Abstract
Background
Marine actinomycetes are efficient producers of new secondary metabolites that show different biological activities, including antibacterial, antifungal, anticancer, insecticidal, and enzyme inhibition activities.
Methods
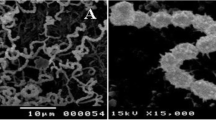
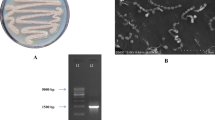
The morphological, physiological, and biochemical properties of the strain Streptomyces sp. VITPSA were confirmed by conventional methods. Antibacterial, anti-oxidant, anti-inflammatory, anti-diabetic, and cytotoxic activities of Streptomyces sp. VITPSA extract were determined. The media were optimized for the production of secondary metabolites. Characterization and identification of secondary metabolites were conducted by high-performance liquid chromatography, gas chromatography-mass spectroscopy, and Fourier transform infrared spectroscopy analysis.
Results
The strain showed significant antibacterial, anti-oxidant, and cytotoxic activities, moderate anti-inflammatory activity, and no satisfactory anti-diabetic activity. The ethyl acetate extract of Streptomyces sp. VITPSA showed maximum antibacterial activity against two gram-positive and gram-negative bacteria at 0.5 mg/mL. The antioxidant potential of the crude extract exhibited strong reducing power activity at 0.5 mg/mL with 95.1% inhibition. The cytotoxic effect was found to be an IC50 of 50 μg/mL on MCF-7 cell lines. Experimental design of optimization by one-factor analysis revealed the most favorable sucrose, yeast extract, pH (7.25), and temperature (28°C) conditions for the effective production of secondary metabolites.
Conclusion
This study revealed that Streptomyces sp. VITPSA is an excellent source of secondary metabolites with various bioactivities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arijit D, Sourav B, Abuelgasim Y, Hassan M, Subbaramiah S (2014). In vitro antimicrobial activity and characterization of mangrove isolates of Streptomycetes effective against bacteria and fungi of nosocomial origin. Braz Arch Biol Technol, 57(3): 349–356
Cheminformatics M, Nova ulica, SK-900 26 Slovensky Grob, Slovak Republic. [Online] Available from: http://www.molinspiration.com
Dash S, Jin C, Lee O O, Xu Y, Qian P Y (2009). Antibacterial and antilarval-settlement potential and metabolite profiles of novel sponge-associated marine bacteria. J Ind Microbiol Biotechnol, 36(8): 1047–1056
Deepika T L, Kannabiran K (2009). A report on antidermatophytic activity of actinomycetes isolated from Ennore coast of Chennai, Tamil Nadu, India. Int J Integrat Biol, 6(3): 132–136
Deepika T L, Kannabiran K (2009). A morphological, biochemical and biological studies of halophilic Streptomyces sp. isolated from saltpan environment. Am J Infect Dis, 5(3): 207–213
Hotam S, Jayprakash C, Anju Y, Shrivastava R, Smriti S, Anil K S, Natrajan G (2013). J Adv Pharm Technol Res, 4(2): 118–123
Kekuda P T R, Shobha K S, Onkarappa R (2010). Studies on antioxidant and anthelmintic activity of two Streptomyces species isolated from Western Ghat soil of Agumbe, Karnataka. J Pharm Res, 3: 26–29
Lynch D L, Worthy T E, Kresheck G C (1968). Chromatographic separation of the pigment fractions from a Serratia marcescens strain. Appl Microbiol, 16(1): 13–20
Man-Chul K, Jeongmin L, Dong-Hwi K, Hong-Joo S, Moon-Soo H (2014). Isolation and identification of antioxidant producing marinesource actinomycetes and optimal medium conditions. Food Sci Biotechnol. 23(5): 1629–1635
Melo I S, Santos S N, Rosa L H, Parma M M, Silva L J, Queiroz S C, Pellizari V H (2014). Isolation and biological activities of an endophytic Mortierella alpina strain from the Antarctic moss Schistidium antarctici. Extremophiles, 18(1): 15–23
Mosmann T (1983). Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods, 65(1-2): 55–63
Prakash G, Yoganandam I, Ilango K, Sucharita D (2003). Evaluation of Anti-inflammatory and Membrane Stabilizing Properties of various extracts of Punica granatum L.(Lythraceae). Int J Pharm Tech Res, 2(2): 260–1263
Qasim S Z (1999). The Indian Ocean: images and realities. New Delhi: Oxford and IBHp. 57–90
Solanki R, Khanna M, Lal R (2008). Bioactive compounds from marine actinomycetes. Indian J Microbiol, 48(4): 410–431
Suthindhiran K, Kannabiran K (2009). Cytotoxic and antimicrobial potential of Actinomycete species Saccharopolyspora salina VITSDK4 isolated from Bay of Bengal coast of India. Am J Infect Dis, 5(2): 90–98
Thirumalairaj J, Shanmugasundaram T, Sivasankari K, Natarajaseenivasan K, Balagurunathan R (2015). Isolation, screening and characterization of Potent marine Streptomyces sp. Pm105 against antibiotic resistant pathogens. Asian J Pharm Clin Res., 8(2): 439–443
Wang Y, Zhang Z S, Ruar T S, Wang Y M, Ali SM (1999). Investigation of actinomycetes diversity in the tropical rain forests of Singapore. J Ind Microbiol Biotechnol, 23(3): 178–187
Weissmann G (2006). Homeopathy: Holmes, Hogwarts, and the Prince of Wales. FASEB J, 20(11): 1755–1758
Williams S T, Goodfellow M, Alderson G, Wellington E M H, Sneath P H A, Sackin M J (1983). Numerical classification of Streptomyces and related genera. J Gen Microbiol, 129(6): 1743–1813
Zothanpuia A K P, Chandra P, Leo V V, Mishra V K, Kumar B, Singh B P (2017). Production of potent antimicrobial compounds from Streptomyces cyaneofuscatus associated with Fresh Water Sediment. Front Microbiol, https://doi.org/10.3389/fmicb.2017.00068
Acknowledgments
The authors acknowledge the management of VIT University for providing all necessary facilities to carry out the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pooja, S., Aditi, T., Naine, S.J. et al. Bioactive compounds from marine Streptomyces sp. VITPSA as therapeutics. Front. Biol. 12, 280–289 (2017). https://doi.org/10.1007/s11515-017-1459-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-017-1459-x