division, on dead twigs of Picea morinda, 8.XI.1957, P.S. Rehill (isotype in TAA!); on fallen angiosperm branch, 5.XI.1953, P.S. Rehill (paratype in TAA!).
Hymenochaetopsis intricata (Lloyd) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814948
Basionym: Stereum intricatum Lloyd, Mycol. Writ. 7: 1157, 1922
Synonyms: Hymenochaete intricata (Lloyd) S. Ito, Bot. Mag., Tokyo 44: 156, 1930; Pseudochaete intricata (Lloyd) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis lamellata (Y.C. Dai & Niemelä) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814949
Basionym: Cyclomyces lamellatus Y.C. Dai & Niemelä, in Dai, Härkönen & Niemelä, Ann. Bot. Fenn. 40: 384, 2003
Synonym: Pseudochaete lamellata (Y.C. Dai & Niemelä) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis latesetosa (S.H. He & Hai J. Li) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814989
Basionym: Pseudochaete latesetosa S.H. He & Hai J. Li, Mycol. Progr. 12: 333, 2013
Hymenochaetopsis lenta (G.A. Escobar ex J.C. Léger) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814952
Basionym: Hymenochaete lenta G.A. Escobar ex J.C. Léger, Cryptog. Mycol. 11: 302, 1990
Synonym: Pseudochaete lenta (G.A. Escobar ex J.C. Léger) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis olivacea (Schwein.) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814953
Basionym: Sistotrema olivaceum Schwein., Schr. Naturf. Ges. Leipzig 1: 101, 1822
Synonyms: Hydnum olivaceum (Schwein.) Fr., Elench. Fung. 1: 134. 1828; Hydnochaete olivacea (Schwein.) Banker, Mycologia 6: 234, 1914; Pseudochaete olivacea (Schwein.) Parmasto, in Parmasto, Saar, Larsson & Rummo, Mycol. Progr. 13: 61, 2014
Hymenochaetopsis rigidula (Berk. & M.A. Curtis) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814954
Basionym: Hymenochaete rigidula Berk. & M.A. Curtis, J. Linn. Soc., Bot. 10: 334, 1868 [1869]
Synonym: Pseudochaete rigidula (Berk. & M.A. Curtis) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis subrigidula (S.H. He & Hai J. Li) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814990
Basionym: Pseudochaete subrigidula S.H. He & Hai J. Li, Mycol. Progr. 12: 335, 2013
Hymenochaetopsis tabacina (Sowerby) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814955
Basionym: Auricularia tabacina Sowerby, Col. Fig. Engl. Fung. 1: 14, 1797
Synonyms: Thelephora tabacina (Sowerby) Fr., Syst. Myc. 1: 437, 1821; Stereum tabacinum (Sowerby) Fr., Epicr. Syst. Mycol.: 550, 1838; Hymenochaete tabacina (Sowerby) Lév., Ann. Soc. Nat., Bot III, 5: 152, 1846
Hymenochaetopsis tabacinoides (Yasuda) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814956
Basionym: Irpex tabacinoides Yasuda, Bot. Mag., Tokyo 33: 189, 1919
Synonym: Pseudochaete tabacinoides (Yasuda) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Hymenochaetopsis yasudae (Imazeki) S.H. He & Jiao Yang, comb. nov.
MycoBank: MB 814960
Basionym: Hymenochaete yasudae Imazeki [as ‘yasudai'], Bull. Tokyo Sci. Mus. 2: 6, 1940
Synonym: Pseudochaete yasudae (Imazeki) S.H. He & Y.C. Dai, Fungal Diversity 56: 89, 2012
Key to species of Hymenochaetopsis
-
1. Hymenophore hydnoid or lamellate ---------2
-
1. Hymenophore smooth--------------4
-
2. Hymenophore effused or effused-reflexed, hydnoid, American species-------------H. olivacea
-
2. Hymenophore effused-reflexed or pileate, lamellate or hydnoid, Asian species---------------3
-
3. Hymenophore lamellate---------H. lamellata
-
3. Hymenophore hydnoid or semi-lamellate-------H. tabacinoides
-
4. Growth exclusively on gymnospermous wood-----5
-
4. Growth on angiospermous wood, or occasionally on gymnospermous wood---------------6
-
5. On Pinus or Abies, submoniliform hyphidia numerous-------------------H. yasudae
-
5. On Larix, submoniliform hyphidia absent-------H. laricicola
-
6. Basidiocarps strictly resupinate or slightly effused-reflexed----------------------7
-
6. Basidiocarps effused-reflexed or pileate-------11
-
7. Hyphal layer absent---------------H. corrugata
-
7. Hyphal layer present---------------8
-
8. Setae up to 14 μm wide--------------9
-
8. Setae up to 24 μm wide-------------10
-
9. Setae 40–60 × 7–12 μm, basidiospores 3.7–5 × 1.5–2.3 μm-----------------H. rigidula
-
9. Setae 35–135 × 7–14 μm, basidiospores 5–7 × 1.5–2 μm-----------------H. subrigidula
-
10. Setae 50–150 × 11–23, tropical species-------H. latesetosa
-
10. Setae 80–190 × 9–24, temperate species-------H. gigasetosa
-
11. Subicular hyphae hyaline-------------H. intricata
-
11. Subicular hyphae yellowish brown--------12
-
12. Setal hyphae present, setae 60–120 × 7–15 μm, cosmopolitan species-------------H. tabacina
-
12. Setal hyphae absent, setae 40–55 × 9–15 μm, South America species---------------H. lenta
Discussion
Like those of Hymenochaete, species of Hymenochaetopsis have usually brown colored basidiocarps turning black in KOH, smooth, lamellate or hydnoid hymenophores, simple-septate generative hyphae, a monomitic or subdimitic hyphal system, characteristic subulate setae, smooth and thin-walled basidiospores negative in cotton blue and Melzer’s solution. They cause white rots on both angiospermous and gymnospermous wood from boreal to tropical areas.
Hymenochaetopsis laricicola is characterized by inhabiting gymnospermous trees, effused-reflexed to pileate basidiocarps, presence of a cortex and hyphal layer, relatively small hymenial setae, and short cylindrical basidiospores. Three species, Hymenochaetopsis intricata (Fig. 2a), H. lenta (Fig. 2d), and H. tabacina (Fig. 2e) with effused-reflexed to pileate basidiocarps and smooth hymenophores that are most similar to H. laricicola. However, H. intricata differs from H. laricicola in having a hyaline hyphal layer, more scattered and wider setae, and growth on angiospermous wood; H. lenta differs in having larger basidiocarps with yellowish hymenophores, no setal hyphae, and a distribution in South America; and H. tabacina differs in having larger setae and growth mainly on angiospermous wood. Parmasto (2001) mentioned that Larix is a common host for H. tabacina in North and East Asia. However, according to our investigations in northeast China, H. tabacina was found on several angiosperm trees, e.g., Acer, Betula, Quercus, etc., but never on Larix. Previously, only Hymenochaetopsis yasudae (Fig. 2g) has been reported to grow strictly on conifer trees such as Pinus and Abies, but this species differs from H. laricicola by having larger hymenial setae and submoniliform hyphidia.
Hymenochaetopsis gigasetosa has been previously reported from India only (Parmasto 2003). It is characterized by large setae, presence of setal hyphae and narrowly cylindrical basidiospores. Parmasto did not find basidiospores when he described this species, but gave the data measured by the collector (6.1–8.4 × 1.75–2.1 μm or 5.25–8.75 × 1.75–2.2 μm, Parmasto 2003). The Chinese specimens fit very well with the characters of the Indian materials, except that Chinese specimens have thinner basidiocarps. However, this might be due to the juvenile stage of Chinese specimens. So far, except for the two species, H. lenta (Fig. 2d) and H. olivacea (Fig. 2f) from America, all the other 11 species of Hymenochaetopsis have been recorded in China (Dai & Li 2010; He & Dai 2012; He & Li 2013).
Although ecologically and morphologically Hymenochaetopsis is very similar to Hymenochaete, it always formed a distinct sister clade with Hymenochaete in the phylogenetic trees of Hymenochaetaceae (Wagner & Fischer 2002; Larsson et al. 2006; He & Dai 2012; Parmasto et al. 2014). He & Dai (2012) found that all the species of Hymenochaetopsis have narrowly and shortly cylindrical to allantoid basidiospores with a maximum length of 7 μm and width of 2.5 μm, which might be a useful character to distinguish it from Hymenochaete (basidiospores cylindrical, ellipsoid to subglobose). However, in order to delimit well these two sibling genera, more species in Hymenochaete s.l. need to be sequenced.
References
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG, Thompson JD (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500. doi:10.1093/nar/gkg500
Dai YC (2010) Hymenochaetaceae (Basidiomycota) in China. Fungal Divers 45:131–343. doi:10.1007/s13225-010-0066-9
Dai YC, Li HJ (2010) Notes on Hydnochaete (Hymenochaetales) with a seta-less new species discovered in China. Mycotaxon 111:481–487. doi:10.5248/111.481
Felsenstein J (1985) Confidence intervals on phylogenetics: an approach using bootstrap. Evolution 39:783–791. doi:10.2307/2408678
Guiry MD, Guiry GM (2015) AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. http://www.algaebase.org
Hall TA (1999) Bioedit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series No. 41: 95–98
He SH, Dai YC (2012) Taxonomy and phylogeny of Hymenochaete and allied genera of Hymenochaetaceae (Basidiomycota) in China. Fungal Divers 56:77–93. doi:10.1007/s13225-012-0174-9
He SH, Li HJ (2011) Three species of Hymenochaete (Hymenochaetales) new to China. Mycosystema 30:644–648
He SH, Li HJ (2013) Pseudochaete latesetosa and P. subrigidula spp. nov. (Hymenochaetales, Basidiomycota) from China based on morphological and molecular characters. Mycol Prog 12:331–339. doi:10.1007/s11557-012-0838-6
Kornerup A, Wanscher JH (1978) Methuen Handbook of Colour, 3rd edn. E. Methuen and Co., Ltd., London, 252 P
Larsson KH, Parmasto E, Fischer M, Langer E, Nakasone K, Redhead S (2006) Hymenochaetales: a molecular phylogeny for the hymenochaetoid clade. Mycologia 98:926–936. doi:10.3852/mycologia.98.6.926
Page RMD (1996) Treeview: An application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Parmasto E (2001) Hymenochaetoid fungi (Basidiomycota) of North America. Mycotaxon 79:107–176
Parmasto E (2003) Two new species of Hymenochaete (Hymenomycetes, Basidiomycota) from India. Folia Cryptog Estonica 40:41–42
Parmasto E, Saar I, Larsson E, Rummo S (2014) Phylogenetic taxonomy of Hymenochaete and related genera (Hymenochaetales). Mycol Prog 13:55–64. doi:10.1007/s11557-013-0891-9
Swofford DL (2002) PAUP*: Phylogenetic analysis using parsimony (*and other methods). Version 4.0b10. Sinauer Associates, Sunderland, Massachusetts
Thiers B (2015) Index Herbariorum: a global directory of public herbaria and asscociated staff. New York Botanical Garden’s Virtual Herbarium. http://sweetgum.nybg.org/ih/
Wagner T, Fischer M (2002) Classification and phylogenetic relationships of Hymenochaete and allied genera of the Hymenochaetales, inferred from rDNA sequence data and nuclear behaviour of vegetative mycelium. Mycol Prog 1:93–104. doi:10.1007/s11557-006-0008-9
West W, West GS (1903) Notes on freshwater algae III. J Botany, London 41:32–41, 74–82, pls 446–448
White TJ, Bruns TD, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols, a guide to methods and applications. Academic, San Diego, pp 315–322
Acknowledgments
The authors would like to express their deep thanks to Drs. Karl-Henrik Larsson (O) and Erast Parmasto (TAA) for loan of specimens. Dr. Yu-Cheng Dai (Beijing Forestry University, China) and two anonymous reviewers provided many suggestions for improving the manuscript. Dr. Hai-Jiao Li (Chinese Center for Disease Control and Prevention, China) helped in the line drawings. This study was supported by the National Natural Science Foundation of China (No. 31470144).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Franz Oberwinkler
Rights and permissions
About this article
Cite this article
Yang, J., Dai, LD. & He, SH. Hymenochaetopsis nom. nov. proposed to replace Pseudochaete (Hymenochaetales, Basidiomycota) with descriptions of H. laricicola sp. nov and H. gigasetosa new to China. Mycol Progress 15, 13 (2016). https://doi.org/10.1007/s11557-015-1153-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-015-1153-9