Abstract
Background
Cardiac CT with late iodine enhancement (LIE-CT) may characterize the scarred myocardium, but the role of readers’ experience and scar pattern on LIE-CT diagnostic performance is unknown. Aim was to assess the diagnostic performance of LIE-CT according to readers’ experience, scar pattern and contrast-to-noise ratio (CNR) using late gadolinium enhancement MRI (LGE-MRI) as reference.
Methods
LIE-CT and LGE-MRI images of 40 consecutive patients were analyzed. Two readers with different experience (8 and 2 years) independently analyzed LIE-CT images defining the presence/absence of scar and scar CNR, segmental involvement, transmural pattern and scar etiology. The same parameters were extracted from LGE-MRI by two expert readers in consensus, blinded to the LIE-CT results.
Results
Scars were identified at LGE-MRI in 29/40 patients and 141/680 segments. Scar burden at LIE-CT versus LGE-MRI correlated better for the most experienced reader than for the least experienced one (ρ = 0.954 and ρ = 0.797, p < 0.001). The most experienced reader missed scars in 2 patients and in 21/141 segments; the least experienced in 5 patients and 53/141 segments. The most experienced reader showed higher accuracy and sensitivity compared to the least experienced in per-patient (accuracy: 95% vs. 88%; sensitivity: 93% vs. 83%) and per-segment analysis (accuracy: 96% vs. 92%; sensitivity: 85% vs. 62%). Specificity was excellent (100% per-patient, 99% per-segment,) regardless of readers’ experience. Missed scars had non-ischemic pattern, low scar burden (< 6%) and lower CNR compared to ischemic scars (2.33 vs. 3.54, p = 0.005).
Conclusion
LIE-CT represents an alternative to LGE-MRI, although the impact of readers’ experience on sensitivity for small non-ischemic scars should be considered.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In clinical routine, cardiac magnetic resonance (CMR) with late gadolinium enhancement (LGE) has a crucial role in noninvasive characterization of myocardial structure, identifying typical patterns of both ischemic and non-ischemic scars, revealing the etiology of underlying myocardial damage [1].
LGE is also fundamental for risk stratification, being a predictor of adverse cardiovascular outcomes [2]. LGE extent is associated with adverse ventricular remodeling, ventricular arrhythmias [3], increased risk of heart failure and sudden cardiac death [4]. Therefore, an accurate and prompt identification of myocardial scars is fundamental to diagnose ischemic and non-ischemic cardiomyopathies and to address the best clinical management for each patient.
However, regardless of its indisputable clinical role, CMR is not so widely available, especially in the acute setting. Also, its application is limited in some patients, for example in those with cardiac devices. Although more recent devices are MR-conditional, the presence of implantable cardioverter defibrillator (ICD) or permanent pacemaker (PPM) may significantly impair LGE images assessability due to generator related artifacts [5].
Moreover, patients suffering from claustrophobia or patients unable to tolerate long scanning time are not able to receive CMR.
Hence, a valid and effective imaging alternative to LGE-MRI is useful. In recent years, thanks to technological improvement, computed tomography (CT) has progressively transformed from noninvasive coronary angiography into an imaging tool able to comprehensively assess the heart, offering also the possibility to identify and characterize myocardial scars.
Iodinated contrast media share the same kinetics and dynamics with gadolinium chelates, with a delayed wash-out in scarred myocardium compared to normal myocardium. This generates different iodine concentrations between scarred and non-scarred areas that can be highlighted as areas of late iodine enhancement (LIE) with a 10–15 min delayed CT scan (LIE-CT) [6].
Based on some recent studies which showed the possibility to identify myocardial scars in CT with promising results, LIE-CT could be used in case of CMR contraindications [7]. However, previous studies always investigated LIE in specific clinical settings or referring to a specific cardiomyopathy, like sarcoidosis [8], hypertrophic cardiomyopathy (HCM) [9], myocardial infarction [10, 11] and heart failure [12].
Despite the promising results highlighted, the diagnostic value of LIE-CT in the clinical routine is not completely established, namely in unselected patients with different cardiac diseases reported by physicians with variable expertise.
The question is: “is LIE-CT a robust diagnostic alternative for myocardial scar imaging? How much does the cardiac disease and the reader’s experience impact on the reliability of the diagnostic evaluation performed using LIE-CT?”
The purpose of the present study was to evaluate the role of LIE-CT in the detection and characterization of myocardial scars, using LGE-MRI as standard of reference, in a population of consecutive unselected patients.
Materials and methods
The study was piloted in agreement with the 1964 Helsinki declaration and its later amendments, and it was approved by the institutional review board. All subjects provided written informed consent.
This is a single-center observational study.
LIE-CT and LGE-MR images, acquired between March 2016 and July 2019 in forty consecutive adults (> 18 years) without contraindication to contrast agent administration or impaired renal function (eGFR ≤ 30 mL/min) at a tertiary referral center and university hospital, for routine clinical exclusion of coronary artery disease and structural cardiomyopathy, were retrospectively analyzed. Patients suffered from suspected coronary artery disease (15/40), ventricular arrhythmias (12/40), dilated cardiomyopathy of unknown origin (6/40), acute chest pain (3/40), atrial fibrillation (2/40), resuscitated sudden cardiac death (2/40).
Cardiac CT protocol
CT was acquired on a second-generation dual-source scanner (SOMATOM Definition Flash, Siemens, Germany) during a triphasic bolus injection of iodinated contrast agent (CA) (Ultravist 370, Bayer Healthcare Pharmaceutical; 370 mg iodine/ml). The injection scheme consisted of 75 ml CA, followed by 40 ml of mixed solution at 25% CA and 75% saline, followed by further 40 ml of pure saline. In all patients, CA was administered via an antecubital vein using a 20G cannula.
Prospective ECG-triggering or retrospective gating was used according to patient heart rate. A second dose of CA was injected immediately after the acquisition of the CTA, in order to reach a total iodine dose of 0.6 iodine grams per kilogram of body weight [13] (range 97–140 ml of total CA). LIE was acquired 10 min after the second administration of CA, using a low voltage axial prospective scan at 75% phase of cardiac circle. Other scan parameters were as follow: kV according to patient’s BMI (BMI < 30 = 80 kV; BMI ≥ 30 = 100 kV); rotation time 0.28 s; detector collimation 128 × 0.6 mm; matrix size 512 × 512; display field of view limited to the heart.
The median dose length product (DLP) was 195 mGy cm [IQR 104–368 mGy cm] for the CTA scan acquired with prospective trigger and 441 mGy cm [IQR 263–772 mGy cm] for CTA scan acquired with retrospective gating.
For the LIE-CT scan, the median DLP was 115 mGy cm [IQR 54–156 mGy cm].
Total median DLP was 459 mGy cm [IQR 208–691.5 mGy cm].
Cardiac MR protocol
All patients underwent CMR on a 1.5 T scanner (Ingenia, Philips Medical Systems, Eindhoven, The Netherlands) equipped with a 32-channel body coil, within 2 weeks after CT. CMR was performed using a comprehensive protocol including long- and short-axis cine images, T2w-STIR sequences and LGE for the determination of presence, distribution and pattern of myocardial scar.
LGE was acquired 10 min after intravenous injection of 0.15 mmol/kg of body weight of gadolinium-chelate CA (Gadovist 1.0 mmol/ml, Bayer Healthcare Pharmaceutical) in the short- and long-axis planes, using 2D inversion recovery gradient-echo sequence (slice thickness = 8 mm; gap = 0 mm; voxel size = 1.6 × 1.8 mm; repetition time ms/echo time ms = 6/3; FA = 30°; FOV = 370 × 331 × 64–96 mm, NAS = 1) with inversion time optimized to null normal myocardium, covering the entire left ventricle (LV).
Image analysis
Two observers (AP and DV, with 8 and 2 years of experience in cardiac imaging) independently evaluated LIE-CT images blinded to other imaging data and to any patient-related information and provided a LIE-CT-based diagnosis.
LGE-MRI images were evaluated by the consensus of two independent experienced radiologists (AE and FDC, 15 and 20 years of experience in cardiac imaging, respectively) and provided an LGE-MRI-based final diagnosis.
LGE-MRI was considered the standard of reference.
The presence of LIE was visually evaluated according to the American Heart Association (AHA) 17-segment model on LIE-CT images reconstructed with B26f soft kernel and SAFIRE 4 and reformatted in the short-axis plane (8 mm slice thickness; gap 0 mm) in average mode. Segmental LIE was also classified according to its transmural pattern (subendocardial, mesocardial, subepicardial, transmural). Myocardial scars identified on LIE images were manually segmented by both observers using a dedicated software (Intellispace Portal v.9.0, Philips). The percentage of LIE was quantified as the ratio of LIE to entire LV mass.
As for LIE, LGE was classified according to the segmental distribution and the transmural pattern.
LGE was semiautomatically quantified using a dedicated software (Intellispace Portal v. 9.0, Philips) with a full width half maximum method [14].
The contrast-to-noise ratio (CNR) was calculated as the difference in CT attenuation or MR signal intensity between hyperenhanced (scarred) and normal myocardium, divided by the standard deviation (SD) of normal myocardium attenuation, the latter obtained with a circular region of interest (> 10 mm2) drawn in normal myocardium [8]. Normal myocardium was defined qualitatively as a region of myocardium without any apparent LGE.
Statistical analysis
Continuous data are expressed as mean ± SD or median (interquartile range (IQR)), according to their distribution. The correlation between scar burden (%) in LIE-CT and LGE-MRI was assessed using Spearman’s correlation test.
Cohen’s kappa (κ) coefficient (< 0.20: poor; 0.20–0.40: fair; 0.40–0.60: moderate; 0.60–0.80: good; 0.80–1.00: very good agreement) was calculated for the assessment of inter-observer agreement in LIE-CT evaluation and for the assessment of segmental and per-patient agreement between LIE-CT and LGE-MRI.
Mann–Whitney U test was used to compare the CNR of LIE-CT according to scar transmurality and clinical diagnosis.
Statistical analyses were performed using SPSS v.25.0.
A p value of < 0.05 was considered statistically significant.
Results
The patients’ demographics and clinical characteristics are shown in Table 1.
29/40 (72.5%) patients had LGE at MRI, mostly with non-ischemic pattern (20/29, 69%) rather than ischemic pattern (9/29, 31%). LGE-MRI was negative in the remaining 11 patients (27.5%).
Among non-ischemic cardiomyopathies (NICM), LGE pattern was suggestive for: myocarditis in 13/20 (65%) patients, idiopathic dilated cardiomyopathy (DCM) in 4/20 (20%) and HCM in 3/20 (15%) patients.
Median scar burden at LGE-MRI was 6% [IQR 3–16%]. Scar burden was slightly but not significantly smaller for NICM scars in respect to ischemic cardiomyopathy (ICM) scars (5% [IQR 3–11.5%] vs. 11% [IQR 7–16%], p > 0.05).
Median scar burden at LIE-CT was 5.6% [IQR 3.2–13%] as observed by the most experienced reader and 3.8% [IQR 2.5–10%] by the least experienced reader.
The correlation between scar burden at LGE-MRI and at LIE-CT was excellent when LIE-CT was evaluated by the most experienced reader (ρ = 0.929, 0 < 0.001) and good for the least experienced reader (ρ = 0.797, 0 < 0.001).
LIE-CT versus LGE-MR: per-patient analysis
The most experienced reader correctly classified 38/40 (95%) patients for the presence/absence of myocardial scar and scar pattern on LIE-CT images, while the least experienced reader correctly classified 35/40 (88%) patients.
Overall per-patient analysis documented a higher accuracy, sensitivity and NPV for the most experienced reader compared to the least experienced one (accuracy 95% vs. 88%; sensitivity 93% vs. 83%; NPV 85% vs. 69%) (Fig. 1a). Specificity (100%) and PPV (100%) were excellent regardless of reader’s experience (Fig. 1a).
Two cases with thin subepicardial post-myocarditis scars, involving only 2% and 3% of myocardial mass at LGE-MRI, were missed by both the readers.
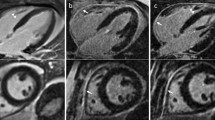
The least experienced reader missed three more cases: 2 small subepicardial post-myocarditis scars, involving 3% and 6% of myocardial mass, respectively, and 1 mesocardial scar in the basal posterior septum, involving 3% of LV myocardial mass in a HCM patient (Fig. 2).
LGE-MR and LIE-CT in a 56-year-old female with hypertrophic cardiomyopathy (HCM). Short-axis LGE. a A hypertrophic myocardium with increased thickness in the posterior septum were a shadowed mesocardial scar is present (arrow). The same scar can be recognized in LIE-CT (arrow in b). This scar was missed by less experienced reader
Therefore, the agreement between LIE-CT and LGE-MRI in evaluating the presence/absence of LIE and the scar pattern was 100% in ICM cases (Fig. 3) independently by readers’ experience while it decreased to 90% and 75% in NICM scars (Fig. 4) for the most and the least experienced reader, respectively.
LIE-CT and LGE-MRI in a 62-year-old male with ischemic cardiomyopathy (ICM). Short-axis (a, b) and long-axis (c). LIE-CT images show a large post-ischemic scar with subendocardial distribution involving the mid- and basal inferolateral wall (arrows in a–c), associated to a thin mesocardial scar in the basal septum (arrowheads in a–c). Myocardial scars at LIE-CT closely agree with corresponding myocardial scars in short-axis (d, e) and long-axis (f) LGE-MRI images
LIE-CT and LGE-MRI in a 53-year-old man with myocardial scars suggestive of non-ischemic etiology. Short-axis LIE-CT (a) and LGE-MRI images (b) show patchy septal enhancement (arrowheads), especially prominent in the anterior and posterior right ventricle insertion point, associated to asymmetric hypertrophy of the septum, suggestive of HCM
LIE-CT versus LGE-MRI: per-segment analysis
A total of 680 myocardial segments were evaluated. One hundred forty-one (21%) segments showed positive LGE-MRI, mainly with a subepicardial pattern (subendocardial LGE 18/141, 12.8%; mesocardial LGE 45/141, 31.9%; subepicardial LGE 57/141, 40.4%; transmural LGE 21/141, 14.9%).
At LIE-CT, the most experienced reader missed 14.9% (21/141) of scarred segments when compared to LGE-MRI. The rate of false negative segments increased to 37.6% (53/141) for the least experienced reader.
The segmental agreement between LIE-CT and LGE-MRI for scar detection or exclusion ranged from very good (k = 0.883; p < 0.001) to good (k = 0.694; p < 0.001), respectively, for the most and the least experienced reader, with a good inter-observer agreement for LIE-CT (k = 0.790; p < 0.001).
Figure 5 shows the detection rate of LIE patterns at segmental analysis in comparison to LGE, according to readers’ experience.
Highest detection rate was obtained for transmural scars, regardless of readers’ experience, with 90% (19/21) of segments with transmural scar identified by the most experienced reader and 71% (15/21) segments identified by the least experienced reader (Fig. 5). The lowest detection rate was achieved for subepicardial scars, with a 71% (46/57) and a 54% (31/57) segmental detection rate by the most and by the least experienced reader, respectively.
Figure 1B shows the diagnostic accuracy of LIE at segmental evaluation in comparison to LGE, according to readers’ experience.
The most experienced reader showed higher diagnostic accuracy (96%) in the identification of scarred segments by LIE-CT, with higher sensitivity (85%) and negative predictive value (NPV) (96%) when compared to the least experienced reader (Fig. 1b). Both readers showed excellent specificity and positive predictive value (PPV) (99% and 97% for the most experienced reader and 99% and 95% for the least experienced reader) (Fig. 1b).
LIE-CT versus LGE-MR: CNR in myocardial scar
In the entire population median CNRMR was 10.6 (IQR 7.15–17.97), and median CNRCT was 2.85 (IQR 1.99–3.53).
Median signal intensity of scarred myocardium was 1086 (IQR 823–1345) and of non-scarred myocardium 161 (IQR 117–202). Median attenuation of scarred myocardium was 127 (IQR 112–150) HU and of non-scarred myocardium 88 (IQR 76–102) HU.
Transmural scar showed higher CNR compared to scars with other patterns both at LIE-CT (median 6.36 [IQR 4.70–6.6] vs. median 2.76 [IQR 1.83–3.52], p = 0.036 Fig. 6a) and at LGE-MR (median 23.64 [IQR 22.27–24.27]) vs. median 10.32 [IQR 6.97–14.55], p = 0.010).
Contrast-to-noise ratio (CNR) in LIE-CT. The box-plots of CNR in relation to the transmural pattern of the scar (a) show higher CNR in transmural rather than in subendocardial, mesocardial and epicardial scars. Subendocardial scars have a slightly higher CNR than mesocardial and epicardial scars, although this difference does not reach statistical significance. CNR was higher in post-ischemic scars (ICM) than in non-ischemic scars (NICM) (b)
CNR of ischemic scars was significantly higher than those of other cardiomyopathies both at LIE-CT (median 3.54 [IQR 3.03–5.34] vs. 2.33 [IQR 1.32–3.01], p = 0.005 Fig. 6b) and at LGE-MR (median 20.90 [IQR 12.84–22.67] vs. 8.73 [IQR 6.62–11.72], p = 0.001).
BMI resulted inversely correlated to CNRCT (R = −0.460, p = 0.016), whereas some correlation was found with CNRMR (R = −0.172, p = 0.372). However, BMI in false negative patients at LIE-CT were not significantly different from true positive patients for the most experienced (p = 0.858) and the least experienced (p = 0.435) reader.
No correlation was found between BSA and CNRCT (R = −0.290, p = 0.142) and between BSA and CNRMR (R = 0.054, p = 0.782), as well as between false negative and true positive patients (p = 0.929 and p = 0.435 for the most and the least experienced reader, respectively).
Discussion
LGE-MRI is a well-established diagnostic and prognostic technique to detect myocardial scars and to define the pattern of LV wall involvement, which is of pivotal importance to address the correct diagnosis, suggesting the underlying etiological mechanism [15], and to guide the more adequate therapeutic management. Moreover, LGE amount is associated to incremental risk of major adverse cardiac events and adverse ventricle remodeling [16, 17].
The continuous improvement in CT technology, in terms of increased spatial and temporal resolution, has led to a further expansion of clinical application of CT. This combined with improvements in detectors technology and iterative reconstruction algorithm allowed a significant noise reduction for images acquired at low energy, expanding the application of CT to myocardial scar characterization.
Similarly to gadolinium-based CA, iodinated CA accumulates in scarred myocardium at equilibrium phase [6], and a low-energy CT scan is able to enhance differences in iodine concentration between scarred and non-scarred myocardium.
A combined evaluation of myocardial scar and coronary arteries anatomy and patency is highly desirable, allowing to simultaneously detect the culprit lesion and its associated myocardial infarction [18], to identify non-ischemic causes of acute chest pain [18], and to evaluate patients with contraindication to MRI (i.e., ICD, PPM, claustrophobia, low compliance) [13] with a short examination time.
The routine application of LIE-CT in the clinical practice is still limited. This is mainly due to the lack of data confirming the diagnostic value of LIE-CT in unselected patient populations with clinical indication for scar imaging.
Considering the potential relevance of scar imaging with CT in clinical practice, the aim of the present study was to evaluate the diagnostic accuracy of LIE-CT in the identification of scarred myocardium in a “real-world” population, also in relation to different readers’ experience.
The results of our study showed that the agreement between LIE-CT and LGE-MRI is dependent on reader’s experience, with a per-patient overall accuracy of 95% and 88% for the most and the least experienced observer, respectively. The agreement between LIE-CT and LGE-MRI resulted to be lower for small thin scars, especially when subepicardial. Subepicardial scars were also associated to a lower CNR when compared to other scars. In both per-segment and per-patient analyses, the specificity and the PPV were excellent regardless of reader’s experience. Moreover, when a myocardial scar was identified on LIE-CT, the scar pattern was perfectly defined. Therefore, LIE-CT can correctly determine the cardiomyopathy underlying the detected scars.
LIE-CT is notoriously affected by lower contrast resolution in respect to LGE-MRI, due to the inability of CT to null the normal myocardium, resulting in significantly lower CNR values and a more challenging identification of scars. The possibility of acquiring a CT scan at the equilibrium phase with low voltages has the double advantage of improving CT delayed scan contrast resolution and of saving dose, in fact the reduction in tube voltage from 120 to 80 kVp accompanies a change in photon energy levels from approximately 66 to 52 keV and improves the attenuation of iodine [19], enhancing scarred myocardium. However, kVp reduction as well as BMI increase are both associated to an increase in image noise. Hence, a trade-off between contrast resolution and noise must be found. For this reason, patients with low BMI have been scanned at 80 kVp, while beam energy was increased to 100 kVp in patients with BMI ≥ 30.
The median CNR value found in our study is comparable with previous studies which were using single-energy low-dose LIE scan (80 kVp) [20]. However, comparing different types of scars, we found that CNR was lower in NICM scars than in ICM scars, thus leading to a lower detection rate for NICM scars. The reason of lower CNR in NICM scars could be explained by the well-known coexistence of fibrotic tissue embedding a variable number of viable myocytes within the NICM scars, while the ICM scars usually include a large central dense scar eventually surrounded by a smaller area of less-dense “border-zone” scar. In fact, non-ischemic scars demonstrated a lower CNR than ischemic scars also at LGE-MRI, but differently form CT, the median CNR of LGE-CMR remain in any case sufficient to detect non-ischemic scars thanks to the capability of LGE-MRI inversion recovery sequences to null the signal of normal myocardium. Moreover, missed scar at LIE-CT were characterized by low scar burden (always < 6%), making it challenging to be distinguished from the grainy background, which can be sometimes experienced in low-energy scans [21].
Reader’s experience seems able to partially overcome the CNR limits in small and shaded scar. In fact, the most experienced reader missed only two scars, which were subepicardial and involving a very small amount of myocardial mass (2%), whereas the least experienced reader missed 4 subepicardial and 1 mesocardial scars, all involving a small percentage of LV mass.
Regardless of similar CNR and segmental extent, the detection rate of mesocardial scars was slightly higher than for subepicardial scars, probably because the surrounding normal myocardium improves the detectability of mesocardial hyperdensity.
Some limitation must be considered when interpreting the present results. Specifically, our study is a single-center study with a relatively small cohort of patients. Larger sample size is required to confirm our results. Second, we did not perform an analysis of inter-observer agreement between two expert readers. The inter-observer agreement among expert readers is beyond the scope of the present study, but it remains an important issue considering the good results achieved by our single expert observer. Furthermore, LIE-CT scan was acquired with a single-energy acquisition mode, although dual-energy mode may increase CNR [12]. The reason of this choice lies on different considerations: Scanners with dual-energy capability based on rapid kVp switching, dual-layer detectors or dual-source technology do not have a widespread diffusion. More importantly, dual-energy LIE-CT imaging results in quite high radiation dose. For example, Ohta et al. [12] reported a median DLP of 258.3 mGy cm (IQR: 258.3–285.3 mGy cm), considerably higher than what we found in our study, despite comparable populations in terms of BMI. Finally, the advantages of the dual-energy approach for LIE detection and quantification are not clearly established, especially on dual-source scanners, because of the halving of temporal resolution and the increase in motion artifacts, originating by the overlapping of two beam energies temporally misaligned by 75 ms [20], in respect to the single-energy approach.
Conclusion
In conclusion, our results suggest that LIE-CT may represent an excellent alternative to LGE-MRI, especially in expert hands. In particular, LIE-CT showed an excellent PPV and capability to define the correct scar pattern. Nevertheless, the NPV resulted significantly affected by the reader’s experience and was particularly limited in case of small subepicardial non-ischemic scars. Hence, a positive LIE-CT scan can be considered reliable for the final diagnosis. A negative LIE-CT scan should always be reviewed by a highly experienced reader; nevertheless, it cannot be considered completely reliable in ruling out the presence of small and thin non-ischemic scars.
Abbreviations
- MRI:
-
Magnetic resonance imaging
- CMR:
-
Cardiac magnetic resonance
- LGE:
-
Late gadolinium enhancement
- ICD:
-
Implantable cardioverter defibrillator
- PPM:
-
Permanent pacemaker
- CT:
-
Computed tomography
- LIE:
-
Late iodine enhancement
- HCM:
-
Hypertrophic cardiomyopathy
- CTA:
-
Computed tomography angiography
- CA:
-
Contrast agent
- DLP:
-
Dose length product
- SD:
-
Standard deviation
- LV:
-
Left ventricle
- AHA:
-
American heart association
- CNR:
-
Contrast-to-noise ratio
- IQR:
-
Interquartile range
- NICM:
-
Non-ischemic cardiomyopathy
- DCM:
-
Dilated cardiomyopathy
- ICM:
-
Ischemic cardiomyopathy
- NPV:
-
Negative predictive value
- PPV:
-
Positive predictive value
References
Mewton N, Liu CY, Croisille P, Bluemke D, Lima JA (2011) Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol 57(8):891–903. https://doi.org/10.1016/j.jacc.2010.11.013
Kuruvilla S, Adenaw N, Katwal AB, Lipinski MJ, Kramer CM, Salerno M (2014) Late gadolinium enhancement on cardiac magnetic resonance predicts adverse cardiovascular outcomes in nonischemic cardiomyopathy: a systematic review and meta-analysis. Circ Cardiovasc Imaging 7(2):250–258. https://doi.org/10.1161/CIRCIMAGING.113.001144
Mavrogeni S, Petrou E, Kolovou G, Theodorakis G, Iliodromitis E (2013) Prediction of ventricular arrhythmias using cardiovascular magnetic resonance. Eur Heart J Cardiovasc Imaging 14(6):518–525. https://doi.org/10.1093/ehjci/jes302
Weng Z, Yao J, Chan RH, He J, Yang X, Zhou Y, He Y (2016) Prognostic value of LGE-CMR in HCM: a meta-analysis. JACC Cardiovasc Imaging 9(12):1392–1402. https://doi.org/10.1016/j.jcmg.2016.02.031
Dickfeld T, Tian J, Ahmad G, Jimenez A, Turgeman A, Kuk R, Peters M, Saliaris A, Saba M, Shorofsky S, Jeudy J (2011) MRI-Guided ventricular tachycardia ablation: integration of late gadolinium-enhanced 3D scar in patients with implantable cardioverter-defibrillators. Circ Arrhythm Electrophysiol 4(2):172–184. https://doi.org/10.1161/CIRCEP.110.958744
Gerber BL, Belge B, Legros GJ, Lim P, Poncelet A, Pasquet A, Gisellu G, Coche E, Vanoverschelde JL (2006) Characterization of acute and chronic myocardial infarcts by multidetector computed tomography: comparison with contrast-enhanced magnetic resonance. Circulation 113(6):823–833. https://doi.org/10.1161/CIRCULATIONAHA.104.529511
di Cesare E, Carbone I, Carriero A, Centonze M, De Cobelli F, De Rosa R, Di Renzi P, Esposito A, Faletti R, Fattori R, Francone M, Giovagnoni A, La Grutta L, Ligabue G, Lovato L, Marano R, Midiri M, Natale L, Romagnoli A, Russo V, Sardanelli F, Cademartiri F, Working Group of the Cardiac Radiology Section of the Italian Society of Medical R (2012) Clinical indications for cardiac computed tomography. From the Working Group of the Cardiac Radiology Section of the Italian Society of Medical Radiology (SIRM). Radiol Med 117(6):901–938. https://doi.org/10.1007/s11547-012-0814-x
Aikawa T, Oyama-Manabe N, Naya M, Ohira H, Sugimoto A, Tsujino I, Obara M, Manabe O, Kudo K, Tsutsui H, Tamaki N (2017) Delayed contrast-enhanced computed tomography in patients with known or suspected cardiac sarcoidosis: a feasibility study. Eur Radiol 27(10):4054–4063. https://doi.org/10.1007/s00330-017-4824-x
Zhao L, Ma X, Delano MC, Jiang T, Zhang C, Liu Y, Zhang Z (2013) Assessment of myocardial fibrosis and coronary arteries in hypertrophic cardiomyopathy using combined arterial and delayed enhanced CT: comparison with MR and coronary angiography. Eur Radiol 23(4):1034–1043. https://doi.org/10.1007/s00330-012-2674-0
Tanabe Y, Kido T, Kurata A, Kouchi T, Fukuyama N, Yokoi T, Uetani T, Yamashita N, Miyagawa M, Mochizuki T (2018) Late iodine enhancement computed tomography with image subtraction for assessment of myocardial infarction. Eur Radiol 28(3):1285–1292. https://doi.org/10.1007/s00330-017-5048-9
Rodriguez-Granillo GA (2017) Delayed enhancement cardiac computed tomography for the assessment of myocardial infarction: from bench to bedside. Cardiovasc Diagn Ther 7(2):159–170. https://doi.org/10.21037/cdt.2017.03.16
Ohta Y, Kitao S, Yunaga H, Fujii S, Mukai N, Yamamoto K, Ogawa T (2018) Myocardial delayed enhancement CT for the evaluation of heart failure: comparison to MRI. Radiology 288(3):682–691. https://doi.org/10.1148/radiol.2018172523
Esposito A, Palmisano A, Antunes S, Maccabelli G, Colantoni C, Rancoita PMV, Baratto F, Di Serio C, Rizzo G, De Cobelli F, Della Bella P, Del Maschio A (2016) Cardiac CT with delayed enhancement in the characterization of ventricular tachycardia structural substrate: relationship between CT-segmented scar and electro-anatomic mapping. JACC Cardiovasc Imaging 9(7):822–832. https://doi.org/10.1016/j.jcmg.2015.10.024
Mikami Y, Kolman L, Joncas SX, Stirrat J, Scholl D, Rajchl M, Lydell CP, Weeks SG, Howarth AG, White JA (2014) Accuracy and reproducibility of semi-automated late gadolinium enhancement quantification techniques in patients with hypertrophic cardiomyopathy. J Cardiovasc Magn Reson 16:85. https://doi.org/10.1186/s12968-014-0085-x
Cummings KW, Bhalla S, Javidan-Nejad C, Bierhals AJ, Gutierrez FR, Woodard PK (2009) A pattern-based approach to assessment of delayed enhancement in nonischemic cardiomyopathy at MR imaging. Radiographics 29(1):89–103. https://doi.org/10.1148/rg.291085052
Gulati A, Jabbour A, Ismail TF, Guha K, Khwaja J, Raza S, Morarji K, Brown TD, Ismail NA, Dweck MR, Di Pietro E, Roughton M, Wage R, Daryani Y, O’Hanlon R, Sheppard MN, Alpendurada F, Lyon AR, Cook SA, Cowie MR, Assomull RG, Pennell DJ, Prasad SK (2013) Association of fibrosis with mortality and sudden cardiac death in patients with nonischemic dilated cardiomyopathy. JAMA 309(9):896–908. https://doi.org/10.1001/jama.2013.1363
Di Marco A, Anguera I, Schmitt M, Klem I, Neilan TG, White JA, Sramko M, Masci PG, Barison A, McKenna P, Mordi I, Haugaa KH, Leyva F, Rodriguez Capitan J, Satoh H, Nabeta T, Dallaglio PD, Campbell NG, Sabate X, Cequier A (2017) Late gadolinium enhancement and the risk for ventricular arrhythmias or sudden death in dilated cardiomyopathy: systematic review and meta-analysis. JACC Heart Fail 5(1):28–38. https://doi.org/10.1016/j.jchf.2016.09.017
Esposito A, Palmisano A, Barbera M, Vignale D, Benedetti G, Spoladore R, Ancona MB, Giannini F, Oppizzi M, Del Maschio A, De Cobelli F (2019) Cardiac computed tomography in troponin-positive chest pain: sometimes the answer lies in the late iodine enhancement or extracellular volume fraction map. JACC Cardiovasc Imaging 12(4):745–748. https://doi.org/10.1016/j.jcmg.2018.08.013
Mahnken AH, Bruners P, Muhlenbruch G, Emmerich M, Hohl C, Gunther RW, Wildberger JE (2007) Low tube voltage improves computed tomography imaging of delayed myocardial contrast enhancement in an experimental acute myocardial infarction model. Invest Radiol 42(2):123–129. https://doi.org/10.1097/01.rli.0000251577.68223.84
Truong QA, Thai WE, Wai B, Cordaro K, Cheng T, Beaudoin J, Xiong G, Cheung JW, Altman R, Min JK, Singh JP, Barrett CD, Danik S (2015) Myocardial scar imaging by standard single-energy and dual-energy late enhancement CT: comparison with pathology and electroanatomic map in an experimental chronic infarct porcine model. J Cardiovasc Comput Tomogr 9(4):313–320. https://doi.org/10.1016/j.jcct.2015.03.003
Reimann AJ, Kuettner A, Klumpp B, Heuschmid M, Schumacher F, Teufel M, Beck T, Burgstahler C, Schroder S, Claussen CD, Kopp AF (2008) Late enhancement using multidetector row computer tomography: a feasibility study with low dose 80 kV protocol. Eur J Radiol 66(1):127–133. https://doi.org/10.1016/j.ejrad.2007.04.013
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
All procedures performed in studies involving human participants were in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Palmisano, A., Vignale, D., Benedetti, G. et al. Late iodine enhancement cardiac computed tomography for detection of myocardial scars: impact of experience in the clinical practice. Radiol med 125, 128–136 (2020). https://doi.org/10.1007/s11547-019-01108-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-019-01108-7