Abstract
With rapid urbanization, built environment has emerged as a set of modifiable factors of cardiovascular disease (CVD) risks. We conducted a systematic review to synthesize evidence on the associations of attributes of urban built environment (e.g. residential density, land use mix, greenness and walkability) with cardiovascular risk factors (e.g. hypertension and arterial stiffness) and major CVD events including mortality. A total of 63 studies, including 31 of cross-sectional design and 32 of longitudinal design conducted across 21 geographical locations and published between 2012 and 2023 were extracted for review. Overall, we report moderately consistent evidence of protective associations of greenness with cardiovascular risks and major CVD events (cross-sectional studies: 12 of 15 on hypertension/blood pressure (BP) and 2 of 3 on arterial stiffness; and longitudinal studies: 6 of 8 on hypertension/BP, 7 of 8 on CVD mortality, 3 of 3 on ischemic heart disease mortality and 5 of 8 studies on stroke hospitalization or mortality reporting significant inverse associations). Consistently, walkability was associated with lower risks of hypertension, arterial stiffness and major CVD events (cross-sectional studies: 11 of 12 on hypertension/BP and 1 of 1 on arterial stiffness; and longitudinal studies: 3 of 6 on hypertension/BP and 1 of 2 studies on CVD events being protective). Sixty-seven percent of the studies were rated as “probably high” risk of confounding bias because of inability to adjust for underlying comorbidities/family history of diseases in their statistical models. Forty-six percent and 14% of the studies were rated as “probably high” risk of bias for exposure and outcome measurements, respectively. Future studies with robust design will further help elucidate the linkages between urban built environment and cardiovascular health, thereby informing planning policies for creating healthy cities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular disease (CVD) is the leading cause of death worldwide [1]. In 2019 alone, an estimated 17.9 million people died from CVDs, accounting for over 30% of deaths globally [2]. CVDs, including ischemic heart disease (IHD) and stroke are complex chronic diseases with multifactorial aetiology and long latency. Well-known risk factors for CVDs include lifestyle and behavioural factors, including dietary habits [3, 4], cigarette smoking [5], alcohol uptake [6], hereditary influences [7], physical activity [8], sleeping quality [9] and socioeconomic status [10]. The role of gene-to-gene and gene-to-environment interactions has also been studied [11]. Among all the CVD risk markers, blood pressure (BP), a circulatory marker has been identified as a primary predictor of heart failure, atrial fibrillation and chronic syndrome disease with the strongest evidence of causation [12, 13]. In addition to BP, other hemodynamic measures such as arterial stiffness have also emerged as objective markers of cardiovascular risk and vascular ageing [14]. Stiffer arteries have long lasting arterial wall damage and relatively reduced capability to expand and contract in response to pressure changes associated with cardiac flows and have been associated with atherosclerosis, CVD events and inflammatory disorders [15]. With an increasing proportion of global population suffering from CVDs, there has been a growing number of studies examining non-pharmacological determinants of cardiovascular risk factors including BP and arterial stiffness.
Relatedly, one of the seventeen sustainable development goals (SDG) framed by United Nations pertains to “Good Health and Well-being”, with an aim to reduce one third of premature mortality from non-communicable diseases including CVDs via a mix of preventive and treatment approaches by 2030 [16]. Reducing cardiovascular risks through urban interventions is of particular importance in cities, given that urban dwellers are likely to be exposed to higher levels of adverse environmental and psychosocial stressors [17, 18]. One of the earliest studies published in 1962 found that Zulu adults who spent more time in town throughout their lifetime reported higher risk of hypertension compared to counterparts who spent a shorter period of their lives in town [19]. In the USA alone, there were 16.1 million deaths attributable to CVD over the period between 1999 and 2017, and 80% of which occurred in metropolitan settings [20].
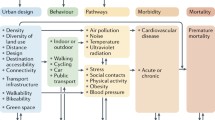
It has been suggested that attributes of built environment in urbanized settings may constitute modifiable risk factors of CVD and CVD risks. A well-designed neighbourhood with appropriate allocations of land uses and street layout, for instance, has the potential to enable easy access to nearby services and key destinations and promote active travel, thereby enhancing physical activity [21]. Previous findings have shown the beneficial associations between residential density and cardiovascular risk factors including physical activity and obesity [22, 23]. Residential proximity to and density of green areas and public open space may promote active lifestyles, relieve stress and improve physical and cardio-respiratory fitness [24, 25]. Exposure to greenspace at standards recommended by the World Health Organization has been estimated to prevent 42,000 deaths annually in European cities [26]. Compact urban design with higher accessibility to bus stops near home and mixed land use (which increases walkable destinations compared to single land use) can also facilitate active travel, enhancing daily physical health thereby reducing cardiovascular risks [27, 28].
There has been accumulating evidence of the associations between urban environmental attributes and CVD [29, 30]. Nonetheless, existing systematic reviews have tended to focus on adiposity and related end points, synthesizing evidence primarily on physical activity, body mass index, obesity, type 2 diabetes and metabolic syndrome [25, 29, 30]. The associations of objectively measured attributes of built environment with hemodynamic measures of CVDs and specific CVD events such as stroke and IHD have remained understudied. In order to better understand underlying aetiology of CVDs and potential risk-minimizing interventions in built environment (including design, configurations and green exposure)[31], we present the first systematic synthesis of evidence of associations of objectively measured built environment exposure measures with key CVD risk factors including hypertension and arterial stiffness and major CVD events including mortality. The included studies were systematically appraised to identify potential methodological biases.
Methodology
Systematic Search and Screening
A systematic search was conducted to summarize existing evidence on the associations of urban environments with cardiovascular risk factors and major CVD events. Journal articles comprising keywords of urban environments (“built environment”, “residential density”, “housing density”, “land use mix”, “greenspace”, “greenness” “walkability”, “street layout”, “land use”, “street connectivity”, “intersection density”, “parks” or “sprawl”) in combination with those related to key cardiovascular risk factors and events (“hypertension”, “blood pressure”, “arterial stiffness”, “stroke”, “coronary heart disease (CHD)”, “myocardial infarction (MI)” or “CVD mortality”) in title or abstract were searched from PubMed, Web of Science and Scopus databases with a wide collection of clinical journal articles. All references were exported to Endnote, followed by a preliminary screening based on title and abstract. Full-text articles were downloaded for further screening in reference to the selection criteria.
Inclusion and Exclusion Criteria
Studies were included for review if they (1) were in English language; (2) objectively measured attributes of residential urban built environment as exposures and (3) included CVD risk factors of hypertension, BP or arterial stiffness or major CVD events as outcomes. We restricted this review to focus only on objectively measured attributes of built environment, given that these constitute systematically measured standardized built environment exposure metrics, easily scalable and replicable in large population-based cohorts. These objectively measured attributes have been known to have inherent ability to explain spatial patterning of health [32, 33]. Review articles, abstracts and reports were excluded. With respect to study design, ecological studies (in which adjustments for individual-level confounders cannot be made) were not included in our review. Studies employing only a composite index of metabolic health as an outcome (such as metabolic syndrome involving systolic and diastolic BP (SBP and DBP), body fat, cholesterol and blood sugar) or measuring urban environment around activity spaces other than residential space (such as around schools or workplace) were also excluded. Key characteristics of the studies pooled (including authoring team, year of publication, geographic setting, study design, sample size, age, sex, outcome, exposure and findings) were entered into a predefined table and presented by study design (cross-sectional and longitudinal design).
Quality Assessment
A systematic appraisal was conducted employing the risk of bias rating tool adopted by the National Toxicology Program’s Office of Health Assessment and Translation, to yield a more nuanced understanding of the study quality [34]. Study quality was evaluated based on seven criteria, including selection bias, confounding bias, attrition/exclusion bias, detection bias for exposure, selective reporting bias and other bias (Text S1). (a) Selection bias occurs if selections of the exposed and non-exposed participants were comparable in terms of, for example, eligibility, inclusion and exclusion criteria, health status and period of recruitment. (b) Confounding bias pertains to residual confounding when key confounders and modifiers (including age, sex, education, income and comorbidities/family history of diseases) were not adjusted for in the analyses resulting in poor internal validity. (c) For cohort studies, substantial loss to follow-up can result in attrition bias. For cross-sectional studies, reasons for sample exclusion should be documented to avoid exclusion bias. (d) Ensuring the accuracy of the exposure measurements can help avoid detection bias for exposure characterization and resulting misclassification. (e) Assessing an outcome by using well-established methods is also beneficial to lowering the risk of detection bias. (f) Selective reporting bias may occur when there is direct evidence that the study’s outcomes outlined in the protocol or abstract are not reported. (g) Lastly, bias may also occur if there are other potential threats to internal validity such as those related to statistical rigor [35]. Each assessment criteria was rated as “definitely low”, “probably low”, “probably high” or “definitely high” risk of bias. The results of the overall risk of bias assessment were presented in a table with symbols as indicated in the rating tool.
Results
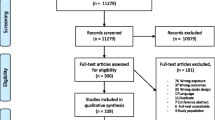
Of the 5543 articles being exported from the systematic search (Fig. 1), 2058 and 3266 articles were excluded due to duplicates and upon primary screening based on title and abstract, resulting in 219 articles for full-text screening. One hundred fifty-six articles were further excluded upon assessments for eligibility, yielding a total of 63 studies (1.8%) for this review.
Of the 63 studies reviewed, 31 (49.2%) were of cross-sectional design, 28 (44.4%) of longitudinal design (cohort studies) and the remaining four (6.3%) comprised of both cross-sectional and longitudinal analyses. Recruited participants were aged ≥15 years in all studies.
Evidence from Studies with Cross-Sectional Design
Tables 1, 2 and 3 show the summary characteristics of the 35 studies of cross-sectional design. These were mostly conducted in multi-context settings, including in the USA (n=9) [39, 46, 48, 49, 53,54,55, 63, 71], China (n=6) [41, 43, 44, 50, 56, 57], Canada (n=4) [36, 60,61,62], the UK (n=3) [51, 52, 66], France (n=2) [59, 64], India (n=2) [45, 70]; and one each in South Africa [67], Ghana [38], Australia [65], Austria [40], Germany [42], South Korea [68], Hong Kong [69] and Spain [47]; and one included data from both Spain and Belgium [37]. All studies comprised both male and female participants, excepting one that included only female (n=1) [42]. With respect to study size, three studies recruited ≤1000 [40, 53, 67], two between 1001 and 2500 [43, 47], six between 2501 and 5000 [42, 45, 49, 51, 59, 71], six between 5001 and 10,000 [37, 38, 48, 64, 65, 70], ten between 10,001 and 100,000 [36, 41, 44, 50, 56, 57, 60, 62, 68, 69], while eight had more than 100,001 participants [39, 46, 52, 54, 55, 61, 63, 66].
Regarding the measurement of outcome variables, 18/35 studies used measured SBP and/or DBP [38, 41, 43,44,45, 47,48,49,50, 53, 59, 60, 62, 64, 66, 67, 69, 71], five employed self-reported doctor-diagnosed hypertension [36, 37, 42, 65, 68], and four defined hypertension using either hospital record [46, 61, 63] or health insurance record [39]. One study included BP readings, self-reported doctor-diagnosed hypertension and self-reported hypotension as outcome variables [40]. Four studies measured arterial stiffness employing either carotid-femoral pulse wave velocity [51, 70] or augmentation index [53] or arterial stiffness index measured from pulse waveform [52]. Among the 19 studies that measured BP, hypertension was defined using cut-off points for SBP and DBP of ≥140/90 mm Hg (n=12) [38, 41, 43, 44, 47, 49, 50, 59, 64, 66, 67, 69] or ≥130/85 mm Hg (n=1) [71], while the remaining six did not define any cut-off point [40, 45, 48, 53, 60, 62]. Of the 19 studies employing measured SBP and/or DBP, BP measurements were repeated three times in nine studies [38, 41, 44, 45, 47, 49, 50, 64, 71], two times in six studies [40, 53, 59, 66, 67, 69], once in one study [48], while relevant information were not provided in the remaining three studies [43, 60, 62]. Of the 13 studies using BP cut-off points to define hypertension, 11 additionally employed antihypertensive medication intake as a criterion [41, 43, 44, 47, 49, 50, 59, 64, 66, 69, 71]. Five studies examined CVD events, which were assessed using either health insurance record [54, 55], imaging techniques [43] or self-reported questionnaire [56, 57]. Specific CVD events included composite CVD endpoints (n=3) [55,56,57], CHD (n=1) [43], stroke/ischemic stroke (n=3) [43, 54, 56], MI, IHD, heart failure and atrial fibrillation (n=1) [55].
A majority of the studies (n=33) employed geographic information system (GIS) to define participants’ residential neighbourhood, one employed Google Street View data [46], and the remaining one employed both [56]. 17/34 studies employed straight-line (Euclidean) buffers to define residential neighbourhoods. The majority of studies used catchment radii of 500 m (n=10) [37, 40,41,42, 44, 50,51,52, 57, 59] and 1000 m (n=8) [40, 41, 45, 50, 51, 53, 56, 57]. A few studies employed street network buffers (n=8) of multiple catchment radii; namely, 800 m (n=3) [61, 65, 69], 1000 m (n=4) [36, 47, 52, 66] and 1600 m (n=3) [59, 65, 69]. One study employed 1250 m by 1250-m2 buffers [48]. Five studies employed other predefined census geographies for exposure measurement including census block (n=3) [39, 54, 55], administrative district (n=1) [68] and dissemination area (n=1) [60]. Another five studies employed publicly available Walk score index which were linked to participants’ postal codes [62], zip code [63] or residential addresses [43, 59, 71]. Relevant data were not found in one study [70].
Among the built environmental exposures, a majority of the studies measured residential greenness from multispectral remote sensing data, modelled in terms of metrics of normalized difference vegetation index (NDVI) (n=19) [37,38,39,40,41,42,43,44,45, 48,49,50,51,52,53,54,55,56,57], soil-adjusted vegetation index (SAVI) [37, 40, 57] or enhanced vegetation index (EVI) [38, 44, 51]. One study employed green land cover data (n=1) [47]. 17/19 studies measured NDVI greenness from satellite data including Landsat (n=9) [37, 38, 40,41,42, 49, 50, 56, 57], Moderate Resolution Imaging Spectroradiometer (MODIS) (n=6) [38, 43,44,45, 48, 51] and Advanced Spaceborne Thermal Emission and Reflection Radiometer (ASTER) (N=3) [39, 54, 55]. One study employed NDVI derived from 0.50-m spatial resolution colour infrared imagery [52]. Another study used data derived from the US Geological Survey [53]. Eight studies employed annual composite value to measure greenness exposure (n=8) [38, 39, 42,43,44, 49, 54, 55], while some selected greenness data from specific months of a year (n=8) [37, 40, 41, 45, 50,51,52, 57], or employed average greenness value over a 16-day period (n=1) [48]. The relevant information was not available in two studies [53, 56]. Of the studies employing data of multiple months selectively, seven selected only data from months during summer time [37, 40, 41, 50,51,52, 57], and one selected two images each from summer and winter time [45].
Walkability was the second most studied environment exposure (n=13). The majority of studies examining neighbourhood-level walkability developed overall metrics of walkability comprising multiple residential environmental attributes (n=7) [36, 49, 52, 60, 61, 65, 66] or employed a Walk score index (n=5) [43, 62,63,64, 71], and one study included both measures [59]. Walk score is a composite walkability index tagged to properties in USA and Canada and comprises distance to multiple amenities, block length and intersection density measured on either street network [43, 62, 71] or Euclidean buffers [63, 64] from each origin. Walk scores developed post-2010 such as Street Smart Walk score provides a more comprehensive measure of walkability using distance to amenities based on pedestrian-friendly street networks [72]. Other built-up environmental attributes included distance to greenspace/public parks (n=2) [37, 68], number of parks (n=1) [36], residential density (n=2) [68, 69], density of physical activity facilities (n=1) [68], impervious surface area (n=1) [45] and land cover type (n=1) [70].
Overall, the protective associations between exposure to greenness and hypertension or BP outcomes were evident in 12 out of 15 (80%) cross-sectional studies [36, 38,39,40,41, 43,44,45,46,47,48, 50], while three (20%) showed null results [37, 42, 49]. Eleven out of 12 (92%) cross-sectional studies reported that higher walkability was beneficially associated with hypertension or BP outcomes [36, 43, 49, 59,60,61,62,63,64, 66, 71], while one (8%) showed null results [65]. Evidence from two studies reported beneficial associations of walkability with arterial stiffness [52] and CHD [43]. Greenness exposure was also associated with lower arterial stiffness in two studies [52, 53], but another one showed insignificant results [51]. Greater greenness exposure was also found to protect against composite CVDs [55,56,57], CHD [43], transient ischemic attack [54], IHD and heart failure [55], but reported null results for MI and atrial fibrillation [55]. One study showed protective association of greenness with stroke [43], and two reported null results for stroke [56] and ischemic stroke [54]. Among the other attributes, longer street distance from home to the nearest community centre and proportion of impervious surface area were associated with higher blood pressure outcomes [45, 67]. Land-use mix (n=1) [68] within residential neighbourhood did not show statistically significant associations with hypertension. Longer distance to greenspace from home was found to be associated with a higher risk of hypertension [37], while one study reported null results [68]. One study found an inverse association between distance to urban centre and carotid-femoral pulse wave velocity [70]. One study showed beneficial association between residential density and hypertension [69], while one showed null results [68].
Evidence from Studies of Longitudinal Design
Tables 4, 5 and 6 show the summary characteristics of longitudinal studies. The 32 longitudinal studies were mostly conducted in Western settings, namely USA (n=10) [49, 58, 71, 75, 84,85,86, 95, 96, 99], Canada (n=5) [82, 83, 88, 91, 94] and Australia (n=2) [77, 93], and one each in Belgium [73], Italy [87], Lithuania [74], Luxembourg [78], Spain [81], Thailand [76], Switzerland [90] and UK [51]. Others were conducted in Asia, including mainland China (n=3) [79, 80, 97], and one each in Hong Kong [69], India [98], South Korea [89] and Israel [92]. Three studies [85, 96, 99] involved only female participants, while two [84, 93] included only male participants. With respect to the number of participants, five studies had a sample size of ≤1000 [71, 73,74,75, 78], three had between 1001 and 2500 [58, 77, 94], two between 2501 and 5000 [49, 51], five between 5001 and 10,000 [79, 80, 93, 95, 98], seven between 10,001 and 100,000 [69, 76, 86, 92, 96, 97, 99] and ten included participants more than 100,001 [81,82,83,84,85, 87,88,89,90,91].
The outcome variables included measured hypertension and/or BP outcomes (n=13) [49, 58, 69, 71, 73,74,75, 77,78,79,80, 95, 98], self-reported hypertension (n=1) [76], hypertension derived from hospital admission and annual health examination records (n=1) [94], arterial stiffness (n=1) [51] and cardiovascular events derived from medical records (n=15) [81,82,83,84,85,86,87,88,89,90,91,92,93, 96, 97] or self-reported questionnaire (n=1) [99]. Specific CVD events included composite CVD endpoints [mortality: [82,83,84, 86, 87, 93, 96]; both hospitalization and mortality: [89]], IHD mortality [87, 90, 91], cerebrovascular mortality [87], hypertension-related mortality [90], CHD [mortality: [85, 99]; both hospitalization and mortality: [89, 99]], CHD or MI [99], MI [hospitalization: [82, 92]; both hospitalization and mortality: [89]], heart failure [82], total stroke [hospitalization: [87, 88, 92]; mortality: [85, 90, 91, 93]; both hospitalization and mortality: [89]], hemorrhagic stroke [both hospitalization and mortality: [89]] and ischemic stroke and/or transient ischemic attack [hospitalization: [81]; both hospitalization and mortality: [89, 97]]. The follow-up periods of the studies were 2.2–40 years, 4 years and 2–27 years for hypertension, arterial stiffness and cardiovascular events respectively. The outcome variables were measured at multiple time points (i.e. at each follow-up) in 9 of 13 longitudinal studies for hypertension [49, 58, 69, 71, 77,78,79,80, 95], and one study for arterial stiffness [51]. Among the 13 studies that measured BP, cut-off points for SBP and DBP of either ≥140/90 mm Hg [49, 69, 74, 79, 80, 95] or ≥130/85 mm Hg [71, 77] were employed, one study measured ambulatory BP during day and night time [73], while four did not define any cut-off point [58, 75, 78, 98]. Of the 13 studies that measured SBP and/or DBP, BP measurements were repeated three times in seven studies [49, 58, 71, 75, 78, 80, 98] and two times in four studies [69, 74, 79, 95]. The outcomes were measured every 15 min or 30 min during day and night time, respectively in one study [73], and the remaining one did not specifically mention about repeated measurements [77]. Seven studies additionally employed antihypertensive medication as a criteria to define hypertension [49, 69, 71, 74, 77, 80, 95].
All the longitudinal studies employed GIS to define residential neighbourhoods. The majority of studies employed straight-line residential catchments (n=17), mostly of buffer radius 500 m (n=10) [51, 73, 79, 81, 83, 86, 90, 91, 93, 98], 1000 m (n=5) [51, 73, 86, 87, 96], 300 m (n=5) [73, 81, 87, 93, 98] and 250 m (n=5) [82, 83, 85, 86, 88]. Only four studies employed street network catchment of buffer sizes 500 m [78], 800 m [69, 78] and 1000 m [75, 77, 78]. Two studies assigned a 250-m2 pixel [84] and a 30-m pixel [92] containing participants’ home address. Four studies employed a publicly available Walk score index linked to individual health data via postal codes [94] or residential locations [71, 95, 97]. Four studies employed other predefined census geographies for exposure measurement including city [80], sub-district [76], administrative district [89] and metropolitan statistical area [99]. Two studies measured distance to multiple attributes of built environment [74, 75].
Among the urban environmental exposures, a majority of the studies examined the associations with residential greenness in terms of NDVI (n=17) [51, 76, 77, 79,80,81,82,83,84,85,86,87,88, 90,91,92,93], EVI [51, 76, 79] or SAVI [78]. Other measures of greenness and urban land use included greenspace coverage [73, 75, 89, 90], greenspace count [75], distance to greenspace [74, 75], density of wooded environment [78] and satellite-derived built-up land-use [98]. Several studies examined associations of CVD risks and events with neighbourhood-level walkability (n=8) [49, 58, 71, 77, 94,95,96,97]. Besides greenness and walkability, one study additionally measured food environment and count, median size and type of public open spaces [77]. Multi-scalar residential density expressed in terms of floor area of the apartment, building block density and neighbourhood level density was measured in one study [69]. One measured distance to major road [74] and another one measured compactness modelled from street connectivity, mixed land use, centredness and residential density [99]. All the 17 studies measuring greenness employed satellite data of moderate to low spatial resolution; 10 used Landsat data [77, 81, 82, 86,87,88, 90,91,92,93], six used MODIS [51, 76, 79, 83,84,85] and one derived data from other resource [80]. Eleven of the 17 studies employed annual composite value to measure greenness exposure [51, 76, 79, 80, 82,83,84,85,86, 88, 91], three selected greenness data from specific years over the follow-up period [90, 92, 93], and three measured the exposure at baseline only [77, 81, 87]. Of the 17 studies, eight selected only data from months in summer time [51, 77, 81, 83, 86, 87, 90, 93], one employed monthly data [80], two derived data of representative months to develop a seasonally-time varying measure for every year [84, 85], and six did not report information with regard to the months of the year in which greenness was assessed [76, 79, 82, 88, 91, 92]. For studies examining neighbourhood-level walkability, the majority developed overall metrics of walkability comprising multiple component built environment attributes measured from participants’ residential locations (n=4) [49, 58, 77, 96] or employed Walk score (n=4) [71, 94, 95, 97]. Three of the eight studies on walkability employed quasi-experimental design examining the effect of moving addresses from low to high walkable neighbourhoods on hypertension [94] and BP reductions [58, 71].
With respect to the temporal frequency of exposure assessment, nine of the 25 studies examining the associations with greenness and walkability measured the exposure at a single time point [49, 77, 81, 87, 90, 92, 95,96,97], and 16 at multiple time points [51, 58, 71, 76, 78,79,80, 82,83,84,85,86, 88, 91, 93, 94]. Of the nine studies employing a single-time exposure measurement, two restricted analyses to only non-movers [49, 96]. Of the 16 studies measuring the exposure at multiple time points, time-varying data were employed in 15 studies, and one employed data of a single time point [71]. The exposure value was assigned for each address at each follow-up in five studies [51, 82, 83, 85, 88], three included only participants who moved residence [58, 71, 94], three included non-movers only [76, 79, 93], and mobility data was not available in another five studies [78, 84, 86, 91, 92]. Among the studies examining other attributes of urban environment, three measured built environment (including distance to major road, food environment, public open space and compactness) at a single time point [74, 77, 99], another employed yearly Landsat data to measure land use change over time [98], while another examined associations of multi-level density on hypertension across two waves of the cohort [69].
Six out of eight longitudinal studies (75%) showed protective associations of higher exposure to greenness with hypertension or BP [73,74,75,76, 79, 80], while two showed null results [77, 78]. Of the eight longitudinal studies examining links between greenness and cardiovascular mortality, a protective association was observed in seven (88%) studies [82,83,84, 86, 87, 90, 100], while one reported null results [93]. Five studies reported association between greenness and total stroke hospitalization/mortality [87,88,89,90,91], while three reported null results [85, 92, 93]. The protective associations of greenness with MI [82, 89, 92], ischemic stroke and/or transient ischemic attack [81, 89, 97] and heart failure[82] were evident. The analysis by CVD mortality/event sub-types also showed protective associations of greenness with IHD mortality [87, 90, 91], cerebrovascular mortality [87] and hypertension-related mortality [90]. One study showed protective association between greenness and CHD [89], while one showed null findings [85]. Evidence from two studies also showed null associations of greenness with hemorrhagic stroke [89] and arterial stiffness [51].
Three out of six longitudinal studies (50%) reported that residing in neighbourhoods with higher walkability was beneficially associated with hypertension or BP outcomes [58, 94, 95]. Three of these studies employed a quasi-experimental design following-up participants who moved residential address by measuring walkability before and after the move and reported protective association of increase in walkability with BP outcomes [58, 94], but one study found counterintuitive results [71]. One study found null associations between walkability and cardiovascular mortality [96], whereas one reported reduced risk of ischemic stroke event with higher walkability [97]. Among the other attributes of urban environments, one study reported beneficial associations of compactness with CHD mortality and incident MI [99]. Another study showed non-beneficial association between built-up land use and BP outcomes [98], and one did not find statistically significant associations of food environment index with BP outcomes [77]. Another study reported protective association of residential floor area with hypertension and BP outcomes, while a positive association was reported with building block density. Participants moving to residences of lower floor area had a higher odds of hypertension in reference to those who did not move [69].
Study Quality
Risk of biases pertaining to selection, selective reporting and other bias were rated as “probably low” in the 63 studies reviewed (Table S1). Forty-two out of 63 studies (67%) were rated as “probably high” risk of confounding bias because of being unable to account for basic socioeconomic status (i.e. age, sex, education and income) and/or comorbidities/family history of diseases in their statistical models. All but one of the studies explicitly specified about reasonable inclusion and exclusion criteria for participant recruitment and/or analysis (62/63, 98%). The remaining one study excluded participants under the age of 40 which accounted for 34% of the original dataset [43] and was considered to have “probably high” risk of exclusion bias. Twenty-nine out of 63 studies (46%) were rated as “probably high” risk of detection bias for exposure due to using only baseline exposure in longitudinal studies [49, 77, 87, 95,96,97] and/or modelling exposures based on geocodes of pre-defined administrative units [38, 39, 41, 47, 50, 51, 54,55,56,57, 60,61,62,63, 68, 76, 79, 80, 82, 83, 89, 91, 99]. Nine studies (14%) were rated as “probably high” risk of bias for outcome ascertainment owing to using self-reported data to identify hypertensive patients [36, 37, 42, 65, 68, 76] and CVDs [56, 57, 99]. Detailed profiles of each study included in this review have been summarized in Tables S2–S64.
Discussion
The review summarizes current evidence examining associations of urban built environment with cardiovascular risks and events. Overall, we report moderately-consistent evidence of associations of greenness with cardiovascular risks and major CVD events (cross-sectional studies: 12 of 15 on hypertension/BP and 2 of 3 on arterial stiffness; and among longitudinal studies: 6 of 8 on hypertension/BP, 7 of 8 on CVD mortality, 3 of 3 on IHD mortality and 5 of 8 on stroke hospitalization or mortality reporting inverse associations). A moderately-consistent protective association was also reported for walkability (cross-sectional studies: 11 of 12 on hypertension/BP and 1 of 1 on arterial stiffness; and in longitudinal studies: 3 of 6 on hypertension/BP and 1 of 2 studies on CVD events being protective).
Study design is an important criterion for determining the validity of evidence. In this review, 49% of the studies examining the association between urban environment and cardiovascular health employed cross-sectional design. Cross-sectional study design is prone to bias associated with common method variance emerging from the use of data measured at a single time point [101], implying that causal inference cannot be established. Additionally, the majority of studies were conducted in Western settings (76%), while studies from Asia accounted for 24% of the remainder. Future studies should rely on longitudinal data, with the assessment of cardiovascular health and measurements of exposures conducted at multiple time points to enable a better understanding of the potential causal relationships between urban environment and CVD risks. Quasi-experimental study design or natural urban experiments, appropriately supported by advanced modelling techniques (such as propensity score matching and inverse probability weighting) may also be leveraged to support causal explanation of associations of built environment interventions on CVD risks; such as the health effects of moving from one neighbourhood setting to another. There is a need to collect evidence from varied geographical contexts for greater external validity.
Rigorous health outcome assessment is fundamental to conducting robust health-related research. Ten percent of the studies relied on questionnaire-based methods to identify participants’ hypertension status. This may be prone to a degree of subjectivity associated with recall bias, thereby affecting the validity of outcome assessment. Commonly used valid diagnostic methods in clinical settings include measuring health parameters (such as BP using electronic monitors) by trained interviewers or nurses or obtaining data directly from health registries and hospital records (enabling data analyses at multiple time points). The use of data from health insurance agencies has also become more common, as in one of the reviewed studies from Florida using annual claims of chronic conditions for all beneficiaries [39]. Besides the use of circulatory markers of CVD risks (like BP) which are transient in nature and thus prone to diurnal fluctuations based on surrounding environment and activities, more stable markers such as arterial stiffness and other CVD biomarkers may produce robust results.
Accurate definition of functional activity neighbourhood for measuring built environment is an important factor determining reliability of the reported results. In the present review, neighbourhoods were defined in terms of buffers of different shapes (primarily Euclidean buffers and some employing street network buffers) and of different spatial scales (buffer radii), which may have contributed to some of the inconsistencies in the reported findings. Street network buffers are a more accurate measure of participants’ activity neighbourhoods, based on actual potential routes available (along a street network) as compared to the traditional circular (Euclidean) buffers [29]. Also, 25 of the 63 studies were rated as “probably high” risk of bias due to modelling exposures based on geocodes of pre-defined administrative units. Census-defined neighbourhoods cannot accurately reflect individual activity neighbourhoods, because of the heterogeneity of built environment conditions within them (such as a census tract area). The value of accurate individualized activity neighbourhoods (street buffers around geocoded dwellings) should be emphasized. The spatial scale of these neighbourhoods (buffer radius) is a crucial criterion for accurate exposure assessment and must be based on prior proof of concept. While there is no standardized guideline for the use of buffer size, it is good practice to conduct sensitivity tests with buffers of different scales, to enhance rigor of evidence. Specifically, attention should be paid to issues such as (1) sufficiency of participants having access to specific destinations within the buffer and (2) sufficiency of these destinations within the sampled area [102]. Besides residential neighbourhoods, future studies should also examine the role of built environment attributes within work place- and school-neighbourhoods in influencing cardiovascular risks, given that a significant proportion of time are also spent within them over the life course.
Accurate assessment of built environment exposures over time in observational cohort studies has been a challenge in environmental epidemiology [33]. The application of multispectral satellite and geospatial data facilitates the creation of standardized metrics to objectively measure and quantify participants’ neighbourhood built environment, which can be scaled-up and replicated across cohorts at relatively low cost [103]. Due consideration should be made with respect to the spatial and temporal resolution of the data employed. Poor spatial resolution of satellite-derived built environment metrics may result in more study participants being linked to same exposure characteristics (homogeneity) and consequent exposure misclassification. Residential greenness measures derived from MODIS data produce NDVI grids of relatively low resolution ranging between 250 and 500 m, and at the same time, its high temporal resolution implies that it can support almost daily observations. Trade-offs between spatial and temporal resolution should be carefully guided by rate of change of landscape characteristics being measured. On the other hand, studies employing NDVI generated from Landsat and ASTER data have moderate spatial resolution of 30 and 15 m, respectively. Other criteria related to the quality of satellite data such as extent of cloud cover, atmospheric distortion, seasonality and degree of temporal match between outcome ascertainment and exposure assessment [104] are important determinants of accuracy of exposure measurement. In addition to reliance on objective measurements of built environments, future studies should also explore of individual perceptions in influencing usability of built environment and their associations with cardiovascular risks [105].
The temporal frequency of exposure assessment is an important consideration in longitudinal studies. One-third (8/24) of the longitudinal studies examining associations between greenness and walkability measured exposures at a single time point. Of the 16 studies that measured exposures at multiple time points, only five studies assigned the exposure value for each address at each follow-up (to account for changes in residential movement). Changes in urban environments over time as well as participants’ long-term trajectories of residential mobility should be considered in longitudinal studies to enhance the precision of investigating long-term effects of urban environments on disease development [29, 106].
The underlying mechanisms of the association between urban built environment and cardiovascular health have not yet been well-established and is an active area of research. One of the possible pathways from urban built environment exposures to cardiovascular health is likely to be mediated via chronic stress. It has been suggested that chronic stress may activate and ignite reactions in the primary biological system — the hypothalamic-pituitary-adrenocortical and sympatho-adrenomedullary axes, which in turn initiates physiological alterations including increased heart rate and energy consumption, thereby increasing risks of CVDs [107]. Exposure to residential greenness maybe associated with CVD risks via their effects on the oxidative stress [108, 109] and DNA methylation [110]. Greater accessibility to neighbourhood greenspace has also been known to positively influence intention to visit and facilitate mental recovery and restoration [111, 112]. The presence of public open space around home may also help create informal encounters as well as opportunities for social interactions, which may benefit psychological well-being and reduce risks of CVDs [113, 114]. The observed protective effects may also be explained via a physical activity-mediated mechanism; residing in neighbourhoods of higher green exposure and walkability has been evident to be associated with higher physical activity [100, 115, 116]. The protective effects of physical activity on cardio-respiratory fitness, hypertension and arterial wall damage are well-evidenced [117, 118]. Increased intensity and volume of physical activity have been found to be associated with consistent reduction in oxidative stress [119]. The oxidative process induced by reactive oxygen species is important in the pathogenesis of atherosclerosis, the primary disorder resulting in heart attack and ischemic stroke [120]. A well-designed neighbourhood with connected walkways and adequate provisions of services and destinations may incentivize active travel and play an antioxidant role in maintaining cardiovascular health [31]. Further studies are needed to elucidate the underlying mechanisms for the associations between urban built environment and cardiovascular health.
Conclusion
CVDs constitute the leading cause of mortality and morbidity globally and is characterized by complex multi-factorial aetiology. This systematic review found moderately consistent evidence of the protective associations of walkability and greenness with cardiovascular risks and major CVD events. Downstream built environment interventions in the form of well-designed multifunctional green and walkable neighbourhoods may be long lasting environmental exposures for good that affect significant proportion of the population. Planning and provisioning healthy urban spaces have the potential for direct long-term cumulative effects on population health, as well as indirect ones, in terms of configuring other exposures such as pollution, degree and quality of social connections. Though the effect sizes of the associations with built environment may be small, interventions in built environment have the potential to lower the population distribution of chronic diseases including CVD risks, thereby reducing the associated health burdens [121, 122].
References
Wang H, Naghavi M, Allen C, et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet. 2016;388(10053):1459–544. https://doi.org/10.1016/S0140-6736(16)31012-1.
World Health Organization. Cardiovascular diseases (CVDs) 2021.https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds). Accessed 18 Jan 2022.
Strazzullo P, D’Elia L, Kandala N-B, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ. 2009;339:b4567. https://doi.org/10.1136/bmj.b4567.
Martínez-González MA, Gea A, Ruiz-Canela M. The Mediterranean diet and cardiovascular health. Circ Res. 2019;124(5):779–98. https://doi.org/10.1161/CIRCRESAHA.118.313348.
Messner B, Bernhard D. Smoking and cardiovascular disease. Arterioscler Thromb Vasc Biol. 2014;34(3):509–15. https://doi.org/10.1161/ATVBAHA.113.300156.
Sesso HD. Alcohol and cardiovascular health. Am J Cardiovasc Drugs. 2001;1(3):167–72. https://doi.org/10.2165/00129784-200101030-00002.
Day INM, Wilson DI. Genetics and cardiovascular risk. BMJ. 2001;323(7326):1409. https://doi.org/10.1136/bmj.323.7326.1409.
Ahmed HM, Blaha MJ, Nasir K, Rivera JJ, Blumenthal RS. Effects of physical activity on cardiovascular disease. Am J Cardiol. 2012;109(2):288–95. https://doi.org/10.1016/j.amjcard.2011.08.042.
Narang I, Manlhiot C, Davies-Shaw J, et al. Sleep disturbance and cardiovascular risk in adolescents. Can Med Assoc J. 2012;184(17):E913. https://doi.org/10.1503/cmaj.111589.
Khaing W, Vallibhakara SA, Attia J, McEvoy M, Thakkinstian A. Effects of education and income on cardiovascular outcomes: a systematic review and meta-analysis. Eur J Prev Cardiol. 2017;24(10):1032–42. https://doi.org/10.1177/2047487317705916.
Stephens JW, Humphries SE. The molecular genetics of cardiovascular disease: clinical implications. J Intern Med. 2003;253(2):120–7. https://doi.org/10.1046/j.1365-2796.2003.01104.x.
Fuchs FD, Whelton PK. High blood pressure and cardiovascular disease. Hypertension. 2020;75(2):285–92. https://doi.org/10.1161/HYPERTENSIONAHA.119.14240.
Wu S, Xu Y, Zheng R, et al. Hypertension defined by 2017 ACC/AHA guideline, ideal cardiovascular health metrics, and risk of cardiovascular disease: a nationwide prospective cohort study. The Lancet Regional Health - Western Pacific. 2022;20:100350. https://doi.org/10.1016/j.lanwpc.2021.100350.
Mitchell GF, Hwang S-J, Vasan RS, et al. Arterial stiffness and cardiovascular events. Circulation. 2010;121(4):505–11. https://doi.org/10.1161/CIRCULATIONAHA.109.886655.
Said MA, Eppinga RN, Lipsic E, Verweij N, van der Harst P. Relationship of arterial stiffness index and pulse pressure with cardiovascular disease and mortality. J Am Heart Assoc. 2018;7(2):e007621. https://doi.org/10.1161/JAHA.117.007621.
World Health Organization. Fact sheets on sustainable development goals: health targets: Noncommunicable Diseases 2017. https://www.euro.who.int/__data/assets/pdf_file/0007/350278/Fact-sheet-SDG-NCD-FINAL-25-10-17.pdf. Accessed 18 Jan 2022.
Galea S, Freudenberg N, Vlahov D. Cities and population health. Soc Sci Med. 2005;60(5):1017–33. https://doi.org/10.1016/j.socscimed.2004.06.036.
Vlahov D, Freudenberg N, Proietti F, et al. Urban as a determinant of health. J Urban Health. 2007;84(1):16–26. https://doi.org/10.1007/s11524-007-9169-3.
Gampel B, Slome C, Scotch N, Abramson JH. Urbanization and hypertension among Zulu adults. J Chronic Dis. 1962;15(1):67–70. https://doi.org/10.1016/0021-9681(62)90102-9.
Cross SH, Mehra MR, Bhatt DL, et al. Rural-urban differences in cardiovascular mortality in the US, 1999-2017. JAMA. 2020;323(18):1852–4. https://doi.org/10.1001/jama.2020.2047.
Sallis JF, Floyd MF, Rodríguez DA, Saelens BE. Role of Built Environments in Physical Activity, Obesity, and Cardiovascular Disease. Circulation. 2012;125(5):729–37. https://doi.org/10.1161/CIRCULATIONAHA.110.969022.
Saelens BE, Sallis JF, Black JB, Chen D. Neighborhood-based differences in physical activity: an environment scale evaluation. Am J Public Health. 2003;93(9):1552–8. https://doi.org/10.2105/AJPH.93.9.1552.
Sarkar C, Webster C, Gallacher J. Association between adiposity outcomes and residential density: a full-data, cross-sectional analysis of 419 562 UK Biobank adult participants. Lancet Planet Health. 2017;1(7):e277–88. https://doi.org/10.1016/S2542-5196(17)30119-5.
Sarkar C, Webster C, Gallacher J. Residential greenness and prevalence of major depressive disorders: a cross-sectional, observational, associational study of 94 879 adult UK Biobank participants. Lancet Planet Health. 2018;2(4):e162–73. https://doi.org/10.1016/S2542-5196(18)30051-2.
Lachowycz K, Jones AP. Greenspace and obesity: a systematic review of the evidence. Obes Rev. 2011;12(5):e183–9. https://doi.org/10.1111/j.1467-789X.2010.00827.x.
Barboza EP, Cirach M, Khomenko S, et al. Green space and mortality in European cities: a health impact assessment study. Lancet Planet Health. 2021;5(10):e718–30. https://doi.org/10.1016/S2542-5196(21)00229-1.
Ewing R, Cervero R. “Does compact development make people drive less?” The answer is yes. J Am Plann Assoc. 2017;83(1):19–25. https://doi.org/10.1080/01944363.2016.1245112.
Laverty AA, Mindell JS, Webb EA, Millett C. Active travel to work and cardiovascular risk factors in the United Kingdom. Am J Prev Med. 2013;45(3):282–8. https://doi.org/10.1016/j.amepre.2013.04.012.
Chandrabose M, Rachele JN, Gunn L, et al. Built environment and cardio-metabolic health: systematic review and meta-analysis of longitudinal studies. Obes Rev. 2019;20(1):41–54. https://doi.org/10.1111/obr.12759.
Malambo P, Kengne AP, De Villiers A, Lambert EV, Puoane T. Built environment, selected risk factors and major cardiovascular disease outcomes: a systematic review. PloS One. 2016;11(11):e0166846. https://doi.org/10.1371/journal.pone.0166846.
Nieuwenhuijsen MJ. Influence of urban and transport planning and the city environment on cardiovascular disease. Nat Rev Cardiol. 2018;15(7):432–8. https://doi.org/10.1038/s41569-018-0003-2.
Thornton LE, Pearce JR, Kavanagh AM. Using geographic information systems (GIS) to assess the role of the built environment in influencing obesity: a glossary. Int J Behav Nutr Phys Act. 2011;8(1):71. https://doi.org/10.1186/1479-5868-8-71.
Jia P, Lakerveld J, Wu J, et al. Top 10 research priorities in spatial lifecourse epidemiology. Environ Health Perspect. 2019;127(7):074501. https://doi.org/10.1289/EHP4868.
Rooney AA, Boyles AL, Wolfe MS, Bucher JR, Thayer KA. Systematic review and evidence integration for literature-based environmental health science assessments. Environ Health Perspect. 2014;122(7):711–8. https://doi.org/10.1289/ehp.1307972.
Johnson PI, Koustas E, Vesterinen HM, et al. Application of the navigation guide systematic review methodology to the evidence for developmental and reproductive toxicity of triclosan. Environ Int. 2016;92-93:716–28. https://doi.org/10.1016/j.envint.2016.03.009.
Adhikari B, Delgado-Ron JA, Van den Bosch M, et al. Community design and hypertension: walkability and park access relationships with cardiovascular health. Int J Hyg Environ Health. 2021;237:113820. https://doi.org/10.1016/j.ijheh.2021.113820.
Bauwelinck M, Zijlema WL, Bartoll X, et al. Residential urban greenspace and hypertension: A comparative study in two European cities. Environ Res. 2020;191:110032. https://doi.org/10.1016/j.envres.2020.110032.
Boakye KA, Iyanda AE, Oppong JR. Urban greenness and hypertension among Ghanaian adults. African Geographical Review. 2023;42(1):72–84. https://doi.org/10.1080/19376812.2021.1970596.
Brown SC, Lombard J, Wang K, et al. Neighborhood greenness and chronic health conditions in Medicare beneficiaries. Am J Prev Med. 2016;51(1):78–89. https://doi.org/10.1016/j.amepre.2016.02.008.
Dzhambov AM, Markevych I, Lercher P. Greenspace seems protective of both high and low blood pressure among residents of an Alpine valley. Environ Int. 2018;121:443–52. https://doi.org/10.1016/j.envint.2018.09.044.
Huang B, Xiao T, Grekousis G, et al. Greenness-air pollution-physical activity-hypertension association among middle-aged and older adults: evidence from urban and rural China. Environ Res. 2021;195:110836. https://doi.org/10.1016/j.envres.2021.110836.
Jendrossek M, Standl M, Koletzko S, et al. Residential air pollution, road traffic, greenness and maternal hypertension: results from GINIplus and LISAplus. J Occup Environ Med. 2017;8(3):131–42. https://doi.org/10.15171/ijoem.2017.1073.
Jia X, Yu Y, Xia W, et al. Cardiovascular diseases in middle aged and older adults in China: the joint effects and mediation of different types of physical exercise and neighborhood greenness and walkability. Environ Res. 2018;167:175–83. https://doi.org/10.1016/j.envres.2018.07.003.
Jiang J, Chen G, Li B, et al. Associations of residential greenness with hypertension and blood pressure in a Chinese rural population: a cross-sectional study. Environ Sci Pollut Res. 2021;28(37):51693–701. https://doi.org/10.1007/s11356-021-14201-0.
Lane KJ, Stokes EC, Seto KC, Thanikachalam S, Thanikachalam M, Bell ML. Associations between greenness, impervious surface area, and nighttime lights on biomarkers of vascular aging in Chennai, India. Environ Health Perspect. 2017;125(8):087003. https://doi.org/10.1289/ehp541.
Nguyen TT, Nguyen QC, Rubinsky AD, et al. Google street view-derived neighborhood characteristics in california associated with coronary heart disease, hypertension, diabetes. Int J Environ Res Public Health. 2021;18(19) https://doi.org/10.3390/ijerph181910428.
Plans E, Gullon P, Cebrecos A, et al. Density of green spaces and cardiovascular risk factors in the city of Madrid: the Heart Healthy Hoods study. Int J Environ Res Public Health. 2019;16(24) https://doi.org/10.3390/ijerph16244918.
Poulsen MN, Schwartz BS, Nordberg C, et al. Association of greenness with blood pressure among individuals with type 2 diabetes across rural to urban community types in Pennsylvania, USA. Int J Environ Res Public Health. 2021;18(2) https://doi.org/10.3390/ijerph18020614.
Savin KL, Roesch SC, Oren E, et al. Social and built neighborhood environments and blood pressure 6 years later: results from the Hispanic Community Health Study/Study of Latinos and the SOL CASAS ancillary study. Soc Sci Med. 2021:114496. https://doi.org/10.1016/j.socscimed.2021.114496.
Yang B-Y, Markevych I, Bloom MS, et al. Community greenness, blood pressure, and hypertension in urban dwellers: the 33 communities Chinese health study. Environ Int. 2019;126:727–34. https://doi.org/10.1016/j.envint.2019.02.068.
de Keijzer C, Foraster M, Busagana X, et al. Long-term greenspace exposure and progression of arterial stiffness: the Whitehall II cohort study. Environ Health Perspect. 2020;128(6):67014. https://doi.org/10.1289/EHP6159.
Lai KY, Kumari S, Gallacher J, Webster C, Sarkar C. Associations of residential walkability and greenness with arterial stiffness in the UK Biobank. Environ Int. 2022;158:106960. https://doi.org/10.1016/j.envint.2021.106960.
Riggs DW, Yeager R, Conklin DJ, et al. Residential proximity to greenness mitigates the hemodynamic effects of ambient air pollution. Am J Physiol Heart Circ Physiol. 2021;320(3):H1102–11. https://doi.org/10.1152/ajpheart.00689.2020.
Brown SC, Aitken WW, Lombard J, et al. Precision greenness and stroke/transient ischemic attack in 249,405 US Medicare beneficiaries. J Stroke. 2023;25(1):173–6. https://doi.org/10.5853/jos.2022.02922.
Wang K, Lombard J, Rundek T, et al. Relationship of neighborhood greenness to heart disease in 249 405 US Medicare beneficiaries. J Am Heart Assoc. 2019;8(6):e010258. https://doi.org/10.1161/jaha.118.010258.
Wang R, Dong P, Dong G, et al. Exploring the impacts of street-level greenspace on stroke and cardiovascular diseases in Chinese adults. Ecotoxicol Environ Saf. 2022;243:113974. https://doi.org/10.1016/j.ecoenv.2022.113974.
Yang BY, Hu LW, Jalaludin B, et al. Association between residential greenness, cardiometabolic disorders, and cardiovascular disease among adults in China. JAMA Netw Open. 2020;3(9):e2017507. https://doi.org/10.1001/jamanetworkopen.2020.17507.
Braun LM, Rodriguez DA, Song Y, et al. Changes in walking, body mass index, and cardiometabolic risk factors following residential relocation: longitudinal results from the CARDIA study. J Transp Health. 2016;3(4):426–39. https://doi.org/10.1016/j.jth.2016.08.006.
de Courrèges A, Occelli F, Muntaner M, Amouyel P, Meirhaeghe A, Dauchet L. The relationship between neighbourhood walkability and cardiovascular risk factors in northern France. Sci Total Environ. 2021;772:144877. https://doi.org/10.1016/j.scitotenv.2020.144877.
Howell NA, Tu JV, Moineddin R, Chu A, Booth GL. Association between neighborhood walkability and predicted 10-year cardiovascular disease risk: the CANHEART (Cardiovascular Health in Ambulatory Care Research Team) cohort. J Am Heart Assoc. 2019;8(21):e013146. https://doi.org/10.1161/JAHA.119.013146.
Howell NA, Tu JV, Moineddin R, et al. Interaction between neighborhood walkability and traffic-related air pollution on hypertension and diabetes: the CANHEART cohort. Environ Int. 2019;132:104799. https://doi.org/10.1016/j.envint.2019.04.070.
Loo CKJ, Greiver M, Aliarzadeh B, Lewis D. Association between neighbourhood walkability and metabolic risk factors influenced by physical activity: a cross-sectional study of adults in Toronto, Canada. BMJ Open. 2017;7(4):e013889. https://doi.org/10.1136/bmjopen-2016-013889.
Makram OM, Nwana N, Nicolas JC, et al. Favorable Neighborhood walkability is associated with lower burden of cardiovascular risk factors among patients within an integrated health system: the Houston Methodist Learning Health System Outpatient Registry. Curr Probl Cardiol. 2023;48(6):101642. https://doi.org/10.1016/j.cpcardiol.2023.101642.
Méline J, Chaix B, Pannier B, et al. Neighborhood walk score and selected cardiometabolic factors in the French RECORD cohort study. BMC Public Health. 2017;17(1):960. https://doi.org/10.1186/s12889-017-4962-8.
Müller-Riemenschneider F, Pereira G, Villanueva K, et al. Neighborhood walkability and cardiometabolic risk factors in australian adults: an observational study. BMC Public Health. 2013;13(1):755. https://doi.org/10.1186/1471-2458-13-755.
Sarkar C, Webster C, Gallacher J. Neighbourhood walkability and incidence of hypertension: findings from the study of 429,334 UK Biobank participants. Int J Hyg Environ Health. 2018;221(3):458–68. https://doi.org/10.1016/j.ijheh.2018.01.009.
Malambo P, De Villiers A, Lambert EV, Puoane T, Kengne AP. The relationship between objectively-measured attributes of the built environment and selected cardiovascular risk factors in a South African urban setting. BMC Public Health. 2018;18(1):847. https://doi.org/10.1186/s12889-018-5772-3.
Lee EY, Choi J, Lee S, Choi BY. Objectively measured built environments and cardiovascular diseases in middle-aged and older Korean adults. Int J Environ Res Public Health. 2021;18(4) https://doi.org/10.3390/ijerph18041861.
Sarkar C, Lai KY, Ni MY, Kumari S, Leung GM, Webster C. Liveable residential space, residential density, and hypertension in Hong Kong: a population-based cohort study. PLoS Med. 2021;18(11):e1003824. https://doi.org/10.1371/journal.pmed.1003824.
Corlin L, Lane KJ, Sunderarajan J, et al. Urbanization as a risk factor for aortic stiffness in a cohort in India. PLoS One. 2018;13(8):e0201036. https://doi.org/10.1371/journal.pone.0201036.
Braun LM, Rodríguez DA, Evenson KR, Hirsch JA, Moore KA, Diez Roux AV. Walkability and cardiometabolic risk factors: cross-sectional and longitudinal associations from the Multi-Ethnic Study of Atherosclerosis. Health Place. 2016;39:9–17. https://doi.org/10.1016/j.healthplace.2016.02.006.
Hall CM, Ram Y. Walk score® and its potential contribution to the study of active transport and walkability: a critical and systematic review. Transp Res D: Transp Environ. 2018;61:310–24. https://doi.org/10.1016/j.trd.2017.12.018.
Bijnens EM, Nawrot TS, Loos RJ, et al. Blood pressure in young adulthood and residential greenness in the early-life environment of twins. Environ Health. 2017;16(1):53. https://doi.org/10.1186/s12940-017-0266-9.
Braziene A, Vencloviene J, Tamosiunas A, Dedele A, Luksiene D, Radisauskas R. The influence of proximity to city parks and major roads on the development of arterial hypertension. Scand J Public Health. 2018;46(6):667–74. https://doi.org/10.1177/1403494817751756.
Jimenez MP, Wellenius GA, James P, et al. Associations of types of green space across the life-course with blood pressure and body mass index. Environ Res. 2020:185. https://doi.org/10.1016/j.envres.2020.109411.
Paoin K, Pharino C, Phosri A, et al. Association between greenness and cardiovascular risk factors: results from a large cohort study in Thailand. Environ Res. 2023:220. https://doi.org/10.1016/j.envres.2023.115215.
Paquet C, Coffee NT, Haren MT, et al. Food environment, walkability, and public open spaces are associated with incident development of cardio-metabolic risk factors in a biomedical cohort. Health Place. 2014;28:173–6. https://doi.org/10.1016/j.healthplace.2014.05.001.
Tharrey M, Klein O, Bohn T, Malisoux L, Perchoux C. Nine-year exposure to residential greenness and the risk of metabolic syndrome among Luxembourgish adults: a longitudinal analysis of the ORISCAV-Lux cohort study. Health Place. 2023;81:103020. https://doi.org/10.1016/j.healthplace.2023.103020.
Wensu Z, Wenjuan W, Fenfen Z, Wen C, Li L. The effects of greenness exposure on hypertension incidence among Chinese oldest-old: a prospective cohort study. Environ Health. 2022;21(1):66. https://doi.org/10.1186/s12940-022-00876-6.
Yang T, Wang J, Xu Z, et al. Associations between greenness and blood pressure and hypertension in Chinese middle-aged and elderly population: a longitudinal study. Environ Res. 2022;212(Pt D):113558. https://doi.org/10.1016/j.envres.2022.113558.
Avellaneda-Gomez C, Vivanco-Hidalgo RM, Olmos S, et al. Air pollution and surrounding greenness in relation to ischemic stroke: a population-based cohort study. Environ Int. 2022:161. https://doi.org/10.1016/j.envint.2022.107147.
Chen H, Burnett Richard T, Bai L, et al. Residential greenness and cardiovascular disease incidence, readmission, and mortality. Environ Health Perspect. 2020;128(8):087005. https://doi.org/10.1289/EHP6161.
Crouse DL, Pinault L, Balram A, et al. Urban greenness and mortality in Canada’s largest cities: a national cohort study. Lancet Planet Health. 2017;1(7):e289–97. https://doi.org/10.1016/S2542-5196(17)30118-3.
Iyer HS, Valeri L, James P, et al. The contribution of residential greenness to mortality among men with prostate cancer: a registry-based cohort study of Black and White men. Environ Epidemiol. 2020;4(2):e087. https://doi.org/10.1097/EE9.0000000000000087.
James P, Hart Jaime E, Banay Rachel F, Laden F. Exposure to greenness and mortality in a nationwide prospective cohort study of women. Environ Health Perspect. 2016;124(9):1344–52. https://doi.org/10.1289/ehp.1510363.
Liao NS, Van den Eeden SK, Sidney S, et al. Joint associations between neighborhood walkability, greenness, and particulate air pollution on cardiovascular mortality among adults with a history of stroke or acute myocardial infarction. Environ Epidemiol. 2022;6(2) https://doi.org/10.1097/EE9.0000000000000200.
Orioli R, Antonucci C, Scortichini M, et al. Exposure to residential greenness as a predictor of cause-specific mortality and stroke incidence in the Rome longitudinal study. Environ Health Perspect. 2019;127(2):027002. https://doi.org/10.1289/EHP2854.
Paul LA, Hystad P, Burnett RT, et al. Urban green space and the risks of dementia and stroke. Environ Res. 2020;186:109520. https://doi.org/10.1016/j.envres.2020.109520.
Seo S, Choi S, Kim K, Kim SM, Park SM. Association between urban green space and the risk of cardiovascular disease: a longitudinal study in seven Korean metropolitan areas. Environ Int. 2019;125:51–7. https://doi.org/10.1016/j.envint.2019.01.038.
Vienneau D, de Hoogh K, Faeh D, et al. More than clean air and tranquillity: residential green is independently associated with decreasing mortality. Environ Int. 2017;108:176–84. https://doi.org/10.1016/j.envint.2017.08.012.
Villeneuve PJ, Jerrett MG, Su J, et al. A cohort study relating urban green space with mortality in Ontario, Canada. Environ Res. 2012;115:51–8. https://doi.org/10.1016/j.envres.2012.03.003.
Yitshak-Sade M, Kloog I, Novack V. Do air pollution and neighborhood greenness exposures improve the predicted cardiovascular risk? Environ Int. 2017;107:147–53. https://doi.org/10.1016/j.envint.2017.07.011.
Zijlema WL, Stasinska A, Blake D, et al. The longitudinal association between natural outdoor environments and mortality in 9218 older men from Perth. Western Australia Environ Int. 2019;125:430–6. https://doi.org/10.1016/j.envint.2019.01.075.
Chiu M, Rezai M-R, Maclagan Laura C, et al. Moving to a highly walkable neighborhood and incidence of hypertension: a propensity-score matched cohort study. Environ Health Perspect. 2016;124(6):754–60. https://doi.org/10.1289/ehp.1510425.
Jones AC, Chaudhary NS, Patki A, et al. Neighborhood walkability as a predictor of incident hypertension in a national cohort study. Frontiers. Public Health. 2021;9(2) https://doi.org/10.3389/fpubh.2021.611895.
India-Aldana S, Rundle AG, Zeleniuch-Jacquotte A, et al. Neighborhood walkability and mortality in a prospective cohort of women. Epidemiology. 2021;32(6):763–72. https://doi.org/10.1097/EDE.0000000000001406.
Yang Z, Wu M, Lu J, et al. Interaction between walkability and fine particulate matter on risk of ischemic stroke: a prospective cohort study in China. Environ Pollut. 2022;292(Pt B):118482. https://doi.org/10.1016/j.envpol.2021.118482.
Milà C, Ranzani O, Sanchez M, et al. Land-use change and cardiometabolic risk factors in an urbanizing area of South India: a population-based cohort study. Environ Health Perspect. 2020;128(4):047003. https://doi.org/10.1289/EHP5445.
Griffin BA, Eibner C, Bird CE, et al. The relationship between urban sprawl and coronary heart disease in women. Health Place. 2013;20:51–61. https://doi.org/10.1016/j.healthplace.2012.11.003.
Villeneuve PJ, Jerrett M, Su JG, Weichenthal S, Sandler DP. Association of residential greenness with obesity and physical activity in a US cohort of women. Environ Res. 2018;160:372–84. https://doi.org/10.1016/j.envres.2017.10.005.
Rindfleisch A, Malter AJ, Ganesan S, Moorman C. Cross-sectional versus longitudinal survey research: concepts, findings, and guidelines. J Market Res. 2008;45(3):261–79. https://doi.org/10.1509/jmkr.45.3.261.
McCormack GR, Giles-Corti B, Bulsara M. The relationship between destination proximity, destination mix and physical activity behaviors. Prev Med. 2008;46(1):33–40. https://doi.org/10.1016/j.ypmed.2007.01.013.
Sarkar C, Webster C. Healthy cities of tomorrow: the case for large scale built environment–health studies. J Urban Health. 2017;94(1):4–19. https://doi.org/10.1007/s11524-016-0122-1.
Buzzelli M, Su J. Multi-level modelling in health research: a caution and rejoinder on temporally mismatched data. Soc Sci Med. 2006;62(5):1215–8. https://doi.org/10.1016/j.socscimed.2005.06.056.
Ma L, Dill J. Associations between the objective and perceived built environment and bicycling for transportation. J Transp Health. 2015;2(2):248–55. https://doi.org/10.1016/j.jth.2015.03.002.
Clarke P, Ailshire JA, Lantz P. Urban built environments and trajectories of mobility disability: findings from a national sample of community-dwelling American adults (1986–2001). Soc Sci Med. 2009;69(6):964–70. https://doi.org/10.1016/j.socscimed.2009.06.041.
Steptoe A, Kivimäki M. Stress and cardiovascular disease. Nat Rev Cardiol. 2012;9(6):360–70. https://doi.org/10.1038/nrcardio.2012.45.
Yeager R, Riggs DW, DeJarnett N, et al. Association between residential greenness and cardiovascular disease risk. J Am Heart Assoc. 2018;7(24):e009117. https://doi.org/10.1161/JAHA.118.009117.
Squillacioti G, Carsin A-E, Bellisario V, Bono R, Garcia-Aymerich J. Multisite greenness exposure and oxidative stress in children. The potential mediating role of physical activity. Environ Res. 2022;209:112857. https://doi.org/10.1016/j.envres.2022.112857.
Jeong A, Eze IC, Vienneau D, et al. Residential greenness-related DNA methylation changes. Environ Int. 2022;158:106945. https://doi.org/10.1016/j.envint.2021.106945.
Barton J, Rogerson M. The importance of greenspace for mental health. BJPsych International. 2017;14(4):79–81. https://doi.org/10.1192/S2056474000002051.
Houlden V, Weich S, Porto de Albuquerque J, Jarvis S, Rees K. The relationship between greenspace and the mental wellbeing of adults: a systematic review. PloS One. 2018;13(9):e0203000. https://doi.org/10.1371/journal.pone.0203000.
Tamosiunas A, Grazuleviciene R, Luksiene D, et al. Accessibility and use of urban green spaces, and cardiovascular health: findings from a Kaunas cohort study. Environ Health. 2014;13(1):20. https://doi.org/10.1186/1476-069X-13-20.
Dalton AM, Jones AP. Residential neighbourhood greenspace is associated with reduced risk of cardiovascular disease: a prospective cohort study. PloS One. 2020;15(1):e0226524. https://doi.org/10.1371/journal.pone.0226524.
McMorris O, Villeneuve PJ, Su J, Jerrett M. Urban greenness and physical activity in a national survey of Canadians. Environ Res. 2015;137:94–100. https://doi.org/10.1016/j.envres.2014.11.010.
Rundle AG, Sheehan DM, Quinn JW, et al. Using GPS Data to study neighborhood walkability and physical activity. Am J Prev Med. 2016;50(3):e65–72. https://doi.org/10.1016/j.amepre.2015.07.033.
Boreham CA, Ferreira I, Twisk JW, Gallagher AM, Savage MJ, Murray LJ. Cardiorespiratory fitness, physical activity, and arterial stiffness. Hypertension. 2004;44(5):721–6. https://doi.org/10.1161/01.HYP.0000144293.40699.9a.
Hegde SM, Solomon SD. Influence of physical activity on hypertension and cardiac structure and function. Curr Hypertens Rep. 2015;17(10):77. https://doi.org/10.1007/s11906-015-0588-3.
Ignarro LJ, Balestrieri ML, Napoli C. Nutrition, physical activity, and cardiovascular disease: An update. Cardiovasc Res. 2007;73(2):326–40. https://doi.org/10.1016/j.cardiores.2006.06.030.
Dhalla NS, Temsah RM, Netticadan T. Role of oxidative stress in cardiovascular diseases. J Hypertens. 2000;18(6): 655–73. Available at: https://journals.lww.com/jhypertension/Abstract/2000/18060/Role_of_oxidative_stress_in_cardiovascular.2.aspx
Rose GA, Khaw K-T, Marmot MG. Rose’s strategy of preventive medicine: the complete original text. New ed. Oxford: Oxford University Press; 2008.
Rose G. Mental disorder and the strategies of prevention. Psychol Med. 1993;23(3):553–5. https://doi.org/10.1017/S0033291700025320.
Acknowledgments
C.S. acknowledges support from Hong Kong Research Grant Commission and General Research Fund (GRF grant 17613220) and the US National Academy of Medicine — The University of Hong Kong Fellowship in Global Health Leadership.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 506 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lai, K.Y., Webster, C., Gallacher, J.E. et al. Associations of Urban Built Environment with Cardiovascular Risks and Mortality: a Systematic Review. J Urban Health 100, 745–787 (2023). https://doi.org/10.1007/s11524-023-00764-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11524-023-00764-5