Abstract
Investment in reproduction is predicted to accelerate ageing, but the link between reproductive investment and lifespan can be sex- and context-specific. In mammals, female reproductive costs are linked to pregnancy and lactation, but in males substantial reproductive allocation is required for a range of pre- and post-copulatory reproductive traits. Such traits include male-specific increased body size, olfactory signalling and territory defence—traits often expressed under androgen-dependent control. In this experimental study, we explored how reproduction influences lifespan in male mice, contrasting this to the established lifespan costs of reproduction in females. In a 2 × 2 factorial design, we gave either castrated or intact males (factor 1) access to a female or a male cage-mate across their entire life (factor 2). Neither castration nor access to females influenced median lifespan in male mice, but maximal lifespan was increased by either castration or reproduction when compared to intact males housed in male groups (standard male housing conditions). In females, mating significantly reduced lifespan, and while both sexes had similar lifespans in non-reproductive environments, males had a much longer lifespan when allowed mating. This data highlights the sex-specific nature of social environments and reproduction on lifespan, and the role of these conditions in promoting sexual dimorphism in ageing.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trade-offs between reproduction and longevity underpin some important evolutionary theories for the evolution of lifespan and the presence and variation of ageing [1, 2]. Negative relationships between reproductive output and lifespan are observable across species, with long-lived species having later ages at sexual maturity, and producing fewer offspring at a slower temporal rate [3, 4].
Trade-offs between reproduction and lifespan are also thought to occur on an intra-individual scale [2, 5]. In situations where an organism increases or decreases its level of reproductive allocation, this is expected to have consequences for investment in other aspects of life history, including subsequent survival. There are still unresolved issues surrounding the presence and causes of intra-individual trade-offs. In particular, the physiological mechanisms that underlie these trade-offs are poorly understood [6, 7]. Exactlywhich aspects of reproduction, or traits relating to reproduction, contribute to these trade-offs is also debated [8]. Work in wild animals has shown negative correlations between investment in early life reproductive traits (e.g. an early age at first reproduction, high early life offspring production) and later life survival [9]. In laboratory animals, experimentally elevating reproduction can also lead to increased mortality [5]. In mammals, manipulative experiments testing for trade-offs between reproduction and survival have predominantly been conducted in females, where manipulating the ability to mate has been found to shorten adult lifespan in some instances [10, 11].
In male mammals, reproductive investment encompasses production of sperm and mating, but also allocation to sexually selected traits that increase a male’s ability to access and/or attract mates. Many of these traits are under androgen-dependent control, and include the expression of aggressive behaviour, male-specific increases in body size, production of olfactory signals and territory defence [8]. The acts of mating and sperm production, therefore, while predicted to have evolutionarily relevant costs [12], are sometimes thought of as trivial relative to the costs of traits and behaviours that are required for access to mates. Recent work in Drosophila melanogaster has suggested that being allowed mating might be beneficial for male survival [13], the opposite response to the well-established costs of mating that occur in females of the same species [5]. An early study in rats also reported a 15% longer lifespan in males allowed mating compared to controls [14], suggesting that mating may also have benefits for male mammals in some contexts.
Castration of companion and domestic animals is linked to longer lifespans [15,16,17], and human castration has been associated with enhanced survival in Korean eunuchs [18] and institutionalised mentally ill men [19] (although not castrato opera singers [20]). Controlled studies in rodents were conducted prior to 1970: in rats, castration was reported to extend mean lifespan in one study [14], but not significantly in another, although maximum lifespan appears improved [21]. These experimental studies suggest that castration, in addition to mating [14], could directly influence male survival, although the median lifespan of control rats in each study was less than 2 years of age, reflecting the suboptimal housing conditions for rodent lifespan assessments at the time period [10]. Studies of castration have also been conducted in male only housing environments, rather than making comparisons in an environment where males are allowed to reproduce, and the costs and benefits of female exposure would be expected to operate. Clearly, scope exists for studies of the effects of castration, mating and reproduction on male longevity, and how such effects differ from those observed in females.
In this study, we tested whether castration influences the longevity of C57BL/6J mice, the most commonly used mouse model in ageing research. To further test the effects of reproduction on male lifespan, we tested whether giving males access to mates influences lifespan, and whether any effects of castration on lifespan were observable when males were housed in environments containing a specific sex. We also compared the relative effects of reproduction on lifespan in females, to compare the strength of any effects of reproduction on ageing in the two sexes.
Methods
Animals
This research was approved by the University of New South Wales (UNSW) Animal Care and Ethics Committee, approval 15/70B. Mice were of the C57BL/6Jausb strain, which are maintained as a stock of C57BL/6J mice at the Australian BioResource Center (ABR), with regular imports of the same strain of animals from the Jackson Laboratory (Bar Harbor, Maine, USA). C57BL/6J is a strain in which the consequences of castration on male mating behaviour and androgen-dependent sexual signalling are well established, and castration is known to rapidly reduce male mating behaviour [22, 23]. Experimental mice were purchased from ABR (ABR, Moss Vale, NSW, Australia) and shipped to UNSW. Before being shipped, 7-week-old male mice were either castrated or sham castrated, with the testes fully removed (castrated), or exposed and then replaced (sham castrated, intact). For all surgeries, animals were anaesthetised with a ketamine and medetomidine mix, then administered ketoprofen as analgesia. An incision in the abdomen was made and in castrated males both testes were also removed during the procedure. Intact (sham) males underwent the same surgical incision with the testes revealed but not removed. Animals were then sent to UNSW and allowed 1 week to habituate. Mice were maintained at 22 ± 2 °C on a 12:12-h light/dark cycle, with the dark period starting at 9 am to allow manipulations to be conducted in the dark period (under dim red light). Animals were housed in HD polyethylene cages (Mascot Wire Works 18 series, dimensions: 51.4 × 21.3 × 13.0 cm) with a raised wire lid. Cages contained corn cob substrate (Bed-o’Cobs 1/8″), nesting material (shredded paper and tissue), a cardboard shelter (toilet paper roll), a climbing ring and sunflower seeds scattered throughout the bedding. Animals were fed the mouse and rat maintenance pellets purchased from Gordon’s Speciality Stock Feeds (Yanderra, New South Wales, Australia). Animals were maintained in conventional housing conditions with pathogen status not monitored.
Experimental procedures
At 8 weeks of age, male mice were allocated to either a female housing (reproductive) environment (housed with two females—three mice per cage) or a male-male environment where males remained with two siblings (three mice per cage) of the same surgery type. Whether males were allocated to male or female housing was randomly determined, but with a bias to more males being housed in the male-male environment. A total of 27 males per surgical status were allocated to female housing, while 32 intact and 36 castrated males were housed in a male-male environment. A sample size of 30 per group, assuming a mean lifespan of 912 ± 143 SD days, provides 80% power to detect a 15% change in lifespan [24], which is approximately the degree of lifespan extension observed with mating and castration in a previous study in rats [14]. Thus, our study was designed to detect changes in lifespan of a similar magnitude within one environment, in addition to providing potential to detect smaller changes in lifespan if they occur similarly in both environments. We note that the power analysis and study design conducted here, like most mouse lifespan studies, did not account for any correction for multiple comparison in the statistical procedures, and therefore, we do not apply any such corrections in this study. Our results should be considered in light of this, where post hoc group comparisons were made.
Males in the reproductive environment were housed with two 4-week-old females. Males were allowed breeding with these females continuously, with offspring removed from the cage at 21 days old. When these female housing partners reached 300 days of age, they were removed and two new 28-day-old females were added. This second set of females remained with males until any of the animals died or were considered moribund. All males successfully reproduced, with males siring a mean of 38.3 litters across life (min: 16, max: 47), with a mean litter size of 4.7. The average age at which males stopped siring litters was 696 days (min: 278, max: 867). For the assessment of the effects of reproduction on female lifespan, females were randomly housed in either reproductive or non-reproductive environments. Females in the reproductive environment were housed with one female sibling and one unrelated male (8 weeks old at time of pairing) from 4 weeks of age. At 300 days, males were removed and a new 8-week-old male was added to eachsibling pair, and this male remained with the females until death. All females successfully reproduced, producing a mean of 11.5 litters (min: 5, max: 16), with a mean litter size of 4.8. The average age that females stopped reproducing was 396 days (min: 237, max: 536). Females in the non-reproductive environments were housed with one sibling and one unrelated female from 4 weeks to 300 days of age, with the unrelated female removed from the cage at 300 days of age.
Mice remained in the described conditions until death, or until they were considered to be so severely moribund that they were unlikely to survive an additional 48 h [25]. Severe moribundity was indicated by one or more of the following clinical signs: rapid weight loss; lack of responsiveness to manual stimulation; trembling/hunched or immobile posture; laboured and irregular breathing; severe abdominal enlargement (unrelated to pregnancy); a severely ulcerated tumour. The age at which a moribund mouse was euthanised was used as an estimate of its lifespan, and mice found dead were also noted at each daily inspection. Mice were not treated with any medication across the course of the study.
Grip strength
When animals were between 675 and 800 days old, we tested the grip strength of male mice using a hang wire test [26]. The mice were placed on a wire cage lid that was then inverted, and the latency to release the wire and fall was recorded. Each mouse was tested three times with a 20-min rest between each. If animals maintained grip for 60 s, this was used as a maximum value. The maximum value recorded in the three trials was used in the analysis.
Statistics
Overall differences in survival between factors were tested using the Cox regression survival analysis (SPSS, version 25), including social environment and surgery status as separate fixed factors. Follow-up survival analysis between two specific groups was conducted using a log-rank test (Prism, version 8). We planned to censor any animals that died early in life due to fighting; however, these deaths were not detected in males. One female died in the reproductive group at 399 days of age because of a birthing difficulty, this animal was censored in the lifespan analysis. To assess differences in maximum lifespan, we first used a 3-dimensional contingency table analysis in R [27] to test whether there was an overall effect of castration across both environments, using 90% experiment-wide mortality (e.g. from across mice of both surgery and social environments) as the cutoff for maximal survival. To follow up on differences in maximal survival betweentwo specific groups, we used Fisher’s exact test (OASIS: online application [28]), using the 90% mortality from the two groups being compared as the cutoff for maximal survival in each separate analysis. Body mass data was analysed using SPSS version 26, using a general linear mixed model for repeated measures to assess changes in body weight between ages 3 and 24 months, in relation to treatment. We fitted individual as a subject term, and the repeat measures with a 1st-order autoregression covariance structure.
Differences in grip strength were tested using a nonparametric aligned rank 2-way ANOVA [29], including surgery and social environment as treatments, following up with a Mann-Whitney U test to compare differences betweentwo specific groups. The relationship between hang duration and either body weight or age was determined with a Spearman rank correlation. For the relationship between hand duration and body weight, we also calculated the unstandardised residuals from the relationship between hand duration and body weight, using a linear regression, and compared the differences in these residuals between the two groups in question with a Mann-Whitney U test.
Results
Castration extends maximal but not median lifespan in male mice
The median lifespan of sham surgery males housed in male-male groups (e.g. standard housing conditions) was 952 days. We used the Cox regression survival analysis to test whether either castration or sham surgery (intact) status or social environment (male or female housing) had a significant overall effect on survival as factors in the two-by-two design. Neither castration status (Wald = 0.229, df = 1, P = 0.632) nor social environment (Wald = 0.022, df = 1, P = 0.883) had significant effects on overall survival (and there was no interaction between the two factors (Wald = 0.400, df = 1, P = 0.527)).The overall survival distribution of mice castrated at 8 weeks of age and housed in male-male groups also did not differ significantly from that of sham surgery males in the same social environment when considering this specific two groups contrast (log-rank test: P = 0.17).
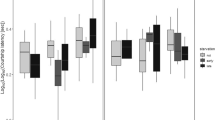
Analysis of maximal lifespan, defined as survival at 90% of total group mortality, showed a marginal overall effect of castration across both environments (Cochran-Mantel-Haenszel test χ2[1] = 0.059). Examining the effects of castration in each social environment separately, this marginal effect was due to a significant increase in late life survival in castrated males compared to sham surgery intact males when kept with other males (Fisher’s exact test: P = 0.026; Fig. 1; Table 1), but there was no change in maximal lifespan in castrated animalswhen kept with females (Fisher’s exact test: P = 0.351; Fig. 1; Table 1). Indeed, intact males housed with females also showed an increase in maximal survival when comparing this group to intact males housed with other males in a Fisher exact test (Fisher’s exact P = 0.039; Fig. 1; Table 1). Intact males housed with other males therefore had the shortest maximal lifespan, and this was extended by either castrating males or allowing them to mate (Table 1).
Housing with females reduces body weight across life and improves grip strength during ageing
There was a significant interaction between social housing condition and surgery treatment on weight (Table 2; Fig. 2a), largely due to the fact that intact males housed with other males were heavier than those of the other three treatments earlier in life. For example, intact males housed with males were significantly heavier than intact males housed with females at the 3-, 4-, 6-, and 12-month time points, but not at later time points (P < 0.05, Student’s t test). There was also a significant interaction between age and surgery due to the fact that castrated males were lighter at 6 months but after this point gained more weight than intact males. In the light of these two interactions, the significant main effect for castration in the model outcomes (Table 2) should be interpreted with caution. The significant linear and quadratic effects of age are linked to the occurrence of weight peaking at 18 months (Fig. 2a).
Effects of castration and reproduction on body weight and grip strength in male mice. a Body weight at 2, 3, 4, 6, 12, 18, and 24 months in intact and castrated individuals in male (non-reproductive) and female (reproductive environments). b Ability of mice to hang on an inverted wire lid. Mice were recorded for their ability to hang on an inverted lid three times, and each dot represents the mean value for a different individual. Error bars represent standard error of the mean. P value calculated from a Student test. In a, N = 36 for castrated males with males at the start of the study, reduced to N = 30 by the 24-month time point. N = 32 for intact males with other males at the start of the study, reduced to 29 by the 24-month time point. N = 27 for castrated males with females, reduced to 22 by the 24-month time point. N = 27 for intact males with females, reduced to 24 by the 24-month time point. In b, N = 28 for intact and castrated males housed with males, N = 24 for intact males with females, and N = 22 for castrated males with females
To test whether an aspect of physical function was influenced by castration or housing with females, we also assessed the grip strength of males when they were between 675 and 800 of age using the wire grip hang strength test [26]. Castration (F1,99 = 2.07, P = 0.154) did not influence hang duration across both environments, nor did female housing (F1,99 = 1.98, P = 0.162), but there was a significant interaction between these two factors (F1,99 = 5.31, P = 0.023). The interaction between castration status and social environment was mainly caused by intact males that were housed with females showing improved hang duration when compared to intact males housed with other males (Fig. 2b). Castration had no significant effect on grip strength in either social environment (P = 0.59 in the male environment, P = 0.075 in the female environment). Age had no significant effect on hang duration (n = 103, Rs = − 0.12, P = 0.025). Body weight was significantly albeit weakly negatively related to hang duration (n = 103, Rs = − 0.24, P = 0.014; Fig. S1A), but males housed with females had a greater hang duration than non-reproductive intact males when the relationship between body weight and grip strength is accounted for (Fig. S1B).
Reproduction has sex-specific effects on lifespan in mice
To understand whether the effects of reproduction on lifespan are sex-specific in mice, we also assessed the lifespan of females in either mating or non-mating environments. In contrast to the effects of reproduction in males, females allowed mating across life had significantly decreased survival as assessed by the log-rank test (Fig. 3; P = 0.034) when compared to non-reproductive females, although showed no significant change in maximal lifespan (P = 0.21). Comparing the effects of reproduction for the relative lifespans of males and females, we observe that there is little difference in lifespan between the sexes in non-reproductive environments, but when allowed mating, a robust difference in lifespan between the sexes is observed (Fig. 3).
Reproduction has sex-specific effects on lifespan in mice. Lifespan of male and female mice housed in either reproductive (with 2 members of the opposite sex) or non-reproductive (with 2 members of the same sex) conditions. Each dot represents the time point when an individual mouse died. N = 27 for intact males with females, N = 32 for intact males with males, N = 30 for females with females, and N = 24 for females with males (including one censored)
Discussion
In this study, we find no evidence that median lifespan is affected by castration or access to females in male mice, but maximal lifespan (90% survival) is significantly increased by both treatments when compared to intact males housed with other males. For the castration comparison in male-male housing conditions, approximately 20% of the castrated males were still alive when all of the intact individuals had died, suggesting castration may extend lifespan to late ages. The presence of male testes and associated hormone production may therefore constrain survival of males, consistent with evolutionary theories of ageing and the predicted lifespan trade-offs with reproduction. However, housing males with two females and allowing reproduction across life also led to a small increase in maximal lifespan, suggesting that mating may also provide survival benefits for male mice.
The lack of change in median survival with castration differs to results from a previous study in rats, where median and maximum lifespan was increased [21]. It is notable that median lifespan of all groups of mice in our study is approximately 1000 days, representing a relatively long lifespan particularly compared to the previous studies that were conducted prior to the 1970s when housing conditions for laboratory rodents were considered suboptimal. One limitation of our work, like previous studies on this question, was that our colony did not undergo quarterly stereological testing and so were not housed in specific pathogen-free (SPF) conditions. Thus, while the long lifespans of the mice in our study would indicate that animals were not dying from specific infectious diseases early in life, and we detected no obvious signs of infection or the atopic dermatitis that can present on this mouse strain, we cannot exclude the possibility of specific pathogens normally screened for being present in our colony. There is increasing interest in the role of gonadal hormones and social conditions in control of lifespans, and future studies conducted in SPF conditions should be of priority. Additionally, further understanding of the robustness of these results would be gained from additional replication in a different strain of mouse, in particular a genetically heterogeneous mouse model, since inbred laboratory strains of mice can show lifespans that are disproportionally influenced by one or a few specific causes of death (e.g. specific cancers).
Effects of castration on lifespan have been documented previously in different contexts, although results from controlled laboratory studies have been limited [8]. Most notably, a historical study of human eunuchs suggested that castration in humans leads to an increased proportion of individuals surviving to be centenarians [18]. Our data is consistent with the idea that survival to very old ages could be increased with castration as a consequence of altered biology, rather than being attributable to socio-demographic factors. Other research conducted since the design of this study has highlighted that sex chromosome complement can also influence survival in mice [30]. In this study, mice with XX chromosomes showed improved survival compared to those with XY chromosomes when they had been genetically manipulated to develop either testes or ovaries on both chromosome backgrounds. Understanding the relative effects of sex hormones and sex chromosomes in control of the ageing process could therefore provide an insight into the causes of sex-specific ageing in mammals, which are seen widely across the mammalian kingdom [31].
Previous research has shown that castration may also provide additional health benefits in old rodents, increasing balance capacity on a rotarod [32] and maintenance of skeletal muscle mass and fibre size at old ages [33]. We did not detect an effect of castration on grip strength in this study, but we highlight that this one assessment of physical function must be interpreted with caution, and future studies warrant a detailed investigation of healthspan using a variety of different approaches, in a dedicated cohort of mice assessed at differing ages. Such a longitudinal assessment may also help to reduce the variability that we observed when assessing animals at a single time point. Interestingly, allowing males to reproduce increased their hanging ability according to the hang wire test. From this single measure, it is not possible to determine whether this reflects an increase of strength during ageing, nor if it reflects changes in hanging endurance or motivation. However, this data highlights that future study of health parameters is warranted to understand how reproduction and mating could influence health, strength and frailty during ageing. Similarly, dedicated pathological assessment of causes of death would be beneficial, helping to elucidate what causes death in different social environments, at different life periods.
The physiological mechanisms that lead to the beneficial effects of castration on ageing are currently unknown, but evidence of anti-ageing effects, including in humans, warrants further research. Testosterone is an anabolic hormone, and reduced androgenic signalling can dampen signalling through additional growth pathways like the growth hormone insulin-like growth factor 1 (GH-IGF1) axis and mechanistic target of rapamycin complex 1(mTORC1) signalling [34]. Suppressed activity of these pathways with castration may contribute to altered lifespan, since both pathways directly influence ageing [35,36,37,38]. Further understanding of the links between androgens, GH-IGF1 and mTOR signalling may also provide an insight into the physiological causes for sexual dimorphism in ageing, given the sex-specific nature of both androgen production and effects of somatotrophic signalling on ageing [35, 39].
Our results also show that allowing males to reproduce can lead to a small but significant increase in maximal lifespan, without a change in median lifespan. The observation that lifespan is not reduced with reproduction in male mice, and instead maximal lifespan is slightly increased, could appear at odds with evolutionary theories for trade-offs between reproduction and lifespan. Indeed, in female mice, we observe that reproduction significantly reduces lifespan, an effect reported previously in laboratory and wild-derived rodents [10, 11]. In our study, males housed with other females were lighter than males housed in male-male environments, suggesting that there is a metabolic response to mating in male mice. However, these metabolic costs of mating are hypothesised to be minor in comparison to the energetic investment that is required to produce traits that aid males in attracting and accessing females, particularly when competing against other individuals of the same sex [40]. Thus, these broader aspects of reproductive investment in males, which are a consequence of sexual selection, may provide the constraint on male longevity.
It has recently been suggested that mating itself may be beneficial for male survival if it relieves life history costs that males experience as a consequence of a lack of access to mating and its associated reward responses, particularly if males perceive females in their environment but cannot access them [13, 41]. This contrasts to the direct costs that females are expected to experience from the act of mating, which leads to associated investment in parental care [5]. Indeed, in our study, allowing males and females to mate promoted a sexual dimorphism in lifespan, with females showing a reduction in lifespan in response to mating. We note that there are some minor methodological differences in the male and female housing conditions that must be considered when contrasting the relative effects of reproductive conditions in each sex: for example, in reproductive conditions, two females were housed together with a male, so males were exposed to two individuals of the opposite sex, while females were exposed to one member of the same sex and a male. These methods were designed to maximise the chance of detecting an effect of social treatment within each sex (e.g. provided males with two females gave double the opportunity to mate), while also being constrained by practical considerations, such as the high possibility that males would fight with each other if housed together in the presence of a female. Nonetheless, our data indicate that social conditions can strongly influence the extent of sex differences in lifespan.
Our previous study in females has shown that mating, even with sterilised males, is capable of reducing female lifespan [42], suggesting that these effects of social conditions may not be linked to reproduction itself, but interactions between individuals of the opposite sex. In relation to this, similar manipulative work would be required to understand the causes of lifespan changes in males in different social conditions. For example, it is possible that, instead of mating increasing late life survival in intact males, housing with other intact males may reduce survival over this period. To distinguish these effects would require manipulation of housing partners, in addition to males themselves, such that intact males are studied with other conspecific males, but who have been manipulated so they do not exert the same potential stressors on their housing partners (for example, through castration). Studies in invertebrates illustrate that a variety of factors occurring with social interactions can influence lifespan [43,44,45], leading to unique social stimuli that differentially influence the lifespan of each sex.
Most work in laboratory rodents is conducted on non-reproductive animals, typically housed with the same sex. Thus, consequences of social conditions could modify the extent or presence of additional sex-specific physiology, health parameters or treatment responses. This could include lifespan and responses to anti-ageing interventions, which are frequently sex-specific [46, 47], in addition to diseases that show sexually dimorphic incidence or severity. For example, susceptibility to obesity is usually more severe in male mice compared to females, but this is typically assessed when mice are housed in non-reproductive environments [48]. Given the spectrum of social environments that both humans and wild animals reside in, understanding and incorporating responses to social interactions under controlled laboratory settings may help to increase the relevance of laboratory work to a wider variety of natural conditions.
References
Kirkwood TBL, Austad SN. Why do we age? Nature. 2000;408:233–8.
Kirkwood TBL, Rose MR. Evolution of senescence: late survival sacrificed for reproduction. Philos Trans R Soc Lond Ser B Biol Sci. 1991;332:15–24.
Promislow DEL, Harvey PH. Living fast and dying young - a comparative-analysis of life-history variation among mammals. J Zool. 1990;220:417–37.
Stearns SC. The evolution of life histories. Oxford: Oxford University Press; 1992.
Barnes AI, Partridge L. Costing reproduction. Anim Behav. 2003;66:199–204.
Speakman JR, Garratt M. Oxidative stress as a cost of reproduction: beyond the simplistic trade-off model. Bioessays. 2014;36:93–106.
Braendle C, Heyland A, Flatt T. Integrating mechanistic and evolutionary analysis of life history variation. In: Mechanisms of life history evolution. The genetics and physiology of life history traits and trade-offs; 2011. p. 3–10.
Brooks RC, Garratt MG. Life history evolution, reproduction, and the origins of sex-dependent aging and longevity. Ann N Y Acad Sci. 2017;1389:92–107.
Lemaître J-F, Berger V, Bonenfant C, Douhard M, Gamelon M, Plard F, et al. Early-late life trade-offs and the evolution of ageing in the wild. Proc R Soc B Biol Sci. 2015;282:20150209.
Green EL. Biology of the laboratory mouse. New York: Dover Publication; 1966.
Flurkey K, Brandvain Y, Klebanov S, Austad SN, Miller RA, Yuan R, et al. PohnB6F1: a cross of wild and domestic mice that is a new model of extended female reproductive life span. J Gerontol Ser A Biol Sci Med Sci. 2007;62:1187–98.
Parker GA. Sperm competition and its evolutionary consequences in insects. Biol Rev Camb Philos Soc. 1970;45:525–67.
Gendron CM, Kuo T-H, Harvanek ZM, Chung BY, Yew JY, Dierick HA, et al. Drosophila life span and physiology are modulated by sexual perception and reward. Science. 2014;343:544–8.
Drori D, Folman Y. Environmental effects on longevity in the male rat: exercise, mating, castration and restricted feeding. Exp Gerontol. 1976;11:25–32.
Jewell PA. Survival and behaviour of castrated Soay sheep (Ovis aries) in a feral island population on Hirta, St. Kilda, Scotland. J Zool. 1997;243:623–36.
Hamilton JB. Relationship of castration, spaying, and sex to survival and duration of life in domestic cats. J Gerontol. 1965;20:96–104.
Hoffman JM, Creevy KE, Promislow DE. Reproductive capability is associated with lifespan and cause of death in companion dogs. PLoS One. 2013;8:e61082.
Min K-J, Lee C-K, Park H-N. The lifespan of Korean eunuchs. Curr Biol. 2012;22:R792–3.
Talbert GB, Hamilton JB. Duration of life in Lewis strain of rats after gonadectomy at birth and at older ages. J Gerontol. 1965;20:489–91.
Nieschlag E, Nieschlag S, Behre HM. Lifespan and testosterone. Nature. 1993;366:215.
Asdell SA, Doornenbal H, Joshi SR, Sperling GA. The effects of sex steroid hormones upon longevity in rats. J Reprod Fertil. 1967;14:113–20.
Clemens LG, Wee BE, Weaver DR, Roy EJ, Goldman BD, Rakerd B. Retention of masculine sexual behavior following castration in male B6D2F1 mice. Physiol Behav. 1988;42:69–76.
McGill TE, Manning A. Genotype and retention of the ejaculatory reflex in castrated male mice. Anim Behav. 1976;24:507–18.
Ladiges W, Van Remmen H, Strong R, Ikeno Y, Treuting P, Rabinovitch P, et al. Lifespan extension in genetically modified mice. Aging Cell. 2009;8:346–52.
Miller RA, Harrison DE, Astle CM, Floyd RA, Flurkey K, Hensley KL, et al. An Aging Interventions Testing Program: study design and interim report. Aging Cell. 2007;6:565–75.
Shoji H, Takao K, Hattori S, Miyakawa T. Age-related changes in behavior in C57BL/6J mice from young adulthood to middle age. Mol Brain. 2016;9:11.
Mangiafico SS. Summary and analysis of extension program evaluation. In: R, version 1.18.1. rcompanionorg/documents/RHandbookProgramEvaluation; 2016.
Yang JS, Nam HJ, Seo M, Han SK, Choi Y, Nam HG, et al. OASIS: online application for the survival analysis of lifespan assays performed in aging research. PLoS One. 2011;6:e23525.
Wobbrock JO, Findlater L, Gergle D, Higgins JJ. The aligned rank transform for nonparametric factorial analyses using only anova procedures. In: Proceedings of the SIGCHI conference on human factors in computing systems; 2011. p. 143–6. https://doi.org/10.1145/1978942.1978963.
Davis EJ, Lobach I, Dubal DB. Female XX sex chromosomes increase survival and extend lifespan in aging mice. Aging Cell. 2019;18:e12871.
Lemaître J-F, Ronget V, Tidière M, Allainé D, Berger V, Cohas A, et al. Sex differences in adult lifespan and aging rates of mortality across wild mammals. PNAS. 2020;117:8546–53.
Perez J, Burunat E, Arevalo R, Rodriguez M. Gonadal influences on behavioral deterioration with aging of male rats. Horm Behav. 1989;23:457–65.
Garratt M, Leander D, Pifer K, Bower B, Herrera JJ, Day SM, et al. 17-alpha estradiol ameliorates age-associated sarcopenia and improves late-life physical function in male mice but not in females or castrated males. Aging Cell. 2019;18:e12920.
Regan JC, Partridge L. Gender and longevity: why do men die earlier than women? Comparative and experimental evidence. Best Pract Res Clin Endocrinol Metab. 2013;27:467–79.
Garratt M, Nakagawa S, Simons MJP. Life-span extension with reduced somatotrophic signaling: moderation of aging effect by signal type, sex, and experimental cohort. J Gerontol A Biol Sci Med Sci. 2017;72:1620–6.
Garratt M, Nakagawa SN, Simons MJ. Comparative idiosyncrasies in life extension by reduced mTOR signalling and its distinctiveness from dietary restriction. Aging Cell. 2016;15:737–43.
Lamming DW, Ye L, Katajisto P, Goncalves MD, Saitoh M, Stevens DM, et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science. 2012;335:1638–43.
Coschigano KT, Holland AN, Riders ME, List EO, Flyvbjerg A, Kopchick JJ. Deletion, but not antagonism, of the mouse growth hormone receptor results in severely decreased body weights, insulin, and insulin-like growth factor I levels and increased life span. Endocrinology. 2003;144:3799–810.
Holzenberger M, Dupont J, Ducos B, Leneuve P, Geloen A, Even PC, et al. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature. 2003;421:182–7.
Bonduriansky R, Maklakov A, Zajitschek F, Brooks R. Sexual selection, sexual conflict and the evolution of ageing and life span. Funct Ecol. 2008;22:443–53.
Harvanek ZM, Lyu Y, Gendron CM, Johnson JC, Kondo S, Promislow DEL, et al. Perceptive costs of reproduction drive aging and physiology in male Drosophila. Nat Ecol Evol. 2017;1:0152.
Garratt M, Try H, Smiley KO, Grattan DR, Brooks RC. Mating in the absence of fertilization promotes a growth-reproduction versus lifespan trade-off in female mice. PNAS. 2020;117(27):15748–54.
Booth LN, Maures TJ, Yeo RW, Tantilert C, Brunet A. Self-sperm induce resistance to the detrimental effects of sexual encounters with males in hermaphroditic nematodes. eLife. 2019;8:e46418.
Booth LN, Shi C, Tantilert C, Yeo RW, Hebestreit K, Hollenhorst CN, et al. Males deploy multifaceted strategies and hijack longevity pathways to induce premature demise of the opposite sex. bioRxiv. 2020. https://doi.org/10.1101/2020.06.30.181008.
Maures TJ, Booth LN, Benayoun BA, Izrayelit Y, Schroeder FC, Brunet A. Males shorten the life span of C elegans hermaphrodites via secreted compounds. Science. 2014;343:541–4.
Garratt M. Why do sexes differ in lifespan extension? Sex-specific pathways of aging and underlying mechanisms for dimorphic responses. Nutr Health Aging. 2019;5:1–13.
Austad S, Bartke A. Sex differences in longevity and in responses to anti-aging interventions: a mini-review. Gerontology. 2015. https://doi.org/10.1159/000381472.
Stubbins R, Holcomb V, Hong J, Núñez N. Estrogen modulates abdominal adiposity and protects female mice from obesity and impaired glucose tolerance. Eur J Nutr. 2012;51:861–70.
Funding
This research was funded by an Australian Research Council Discovery Grant (DP150100676).
Author information
Authors and Affiliations
Contributions
This study was designed by Michael Garratt and Robert Brooks, and conducted by Heather Try. Michael Garratt wrote the manuscript with feedback from the other authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 150 kb)
About this article
Cite this article
Garratt, M., Try, H. & Brooks, R.C. Access to females and early life castration individually extend maximal but not median lifespan in male mice. GeroScience 43, 1437–1446 (2021). https://doi.org/10.1007/s11357-020-00308-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-020-00308-8