Abstract
Purpose
The increasing reuse of wastewater for irrigation introduces surfactants and antibiotics into the environment. How these two kinds of compounds interact with regard to their sorption processes in soil is not clear.
Materials and methods
We performed batch experiments to investigate the sorption of linear alkylbenzene sulfonates (LAS) and its effect on sorption of sulfamethoxazole and ciprofloxacin in irrigated and non-irrigated soils with different organic matter (OM) contents.
Results and discussion
LAS sorption was non-linear in the presence of the antibiotics, and as general trend, it increased with rising OM content of soils. Free LAS was also removed from solution by complexation with Ca2+. Dissolved organic compounds released from soils with OM contents ≥18.4 g kg−1 further reduced LAS sorption. Sorption of sulfamethoxazole was reduced by LAS sorption only in one soil with a small OM content of 9.5 g kg−1.
Conclusions
The strong sorption of ciprofloxacin is not affected by LAS. Sulfamethoxazole sorption only competes with LAS sorption in organic matter-poor soils. Accumulation of organic matter in soils, for example due to long-term wastewater irrigation, provides extra sorption capacity for LAS and sulfamethoxazole so that competition for sorption sites is reduced.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Water supply is closely related with other crucial needs of people, like food production and sanitation. Therefore, in areas with a water deficiency—either in quantity or quality—the reuse of water has become a key issue (Bos et al. 2010). Particularly, in urban and peri-urban areas, the population growth demands an intensive use of high quality water and—at the same time—produces a large volume of wastewater (Jiménez et al. 2010). The irrigation of arable fields with untreated wastewater has been an ancient practice in many cities (Asano and Levine 1996) that is increasing nowadays, especially in many impoverished countries (Qadir et al. 2010). Additionally, irrigation with wastewater is an efficient treatment strategy because many pollutants are either transformed or retained in soil as water percolates through the underground in the so-called soil aquifer treatment (SAT), which contributes to the recharge of aquifers (Drewes et al. 2003).
On the other hand, several drawbacks are linked to wastewater irrigation like an accumulation of pollutants in soil (Gibson et al. 2010; Fatta-Kassinos et al. 2011), an increase in soil salinity (Rusan et al. 2007), and an exposure of farmers and crop consumers to pathogens (Gupta et al. 2009). Pharmaceuticals are present in wastewater (Kümmerer 2009) because most of them are not completely metabolized and are partially excreted together with urine and feces. Surfactants are also commonly present in municipal effluents (Edser 2006), and due to their amphiphilic properties, they can interact with both, polar and non-polar species, in solid phases and in solution as well. These interactions can affect sorption processes and mobility of pollutants in soil (Abu-Zreig et al. 1999; Peña et al. 2011). Such phenomena have already been observed for hydrophobic compounds (Jones-Hughes and Turner 2005), but little is known regarding the interactions between surfactants and hydrophilic pollutants like many pharmaceuticals.
The largest contiguous wastewater-irrigated area worldwide is the Mezquital Valley, which receives wastewater from the Mexico City metropolitan area (MCMA) since more than 100 years (Jiménez et al. 2010). The wastewater is discharged untreated into the semi-arid Mezquital Valley located 80 km North of Mexico City, where it is used for crop irrigation, mainly of alfalfa and maize. Several antibiotics have been found in the wastewater (Siemens et al. 2008; Gibson et al. 2010) and have accumulated in soils over time (Dalkmann et al. 2012). The two antibiotics selected for the study at hand, sulfamethoxazole and ciprofloxacin, were found in wastewater in the path flow from Mexico City to Mezquital Valley at concentrations of 1.21 ± 0.76 and 0.47 ± 0.30 μg L−1, respectively (Supporting information of Dalkmann et al. 2014b). Carrillo et al. (2016) reported a sulfamethoxazole concentration of 1.04 μg L−1 and a ciprofloxacin concentration of 3.47 μg L−1 for Mexico City wastewater. Batch experiments evidenced a weaker sorption of the anionic sulfamethoxazole, but not of the cationic and zwitterionic ciprofloxacin in irrigated soil compared to that in non-irrigated soil (Dalkmann et al. 2014a). The weaker sorption in wastewater-irrigated soil could be due to either (i) an increased negative charge of organic matter (OM) as a consequence of its aging, (ii) to blocking of high-affinity sorption sites by sulfamethoxazole that has been added to the soils with wastewater in the past, and/or (iii) to a competition with other compounds present in wastewater like dissolved organic matter (DOM) including surfactants (Dalkmann et al. 2014a). Linear alkylbenzene sulfonates (LAS) have been the most used surfactants since the 1960s (Schiebel 2004), when they were introduced to the market to replace the poorly biodegradable branched alkylbenzene sulfonates (Berna et al. 1991). LAS are usually degraded during aerobic wastewater treatment (Mungray and Kumar 2009). Nevertheless, there are still many areas like the Mezquital Valley around the globe that receive untreated wastewater for growing crops. Typical concentrations of LAS in treated and untreated wastewater from urban centers ranged from 2 to 32 mg L−1 (Gomez et al. 2011; Clara et al. 2007; Temminik and Klapwijk 2004; Scott and Jones 2000; Holt et al. 1995, 1998). In the case of Mexico City metropolitan area, concentrations in the range of 1.3–35 mg L−1 of methylene blue active substances (MBAS) in wastewater are reported (Mazari 1992). This parameter includes not only LAS but also other types of anionic surfactants. Nevertheless, these values are in good agreement with the previously mentioned studies, especially when taking into account that LAS are actually the most used surfactants. According to Fytianos et al. (1998), the risk of groundwater pollution with LAS is low, as these compounds sorb readily to OM in soil, but little is known about the influence they might have on the sorption and mobilization of other compounds.
Only few studies have focused on the influence of DOM (Haham et al. 2012) or of surfactants (Hari et al. 2005; Yu et al. 2012) on the sorption processes of pharmaceuticals. In the experiments of Haham et al. (2012), pre-conditioning of OM-poor soils with DOM extracted from biosolids or co-introduction of DOM reduced sulfapyridine sorption and enhanced its desorption, suggesting competition for sorption sites. Dissolved organic matter only enhanced sulfapyridine sorption in a soil with a very small specific surface area of 61 m2 g−1. Hari et al. (2005) found an increased sorption of acetaminophen and carbamazepine to river sediment in the presence of the cationic surfactant cetylpyridinium chloride (CPC) and the nonionic Tergitol NP9 surfactant, while the sorption of norfloxacin was not affected by the surfactants. Nalidixic acid sorption was increased at pH > 8 in the presence of CPC. Yu et al. (2012) reported that sodium dodecylbenzene sulfonate can enhance the mobility of enrofloxacin in soils, but that the magnitude of this effect was small.
We studied the sorption of anionic LAS to soils from the Mezquital Valley that were either irrigated or not with wastewater for more than 100 years. We also investigated the effect of LAS on the sorption of two commonly used antibiotics, ciprofloxacin and sulfamethoxazole. We hypothesized that (i) higher soil OM contents in long-term irrigated soils increase LAS sorption and (ii) sorption of anionic LAS competes with sorption of anionic sulfamethoxazole, but not with the strong sorption of cationic or zwitterionic ciprofloxacin.
2 Materials and methods
2.1 Soils
We selected two soils for our experiments that have received untreated wastewater for more than 100 years, referred to as Ulapa (Irr1) and Juandhó (Irr2), and two soils that have received only rainwater, namely Tezontlale (Rnf1) and El Tigre (Rnf2). The topsoils were sampled in August 2009 and March 2011 as composite samples from 0- to 30-cm depth. Physicochemical properties are summarized in Table 1. Soil samples were sieved (2 mm), lyophilized, and kept at −21 °C until the start of the experiment.
2.2 Chemicals
Standards of ciprofloxacin and sulfamethoxazole were obtained from Sigma-Aldrich (Schnelldorf, Germany). Their structures and some chemical properties are summarized in Table 2. As internal standards for the quantification of antibiotic concentrations, we used isotope-labeled ciprofloxacin (carboxyl-13C3, quinolone-15N, ≥98 % pure) and sulfamethoxazole (ring-13C6, ≥98 % pure), both purchased from LGC Standards (Wesel, Germany). We selected sodium dodecylbenzene sulfonate as model surfactant because it is used widely in household cleaning products. It was purchased from Tokyo Chemical Industries (TCI, Eschborn, Germany). All solvents used were of HPLC-grade quality.
2.3 Analysis of supernatants
Concentrations of pharmaceuticals in the liquid phase were measured by liquid chromatography tandem mass spectrometry (LC-MS/MS) with a TSQ Quantum Ultra (Thermo Finnigan, Dreieich, Germany) spectrometer equipped with a heated electrospray ionization ion source (HESI) running in the positive mode as described by Dalkmann et al. (2012, 2014a, 2014b). Routine limits of quantification (equal to the lowest concentration of standard used) were 1 μg L−1 for ciprofloxacin and 0.1 μg L−1 for sulfamethoxazole. Concentrations of LAS were also quantified using the LC-MS/MS system, but running in the negative mode. The acquired surfactant consisted of a mixture of four linear alkylbenzene sulfonates with alkyl chain lengths of C10, C11, C12, and C13. The relative contribution of the four compounds to the total signal intensity in the MS was 19 % for C10, 38 % for C11, 31 % for C12, and 12 % for C13. Assuming that the compounds had similar ionization efficiencies in the HESI source, we first calculated the concentrations of all the four compounds in the supernatant. Afterwards, the concentrations of the four individual compounds were summed up to give the total concentration of LAS in solution. The routine limit of quantification for LAS was 0.1 mg L−1. Details regarding the analysis of pharmaceutical and LAS concentrations can be found in the Electronic supplementary material.
Dissolved organic carbon (DOC) concentrations in the supernatant were determined as non-purgeable organic carbon using a Shimadzu VCPH TOC analyzer (Shimadzu, Duisburg, Germany).
2.4 Batch experiments
Batch experiments were performed in duplicate in 25-mL borosilicate glass vials. To prevent the biodegradation of the tested compounds, the soil was weighted inside the vials and then sterilized using an autoclave (121 °C, 100 kPa for 15 min). To ensure comparability with former studies (Dalkmann et al. 2014a), soil to solution ratios of 1:50 were selected when ciprofloxacin was used and 1:5 in the case of sulfamethoxazole, attending also to recommendations for optimal ratios of sorbed and dissolved pharmaceuticals (OECD 2000). To achieve a pre-equilibration of soil and added solution, the soil was shaken for 24 h at 10 rpm with a 0.01-M aqueous CaCl2 solution, and then, the system was spiked with a mixture of LAS and one of the two tested pharmaceuticals. Nominal surfactant concentrations of 0, 5, 10, and 30 mg L−1 were established, as typical LAS concentrations in wastewater are in the order of several mg L−1 (2 to 32 mg L−1 according to Clara et al. 2007; Scott and Jones 2000; Holt et al. 1995). The concentration of pharmaceuticals was 1.0 mg L−1 in all the experiments according to the procedure described in Dalkmann et al. (2014a). Once spiked, vials were shaken for 48 h at 10 rpm in the dark and then centrifuged (2500× g, 40 min) to separate the soil from the liquid phase. An aliquot of the supernatant was centrifuged at 15,000× g for 20 min to remove fine particles. The supernatant was spiked with the corresponding internal standard for the LC-MS/MS measurements and analyzed for both, surfactants and pharmaceuticals. As the surfactant can form complexes with divalent cations like Ca2+ or Mg2+ (Rico-Rico et al. 2009), we incubated controls of LAS solutions with the background electrolyte and the corresponding pharmaceutical, but without soil to evaluate the effect of the Ca2+ ions on the concentration of LAS under the same conditions as the main samples.
2.5 Data evaluation
The LAS sorption to the soils was described with the Freundlich isotherm model,
where Q is the amount of sorbed compound associated with the solid phase (mg kg−1), c is the concentration of the compound in the liquid phase (mg L−1), KF (mg1 − N LN kg−1) is a constant related to the sorption intensity, and N (dimensionless) is related to the heterogeneity of sorption sites. For the fitting of isotherms and the evaluation of correlations between variables, we used the software SigmaPlot version 11 (Systat, Erkrath, Germany).
3 Results
Measured concentrations of LAS in the controls with background electrolyte (0.01 M aqueous CaCl2) without soil were smaller than the nominal concentrations of 5, 10, and 30 mg L−1 after the equilibration period. As the media were sterile and protected from light, we discarded the possibility of a degradation of the surfactant. The observed reduction in LAS concentrations was thus likely caused by LAS complexation and precipitation with Ca2+. Since sorbed amounts of LAS were calculated as difference between spiked and measured LAS concentrations, this complexation has to be considered when interpreting the isotherms, and the observed reduction in LAS concentrations must be mathematically corrected for complexation-precipitation effects.
Independent of the presence of antibiotics, LAS were in almost all the cases bound more strongly to wastewater-irrigated soils (Irr1 and Irr2) than those to non-irrigated (rain fed) soils (Rnf1 and Rnf2), as shown by the slope of the isotherms (Figs. 1 and 2) and by their KF values (Table 3). At a soil-solution ratio of 1:50, we observed almost linear sorption isotherms with N values close to 1 when ciprofloxacin was not present (Fig. 1a) (Table 3). With a soil-solution ratio of 1:5 in the absence of sulfamethoxazole, we got also nearly linear isotherms for the soil with a OC content >28 g kg−1 (Fig. 2a). In both systems, when the antibiotic was present, the linearity of the LAS sorption isotherms was reduced (Figs. 1b and 2b), which was more pronounced with ciprofloxacin (Fig. 1b; soil-solution ratio of 1:50). At a soil to solution ratio of 1:5 in the presence of sulfamethoxazole, the isotherm for soil Irr1 showed an increase in LAS sorption for the spiking concentration of 30 mg LAS L−1, leading to an N Freundlich exponent larger than 1 (Fig. 2b, Table 3). In the presence of ciprofloxacin (soil-solution ratio of 1:50), KF values ranged from 100 to 276 mg1 − N LN kg−1 (Table 3), exceeding the KF coefficients in the range of 29–124 mg1 − N LN kg−1 that were found in the presence of sulfamethoxazole for a soil-solution ratio of 1:5. When normalized to the OC content, the isotherms for all the soils showed a similar pattern, and the whole set of data fell on a single isotherm for LAS sorption in the presence of sulfamethoxazole or ciprofloxacin and one single isotherm in the absence of the antibiotics (Figs. 1c, d and 2c, d).
Comparison of Freundlich sorption isotherms of linear alkylbenzene sulfonates (LAS) on rainfed (open symbols) and wastewater-irrigated (closed symbols) soils (soil to solution ratio 1:50) in the absence, (a) and (c), and in the presence, (b) and (d), of ciprofloxacin (1 mg L−1). The lower panels, (c) and (d), show the LAS sorption normalized to the organic carbon (OC) content of the four soils. The two replicates for each point of the isotherms are included in the plots. Therefore, no error bars are displayed
Comparison of Freundlich sorption isotherms of linear alkylbenzene sulfonates (LAS) on rainfed (open symbols) and wastewater-irrigated (closed symbols) soils (soil to solution ratio 1:5) in the absence, (a) and (c), and in the presence, (b) and (d), of sulfamethoxazole (1 mg L−1). The lower panels, (c) and (d), show the LAS sorption normalized to the organic carbon (OC) content of the four soils. The two replicates for each point of the isotherms are included in the plots. Therefore, no error bars are displayed
Concentrations of dissolved organic carbon (DOC) in equilibrium with the soils differed widely between the treatments. After equilibration with 0.01 M CaCl2 at a soil to solution ratio of 1:50, small concentrations of 1.3–3.8 mg L−1 DOC were found prior to the addition of LAS and ciprofloxacin, while concentrations of 7.4–15.8 mg L−1 DOC were found in the supernatant at a soil to solution ratio of 1:5 prior to the addition of LAS and sulfamethoxazole, which was an effect of dilution. The addition of LAS and sulfamethoxazole to the OM-rich soils Rnf2, Irr1, and Irr2 induced a further release of OM from soil to the liquid phase, increasing concentrations of soil-borne DOC (LAS carbon subtracted from total DOC) in the solution to values in the range from 31.3 to 47.2 mg L−1.
As hypothesized, the sorption of ciprofloxacin was not affected by the presence of LAS (Fig. 3a), but other than expected, the sorption of the anionic sulfamethoxazole did not decrease with increasing amounts of sorbed LAS in soils Rnf2, Irr1, and Irr2 (Fig. 3b). Only for the soil with the lowest OC content, Rnf1, sorbed amounts of sulfamethoxazole significantly decreased with increasing amounts of sorbed LAS indicating a reduced sorption due to competition for sorption sites (Fig. 3b).
4 Discussion
4.1 LAS sorption
Independent of the presence or absence of the antibiotic, the type of added antibiotic, or the soil to solution ratio, sorption of LAS was stronger to irrigated soils than that to non-irrigated soils. This can be related to the higher OM contents of the irrigated soils, since normalization of the sorbed amounts of LAS to the OC contents of the different soils produced superposed sorption isotherms for all the investigated soils (Figs. 1c, d and 2c, d). We also found a positive correlation between the KF parameter for LAS sorption and the OC content in the presence of ciprofloxacin (Pearson correlation coefficient R = 0.994, p = 0.006). This kind of correlation for LAS sorption has already been observed for sediments (Westall et al. 1999) and soils (Fytianos et al. 1998). Although sorption of anionic surfactants by clay minerals has been reported (Harendra and Vipulanandan 2012), LAS sorb mainly to the soil organic phases (de Wolf and Feijtel 1998). Only when concentrations of LAS are small (<<1 mg L−1), inorganic phases such as clay minerals or oxyhydroxides had a significant effect on LAS sorption (Rico-Rico et al. 2009). This explains why the Rnf2 soil, the most clayey soil, did not show a higher affinity for LAS than the OC-rich soils (KF values in Table 3) as we used always LAS concentrations larger than 1 mg L−1. The large OM content of the soils Irr1 and Irr2 is a consequence of the long-term irrigation in the Mezquital Valley (Dalkmann et al. 2014b). Our results thus illustrate that long-term irrigation indirectly increases LAS sorption in soils as a consequence of soil OM accrual over time.
We suggest that the weaker sorption of LAS at a soil to solution ratio of 1:5 compared to a soil to solution ratio of 1:50 is mainly a consequence of larger DOC concentrations. Although at a smaller soil to solution ratio smaller Kd values for soil organic matter could be expected (You et al. 1999; Yin et al. 2002), the total content of soil and therefore of soil organic matter is larger at the 1:5 soil to solution ratio. This led to a larger concentration of dissolved organic matter released from soil into the solution, which was also indicated by the noticeable darker coloration of the supernatant. This dissolved organic matter might have reduced LAS sorption by (i) direct competition with the surfactant molecules for sorption (Yu et al. 2011) and (ii) interactions with dissolved LAS by forming soluble associations as Yu et al. (2014) suggest for an anionic biosurfactant.
A sorption isotherm with N > 1 was observed for soil Irr1 at a soil to solution ratio of 1:5 in the presence of sulfamethoxazole (Fig. 2b). Such isotherms are typical when solute-solute interactions induce cooperative sorption (Hinz 2001). At an early stage, surfactant molecules attach to the mainly mineral (e.g., Ou et al. 1996) solid surface. As the density of sorbed molecules on soil surfaces increases, the intermolecular forces among the hydrophobic moiety contribute additionally to the sorption process. Hence, the isotherm shows a strong increase at larger dissolved and sorbed concentrations (Zhang and Somasundaran 2006).
4.2 LAS influence on sorption of pharmaceuticals
We observed a noticeable reduction in the linearity of LAS sorption in all the investigated soils when ciprofloxacin was present (Fig. 1b). This indicated a reduction of the homogeneity of sorption sites for LAS when the antibiotic was sorbed. In the presence of ciprofloxacin, sorption of LAS was described with smaller N values, but larger KF constants implying enhanced sorption of LAS at low LAS concentrations. Potential mechanisms causing enhanced sorption at low LAS concentrations could be the formation of ciprofloxacin-LAS associations via cation bridges or the masking of negatively charged clay mineral surfaces by sorbed ciprofloxacin. At higher LAS concentrations (nominal concentration = 30 mg L−1), the surfactant was sorbed in a very similar degree with and without ciprofloxacin (Fig. 1a, b). It seems that at higher LAS concentrations, the intermolecular forces among the hydrophobic moieties of LAS and also between them and the soil solid phase play an increasing role for sorption, so that interactions with ciprofloxacin become less important.
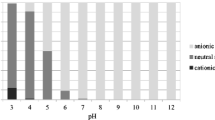
The present study confirmed the expected small effect of LAS on the strong sorption of the fluoroquinolone ciprofloxacin to the Mezquital Valley soils. Similarly, a negligibly small effect of LAS on the leaching of enrofloxacin (also a fluoroquinolone) in column experiments was observed (Yu et al. 2012). Ciprofloxacin sorbs mainly by a cation exchange mechanism (Wu et al. 2013); hence, anionic surfactants like LAS do not compete directly with ciprofloxacin for sorption sites. Although we only performed tests with the anionic surfactant LAS, neither cationic nor non-ionic surfactants had an effect on the sorption of the fluoroquinolone norfloxacin onto an aquifer material studied by Hari et al. (2005). The results of Hari et al. (2005) for norfloxacin can likely be extrapolated to other fluoroquinolones, because the different substituent groups in the chemical structure of fluoroquinolones have little influence on sorption interactions with soil so that they exhibit similar sorption behavior (Figueroa-Diva et al. 2010). Sorption of fluoroquinolones is strongly influenced by their pH-dependent chemical speciation (e.g., Vasudevan et al. 2009). The four studied soils have a pH in the range of 7.22–7.29 (Table 1), but the pH in the sorption experiments were always lower. The pH of the supernatant after the sorption process were in the range of 6.41–6.81 with a mean value of 6.57 (n = 32). At pH values larger than 9, the anionic form of ciprofloxacin predominates (pKa2 = 8.7), and its overall sorption to negatively charged surfaces, is strongly reduced. Therefore, competition between fluoroquinolones and anionic surfactants for sorption sites could take place at pH values larger than their pKa2.
Except for the soil with the lowest OM content (Rnf1), we did not find an evident trend of decreasing sulfamethoxazole sorption with increasing LAS concentrations. Only soil Rnf1 sorbed decreasing amounts of sulfamethoxazole with increasing concentrations of sorbed LAS (Fig. 3b), suggesting that in organic matter-poor soils competitive sorption with LAS might reduce sulfamethoxazole sorption. In addition, the formation of soluble LAS-sulfamethoxazole associations might have limited the sulfamethoxazole sorption in the organic matter-poor Rnf1 soil. The inhibition of sorption of hydrophobic compounds, such as chlorophenols, in sediments in the presence of LAS was attributed to the formation of associations of the hydrophobic compounds with LAS in the aqueous phase (Gao et al. 2012). On the other hand, a similar effect of sodium dodecyl sulfate on the sorption of phenanthrene was explained by the competition of both sorbates for sorption sites (Jones-Hughes and Turner 2005).
5 Conclusions
We conclude that OM is the main sorbent of LAS in the Mezquital Valley soils. In addition to sorption, complexation with Ca2+ significantly contributes to the removal of free LAS from the solution phase. Interactions of LAS with other dissolved organic compounds released from soils reduce LAS sorption. Significant reduction of sulfamethoxazole sorption due to competition with LAS for sorption sites likely occurs only in soils with small OM contents. The accumulated OM in wastewater-irrigated soils offers enough sorption sites for both LAS and sulfamethoxazole so that they no longer compete for sorption sites and the sorption of the antibiotic is not hindered. The effect of the anionic LAS on the sorption of ciprofloxacin in the clayey circumneutral soils of the Mezquital Valley is negligible.
References
Abu-Zreig M, Rudra R, Dickinson W, Evans L (1999) Effect of surfactants on sorption of atrazine by soil. J Contam Hydrol 36:249–263
Asano T, Levine AD (1996) Wastewater reclamation, recycle and reuse: past, present and future. Water Sci Technol 33:1–14
Bos R, Carr R, Keraita B (2010) Assessing and mitigating wastewater-related health risks in low-income countries: an introduction. In: Drechsel P, Scott CA, Raschid-Sally L, Redwood M, Bahri A (eds) Wastewater irrigation and health: assessing and mitigating risk in low-income countries. Earthscan, London, pp 29–47
Berna JL, Moreno A, Ferrer J (1991) The behaviour of LAS in the environment. J Chem Technol Biotechnol 50:387–398
Carrillo M, Braun GC, Siebe C, Amelung S, Siemens J (2016) Desorption of sulfamethoxazole and ciprofloxacin from long-term wastewater-irrigated soils of the Mezquital Valley as affected by water quality. J Soils Sediments 16:966–975
Clara M, Scharf S, Scheffknecht C, Gans O (2007) Occurrence of selected surfactants in untreated and treated sewage. Water Res 41:4339–4348
Dalkmann P, Broszat M, Siebe C, Willaschek E, Sakinc T, Huebner J, Amelung W, Grohmann E, Siemens J (2012) Accumulation of pharmaceuticals, Enterococcus, and resistance genes in soils irrigated with wastewater for zero to 100 years in central Mexico. PLoS One 7(9), e45397
Dalkmann P, Willaschek E, Schiedung H, Bornemann L, Siebe C, Siemens J (2014a) Long-term wastewater irrigation reduces sulfamethoxazole sorption, but not ciprofloxacin binding, in Mexican soils. J Environ Qual 43:964–970
Dalkmann P, Siebe C, Amelung W, Schloter M, Siemens J (2014b) Does long-term irrigation with untreated wastewater accelerates the dissipation of pharmaceuticals in soil? Environ Sci Technol 48:4963–4970
de Wolf W, Feijtel T (1998) Terrestial risk assessment for linear alkyl benzene sulfonate (LAS) in sludge-amended soils. Chemosphere 36:1319–1343
Drewes JE, Reinhard M, Fox P (2003) Comparing microfiltration-reverse osmosis and soil-aquifer treatment for indirect potable reuse of water. Water Res 37:3612–3621
Edser C (2006) Latest market analysis. Focus on Surfactants 5:1–2
Fatta-Kassinos D, Kalavrouziotis IK, Koukoulakis PH, Vasquez MI (2011) The risks associated with wastewater reuse and xenobiotics in the agroecological environment. Sci Total Environ 409:3555–3563
Figueroa-Diva RA, Vasudevan D, MacKay A (2010) Trends in soil sorption coefficients within common antimicrobial families. Chemosphere 79:786–793
Fytianos K, Voudrias E, Papamichali A (1998) Behavior and fate of linear alkylbenzene sulfonate in different soils. Chemosphere 36:2741–2746
Gao P, Feng YJ, Zhang ZH, Liu JF (2012) Effect of heavy metals and surfactants on the adsorption of phenolic compounds on sediment. Int J Environ Sci Technol 9:671–682
Gibson R, Durán-Álvarez JC, León Estrada K, Chávez A, Jiménez-Cisneros B (2010) Accumulation and leaching potential of some pharmaceuticals and potential endocrine disruptors in soils irrigated with wastewater in the Tula Valley, Mexico. Chemosphere 81:1437–1445
Gomez V, Ferreres L, Pocurull E, Borrull F (2011) Determination of non-ionic and anionic surfactants in environmental water matrices. Talanta 84:859–866
Gupta N, Khan DK, Santra SC (2009) Prevalence of intestinal helminth eggs on vegetables grown in wastewater-irrigated areas of Titagarh, West Bengal, India. Food Control 20:942–945
Haham H, Oren A, Chefetz B (2012) Insight into the role of dissolved organic matter in sorption of sulfapyridine by semiarid soils. Environ Sci Technol 46:11870–11877
Harendra S, Vipulanandan C (2012) Determination of sodium dodecyl sulfate (SDS) and biosurfactants sorption and transport parameters in clayey soil. J Surfactants Deterg 15:805–813
Hari AC, Paruchuri RA, Sabatini DA, Kibbey TCG (2005) Effects of pH and cationic and nonionic surfactants on the adsorption of pharmaceuticals to a natural aquifer material. Environ Sci Technol 39:2592–2598
Hinz C (2001) Description of sorption data with isotherm equations. Geoderma 99:225–243
Holt MS, Waters J, Comber MHI, Armitage R, Morris G, Newbery C (1995) AIS/CEISO environmental surfactant monitoring programme. SDIA sewage treatment pilot study on linear alkylbenzene sulphonate (LAS). Water Res 29:2063–2070
Holt MS, Fox KK, Burford M, Daniel M, Buckland H (1998) UK monitoring study on the removal of linear alkylbenzene sulphonate in trickling filter type sewage treatment plants. Contribution to GREAT-ER project #2. Sci Total Environ 210–211:255–269
Jiménez B, Drechsel P, Koné D, Bahri A, Raschi-Sally L, Qadir M (2010) Wastewater, sludge and excreta. Use in developing countries: an overview. In: Drechsel P, Scott CA, Raschid-Sally L, Redwood M, Bahri A (eds) Wastewater irrigation and health: assessing and mitigating risk in low-income countries. Earthscan, London, pp 3–27
Jones-Hughes T, Turner A (2005) Sorption of ionic surfactants to estuarine sediment and their influence on the sequestration of phenanthrene. Environ Sci Technol 39:1688–1697
Kümmerer K (2009) Antibiotics in the aquatic environment—a review—part I. Chemosphere 75:417–434
Mazari-Hiriart M (1992) Potential groundwater contamination by organic compounds in the Mexico City metropolitan area. University of California, Los Angeles, Dissertation
Mungray AK, Kumar P (2009) Fate of linear alkylbenzene sulfonates in the environment: a review. Int Biodeter Biodegr 63:981–987
OECD (2000) Test No. 106: adsorption—desorption using a batch equilibrium method, OECD guidelines for the testing of chemicals, Section 1, OECD Publishing, Paris
Ou Z, Yediler A, He Y, Jia L, Kettrup A, Sun T (1996) Adsorption of linear alkylbenzene sulfonate (LAS) on soils. Chemosphere 32:827–839
Peña A, Palma R, Mingorance MD (2011) Transport of dimethoate through a Mediterranean soil under flowing surfactant solutions and treated wastewater. Colloid Surface A 384:507–512
Qadir M, Wichelns D, Raschid-Sally L, McCornick PG, Drechsel P, Bahri A, Minhas PS (2010) The challenges of wastewater irrigation in developing countries. Agr Water Manage 97:561–568
Rico-Rico A, Temara A, Behrends T, Hermens JLM (2009) Effect of sediment properties on the sorption of C12-2-LAS in marine and estuarine sediments. Environ Pollut 157:377–383
Rusan MJM, Hinnawi S, Rousan L (2007) Long term effect of wastewater irrigation of forage crops on soil and plant quality parameters. Desalination 215:143–152
Schiebel JJ (2004) The evolution of anionic surfactant technology to meet the requirements of the laundry detergent industry. J Surfactants Deterg 7:319–328
Scott MJ, Jones MN (2000) The biodegradation of surfactants in the environment. BBA–Biomembranes 1508:235–251
Siemens J, Huschek G, Siebe C, Kaupenjohann M (2008) Concentrations and mobility of human pharmaceuticals in the world’s largest wastewater irrigation system, Mexico City-Mezquital Valley. Water Res 42:2124–2134
Temminik H, Klapwijk B (2004) Fate of linear alkylbenzene sulfonate (LAS) in activated sludge plants. Water Res 38:903–912
Vasudevan D, Bruland GL, Torrance BS, Upchurch VG, MacKay AA (2009) pH-dependent ciprofloxacin sorption to soils: interaction mechanisms and soil factors influencing sorption. Geoderma 151:68–76
Wu Q, Li Z, Hong H (2013) Adsorption of the quinolone antibiotic nalidixic acid onto montmorillonite and kaolinite. Appl Clay Sci 74:66–73
Westall JC, Chen H, Zhang W, Brownawell BJ (1999) Sorption of linear alkylbenzenesulfonates on sediment materials. Environ Sci Technol 33:3110–3118
Yin Y, Impellitteri CA, You SJ, Allen HE (2002) The importance of organic matter distribution and extract soil:solution ratio on the desorption of heavy metals from soils. Sci Total Environ 287:107–119
You SJ, Yin Y, Allen HE (1999) Partitioning of organic matter in soils: effects of pH and water/soil ratio. Sci Total Environ 227:155–160
Yu H, Huang GH, An CJ, Wei J (2011) Combined effect of DOM extracted from site soil/compost and biosurfactant on the sorption and desorption of PAHs in a soil-water system. J Hazard Mater 190:883–890
Yu Z, Yediler A, Yang M, Schulte-Hostede S (2012) Leaching behavior of enrofloxacin in three different soils and the influence of a surfactant on its mobility. J Environ Sci 24:435–439
Yu H, Huang GH, Xiao H, Wang L (2014) Combined effects of DOM and biosurfactant enhanced biodegradation of polycyclic aromatic hydrocarbons (PAHs) in soil-water systems. Environ Sci Pollut Res 21:10536–10549
Zhang R, Somasundaran P (2006) Advances in adsorption of surfactants and their mixtures at solid/solution interfaces. Adv Colloid Interfac 123–126:213–229
Acknowledgments
We acknowledge funding for a scholarship for MC by the German Academic Exchange Service (DAAD, Ref. Nr. A/11/79684). Furthermore, this work was jointly co-funded by the German Research Foundation (DFG, grant no. SI 1106/5-1,2) and the Mexican Consejo Nacional de Ciencia y Tecnología (CONACyT, grant no. I 0110-193-10). We thank Christian Hoffmann for supporting the LC-MS/MS analysis of LAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Xilong Wang
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 28 kb)
Rights and permissions
About this article
Cite this article
Carrillo, M., Siebe, C., Dalkmann, P. et al. Competitive sorption of linear alkylbenzene sulfonate (LAS) surfactants and the antibiotics sulfamethoxazole and ciprofloxacin in wastewater-irrigated soils of the Mezquital Valley, Mexico. J Soils Sediments 16, 2186–2194 (2016). https://doi.org/10.1007/s11368-016-1418-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-016-1418-1