Abstract
Purpose
Biochar has been suggested as a soil conditioner to improve soil fertility and crop productivity while simultaneously mitigate global climate change by storing carbon in the soil. This study investigated the effect of pine (Pinus radiata) biochar application on soil water availability, nitrogen (N) and carbon (C) pools and growth of C3 and C4 plants.
Materials and methods
In a glasshouse pot trial, a pine biochar (untreated) and nutrient-enriched pine biochar were applied to a market garden soil with C3 (Spinacia oleracea L.) and C4 (Amaranthus paniculatus L.) plants at rates of 0, 1.0, 2.0, and 4.0 % (w/w). Plant biomass, soil pH, moisture content, water holding capacity (WHC), hot water extractable organic C (HWEOC), and total N (HWETN), total C and N, and their isotope compositions (δ 13C and δ 15N) of soils and plants were measured at the end of the experimentation.
Results and discussion
The soil moisture content increased while plant biomass decreased with increasing untreated biochar application rates. The addition of nutrient-enriched biochar significantly improved plant biomass in comparison to the untreated biochar addition at most application rates. Biochar application also increased the levels of labile organic C and N pools as indicated by HWEOC and HWETN.
Conclusions
The results suggested that the addition of pine biochar significantly improved soil water availability but not plant growth. The application of nutrient-enriched pine biochar demonstrated that the growth of C3 and C4 plants was governed by biochar nutrient availability rather than its water holding capacity under the pot trial condition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Biochar is a highly porous, carbon (C)-rich solid residue of pyrolysis of biomass and has recently been suggested and hotly contested for applications in soil as a means of mitigating global climate change while simultaneously improving soil fertility and crop productivity (Lehmann and Joseph 2009; Atkinson et al. 2010; Woolf et al. 2010). During the growth of plant biomass, C is removed from the atmosphere through photosynthesis and sequestrated in the form of biochar after plant biomass is converted to char. Due to its inherently recalcitrant chemical composition, biochar could remain stable in soils for hundreds to thousands of years (Cheng et al. 2008). In addition, adding biochar to soil also has the potential to reduce greenhouse gases (GHGs) such as nitrous oxide (N2O) and methane (CH4) emissions (e.g., Woolf et al. 2010; Anderson et al. 2011; Rogovska et al. 2011).
Biochar addition could also improve the soil’s ability to retain bioavailable water, reduce leaching of nutrients to surface and ground waters, decrease soil bulk density, increase its cation exchange capacity (CEC), and act as a liming agent in the acidic soils (Laird et al. 2010; Rogovska et al. 2011; Novak et al. 2012). Due to its highly porous physical structure and high specific surface area, biochar is considered to have a greater ability to adsorb and retain water and nutrients, and its application may improve soil physical properties and thus soil water content and holding capacity (WHC) (Glaser et al. 2002; Steiner et al. 2007; Baronti et al. 2014). Novak et al. (2009) found that the addition of switchgrass biochar to a Norfolk loamy sandy soil increased soil water retention from 6.7 to 15.9 % relative to the control. Basso et al. (2013) applied biochar produced using red oak (Quercus rubra) feedstock by fast pyrolysis (500 °C) to a sandy loam soil, and found that biochar addition significantly increased gravity-drained water content and could maintain this water in the soil for an extended period of time relative to the control. This appears to be mostly due to the high porosity of the biochar-treated soil that could have allowed more water to be physically retained. The increase in soil CEC, resulting from the oxidation of biochar and the adsorption of organic matter on the biochar surfaces, will increase its ability to retain cations such as NH4 + and thus enhance nutrient retention (Liang et al. 2006; Clough and Condron 2010). Since soil water and nutrient availability are two key factors controlling the plant growth and microbial activities, biochar addition would enhance plant productivity.
Despite the beneficial effects mentioned above, biochar application may also have negative effects on plant growth (Gaskin et al. 2010; Reverchon et al. 2014). Volatile organic compounds associated with biochar could be deleterious to plant growth (Deenik et al. 2010). Moreover, adsorption of nutrient cations such as NH4 + and potassium (K) by biochar from the soil may result in a decline in available nutrients for the plant to uptake. In some cases, yield depression was attributed to the high pH under high rates of biochar application (van Zwieten et al. 2010). As a result, conflicting results about the effect of biochar addition on plant growth and crop productivity have been reported (Gaskin et al. 2010; Zhang et al. 2012; Güereña et al. 2013). To offset the negative effects of biochar addition, it has been proposed to treat biochar with an appropriate nutrient solution or in combination with fertilizers (Rajkovich et al. 2012; Zhang et al. 2012; Reverchon et al. 2014). For example, Taghizadeh-Toosi et al. (2012) demonstrated a proof of concept that ammonia (NH3) emissions could be captured by pine (Pinus radiata) biochar and made bioavailable in the soil for growing perennial ryegrass (Lolium perenne L.). They found that the addition of NH3 adsorbed biochar increased the leaf dry matter yields by two- to threefold and root dry matter yields by twofold relative to the treatments of non-treated biochar addition. Steiner et al. (2007) also showed that charcoal application together with a mineral fertilizer improved plant growth and had almost twice as much yield as the mineral fertilizer application only, because charcoal had the ability to sustain the soil fertility if an additional nutrient source was given. In our recent study, Reverchon et al. (2014) saturated a jarrah biochar and a pine biochar in a nutrient-rich solution (Hoagland’s No. 2 Basal Salt Mixture, Sigma) to investigate the effect of nutrient enrichment on the wheat biomass due to biochar application. They found that both types of the nutrient-enriched biochar had a positive effect on wheat grain biomass when compared with the same application rate of the untreated biochar, believed to be due to the enhanced nutrient availability and reduced N cycling rates in the plant-soil system, and thus reduced the competition for available nutrients between biochar and plants. However, nutrient-enriched biochar was added only at single rate in the previous study, the effect of nutrient-enriched biochar on plant growth following multiple biochar application rates was not examined.
Hot water extractable organic C (HWEOC) and total N (HWETN) are considered as useful indicators of C and N availability and soil fertility (Sparling et al. 1998; Ghani et al. 2003; Chen et al. 2004; Ibell et al. 2010; Jiang et al. 2010). Hot water would not only extract soluble C and N pools, but also components of microbial biomass. Previous studies also showed that HWEOC was highly correlated with soil respired CO2, microbial biomass C and N, mineralizable N, and total carbohydrates (Sparling et al. 1998; Ghani et al. 2003; Chen and Xu 2005). Thus, Ghani et al. (2003) suggested that HWEOC might be used as an integrated indicator of soil quality. In this study, a glasshouse pot experiment was conducted to investigate the effects of untreated and nutrient-enriched biochar application on growth of C3 and C4 plants with different water use efficiencies and growth strategy. The specific objectives were to: (1) evaluate the effect of biochar addition on soil water availability and C and N pools; (2) investigate the effect of untreated and nutrient-enriched biochar addition on C3 and C4 plant biomass; and (3) test which factor influenced by biochar application controlled the growth of C3 and C4 plants. Based on the previous studies, we hypothesized that: (1) biochar addition could enhance soil water availability, but depend on the application rates; (2) the addition of nutrient-enriched biochar could enhance plant growth by increasing nutrient availability for plants; and (3) growth of C3 and C4 plants would respond differently to the biochar application.
2 Materials and methods
2.1 Biochar
As an extension of our previous work, the pine biochar (P. radiata D. Don) used in this study was the same as that used in Reverchon et al. (2014). In brief, the woodchips used to generate the biochar were from a pine plantation near Bunbury, about 200 km south of Perth, Western Australia. The biochar was produced by ANSAC Pty Ltd (www.ansac.com.au) using the ANSAC HK indirectly fired kiln with 20 min residence time and 700 °C nominal temperature of pyrolysis under oxygen depleted condition. The biochar was cooled indirectly to below 90 °C and then discharged in air. In the present study, a portion of the pine-char was treated with Hoagland solution (Hoagland’s no. 2 Basal Salt Mixture, Sigma), a commonly used fertilizer in the horticultural industry principally containing calcium and potassium nitrate, magnesium sulfate and ammonium phosphate (Electronic supplementary material, Table S1). Pine biochar was saturated in Hoagland solution for 24 h, and then thoroughly dried in the oven at 70 °C for 48 h. The characteristics of both untreated and nutrient-enriched biochar were shown in Table 1. Before mixing with the soil, both types of biochar were ground and passed through a 4-mm sieve.
2.2 Pot experimentation
The pot trial was carried out in a glasshouse at The University of Western Australia, Perth, Australia. A single type of soil, the market garden soil (Table 2), was chosen to provide consistency, allowing the study to focus on the effect of addition rates and nutrient treatment of biochar. Different quantities of either untreated or nutrient-enriched pine biochar were mixed with the soil at four rates of 0, 1.0 %, 2.0 % and 4.0 % (w/w). Two types of plants, spinach (Spinacia oleracea L., C3 plant) and amaranth (Amaranthus paniculatus L., C4 plant), were planted. During the whole trial period, each pot was weighed and watered every 2 or 3 days to maintain 80 % of water holding capacity. Each treatment was replicated three times. The detailed management and maintenance of the pot trials were similar to those reported by Reverchon et al. (2014).
Both spinach (SP) and amaranth (AM) plants were harvested after 53 days of growth. During harvesting, each plant was separated into two sections, namely aboveground parts (the leaves and stems) and belowground parts (the roots). The soil from each of the pots was well mixed and fresh subsamples (about 500 g) were collected and stored at 4 °C. The plant materials were dried at 65 °C to a constant weight to determine the dry weights and the root-to-shoot mass ratios (R:S). Then, the oven-dried plant samples and fresh soils were posted from UWA in Perth to Griffith University (Nathan campus, Brisbane, Australia) for further analysis.
2.3 Sample analyses
Soil moisture was measured gravimetrically after drying in the oven at 105 °C for 48 h. Soil WHC was determined by submerging the subsamples in water for 4 h, and subsequent draining for another 24 h, and then drying in the oven at 105 °C for 48 h. A soil suspension was prepared using a soil:water ratio of 1:5 (w:v) to measure soil pH. About 50 g of each soil sample was air-dried and ground to a fine homogenous powder using a Rocklabs™ ring grinder. 60-70 mg of the grounded soil was then weighed and packed into 8 × 5 mm tin capsules for analysis of total C, total N, C isotope composition (δ13C), and N isotope composition (δ15N) using an isotope ratio mass spectrometer (GV Isoprime, Manchester, UK) (Xu et al. 2000, 2003). Hot water extractable organic C (HWEOC) and total N (HWETN) were determined using the method of Sparling et al. (1998) and Chen and Xu (2005). 7.00 g of fresh soil was mixed with 35 mL of distilled water in polypropylene tubes and incubated in the oven at 70 °C for 18 h. The tubes were shaken on an end-over-end shaker for 5 min and then placed in a centrifuge at 10,000 rpm for 10 min. The suspension was filtered through Whatman 42 filter papers into 70 mL plastic containers. Finally, the extract was passed through a 33 mm Millex syringe-driven 0.45-μm filter. HWEOC and HWETN were measured using a TOC/TN analyzer (TOC-VCSH/CSN TOC/N, Shimadzu, Kyoto, Japan). The oven-dried plant materials were also ground to measure total C, total N, δ13C, and δ15N using mass spectrometer as above (Xu et al. 2000, 2003).
2.4 Statistical analyses
Statistical analyses were conducted with Statistix 8.0, and all figures were drawn with OriginPro 8.5 software (OriginLab, USA). Three-way ANOVA were conducted to test the effects of biochar type, biochar application rate, plant species and their interactions on the soil and plant characteristics at P < 0.05. Regression curve estimations were performed to establish the relationships between the rates of biochar application and measured soil and plant variables. The different effects of untreated and nutrient-enriched biochar were tested by pre-forming the effect of treatment contrast on the soil and plant variables by linear regression analysis. All measured variables were analyzed separately for each plant type and considered significant at P < 0.05.
3 Results
3.1 Soil properties
The addition of both untreated and nutrient-enriched biochars resulted in a linear increase in soil moisture content with the application rate with growth of the C3 plant (Electronic supplementary material, Fig. S1a), while moisture content of the soils with growth of the C4 plant only quadratically increased with the rate of untreated biochar addition (Electronic supplementary material, Fig. S1b). For the soils with the C4 plant, the moisture content with nutrient-enriched biochar addition was significantly lower than that with untreated biochar addition at the highest rate (4 % w/w) (Electronic supplementary material, Figs. S1b, d; P < 0.001). WHC of the soils with the C4 plant increased linearly with the rate of untreated biochar addition (Electronic supplementary material, Fig. S1d). For the soils with both C3 and C4 plants, the soil pH and total C increased and soil δ 13C decreased with increasing the rate of biochar addition for both untreated and nutrient-enriched biochar (Table 3). No significant effect of biochar addition on the soil total N and δ 15N was observed (Electronic supplementary material, Fig. S2). There were significant interactions between biochar type, biochar application rate and plant species on soil pH, moisture content, WHC and total C, but not on soil total N, δ 13C and δ 15N (Electronic supplementary material, Table S2), and soil inorganic N contents (Electronic supplementary material, Table S3).
3.2 HWEOC and HWETN
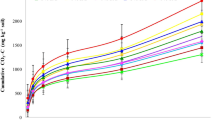
Concentrations of HWEOC in soils with both C3 and C4 plants linearly increased with the rate of untreated biochar addition (Figs. 1a, b). For the soils with both C3 and C4 plants, concentrations of HWEOC with the untreated biochar addition were significantly higher than those of the nutrient-enriched biochar addition (Fig. 1a, b). For the soils with C3 plants, HWETN linearly increased with the rate of nutrient-enriched biochar addition (Fig. 1c), while HWETN of the soils with C4 plants increased linearly with the rate of biochar addition for both untreated and nutrient-enriched biochar (Fig. 1d). For the soils with C4 plants, HWETN of the soils with the untreated biochar addition was significantly higher than that of the soils with the nutrient-enriched biochar addition (Fig. 1d). There were no interactions between biochar type, biochar application rate and plant species on HWEOC and HWETN of the soils (Electronic supplementary material, Table S2).
Hot water extractable organic carbon (HWEOC) and total nitrogen (HWETN) following the rate of biochar application in the soils with C3 and C4 plants in a pot experiment. Nutrient-enriched biochar application is represented in open circles and untreated biochar in filled circles. Values represent the means of triplicates ± standard errors
3.3 Plant biomass
For both C3 and C4 plants, aboveground biomass with the nutrient-enriched biochar addition was significantly higher than that with the untreated biochar addition (Fig. 2a, b). Aboveground biomass of the C4 plant was linearly decreased with the rate of untreated biochar addition (Fig. 2b) and similar trend but to a less extent was observed for the C3 plants (Fig. 2a). Root biomass with nutrient-enriched biochar addition was significantly higher than that with the untreated biochar addition (P < 0.05) (Figs. 2c, d). Root biomass of both C3 and C4 plants was linearly decreased with the rate of untreated biochar addition (Figs. 2c, d), while there were no consistent and significant effects of application rate of nutrient-enriched biochar on root biomass of both C3 and C4 plants (Figs. 2c, d). For the C4 plants, R:S was linearly decreased with the rate of untreated biochar addition (Fig. 2f), but this trend was not observed for the C3 plant (Fig. 2e). There were no interactions between biochar type, biochar application rate and plant species on plant aboveground, root biomass and R:S ratio, except for the significant interactions between biochar type and plant species on root biomass (P < 0.05, Electronic supplementary material, Tables S3 and S4).
3.4 Plant nutrient status
Plant δ 13C and nutrient concentrations versus the rate of biochar addition are shown in Table 3. δ 13C of the aboveground C3 plants decreased linearly with the rate of untreated biochar addition and non-linearly with the rate of nutrient-enriched biochar addition. Total N concentration of the aboveground C3 plants non-linearly decreased with the rate of both untreated and nutrient-enriched biochar addition. Root δ 13C of the C3 plants linearly decreased with the rate of biochar addition for both untreated and nutrient-enriched biochar. Root δ 15N of the C3 plants linearly increased with the untreated biochar addition. Total C concentration of the aboveground C4 plants were linearly increased with the rate of nutrient-enriched biochar addition. The addition of nutrient-enriched biochar significantly increased root total C concentration of the C4 plants in comparison to the addition of untreated biochar (P < 0.05). δ 15N of the aboveground C4 plants non-linearly decreased with the rate of nutrient-enriched biochar addition. There were no interactions between biochar type, biochar application rate and plant species on total C concentration, C content and δ 15N of the aboveground plants, and on root total N concentration and δ 15N, while significant interactions between biochar type, biochar application rate and plant species on total N concentration, N content and δ 13C of the aboveground plants, and on root total C concentration, C content, N content, and δ 13C as well (Supplementary Tables S3 and S4).
When data were pooled together for each species, δ 13C of the plant aboveground parts accounted for 69 % (n = 23, P < 0.001) and 22 % (n = 24, P < 0.05) of the variation in root δ 13C for C3 and C4 plants respectively (Fig. 3a, b). Root δ 15N accounted for 41 % (n = 23, P < 0.001) and 94 % (n = 24, P < 0.001) of the variation in aboveground δ 15N for C3 and C4 plants respectively (Fig. 3c, d).
4 Discussion
In this study, the addition of biochar to a market garden soil was shown to significantly affect soil water availability, labile C and N pools, and the growth of both C3 and C4 plants. Soil water availability and C and N availability were enhanced, while the growth of C3 and C4 plants was reduced with increased rate of untreated biochar addition. Overall, the greater plant biomass of both C3 and C4 plants in nutrient-enriched biochar treatment than in the untreated biochar treatment indicated that the negative effect of biochar on the plant growth could be offset by high nutrient availability.
4.1 Soil water availability
The increased soil moisture content and WHC in response to the biochar amendment in this study confirmed that biochar could affect soil water availability (Electronic supplementary material, Fig. S1), and was consistent with the findings reported in the previous studies (Glaser et al. 2002; Kammann et al. 2011; Basso et al. 2013). The ability of biochar to improve soil water availability was related to its high specific surface area and porous structure (de Melo Carvalho et al. 2014). In the present study, the addition of both untreated and nutrient-enriched biochar increased the moisture content of soil with C3 plants (Electronic supplementary material, Fig. S1a), while moisture content of the soils with C4 plants only increased with the rate of untreated biochar addition (Electronic supplementary material, Fig. S1b). Different responses of water content in the soils with C3 and C4 plants were attributed to their different water use efficiencies. It has been well known that C4 plants have higher water use efficiencies than C3 plants, since C4 plant can maintain high photosynthetic rates even when stomatal conductance is low (Rawson et al. 1977; Morison and Gifford 1983). When the C4 plants were provided with more nutrients as a result of nutrient-enriched biochar addition, more water would be used for the growth (Fig. 2b, d) and result in lower soil moisture content (Electronic supplementary material, Fig. S1b), which could provide an explanation as to why, in this study, water content of the soils with the C4 plants did not increase following the rate of nutrient-enriched biochar application. The significantly lower soil water content under the nutrient-enriched biochar addition (P < 0.001) was also associated with the finding that biomass of the C4 plants was significantly higher under the nutrient-enriched biochar application.
4.2 Plant biomass
Many studies have demonstrated that biochar increased plant biomass and crop yields (Major et al. 2010; Jeffery et al. 2011; Zhao et al. 2014; Xu et al. 2015), however, both no changes and negative effect of biochar application have also been reported (van Zwieten et al. 2010; Lentz and Ippolito 2012). Depending on soil, crop, biochar type and experimental conditions, the effect of biochar on plant growth and crop yields has been attributed to various mechanisms: (i) affecting soil water availability by improving soil water retention or competition with plants for water; (ii) influencing nutrient availability either by serving as a direct source of nutrients for plant uptake and increasing soil nutrient retention, or competing with plants for nutrients by adsorption; and (iii) affecting soil N cycling. In this study, the aboveground and root biomass of both C3 and C4 plants decreased with increasing the application rate of untreated biochar (Fig. 2). This observation may be attributed to: (i) biochar reduced nutrient availability for the plants by competing with the plants for water and nutrient, and a previous study reported that the addition of biochar to calcareous soils may reduce N availability, and require additional soil N inputs to maintain yield targets (Lentz and Ippolito 2012); (ii) volatile organic compounds, heavy metals, and/or other contaminants associated with biochar can be deleterious to plant growth (Chan and Xu 2009; Deenik et al. 2010; Hale et al. 2012); and (iii) high pH as a result of the high rates of biochar application (van Zwieten et al. 2010).
In this study, the plant growth was not limited by soil water availability, since the pot experimentation was conducted under controlled conditions with sufficient soil water. Moreover, the increased soil water availability (Electronic supplementary material, Fig. S1) following biochar application did not enhance the plant growth. However, the opposite results that the plant biomass decreased with increasing the rate of biochar addition (Fig. 2) were observed. Especially for the C3 plants, although the plant water use efficiency as revealed by δ 13C was affected by the biochar-mediated soil moisture change (Table 3), their growth was still not enhanced (Fig. 2a). The increased soil water availability under higher rates of biochar application could not offset the negative effect of other controlling factors such as nutrient availability. This is consistent with the finding of de Melo Carvalho et al. (2014) that the increment of soil moisture following biochar addition did not result in an increase in rice yield, most likely because rainfall during the critical period for rice production exceeded 650 mm in this field study.
To offset the negative effect of biochar on plant growth and crop productivity, both saturating biochar in a nutrient-rich solution and applying in combined with fertilizer have been suggested in previous studies (Steiner et al. 2007; van Zwieten et al. 2010; Reverchon et al. 2014, 2015). A previous study showed that saturating biochar in a nutrient solution prior to application had a positive effect on plant growth, since the adsorption of soil nutrients before application could reduce its competition with plants for soil nutrients. For example, Reverchon et al. (2014) treated two types of biochar prior to application with Hoagland’s solution, which is a widely used fertilizer in the horticultural industry to improve soil fertility. They found that the application of nutrient-enriched biochar had a positive effect on grain dry weight, especially for pine biochar that was the same as used in the present study. The biomass of both C3 and C4 plants with the nutrient-enriched biochar application were significantly higher than those with untreated biochar application (Fig. 2), indicating that nutrient treatment of biochar before application could enhance the growth of C3 and C4 plants by offsetting the competition between biochar and plants for nutrients and thus enhanced soil nutrient availability. Although an increase in plant biomass following the rate of nutrient-enriched biochar application was not observed in this study, our results still demonstrated that soil nutrient availability rather than water availability controlled the growth of both C3 and C4 plants under biochar additions.
4.3 Labile soil C and N pools
Labile pool of soil organic matter is an important fraction that is available for plant and microbial use. HWEOC and HWETN have been suggested as useful indicators of soil fertility and C and N availability, since hot water extraction could not only extract soluble C and N pools, but also components of microbial biomass (Sparling et al. 1998). It has been reported that HWEOC was highly correlated with microbial biomass C and soil respired CO2 and HWETN with microbial biomass N (Sparling et al. 1998; Chen and Xu 2005). Ghani et al. (2003) also suggested that HWEOC could be used as an integrated indicator of soil quality for management practices, since they found that HWEOC was one of the most sensitive and consistent indicators examined at 52 different sites, and HWEOC was positively and significantly correlated with soil microbial biomass C, mineralizable N, and extractable total carbohydrates. Results of this study showed that HWEOC linearly increased with the addition rates of untreated biochar in the soils with both C3 and C4 plants (Fig. 1a, b), indicating that the labile soil C and N pools increased as a result of C and N input from biochar addition. However, concentrations of HWEOC were not enhanced following the rates of nutrient-enriched biochar addition (Fig. 1a, b), showing that increased nutrient input from the nutrient-enriched biochar also increased soil microbial activity with more soil labile organic C (i.e., HWEOC) being utilized. For the soils with the C4 plants, HWETN of the soils with untreated biochar addition was significantly higher than that of the soils with the nutrient-enriched biochar addition, and this might be due to the addition of nutrients from the nutrient-enriched biochar resulting in enhanced uptake of N by plants from the soil.
Our research findings of significant relationships between the aboveground and root δ 13C, and between the root and aboveground δ 15N (Fig. 3) have highlighted that the plant C balance and cycle are regulated by the above ground plant photosynthesis, and that the plant N uptake and cycle are mediated by soil N pools and transformations as well as plant root N uptake. These are consistent with those reported by Ibell et al. 2010, 2013a, 2013b and Tutua et al. (2014).
5 Conclusions
Results from this greenhouse pot experimentation and subsequent analysis have demonstrated that the soil water availability was significantly increased following the application of untreated biochar, while it was accompanied by a decrease in plant biomass. However, the application of the nutrient-enriched biochar clearly offset the negative effect of the untreated biochar application on the plant growth and significantly improved plant biomass. It is concluded that it is the biochar nutrient availability, rather than its water holding capacity, governed the growth of C3 and C4 plants under the pot trial condition with effective water supply. Further study is warranted in order to examine the role of the different types of biochar at different nutrient and water levels for plant growth.
References
Anderson CR, Condron LM, Clough TJ, Fiers M, Stewart A, Hill RA, Sherlock RR (2011) Biochar induced soil microbial community change: implications for biogeochemical cycling of carbon, nitrogen and phosphorus. Pedobiologia 54:309–320
Atkinson C, Fitzgerald J, Hipps N (2010) Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337:1–18
Baronti S, Vaccari FP, Miglietta F, Calzolari C, Lugato E, Orlandini S, Pini R, Zulian C, Genesio L (2014) Impact of biochar application on plant water relations in Vitis vinifera (L.). Europ J Agron 53:38–44
Basso AS, Miguez FE, Laird DA, Horton R, Westgate M (2013) Assessing potential of biochar for increasing water-holding capacity of sandy soils. Glob Change Biol Bioenergy 5:132–143
Chan KY, Xu ZH (2009) Biochar: nutrient properties and their enhancement. In: Lehmann J, Joseph S (eds) Biochar for environmental management, science and technology. Earthscan, London
Chen CR, Xu ZH (2005) Soil carbon and nitrogen pools and microbial properties in a 6-year-old slash pine plantation of subtropical Australia: impacts of harvest residue management. For Ecol Manage 206:237–247
Chen CR, Xu ZH, Mathers NJ (2004) Soil carbon pools in adjacent natural and plantation forests of subtropical Australia. Soil Sci Soc Am J 68:282–291
Cheng C-H, Lehmann J, Thies JE, Burton SD (2008) Stability of black carbon in soils across a climatic gradient. J Geophys Res 113:G02027
Clough TJ, Condron LM (2010) Biochar and the nitrogen cycle: introduction. J Environ Qual 39:1218–1223
de Melo Carvalho MT, de Holanda Nunes Maia A, Madari BE, Bastiaans L, van Oort PAJ, Heinemann AB, Soler da Silva MA, Petter FA, Marimon BH Jr, Meinke H (2014) Biochar increases plant-available water in a sandy loam soil under an aerobic rice crop system. Solid Earth 5:939–952
Deenik JL, McClellan T, Uehara G, Antal MJ, Campbell S (2010) Charcoal volatile matter content influences plant growth and soil nitrogen transformations. Soil Sci Soc Am J 74:1259–1270
Güereña D, Lehmann J, Hanley K, Enders A, Hyland C, Riha S (2013) Nitrogen dynamics following field application of biochar in a temperate North American maize-based production system. Plant Soil 365:239–254
Gaskin JW, Speir RA, Harris K, Das KC, Lee RD, Morris LA, Fisher DS (2010) Effect of peanut hull and pine chip biochar on soil nutrients, corn nutrient status, and yield. Agron J 102:623–633
Ghani A, Dexter M, Perrott KW (2003) Hot-water extractable carbon in soils: a sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol Biochem 35:1231–1243
Glaser B, Lehmann J, Zech W (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—a review. Biol Fertil Soils 35:219–130
Hale SE, Lehmann J, Rutherford D, Zimmerman AR, Bachmann RT, Shitumbanuma V, O’Toole A, Sundqvist KL, Arp HPH, Cornelissen G (2012) Quantifying the total and bioavailable polycyclic aromatic hydrocarbons and dioxins in biochars. Environ Sci Technol 46:2830–2838
Ibell P, Xu ZH, Blumfield TJ (2010) Effects of weed control and fertilization on soil carbon and nutrient pools in an exotic pine plantation of subtropical Australia. J Soils Sediments 10:1027–1038
Ibell P, Xu ZH, Blumfield TJ (2013a) The influence of weed control on foliar δ15N, δ13C and tree growth in an 8-year-old exotic pine plantation of subtropical Australia. Plant Soil 369:199–217
Ibell PT, Xu ZH, Blake T, Blumfield TJ (2013b) Effects of weed control and fertilisation at early establishment on tree nitrogen and water use in an exotic F1 hybrid pine plantation of subtropical Australia. J Soils Sediments 13:1538–1552
Jeffery S, Verheijen FGA, van der Velde M, Bastos AC (2011) A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ 144:175–187
Jiang YM, Chen CR, Xu ZH, Liu YQ (2010) Soil soluble organic carbon and nitrogen pools under mono- and mixed-species forest ecosystems in subtropical China. J Soils Sediments 10:1071–1081
Kammann C, Linsel S, Gößling J, Koyro H-W (2011) Influence of biochar on drought tolerance of Chenopodium quinoa Willd and on soil-plant relations. Plant Soil 345:195–210
Laird D, Fleming P, Wang B, Horton R, Karlen D (2010) Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma 158:436–442
Lehmann J, Joseph S (2009) Biochar for environmental management: an introduction. In: Lehmann J, Joseph S (eds) Biochar for environmental management, science and technology. Earthscan, London
Lentz RD, Ippolito JA (2012) Biochar and manure affect calcareous soil and corn silage nutrient concentrations and uptake. J Environ Qual 41:1033–1043
Liang B, Lehmann J, Solomon D, Kinyangi J, Grossman J, O'Neill B, Skjemstad JO, Thies J, Luizão FJ, Petersen J, Neves EG (2006) Black carbon increases cation exchange capacity in soils. Soil Sci Soc Am J 70:1719–1730
Major J, Rondon M, Molina D, Riha S, Lehmann J (2010) Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant Soil 333:117–128
Morison JI, Gifford RM (1983) Stomatal sensitivity to carbon dioxide and humidity: a comparison of two C3 and two C4 grass species. Plant Physiol 71:789–796
Novak JM, Busscher WJ, Watts DW, Amonette JE, Ippolito JI, Lima IM, Gaskin J, Das KC, Steiner C, Ahmedna M, Rehrah D, Schomberg H (2012) Biochars impact on soil moisture storage in an Ultisol and two Aridisols. Soil Sci 177:310–320
Novak JM, Lima I, Xing B, Gaskin JW, Steiner C, Das KC, Ahmedna M, Rehrah D, Watts DW, Busscher WJ (2009) Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann Environ Sci 3:195–206
Rajkovich S, Enders A, Hanley K, Hyland C, Zimmerman A, Lehmann J (2012) Corn growth and nitrogen nutrition after additions of biochars with varying properties to a temperate soil. Biol Fertil Soils 48:271–284
Rawson HM, Begg JE, Woodward RG (1977) The effect of atmospheric humidity on photosynthesis, transpiration and water use efficiency of leaves of several plant species. Planta 134:5–10
Reverchon F, Flicker RC, Yang H, Yan GJ, Xu ZH, Chen CR, Hosseini Bai S, Zhang DK (2014) Changes in δ 15N in a soil-plant system under different biochar feedstocks and application rates. Biol Fertil Soils 50:275–283
Reverchon F, Yang H, Ho TY, Yan GJ, Wang J, Xu ZH, Chen CR, Zhang DK (2015) A preliminary assessment of the potential of using an acacia-biochar system for spent mine site rehabilitation. Environ Sci Pollut Res 22:2138–2144
Rogovska N, Laird D, Cruse R, Fleming P, Parkin T, Meek D (2011) Impact of biochar on manure carbon stabilization and greenhouse gas emissions. Soil Sci Soc Am J 75:871–879
Sparling G, Vojvodic-Vukovic M, Schipper LA (1998) Hot-water-soluble C as a simple measure of labile soil organic matter: the relationship with microbial biomass C. Soil Biol Biochem 30:1469–1472
Steiner C, Teixeira WG, Lehmann J, Nehls T, de Macedo JLV, Blum WEH, Zech W (2007) Long term effects of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered Central Amazonian upland soil. Plant Soil 291:275–290
Taghizadeh-Toosi A, Clough T, Sherlock R, Condron L (2012) Biochar adsorbed ammonia is bioavailable. Plant Soil 350:57–69
Tutua SS, Xu ZH, Blumfield TJ (2014) Foliar and litter needle carbon and oxygen isotope compositions relate to tree growth of an exotic pine plantation under different residue management regimes in subtropical Australia. Plant Soil 375:189–204
van Zwieten L, Kimber S, Morris S, Chan KY, Downie A, Rust J, Joseph S, Cowie A (2010) Effects of biochar from slow pyrolysis of paper mill waste on agronomic performance and soil fertility. Plant Soil 327:235–246
Woolf D, Amonette JE, Street-Perrott FA, Lehmann J, Joseph S (2010) Sustainable biochar to mitigate global climate change. Nat Commun 1:56
Xu CY, Hosseini-Bai S, Hao YB, Rachaputi RN, Wang HL, Xu ZH, Wallace H (2015) Effect of biochar amendment on yield and photosynthesis of peanut on two types of soils. Environ Sci Pollut Res 22:6112–6125
Xu ZH, Prasolova N, Lundkvist K, Beadle C, Leaman T (2003) Genetic variation in branchlet carbon and nitrogen isotope composition and nutrient concentration of 11-year-old hoop pine families in relation to tree growth in subtropical Australia. For Ecol Manage 186:359–371
Xu ZH, Saffigna PG, Farquhar GD, Simpson JA, Haines RJ, Walker S, Osborne DO, Guinto D (2000) Carbon isotope discrimination and oxygen isotope composition in clones of the F1 hybrid between slash pine and Caribbean pine in relation to tree growth, water-use efficiency and foliar nutrient concentration. Tree Physiol 20:1209–1217
Zhang A, Liu Y, Pan G, Hussain Q, Li L, Zheng J, Zhang X (2012) Effect of biochar amendment on maize yield and greenhouse gas emissions from a soil organic carbon poor calcareous loamy soil from Central China Plain. Plant Soil 351:263–275
Zhao X, Wang J, Wang S, Xing G (2014) Successive straw biochar application as a strategy to sequester carbon and improve fertility: a pot experiment with two rice/wheat rotations in paddy soil. Plant Soil 378:279–294
Acknowledgments
This research was funded by the Australian Research Council under the ARC Linkage Project Scheme (ARC Linkage Project LP100200135), with BHP Billiton Iron Ore Pty Ltd, ANSAC Pty Ltd and ENN as the partner organizations. The authors are grateful to Dr Kai Zeng, Jianbo Wang, Geoffrey Lambert, Rene Diocares, and Carolyn Polson for their technical assistance. We also thank Dr Chengyuan Xu for his valuable comments on this manuscript. Yuzhe Wang also acknowledges Professor Qixing Zhou as his PhD supervisor at his home institution, Nankai University and the scholarship provided by the China Scholarship Council (CSC) to visit Australia as a visiting PhD student.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Jianming Xu
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 391 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Zhang, L., Yang, H. et al. Biochar nutrient availability rather than its water holding capacity governs the growth of both C3 and C4 plants. J Soils Sediments 16, 801–810 (2016). https://doi.org/10.1007/s11368-016-1357-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-016-1357-x