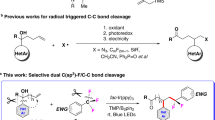
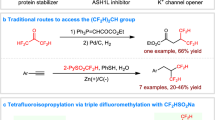
Abstract
Methylenecyclopropanes are among the most robust building blocks in synthetic chemistry, but the study on (difluoromethylene)-cyclopropanes is rather limited, because of the difficulty in the synthesis of these compounds. Herein, we report the invention of a novel carbene precursor, (1-diazo-2,2,2-trifluoroethyl)dimethyl(phenyl)silane (1a) and its application in the synthesis of (difluoromethylene)cyclopropanes. The reaction proceeds through photocatalyzed [2+1] cyclization of readily available alkenes and diazo compound 1a followed by the work-up of the reaction through the elimination of silyl fluoride. Both aromatic and aliphatic alkenes are tolerated by the mild reaction conditions, affording various (difluoromethylene)cyclopropanes in 44%–82% yield (>30 examples). Gram scale reaction and diversified downstream transformations highlight the synthetic potential of this methodology. The experimental and DFT calculations suggest the involvement of triplet carbene intermediate.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
For selected reviews, see: Wei HZ, Shi M, Wei Y. Chem Commun, 2023, 59: 2726–2738
Li D, Zang W, Bird MJ, Hyland CJT, Shi M. Chem Rev, 2021, 121: 8685–8755
Molchanov AP, Efremova MM, Kuznetsov MA. Russ Chem Bull, 2022, 71: 620–650
Cao H, Chen F, Su C, Yu L. Adv Synth Catal, 2020, 362: 438–461
Yu L, Shi M. Chem Eur J, 2019, 25: 7591–7606
Fang W, Shi M. Chem Eur J, 2018, 24: 9998–10005
Yu LZ, Chen K, Zhu ZZ, Shi M. Chem Commun, 2017, 53: 5935–5945
Yu L, Liu M, Chen F, Xu Q. Org Biomol Chem, 2015, 13: 8379–8392
Zhang DH, Tang XY, Shi M. Acc Chem Res, 2014, 47: 913–924
Pellissier H. Tetrahedron, 2014, 70: 4991–5031
Shi M, Lu JM, Wei Y, Shao LX. Acc Chem Res, 2012, 45: 641–652
Yu L, Guo R. Org Prepar Proc Int, 2011, 43: 209–259
Shi M, Shao LX, Lu JM, Wei Y, Mizuno K, Maeda H. Chem Rev, 2010, 110: 5883–5913
Pellissier H. Tetrahedron, 2010, 66: 8341–8375
Brandi A, Cicchi S, Cordero FM, Goti A. Chem Rev, 2003, 103: 1213–1270
Dolbier WR, Battiste MA. Chem Rev, 2003, 103: 1071–1098
Nakamura I, Yamamoto Y. Adv Synthesis Catalyis, 2002, 344: 111–129
Nakamura E, Yamago S. Acc Chem Res, 2002, 35: 867–877
Lautens M, Klute W, Tam W. Chem Rev, 1996, 96: 49–92
Mizuno K, Ichinose N, Yoshimi Y. J Photochem Photobiol C, 2000, 1: 167–193
For recent examples, see: Kimura N, Katta S, Kitazawa Y, Kochi T, Kakiuchi F. J Am Chem Soc, 2021, 143: 4543–4549
Zhao C, Feng Z, Xu G, Gao A, Chen J, Wang Z, Xu P. Angew Chem Int Ed, 2020, 59: 3058–3062
Kong RY, Crimmin MR. J Am Chem Soc, 2020, 142: 11967–11971
Huang J, Ho C. Angew Chem Int Ed, 2020, 59: 5288–5292
Beaumier EP, Ott AA, Wen X, Davis-Gilbert ZW, Wheeler TA, Topczewski JJ, Goodpaster JD, Tonks IA. Chem Sci, 2020, 11: 7204–7209
Yamamoto T, Murakami R, Komatsu S, Suginome M. J Am Chem Soc, 2018, 140: 3867–3870
Liang YF, Müller V, Liu W, Münch A, Stalke D, Ackermann L. Angew Chem Int Ed, 2017, 56: 9415–9419
Pal S, Zhou YY, Uyeda C. J Am Chem Soc, 2017, 139: 11686–11689
Maurer DP, Fan R, Thamattoor DM. Angew Chem Int Ed, 2017, 56: 4499–4501
Felix RJ, Weber D, Gutierrez O, Tantillo DJ, Gagné MR. Nat Chem, 2012, 4: 405–409
Wang R, Ksebati MB, Corbett TH, Kern ER, Drach JC, Zemlicka J. J Med Chem, 2001, 44: 4019–4022
Li C, Prichard MN, Korba BE, Drach JC, Zemlicka J. BioOrg Medicinal Chem, 2008, 16: 2148–2155
Takeuchi D, Osakada K. Polymer, 2008, 49: 4911–4924
Kirsch P. Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications. Weinheim: Wiley-VCH, 2013
Wang J, Sánchez-Roselló M, Aceña JL, del Pozo C, Sorochinsky AE, Fustero S, Soloshonok VA, Liu H. Chem Rev, 2014, 114: 2432–2506
Ni C, Hu M, Hu J. Chem Rev, 2015, 115: 765–825
Decaens J, Couve-Bonnaire S, Charette AB, Poisson T, Jubault P. Chem Eur J, 2021, 27: 2935–2962
Xiao H, Zhang Z, Fang Y, Zhu L, Li C. Chem Soc Rev, 2021, 50: 6308–6319
Dolbier WR Jr., Sellers SF, Al-Sader BH, Smart BE. J Am Chem Soc, 1980, 102: 5398–5399
Dolbier WR Jr., Sellers SF, Al-Sader BH, Fielder THJ, Elsheimer S, Smart BE. Isr J Chem, 1981, 21: 176–184
Dolbier WR Jr., Seabury M, Daly D, Smart BE. J Org Chem, 1986, 51: 974–979
Bunegar MJ, Fields R, Haszeldine RN. J Fluorine Chem, 1980, 15: 497–509
Dolbier WR Jr., Gautriaud E, Cai X. J Fluorine Chem, 2005, 126: 339–343
Hang XC, Gu WP, Chen QY, Xiao JC, Xu WG, Liu S. J Fluorine Chem, 2011, 132: 63–67
Dolbier WR Jr., Burkholder CR. Tetrahedron, 1985, 41: 297–307
For recent reviews on transition metal catalyzed carbene transfer reaction of diazo compounds, see: Wang J. Tetrahedron Lett, 2022, 108: 154135
Lee WCC, Zhang XP. Trends Chem, 2022, 4: 850–851
Liu Z, Sivaguru P, Zanoni G, Bi X. Acc Chem Res, 2022, 55: 1763–1781
Huang MY, Zhu SF. Chem Sci, 2021, 12: 15790–15801
Lübcke M, Szabó KJ. Synlett, 2021, 32: 1060–1071
Zhu D, Chen L, Fan H, Yao Q, Zhu S. Chem Soc Rev, 2020, 49: 908–950
Empel C, Koenigs RM. Synlett, 2019, 30: 1929–1934
Xiang Y, Wang C, Ding Q, Peng Y. Adv Synth Catal, 2019, 361: 919–944
Xia Y, Qiu D, Wang J. Chem Rev, 2017, 117: 13810–13889
Zhang D, Hu W. Chem Rec, 2017, 17: 739–753
Fructos MR, Díaz-Requejo MM, Pérez PJ. Chem Commun, 2016, 52: 7326–7335
Davies HML, Morton D. Chem Soc Rev, 2011, 40: 1857–1869
Jellema E, Jongerius AL, Reek JNH, de Bruin B. Chem Soc Rev, 2010, 39: 1706–1723
Doyle MP, Duffy R, Ratnikov M, Zhou L. Chem Rev, 2010, 110: 704–724
Lee WCC, Wang DS, Zhang C, Xie J, Li B, Zhang XP. Chem, 2021, 7: 1588–1601
Wang J, Xie J, Cindy Lee WC, Wang DS, Zhang XP. Chem Catal, 2022, 2: 330–344
Burdzinski G, Platz MS. J Phys Org Chem, 2010, 23: 308–314
For reviews on visible-light-mediated carbene transfer of diazo compounds, see: Empel C, Pei C, Koenigs RM. Chem Commun, 2022, 58: 2788–2798
Chen Z, Xie Y, Xuan J. Eur J Org Chem, 2022, 2022: e202201066
Durka J, Turkowska J, Gryko D. ACS Sustain Chem Eng, 2021, 9: 8895–8918
Yang Z, Stivanin ML, Jurberg ID, Koenigs RM. Chem Soc Rev, 2020, 49: 6833–6847
Ciszewski ŁW, Rybicka-Jasiñska K, Gryko D. Org Biomol Chem, 2019, 17: 432–448
Hirai K, Itoh T, Tomioka H. Chem Rev, 2009, 109: 3275–3332
For selected examples visible-light-mediated carbene transfer of diazo compounds,, see: Guo W, Zhou Y, Xie H, Yue X, Jiang F, Huang H, Han Z, Sun J. Chem Sci, 2023, 14: 843–848
Jana S, Pei C, Empel C, Koenigs RM. Angew Chem Int Ed, 2021, 60: 13271–13279
Zhou S, Cai B, Hu C, Cheng X, Li L, Xuan J. Chin Chem Lett, 2021, 32: 2577–2581
Yan K, He H, Li J, Luo Y, Lai R, Guo L, Wu Y. Chin Chem Lett, 2021, 32: 3984–3987
Hommelsheim R, Guo Y, Yang Z, Empel C, Koenigs RM. Angew Chem Int Ed, 2019, 58: 1203–1207
Jurberg ID, Davies HML. Chem Sci, 2018, 9: 5112–5118
Wei Y, Tinoco A, Steck V, Fasan R, Zhang Y. J Am Chem Soc, 2018, 140: 1649–1662
Xiao T, Mei M, He Y, Zhou L. Chem Commun, 2018, 54: 8865–8868
Vaske YSM, Mahoney ME, Konopelski JP, Rogow DL, McDonald WJ. J Am Chem Soc, 2010, 132: 11379–11385
Heinz B, Schmidt B, Root C, Satzger H, Milota F, Fierz B, Kiefhaber T, Zinth W, Gilch P. Phys Chem Chem Phys, 2006, 8: 3432–3439
Cai X, Sakamoto M, Hara M, Sugimoto A, Tojo S, Kawai K, Endo M, Fujitsuka M, Majima T. Photochem Photobiol Sci, 2003, 2: 1209–1214
Kretzschmar A, Patze C, Schwaebel ST, Bunz UHF. J Org Chem, 2015, 80: 9126–9131
For reviews on energy transfer catalysis, see: Strieth-Kalthoff F, James MJ, Teders M, Pitzer L, Glorius F. Chem Soc Rev, 2018, 47: 7190–7202
Zhou Q, Zou Y, Lu L, Xiao W. Angew Chem Int Ed, 2019, 58: 1586–1604
Neveselý T, Wienhold M, Molloy JJ, Gilmour R. Chem Rev, 2022, 122: 2650–2694
For recent examples, see: Zhang Y, Zhou G, Gong X, Guo Z, Qi X, Shen X. Angew Chem Int Ed, 2022, 61: e202202175
Zhou G, Shen X. Angew Chem Int Ed, 2022, 61: e202115334
Zhang Y, Zhang Y, Ye C, Qi X, Wu LZ, Shen X. Nat Commun, 2022, 13: 6111
Ye JH, Quach L, Paulisch T, Glorius F. J Am Chem Soc, 2019, 141: 16227–16231
Zhu M, Huang XL, Xu H, Zhang X, Zheng C, You SL. CCS Chem, 2021, 3: 652–664
Ma J, Chen S, Bellotti P, Guo R, Schäfer F, Heusler A, Zhang X, Daniliuc C, Brown MK, Houk KN, Glorius F. Science, 2021, 371: 1338–1345
Molloy JJ, Schäfer M, Wienhold M, Morack T, Daniliuc CG, Gilmour R. Science, 2020, 369: 302–306
Nakafuku KM, Zhang Z, Wappes EA, Stateman LM, Chen AD, Nagib DA. Nat Chem, 2020, 12: 697–704
Becker MR, Wearing ER, Schindler CS. Nat Chem, 2020, 12: 898–905
Cheng Q, Chen J, Lin S, Ritter T. J Am Chem Soc, 2020, 142: 17287–17293
Tian L, Till NA, Kudisch B, MacMillan DWC, Scholes GD. J Am Chem Soc, 2020, 142: 4555–4559
Zheng J, Swords WB, Jung H, Skubi KL, Kidd JB, Meyer GJ, Baik MH, Yoon TP. J Am Chem Soc, 2019, 141: 13625–13634
Chatterjee A, König B. Angew Chem Int Ed, 2019, 58: 14289–14294
Jiang Y, Wang C, Rogers CR, Kodaimati MS, Weiss EA. Nat Chem, 2019, 11: 1034–1040
Ravetz BD, Pun AB, Churchill EM, Congreve DN, Rovis T, Campos LM. Nature, 2019, 565: 343–346
Uoyama H, Goushi K, Shizu K, Nomura H, Adachi C. Nature, 2012, 492: 234–238
Timpe HJ, Kronfeld KP. J PhotoChem PhotoBiol A-Chem, 1989, 46: 253–267
Hock KJ, Mertens L, Koenigs RM. Chem Commun, 2016, 52: 13783–13786
Duan Y, Lin JH, Xiao JC, Gu YC. Chem Commun, 2017, 53: 3870–3873
Ning Y, Zhang X, Gai Y, Dong Y, Sivaguru P, Wang Y, Reddy BRP, Zanoni G, Bi X. Angew Chem Int Ed, 2020, 59: 6473–6481
Zhang H, Wang E, Geng S, Liu Z, He Y, Peng Q, Feng Z. Angew Chem Int Ed, 2021, 60: 10211–10218
Zhang J, Geng S, Feng Z. Chem Commun, 2021, 57: 11922–11934
Totherow WD, Gleicher GJ. J Am Chem Soc, 1969, 91: 7150–7154
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2022YFA1506100), Shenzhen Science and Technology Program (JCYJ20220818100604009), Guangdong Basic and Applied Basic Research Foundation (2021A1515010105, 2023A1515010601).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest A patent application based on this work has been filed and X.S., S.L. and S.C. may benefit from royalty payments.
Additional information
Supporting information
The supporting information is available online at chem.scichina.com and springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
11426_2023_1676_MOESM1_ESM.pdf
Supporting Information: Photocatalyzed [2+1] Cyclization of Alkenes and Silylated Trifluorodiazoethanes: Facile Entry into (Difluoromethylene)cyclopropanes
Rights and permissions
About this article
Cite this article
Chen, S., Zhang, Y., Liu, S. et al. Photocatalyzed [2+1] cyclization of alkenes and silylated trifluorodiazoethanes: facile entry into (difluoromethylene)cyclopropanes. Sci. China Chem. 66, 3141–3147 (2023). https://doi.org/10.1007/s11426-023-1676-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1676-y