Abstract
Polycyclic aromatic hydrocarbons (PAHs) are widely existing organic pollutants in the environment, and their persistence in the environment makes us have to pay continuous attention to their health effects. However, since the American Heart Association updated its definition of hypertension in 2017, few studies have explored the relationship. This study aimed to investigate the relationship between PAH exposure and hypertension after the updated definition of hypertension and explore whether body mass index (BMI) moderates this relationship. A total of 6332 adult participants from the 2009–2016 National Health and Nutrition Examination Survey (NHANES) were examined. Multiple logistic regression and restricted cubic splines were used to analyze the association between urinary polycyclic aromatic hydrocarbon metabolites and hypertension, and the dose–response relationship. Weighted quantile sum (WQS) regression was applied to blood pressure to reveal multiple exposure effects and the relative weights of each PAH. The prevalence of hypertension in the study population was 48.52%. There was a positive dose–response relationship between high exposure to 1-hydroxynaphthalene, 2&3-hydroxyphenanthrene, and the risk of hypertension. Naphthalene metabolites accounted for the most significant proportion of systolic blood pressure, and phenanthrene metabolites accounted for the most significant proportion of diastolic blood pressure. Obese individuals with high PAH exposure were at greater risk for hypertension than individuals with low PAH exposure and normal BMI. Higher prevalence rate and stronger association of metabolites with outcomes were obtained in the general population of the USA under the new guideline. High levels of exposure to PAHs were positively associated with the risk of hypertension, and these effects were modified by BMI.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As a preventable risk factor for cardiovascular disease (CVD), hypertension affects over one billion people worldwide (Beaney, et al. 2019; WHO 2019). In 2017, about 10.4 million people died from hypertension and its complications (coronary heart disease, arteriosclerosis, chronic kidney disease, and others) (Stanaway, et al. 2018). Influencing factors of hypertension include modifiable factors such as obesity, environmental exposure, physical exercise, and unmodifiable factors such as age, gender, race, and genetics (Freire, et al. 2018). Hypertension has become an enormous public health problem because of its high prevalence and low control rate (Freire, et al. 2018; Qu, et al. 2019). The role of air pollution in hypertension has recently attracted increasing attention (Qin, et al. 2021; Yusuf, et al. 2020).
As a class of persistent organic pollutants widely ubiquitous in the environment, polycyclic aromatic hydrocarbons (PAHs) are mainly derived from the incomplete combustion of fossil fuels. PAHs and their metabolites have been detected in blood, urine, hair, and other biological samples (ATSDR 1995). Urinary PAHs (mono-hydroxylated polycyclic aromatic hydrocarbons, OH-PAH) are common contact biomarkers to reflect the exposure of individuals to PAHs (Huang, et al. 2019). Correlative studies have revealed that PAHs can jeopardize cardiovascular health in addition to causing cancer (Mallah, et al. 2021). The cohort study results showed that occupational exposure to PAHs positively correlates with ischemic heart disease (Friesen, et al. 2010). Another study found a link between exposure to PAHs and pre-hypertension in children (Trasande, et al. 2015). Animal and cell experiments suggest that PAHs adhering to particulate matter may be a key determinant of cardiovascular disease (Holme, et al. 2019). Due to their prevalence in the environment, high detection frequency in human urine, and adverse health effects, PAHs in the general population are receiving increasing attention (de Oliveira Galvão, et al. 2020; Zhang, et al. 2019).
As a risk factor for many chronic diseases, obesity is associated with various diseases, such as cancer, dyslipidemia, and cardiovascular diseases (Lauby-Secretan, et al. 2016; Vekic, et al. 2019). Several studies have reported the association between elevated blood pressure and weight gain (Iida, et al. 2019; Sundström, et al. 2020). According to National Health and Nutrition Examination Survey (NHANES) data, obese people have a significantly higher prevalence of hypertension than the general population (Foti, et al. 2022). Because PAHs are lipophilic, they are easily stored in adipose tissue until excreted through the normal bladder and gastrointestinal function. Therefore, PAHs may be more persistent in patients with higher body mass index (BMI) than those with lower BMI, potentially affecting the risk of hypertension.
Although previous studies have investigated the interaction between PAHs and BMI on diabetes and cardiovascular diseases, including hypertension, few investigations conducted into the general population’s dose–response relationships. Furthermore, PAHs are persistent in the environment (Pastor-Belda, et al. 2019). The recent data expect to find higher exposure doses in the general population. Moreover, when new guidelines applied to the general US population, hypertension prevalence increased from 32.0% (based on traditional guideline) to 45.4%, which, if implemented globally, could annually prevent approximately 610,000 CVD events and 334,000 deaths in the USA (Bundy, et al. 2018). Therefore, we wondered whether the relationship between PAHs and hypertension might change under these influence, as well as whether this relationship is independent of BMI.
Methods
Study population
The NHANES (National Health and Nutrition Examination Survey) is a nationally representative survey conducted by the National Institutes of Health of the Centers for Disease Control and Prevention. The survey collected comprehensive data on diet, nutritional status, and chronic diseases to assess the population’s health and nutritional status and exposure to various chemicals, including PAHs (CDC 2017). Survey data have been published every 2 years since 1999, with the most recent PAHs laboratory data available in the 2015–2016 cycle. In addition, sample weights are provided every 2 years.
This study used the survey data from 2009 to 2016, including 40,439 participants ≥ 18 years of age. Participants were included if they had adequate blood pressure, metabolites, and covariate information. Finally, our study enrolled 6332 participants.
Measure of urinary levels of PAH biomarkers
The urine sample was preprocessed using enzymatic hydrolysis, centrifugation, and transfer. All urinary PAH metabolite concentrations were analyzed using high-performance liquid chromatography (detailed testing method and measurement conditions are available from the following official website). The concentrations of metabolites were finally measured in the laboratory, including 1-hydroxynaphthalene, 2-hydroxynaphthalene, 2-hydroxyfluorene 3-hydroxyfluorene, 1-hydroxyphenanthrene, 2&3-hydroxyphenanthrene, and 1-hydroxypyrene. To explain the variation in urine sample volume, exposure variables were corrected for urinary creatinine in all analyses by dividing each of the PAHs (ng/L) by urinary creatinine (mg/dl) and multiplying by 0.01 to result in nanograms of PAHs per gram of creatinine (ng/g) (Alshaarawy, et al. 2014; Everett, et al. 2010; Xu, et al. 2013). Measurements less than LOD were replaced with LOD divided by the square root of 2.
Definition of hypertension
Blood pressure measurements were taken during the NHANES examination visits. After resting quietly in a seated position for 5 min, blood pressure was measured three times using a mercury sphygmomanometer by well-trained examiners. A fourth reading was measured if required. Then, the means of systolic blood pressure (SBP) and diastolic blood pressure (DBP) were calculated. According to the 2017 American College of Cardiology/American Heart Association (ACC/AHA) Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults (Whelton, et al. 2018), hypertension was defined as SBP ≥ 130 mmHg and (or) DBP ≥ 80 mmHg. At the same time, participants who answered “yes” to the question: “Are you now taking prescribed medicine for high BP?” were also defined as having hypertension.
Covariates
Covariates included gender, age, race, education level, family income level, body mass index, smoking, and alcohol use. The categories are as follows: gender (male, female), age (18–39, 40–59, ≥ 60), race (non-Hispanic White; non-Hispanic Black; normal individuals, Mexican American, and Other Hispanic; Other Race, including Multi-Racial), education level (< senior high school, senior high school, > senior high school), the household income to poverty ratio (< 1.3, 1.3–3.5, > 3.5) based on SNAP eligibility recommended by the website, body mass index (underweight: < 18.5, normal weight: 18.5–24.9, overweight: 25.0–29.9, obese: ≥ 30.0), smoking status (never: < 100 cigarettes in life, former: > 100 cigarettes in life and did not smoke at the time of the survey, current: > 100 cigarettes in life and smoked every day in the previous 30 days), and alcohol use (never: < 12 drinks in life, former: > 12 drinks in their life but none in the past year, current: > 12 drinks in the past year and drink alcohol every week).
Statistical analysis
Since there were no separate data for 2-hydroxyphenanthrene and 3-hydroxyphenanthrene exposure in 2013–2014, we combined them into one variable. The distribution of PAH metabolites was described by the geometric mean and standard error. They were then stratified by tertiles, considering the lowest exposure level as the reference group (tertile1) for subsequent analysis. Sample weights, stratification, and clustering design variables were incorporated into the analysis procedures to ensure the correct estimation of sampling error. More specifically, Subsample A weights for 2011–2012, 2013–2014, and 2015–2016, and subsample B weights for 2009–2010 were used in this analysis.
The chi-square test was used to compare the differences of classification variables between the two groups. Multiple logistic regression was used to analyze the correlation between urinary PAH metabolites and hypertension. The covariates having an impact on outcomes were included in the model. The restricted cubic spline (RCS) is a powerful tool to characterize this association between continuous exposure and outcomes (Desquilbet and Mariotti 2010). It is worth noting that the number of nodes has a more non-negligible effect relative to their locations on the dose–response correlation curve (Lusa and Ahlin 2020). To more accurately describe the dose–response relationship between PAHs and hypertension risk, we selected five RCS knots, including 5th, 25th, 50th, 75th, and 95th percentiles.
Weighted quantile sum (WQS) regression is a statistical model used for multivariate regression of high-dimensional data sets common to environmental exposure, epigenetic/genomic, and metabolomics studies. The model constructs a weighted index to estimate the mixed effect of all the predicted variables on the outcome, which can be used in regression models with covariates to test the association of the index with the dependent variable or outcome. In addition, the contribution of each predictor to the overall exponential effect can be assessed by the relative strength of the weight assigned to each variable by the model. The data was divided into 40% for training; 60% for validation and 100 bootstrap samples were assigned for parameter estimation. The cutoff value ττ by default is equal to the reciprocal of the number of elements in the mixture, according to Carrico’s proposed definition (Carrico, et al. 2015).
The SURVEYFREQ program performed statistical descriptions, and the SURVEYLOGISTIC program performed statistical modeling. WQS was implemented by the R packages “gWQS.” P < 0.05 for bilateral tests was considered statistically significant.
Results
Table 1 describes the basic characteristics of the study population. The prevalence of hypertension was 48.52% in 6,332 participants, whose ages ranged from 18 to 80 years old. The adults who were diagnosed with hypertension were more likely than those without hypertension to be in the older age group (43.11% vs. 11.44%, P < 0.0001), non-Hispanic white (69.25% vs. 64.62%, P < 0.0001), men (53.38% vs. 45.87%, P < 0.0001), cigarette smokers (smoking: 47.83% vs. 40.70%, P < 0.001), and higher BMI (47.14% vs. 27.77%, P < 0.0001). However, there was no significant difference in income and alcohol drinking between participants diagnosed with hypertension and those without hypertension.
Table 2 shows the exposure distribution for each quintile of PAHs corrected for urinary creatinine among the study participants. The greatest PAH exposure was observed for 2-hydroxynaphthalene with a geometric mean of 5051 ng/g and a standard deviation of 60 ng/g. The least PAH exposure was observed for 3-hydroxyfluorene of 104 ng/g (Std. deviation = 2). The most extensive exposure range occurred of 1-hydroxynaphthalene was 12,515,700 ng/g.
Table 3 explores the relationship between urinary PAH metabolites and hypertension using multiple logistic regression analyses. After adjusting for confounding factors except for BMI (Model 1), the results revealed that compared with the lowest level of exposure, the highest levels of exposure to 2&3-hydroxyphenanthrene (aOR: 1.47; 95%CI: 1.04–2.07) and tertiles2 (aOR: 1.29; 95%CI: 1.04–1.59) for 1-hydroxyphenanthrene were positively associated with the risk of hypertension. In contrast, 1-hydroxypyrene was negatively related to the risk of hypertension. When the regression model included BMI (Model 2), the highest levels of 1-hydroxynaphthalene (bOR: 1.38; 95%CI: 1.12–1.71) were also positively correlated with the risk of hypertension.
Figure 1 displays a clear relationship between urinary PAH metabolites and hypertension. After adjusting for significant covariates, the model showed a linear dose–response relationship between 1-hydroxynaphthalene, 2&3-hydroxyphenanthrene, and 1-hydroxypyrene and the risk of hypertension (P for overall < 0.05, P for nonlinear > 0.05). There was a negative linear relationship between 1-hydroxypyrene and the outcome.
Restricted square spline analysis for the association between PAH metabolites and hypertension, adjusted for age, gender, education level, race, BMI, smoking status, and other PAH metabolites. The knots placed at 5th, 25th, 50th, 75th, and 95th percentiles of log-transformed PAH metabolite distribution, with the reference values, were set at the 50th percentile. The solid lines and dashed lines represent the odds ratio and corresponding 95% confidence interval for logarithmic transformation
We applied the WQS model to blood pressure to further reveal the multiple exposure effect and the relative weight of each PAH on the outcome. Table 4 shows the WQS results, where only diastolic blood pressure positively correlates with PAHs. The influence of the WQS model on diastolic blood pressure was statistically significant (P < 0.05) (β = 0.192; 95%CI: 0.026–0.357). However, the results showed no significant statistical significance in establishing the WQS model of systolic blood pressure.
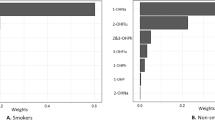
In addition, the WQS model can specify a weight table to measure the importance of each metabolite. For diastolic blood pressure, PAHs’ most prominent weight component in the WQS model was 1-hydroxyphenanthrene (weight = 0.69). The most heavily weighted component for systolic blood pressure was 2-hydroxynaphthalene (weight = 0.52) (Fig. 2A). We further analyzed the contribution of each metabolite to blood pressure in the four cycles and found that the data from 2009 to 2014 showed that 1-hydroxyphenanthrene accounted for a large proportion in diastolic blood pressure (weight = 0.59, 0.49, 0.20), and naphthalene metabolites accounted for a large proportion in systolic blood pressure. Supplement Fig. 1 shows the proportion of blood pressure in the 2015–2016 cycle, with 2-hydroxynaphthalene and 2&3-hydroxyphenanthrene contributing the first weight, respectively.
Contribution of PAHs to WQS percentage for Diastolic and Systolic, NHANES 2009–2010. The dashed red line represents the cutoff value (by default equal to the inverse of the number of elements in the mixture). A The WQS index weights for diastolic blood pressure. B The WQS index weights for systolic blood pressure. The model adjusted for age, gender, education level, race, BMI, and smoking status. Abbreviations: 1-NAP 1-hydroxynaphthalene, 2-NAP 2-hydroxynaphthalene, 2-FLU 2-hydroxyfluorene, 3-FLU 3-hydroxyfluorene, 1-PHE 1-hydroxyphenanthrene, 2&3-PHE 2&3-hydroxyphenanthrene, 1-PYR 1-hydroxypyrene
Finally, because of the impact of BMI on the regression relationship between metabolites and hypertension, we analyzed the impact of their interaction on the risk of hypertension (Fig. 3). The results indicated that individuals with obesity had a greater odds ratio for prevalent hypertension than normal weight individuals. Within the individuals with obesity, those in the tertiles2 of 1-hydroxyphenanthrene (OR: 3.66; 95%CI: 2.79–4.80) and 2&3-hydroxyphenanthrene (OR: 3.40; 95%CI: 2.59–4.46) had the highest odds of hypertension. The additive interaction between metabolites and BMI was not statistically significant, as shown in Supplement Table 1.
Interactions between PAH metabolites and BMI on the risk of hypertension. Urinary PAH levels were divided into three groups by tertiles. BMI was divided into three groups: normal weight (18.5–24.9 kg/m2), overweight (25–29.5 kg/m2), and obese (≥ 30 kg/m.2). Adjust for age, gender, education level, race, smoking status, and other metabolites. *Statistically different from the lowest metabolite level in normal weight group
Discussion
This study used a cross-sectional survey to investigate the effects of urinary PAH exposure on hypertension in American adults. The results indicated a positive correlation between high exposure to PAHs and increased risk of hypertension. Compared with the previous hypertension guideline, we obtained a higher prevalence of hypertension (48.52% vs. 34.67%) and a stronger association of metabolites with outcomes using the new guideline (Supplement Table 2). We found a positive dose–response relationship between high exposure to 2&3-hydroxyphenphene and the risk of hypertension, consistent with previous studies (Bangia, et al. 2015; Hu, et al. 2015; Shiue 2015). In addition, we also found a dose–response relationship between high 1-hydroxynaphthalene exposure and hypertension, which has not been reported in the US population. It may be related to the cumulative effect of PAHs. When we compared the concentrations of PAHs in NHANES from 2001 to 2008 reported by Ranjbar et al., we did find the concentrations of 2-hydroxynaphthalene (4758 ± 69) ng/L, and 1-hydroxypyrene (121 ± 2) ng/L increased. The effect of the updated definition of hypertension on marginalized populations could not be ruled out.
Furthermore, when revealing the relative weights caused by multiple exposures, we found that PAH exposure was associated with increased systolic and diastolic blood pressure. To be precise, phenanthrene (1-hydroxyphenanthrene and 2–3-hydroxyphenanthrene) is primarily responsible for diastolic blood pressure, while naphthalene (1-hydroxynaphthalene and 2-hydroxynaphthalene) is closely related to systolic blood pressure. A study published in 2022 found that exposure to 2-hydroxynaphthalene was associated with an increase in systolic blood pressure (Shahsavani, et al. 2022). Contrary to our results, a recent study in adolescents with metabolic syndrome also applied WQS models to outcomes to reveal multiple exposure effects and the relative weight of each PAH but found no association (K. Li, et al. 2021). It is worth noting that blood pressure outcomes in that study were classified variables, whereas our study used continuous variables. In general, using continuous variables to analyze independent variables is more efficient than using categorical variables to analyze independent variables (Kizilkaya, et al. 2014; Tannenbaum, et al. 2006). The association between metabolites and systolic and diastolic blood pressure needs more explicit support mechanisms.
PAHs resulting from anthropogenic activities can remain for long periods in nature and can be transported over long distances. As organic pollutants, PAHs can be bioaccumulated and bioamplified, thus affecting the quality of the environment (air, water, soil, etc.) and posing detrimental effects on living organisms (Abbas, et al. 2022; Akram, et al. 2018a, 2018b; Khalil, et al. 2022). Naphthalene, a two-ring PAH with the simplest chemical structure, is used in manufacturing processes as a sulfonate, and in dyes, plasticizers, pesticides, and pharmaceuticals (Ifegwu and Anyakora 2016). It is the most abundant PAH in cigarette smoke, especially in diesel and jet fuel exhaust (Ding, et al. 2005). Different from other metabolites, the main source of exposure for naphthalene is inhalation. Although speculative, perhaps the widespread use of naphthalene in commercial applications and its molecular property may have some effect on the growing hypertension status in the USA. The lipophilic nature of phenanthrene makes it easy to access and accumulate in human tissue, which may exert great harmful effects (Marris, et al. 2020).
Urine 1-hydroxypyrene is the most common alternative for evaluating human exposure to BaP and other PAHs. As the only metabolite with a high molecular weight in urine polycyclic aromatic hydrocarbons, its metabolic pathways may differ from those of other low molecular weight substances (Z. Li, et al. 2008). A Middle Eastern study showed a positive correlation between 1-hydroxypyrene and systolic and diastolic blood pressure in adults. In contrast, another survey of occupational exposures showed the opposite, and our results supported the latter. It is worth mentioning that only 200 randomly selected adults were included in the study in the Middle East. Sample representation and environmental exposure differences in different regions cannot be ignored. The association between 1-hydroxypyrene and blood pressure may need to be confirmed in more representative samples and cohort studies.
The association between PAH exposure and hypertension is mainly due to oxidative stress and inflammatory responses induced by particulate matter. Particulate matter exposure disrupts vascular function and systemic hemodynamics and may lead to hypertension (Alshaarawy, et al. 2013; Brook, et al. 2004; Brook and Rajagopalan 2012). According to related studies, PAHs, attached to particulate matter and then diffuse into the blood, caused a rapid increase in Ca2+ in human microvascular endothelial cells (HMEC-1) through aromatic hydrocarbon receptors (AHR) (Brinchmann, et al. 2018). Endothelial dysfunction impairs the ability of endothelial cells to release endothelium-derived relaxing factors (EDRFs), resulting in vasoconstriction and increased blood pressure (Konukoglu and Uzun 2017). Additionally, animal experiments revealed increased Ca2+ influx mediated by store operated calcium entry (SOCE) in monocytes isolated from hypertensive rats (Giachini, et al. 2009; Liu, et al. 2007).
Due to PAHs’ high lipophilicity, they accumulate in adipose tissue and affect adipocyte metabolism, which may be related to estrogen receptor activation and thyroid receptor inhibition (Sun, et al. 2008). Ranjbar’s study revealed that participants with high exposure to 3-hydroxyfluoreneand obesity had an increased risk of developing hypertension (Ranjbar, et al. 2015). Our study also demonstrates that BMI can significantly change the relationships between 1-hydroxynaphthalene with hypertension. Participants exposed to high levels of metabolites and obesity concurrently displayed a significantly increased risk of hypertension. Nevertheless, additional research is required to elucidate the mechanism of interaction between PAHs and BMI on hypertension.
To our knowledge, this is the first study to examine the association between PAHs and hypertension since the AHA updated its definition of hypertension. In addition, our study is based on NHANES, which is designed to be a nationally representative survey data; thus, the results from our study are potentially generalizable across the US population. Furthermore, we used WQS models to reveal the multiple exposure effects and the relative weight of PAHs, which has more significant public health significance for targeted reduction of pollutant exposure.
This study has several limitations. Because of the survey’s restriction on these indicators, the relationship between other significant metabolite indicators (4-OHPH) and hypertension was not examined. Diet and inhaled PAHs were also excluded. Other environmental pollutants such as particulate matter and heavy metals may have synergistic effects with PAHs on hypertension (Tauqeer, et al. 2022a, 2022b). Hence, the effects of mixed exposure to pollutants on hypertension remain to be studied. Our data are from a cross-sectional study; they reflect only recent PAH exposure and fail to investigate the causal relationship between metabolites and hypertension.
In conclusion, under the new hypertension guideline, we found a stronger association between high level exposure to PAHs and hypertension in a dose–response relationship. As both hypertension and PAHs are highly prevalent in the USA, the public health significance of a potential causal relationship between PAH exposure and hypertension should be noted. Regulating atmospheric PAHs may be an efficient strategy to reduce the risk of hypertension and prevent hypertension.
Access to data
All data of the current study are in public and available free of charge in the NHANES repository. (https://wwwn.cdc.gov/nchs/ nhanes/Default.aspx).
References
Abbas MA, Iqbal M, Tauqeer HM, Turan V, Farhad M (2022) Chapter 16 - Microcontaminants in wastewater. In: Hashmi MZ, Wang S and Ahmed Z (ed) Environmental Micropollutants. Elsevier, pp 315–329
Akram R, Turan V, Hammad HM, Ahmad S, Hussain S, Hasnain A et al (2018a) Fate of organic and inorganic pollutants in paddy soils. In: Hashmi MZ and Varma A (eds) Environmental pollution of paddy soils. ChamSpringer International Publishing, pp 197–214
Akram R, Turan V, Wahid A, Ijaz M, Shahid MA, Kaleem S et al (2018b) Paddy land pollutants and their role in climate change. In: Hashmi MZ and Varma A (eds) Environmental pollution of paddy soils. ChamSpringer International Publishing, pp 113–124
Alshaarawy O, Zhu M, Ducatman A, Conway B, Andrew ME (2013) Polycyclic aromatic hydrocarbon biomarkers and serum markers of inflammation. A positive association that is more evident in men. Environ Res 126:98–104. https://doi.org/10.1016/j.envres.2013.07.006
Alshaarawy O, Zhu M, Ducatman AM, Conway B, Andrew ME (2014) Urinary polycyclic aromatic hydrocarbon biomarkers and diabetes mellitus. Occup Environ Med 71:437–441. https://doi.org/10.1136/oemed-2013-101987
ATSDR (1995) Agency for toxic substances and disease registry. Toxicological profile for polycyclic aromatic hydrocarbons (PAHs). U.S. Department of Health and Human Services, Public Health Service, Atlanta. https://wwwn.cdc.gov/TSP/ToxProfiles/ToxProfiles.aspx?id=122&tid=25. Accessed Feb 2021
Bangia KS, Symanski E, Strom SS, Bondy M (2015) A cross-sectional analysis of polycyclic aromatic hydrocarbons and diesel particulate matter exposures and hypertension among individuals of Mexican origin. Environ Health 14:51. https://doi.org/10.1186/s12940-015-0039-2
Beaney T, Burrell LM, Castillo RR, Charchar FJ, Cro S, Damasceno A et al (2019) May Measurement Month 2018: a pragmatic global screening campaign to raise awareness of blood pressure by the International Society of Hypertension. Eur Heart J 40:2006–2017. https://doi.org/10.1093/eurheartj/ehz300
Brinchmann BC, Le Ferrec E, Podechard N, Lagadic-Gossmann D, Shoji KF, Penna A et al (2018) Lipophilic chemicals from diesel exhaust particles trigger calcium response in human endothelial cells via aryl hydrocarbon receptor non-genomic signalling. Int J Mol Sci 19:1429. https://doi.org/10.3390/ijms19051429
Brook RD, Franklin B, Cascio W, Hong Y, Howard G, Lipsett M et al (2004) Air pollution and cardiovascular disease: a statement for healthcare professionals from the Expert Panel on Population and Prevention Science of the American Heart Association. Circulation 109:2655–2671. https://doi.org/10.1161/01.Cir.0000128587.30041.C8
Brook RD, Rajagopalan S (2012) Chronic air pollution exposure and endothelial dysfunction: what you can’t see—can harm you. J Am Coll Cardiol 60:2167–2169. https://doi.org/10.1016/j.jacc.2012.08.974
Bundy JD, Mills KT, Chen J, Li C, Greenland P, He J (2018) Estimating the association of the 2017 and 2014 hypertension guidelines with cardiovascular events and deaths in US adults: an analysis of national data. JAMA Cardiol 3:572–581. https://doi.org/10.1001/jamacardio.2018.1240
Carrico C, Gennings C, Wheeler DC, Factor-Litvak P (2015) Characterization of weighted quantile sum regression for highly correlated data in a risk analysis setting. J Agric Biol Environ Stat 20:100–120. https://doi.org/10.1007/s13253-014-0180-3
CDC (2017) National Center for Health Statistics. About the National Health and Nutrition Examination Survey. https://www.cdc.gov/nchs/nhanes/about_nhanes.htm. Accessed Jan 2021
de Oliveira Galvão MF, Sadiktsis I, Batistuzzo de Medeiros SR, Dreij K (2020) Genotoxicity and DNA damage signaling in response to complex mixtures of PAHs in biomass burning particulate matter from cashew nut roasting. Environ Pollut 256:113381. https://doi.org/10.1016/j.envpol.2019.113381
Desquilbet L, Mariotti F (2010) Dose-response analyses using restricted cubic spline functions in public health research. Stat Med 29:1037–1057. https://doi.org/10.1002/sim.3841
Ding YS, Trommel JS, Yan XJ, Ashley D, Watson CH (2005) Determination of 14 polycyclic aromatic hydrocarbons in mainstream smoke from domestic cigarettes. Environ Sci Technol 39:471–478. https://doi.org/10.1021/es048690k
Everett CJ, King DE, Player MS, Matheson EM, Post RE, Mainous AG 3rd (2010) Association of urinary polycyclic aromatic hydrocarbons and serum C-reactive protein. Environ Res 110:79–82. https://doi.org/10.1016/j.envres.2009.09.010
Foti K, Hardy ST, Chang AR, Selvin E, Coresh J, Muntner P (2022) BMI and blood pressure control among United States adults with hypertension. J Hypertens 40:741–748. https://doi.org/10.1097/hjh.0000000000003072
Freire IV, Casotti CA, Ribeiro ÍJS, Silva JRD, Barbosa AAL, Pereira R (2018) Daily sodium intake influences the relationship between angiotensin-converting enzyme gene insertion/deletion polymorphism and hypertension in older adults. J Clin Hypertens (greenwich) 20:541–550. https://doi.org/10.1111/jch.13224
Friesen MC, Demers PA, Spinelli JJ, Eisen EA, Lorenzi MF, Le ND (2010) Chronic and acute effects of coal tar pitch exposure and cardiopulmonary mortality among aluminum smelter workers. Am J Epidemiol 172:790–799. https://doi.org/10.1093/aje/kwq208
Giachini FR, Webb RC, Tostes RC (2009) STIM and Orai proteins: players in sexual differences in hypertension-associated vascular dysfunction? Clin Sci (lond) 118:391–396. https://doi.org/10.1042/cs20090449
Holme JA, Brinchmann BC, Refsnes M, Låg M, Øvrevik J (2019) Potential role of polycyclic aromatic hydrocarbons as mediators of cardiovascular effects from combustion particles. Environ Health 18:74. https://doi.org/10.1186/s12940-019-0514-2
Hu H, Kan H, Kearney GD, Xu X (2015) Associations between exposure to polycyclic aromatic hydrocarbons and glucose homeostasis as well as metabolic syndrome in nondiabetic adults. Sci Total Environ 505:56–64. https://doi.org/10.1016/j.scitotenv.2014.09.085
Huang X, Deng X, Li W, Liu S, Chen Y, Yang B et al (2019) Internal exposure levels of polycyclic aromatic hydrocarbons in children and adolescents: a systematic review and meta-analysis. Environ Health Prev Med 24:50. https://doi.org/10.1186/s12199-019-0805-9
Ifegwu OC, Anyakora C (2016) Polycyclic aromatic hydrocarbons: part II, urine markers. Adv Clin Chem 75:159–183. https://doi.org/10.1016/bs.acc.2016.03.001
Iida H, Kurita N, Takahashi S, Sasaki S, Nishiwaki H, Omae K et al (2019) Salt intake and body weight correlate with higher blood pressure in the very elderly population: The Sukagawa study. J Clin Hypertens (greenwich) 21:942–949. https://doi.org/10.1111/jch.13593
Khalil M, Iqbal M, Turan V, Tauqeer HM, Farhad M, Ahmed A et al (2022) Chapter 11 - Household chemicals and their impact. In: Hashmi MZ, Wang S and Ahmed Z (eds) Environmental micropollutants. Elsevier, pp 201–232
Kizilkaya K, Fernando RL, Garrick DJ (2014) Reduction in accuracy of genomic prediction for ordered categorical data compared to continuous observations. Genet Sel Evol 46:37. https://doi.org/10.1186/1297-9686-46-37
Konukoglu D, Uzun H (2017) Endothelial dysfunction and hypertension. Adv Exp Med Biol 956:511–540. https://doi.org/10.1007/5584_2016_90
Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K (2016) Body fatness and cancer—viewpoint of the IARC Working Group. N Engl J Med 375:794–798. https://doi.org/10.1056/NEJMsr1606602
Li K, Yin R, Wang Y, Zhao D (2021) Associations between exposure to polycyclic aromatic hydrocarbons and metabolic syndrome in U.S. adolescents: cross-sectional results from the National Health and Nutrition Examination Survey (2003–2016) data. Environ Res 202:111747. https://doi.org/10.1016/j.envres.2021.111747
Li Z, Sandau CD, Romanoff LC, Caudill SP, Sjodin A, Needham LL et al (2008) Concentration and profile of 22 urinary polycyclic aromatic hydrocarbon metabolites in the US population. Environ Res 107:320–331. https://doi.org/10.1016/j.envres.2008.01.013
Liu DY, Thilo F, Scholze A, Wittstock A, Zhao ZG, Harteneck C et al (2007) Increased store-operated and 1-oleoyl-2-acetyl-sn-glycerol-induced calcium influx in monocytes is mediated by transient receptor potential canonical channels in human essential hypertension. J Hypertens 25:799–808. https://doi.org/10.1097/HJH.0b013e32803cae2b
Lusa L, Ahlin Č (2020) Restricted cubic splines for modelling periodic data. PLoS ONE 15:e0241364. https://doi.org/10.1371/journal.pone.0241364
Mallah MA, Mallah MA, Liu Y, Xi H, Wang W, Feng F et al (2021) Relationship between polycyclic aromatic hydrocarbons and cardiovascular diseases: a systematic review. Front Public Health 9:763706. https://doi.org/10.3389/fpubh.2021.763706
Marris C, Kompella SN, Miller M, Incardona JP, Brette F, Hancox JC et al (2020) Polyaromatic hydrocarbons in pollution: a heart-breaking matter. J Physiol 598:227–247
Pastor-Belda M, Campillo N, Arroyo-Manzanares N, Torres C, Pérez-Cárceles MD, Hernández-Córdoba M et al (2019) Bioaccumulation of polycyclic aromatic hydrocarbons for forensic assessment using gas chromatography-mass spectrometry. Chem Res Toxicol 32:1680–1688. https://doi.org/10.1021/acs.chemrestox.9b00213
Qin P, Luo X, Zeng Y, Zhang Y, Li Y, Wu Y et al (2021) Long-term association of ambient air pollution and hypertension in adults and in children: a systematic review and meta-analysis. Sci Total Environ 796:148620. https://doi.org/10.1016/j.scitotenv.2021.148620
Qu Y, Niu H, Li L, Li M, Yan S, Li M et al (2019) Analysis of dose-response relationship between BMI and hypertension in northeastern China using restricted cubic spline Functions. Sci Rep 9:18208. https://doi.org/10.1038/s41598-019-54827-2
Ranjbar M, Rotondi MA, Ardern CI, Kuk JL (2015) Urinary biomarkers of polycyclic aromatic hydrocarbons are associated with cardiometabolic health risk. PLoS ONE 10:e0137536. https://doi.org/10.1371/journal.pone.0137536
Shahsavani S, Fararouei M, Soveid M, Dehghani M, Hoseini M (2022) Exposure to polycyclic aromatic hydrocarbon-induced oxidative stress in Shiraz, Iran: urinary levels, health risk assessment and mediation effect of MDA on the risk of metabolic syndromes. Int Arch Occup Environ Health. https://doi.org/10.1007/s00420-021-01822-8
Shiue I (2015) Are urinary polyaromatic hydrocarbons associated with adult hypertension, heart attack, and cancer? USA NHANES, 2011–2012. Environ Sci Pollut Res Int 22:16962–16968. https://doi.org/10.1007/s11356-015-4922-8
Stanaway JD, Afshin A, Gakidou E, Lim SS, Abate D, Abate KH et al (2018) Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet 392
Sun H, Shen OX, Xu XL, Song L, Wang XR (2008) Carbaryl, 1-naphthol and 2-naphthol inhibit the beta-1 thyroid hormone receptor-mediated transcription in vitro. Toxicology 249:238–242. https://doi.org/10.1016/j.tox.2008.05.008
Sundström J, Lind L, Lampa E, Angerås O, Bachus E, Bergström G et al (2020) Weight gain and blood pressure. J Hypertens 38:387–394. https://doi.org/10.1097/hjh.0000000000002298
Tannenbaum SJ, Holford NH, Lee H, Peck CC, Mould DR (2006) Simulation of correlated continuous and categorical variables using a single multivariate distribution. J Pharmacokinet Pharmacodyn 33:773–794. https://doi.org/10.1007/s10928-006-9033-1
Tauqeer HM, Turan V, Farhad M, Iqbal M (2022a) Sustainable agriculture and plant production by virtue of biochar in the era of climate change. In: M. Hasanuzzaman, G. J. Ahammed and K. Nahar (ed) Managing plant production under changing environment. SingaporeSpringer Nature Singapore, pp 21–42
Tauqeer HM, Turan V, Iqbal M (2022b) Production of safer vegetables from heavy metals contaminated soils: the current situation, concerns associated with human health and novel management strategies. In: J. A. Malik (ed) Advances in bioremediation and phytoremediation for sustainable soil management: principles, monitoring and remediation. ChamSpringer International Publishing, pp 301–312
Trasande L, Urbina EM, Khoder M, Alghamdi M, Shabaj I, Alam MS et al (2015) Polycyclic aromatic hydrocarbons, brachial artery distensibility and blood pressure among children residing near an oil refinery. Environ Res 136:133–140. https://doi.org/10.1016/j.envres.2014.08.038
Vekic J, Zeljkovic A, Stefanovic A, Jelic-Ivanovic Z, Spasojevic-Kalimanovska V (2019) Obesity and dyslipidemia. Metabolism 92:71–81. https://doi.org/10.1016/j.metabol.2018.11.005
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C et al (2018) 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 71:1269–1324. https://doi.org/10.1161/hyp.0000000000000066
WHO (2019) World Health Organisation. Hypertension. Key facts. https://www.who.int/news-room/fact-sheets/detail/hypertension. Accessed 31 March 2021
Xu X, Hu H, Kearney GD, Kan H, Sheps DS (2013) Studying the effects of polycyclic aromatic hydrocarbons on peripheral arterial disease in the United States. Sci Total Environ 461–462:341–347. https://doi.org/10.1016/j.scitotenv.2013.04.089
Yusuf S, Joseph P, Rangarajan S, Islam S, Mente A, Hystad P et al (2020) Modifiable risk factors, cardiovascular disease, and mortality in 155 722 individuals from 21 high-income, middle-income, and low-income countries (PURE): a prospective cohort study. Lancet 395:795–808. https://doi.org/10.1016/s0140-6736(19)32008-2
Zhang Y, Zheng H, Zhang L, Zhang Z, Xing X, Qi S (2019) Fine particle-bound polycyclic aromatic hydrocarbons (PAHs) at an urban site of Wuhan, central China: characteristics, potential sources and cancer risks apportionment. Environ Pollut 246:319–327. https://doi.org/10.1016/j.envpol.2018.11.111
Acknowledgements
Thanks to the NHANES participants and the staff members for their contribution of data collection and for making the data publicly available.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 81502795) and Shanxi Basic Research Project (grant numbers 20210302123317).
Author information
Authors and Affiliations
Contributions
Protocol/project development: Fang Wang, Yuying Wang, and Yu Wang. Data collection or management: Yuying Wang, Li Chang and Jie Ding. Data analysis: Yuying Wang, Li Zhou, and Yu Wang. Manuscript writing/editing: Fang Wang, Yuying Wang and Teng Jia. Supervision: Fang Wang.
Corresponding author
Ethics declarations
Ethics in publishing
This study was done using Public Data from the NCHS program, NHANES. The data have been de-identified, and it has not been merged or augmented in a way that has compromised the privacy of the participants. Therefore, no further approval was required and the study follows ethical guidelines.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, F., Wang, Y., Wang, Y. et al. Urinary polycyclic aromatic hydrocarbon metabolites were associated with hypertension in US adults: data from NHANES 2009–2016. Environ Sci Pollut Res 29, 80491–80501 (2022). https://doi.org/10.1007/s11356-022-21391-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21391-8