Abstract
In recent decades, the biodiversity of freshwater environments has decreased sharply due to anthropogenic disturbances that damaged ecosystem structures and functions. Habitat restoration has emerged as an important method to mitigate the degradation of river ecosystems. Although in many cases a post-project monitoring has been promoted to access the restoration progress, it is still unclear how aquatic community changes following river habitat restoration in China. Macroinvertebrate communities intermediately positioned within ecosystem food webs play a key role in ecosystem processes within river ecosystem, driving energy flow and nutrient cycling. Here, benthic macroinvertebrates are used as bio-indicators to assess the ecosystem health of degraded urban rivers, restored urban rivers, and undisturbed rivers. This study aims to determine (i) how habitat restoration influences macroinvertebrates diversity and how this compared to degraded and reference conditions; (ii) how did macroinvertebrate community compositions differ in restored, degraded, and reference sites; and (iii) the environmental factors shaping macroinvertebrate communities. Habitat restoration significantly increased the diversity and richness of macroinvertebrate community and intolerant species and shifted the community composition towards reference status. Habitat characteristics and water chemistry, including substrate diversity, water velocity, and both nutrients (TN) and organic pollutants (TOC), appeared to shape the turnover of these communities. Habitat characteristics contributed to most of the variation of the entire macroinvertebrate community. Our research indicates that habitat restoration is an efficient approach to restore the aquatic community and hence improve river ecosystem health for freshwater conservation and sustainable management in Zhejiang province. This study strengthens our understanding of the changes of macroinvertebrate community after habitat restoration and important controlling variables that attribute to these changes, which provides an important guidance for future freshwater management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anthropogenic disturbances, such as urbanization, damming, water withdrawal, and pollution, have sharply increased in the past centuries, which markedly damaged freshwater ecosystem structure and decreased biodiversity (Zhang et al. 2019). To mitigate and prevent the degradation of river ecosystems, habitat restoration has emerged as a key activity around the world (Geist and Hawkins 2016). The aim of habitat restoration is to improve the ecosystem health of freshwater systems through enhancing habitat complexity and heterogeneity, thus sustaining the ecosystem from human disturbance. To this end, process-based restoration that focuses on correcting anthropogenic disruptions to driving processes arose as important measure to recover the river habitats in the last 20 years (Beechie et al. 2012). Restoration approach such as river channel re-meandering is applied in some projects to shape the heterogenous river habitat indirectly (Garcia et al. 2012; Lorenz et al. 2016), channel reconfiguration measures include riverbed reconstruction, adding both in-stream islands and aquatic vegetation, and increasing flood plain areas are widely included in restoration strategies of urban rivers to reconstruct the river habitat directly (Bernhardt et al. 2007; Palmer et al. 2014; Martín et al. 2018). In combination, these treatments should enhance substrate and hydraulic heterogeneity, increasing both specific aquatic habitat and food availability (Laasonen et al. 1998; Lepori et al. 2005; Miller et al. 2010).
Different types of riverine habitats are known to influence the community composition of aquatic organisms such as fish and macroinvertebrates, attributing to the variance of river hydromorphology, substrate composition, and environmental condition at the reach scale (Zhang et al. 2009; Kail et al. 2015). Many studies measured benthic biological indicators (i.e., microbes, algae, invertebrates) to assess the structural integrity and ecosystem health following habitat restoration (Frainer et al. 2018; Schmutz et al. 2016; Kail et al. 2016). Evidence accumulated indicated that aquatic rehabilitation would improve habitat condition and water quality for aquatic biota through restructuring heterogeneous habitat, re-introducing aquatic plants, riparian zone reforestation, etc. (Miller et al. 2010; Kail et al. 2015). However, evidence of ecological improvements associated with habitat restoration has been highly varied due to the natural variability of the system studied (Miller et al. 2010; Louhi et al. 2011; Zan et al. 2017), and the response of benthic aquatic communities to habitat restoration remains unclear in China. Therefore, it is imperative to obtain a better understanding of restoration effects and the underlying ecological mechanisms. Some information could be gained to better understand this restoration progress using a before-after (BA), before-after-control-impact (BACI), or control-impact (CI) approach, hence providing sufficient evidence for post river management and improvement of future endeavors.
Macroinvertebrate communities are composed of a range of species that tolerate a wide range of environmental conditions (Plafkin et al. 1989). Intermediately positioned within ecosystem food webs in river ecosystems, macroinvertebrates play a key role in ecosystem processes such as nutrient cycling and energy flow (Zhang et al. 2004; Strayer 2006; Duan et al. 2010). Stream macroinvertebrates are generally recognized as good biological indicators of water quality (Hilsenhoff 1988) and ecosystem health (Karr 1999), because of their availability in most freshwater ecosystems, and their sensitivity to environmental changes such as disturbance, deterioration, and improvement (Zhang et al. 2010; Li et al. 2015). They can reflect the relative long-term temporal and spatial changes of river ecosystems and can be early warning indicators of environmental pressures given that they are such a diverse group containing a high number of species with a large variability in ecological requirements (Smith et al. 1999; Shao et al. 2006; Dos Santos et al. 2011). Hence, macroinvertebrates are frequently used as indicators of restoration efficiency (Spänhoff and Arle 2007; Besacier-Monbertrand et al. 2014).
The use of macroinvertebrates as bio-indicators for restoration has been studied in Europe and North America (Kail et al. 2015; Zan et al. 2017), but there have been few assessments of restoration in Asia and, in particular, China (Li et al. 2015; Lin et al. 2020). Although the restoration-related effect on macroinvertebrate communities should be theoretically positive with the increase of habitat heterogeneity (Miller et al. 2010), as features of river habitat may influence detritus (Douglas and Lake 1994; Taniguchi and Tokeshi 2004), epiphytic algae (Dudley et al. 1986) and form “refuges” from high flow conditions for predators (Lake et al. 2000; Taniguchi and Tokeshi 2004), observed changes have been inconsistent with the scale and specific metrics assessed (Palmer et al. 2010; Ernst et al. 2012). The results may also differ when investigating rivers with diverse and complex conditions, especially in China, where land use change posed varying degree of habitat degradation and water pollution in river ecosystems (Zhang et al. 2010; Knouft and Chu 2015).
In this study, macroinvertebrate communities of three river groups were compared, (1) degraded urban rivers, (2) urban rivers undergoing habitat restoration, and (3) undisturbed rivers (i.e., reference conditions), essentially providing a gradient from severely damaged to near-natural conditions. Within each river, a range of habitat features, physico-chemical factors, and spatial factors were measured, and macroinvertebrate communities were sampled. Through comparing the relationship between macroinvertebrate community composition and environmental variables along this simple gradient, this study intends to determine (i) how habitat restoration impacts on benthic macroinvertebrates diversity and how this compared to degraded and reference conditions; (ii) how did macroinvertebrate community compositions differ in restored relative to degraded and reference sites; and (iii) the environmental factors shaping macroinvertebrate communities across the three river groups. We hypothesized that habitat restoration would shift the benthic macroinvertebrate community composition and the macroinvertebrate diversity and richness would increase, and there would be an improvement in both water quality and availability of aquatic habitats following the restructuring of heterogeneous habitat and re-introducing of aquatic macrophytes and riparian zone reforestation. Moreover, some tolerant species that are dominant in degraded urban rivers will be replaced by Ephemeroptera, Plecoptera, and Trichoptera (EPT) species that are sensitive to external disturbance. Substrate composition, water flow velocity, and physico-chemical variables were hypothesized to be the main factors affecting any change in macroinvertebrate community composition.
Materials and methods
Study sites
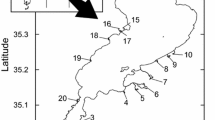
Control-impact approach was used for this study. Accordingly, three groups of rivers selected from the same catchment (Shaoxi River) in Anji, Zhejiang Province, PRC, were investigated from July 8 to August 15, 2018, each group with three different rivers. Three river groups (Fig. 1, Table S1) include (i) undisturbed rivers (reference sites, denoted F), (ii) urban rivers undergoing habitat restoration in the last 7 years (denoted R), and (iii) degraded urban rivers (denoted D). Spatial factors of each river were derived using geographic coordinates (latitude and longitude) measured by a handheld global positioning system (GPS, Trimble Juno SA; Guo et al. 2019; Lin et al. 2020). The investigation was authorized by director Yun Xiang in the general office of Anji County Water Resources Bureau. In summer 2018, the average day/night temperatures of the region were 29°C/21°C, and the average precipitation was 133 mm.
Both degraded rivers and pre-restored urban rivers had similar hydromophological conditions, stream order, slope, and temperature regime (Lin et al. 2019) and were located in the same ecoregion. Straitened and hardened with concrete, these three degraded rivers were covered with mud and were listed as rivers to be restored in the future by the local water conservancy bureau. Two of the degraded rivers are surrounded by suburban areas, and another one is located in the city center. The three restored rivers located in urban areas were at the same elevation with those degraded rivers. With reference to the habitat conditions of reference sites, these rivers have been restored using a similar ecological restoration strategy for up to 7 years. This involved natural reconstruction of the riverbed using diverse substrates (e.g., cobbles and pebbles), the channel was re-connected and re-meandered, floating islands were constructed, aquatic plants including submerged macrophytes and emergent plants were re-introduced, and the riparian zone was re-afforested in an attempt to recover a more natural river form based on their specific river type. Three undisturbed rivers were 40-km upstream of these urban rivers within the same catchment, and these undisturbed rivers were considered approximations to reference sites.
Habitat characteristics
Habitat surveys were performed in July and August 2018. At each river, habitat characteristics (denoted habitat) were measured within a 50-m sampling reach as described in Lin et al. (2019). After visually estimating the reach canopy cover, the water velocity across the channel was measured by Teledyne flow meters (ISCO, Lincoln, NE, USA), the riverbed types including riffle, pool, and island were counted, and the substrate composition was described by random-selecting 100 sediment particles on the riverbed and counting the ratio of substrate classes (boulders, cobbles, pebbles, sand grains) according to Kondolf (1997). The substrate diversity was then calculated by means of the Shannon-Wiener diversity H’ (Shannon 1997) for each site.
Physico-chemical variables
A 100-m tape was used to measure the river width. The river depth was measured at five evenly spaced points across the channel. Three sampling positions were randomly selected within a 50-m sampling reach in each river, and physico-chemical variables (denoted ENV) were monitored by standard methods (Lin et al. 2019). Briefly, (1) temperature, pH, dissolved oxygen (DO), and turbidity were measured in situ using handheld water quality analyzers, and (2) a 1-l water sample was taken from three sampling points, filtered through a 0.45-μm filter and tested within 48 h for ammonium nitrogen (NH4-N), nitrate-nitrogen (NO3-N), total nitrogen (TN), total phosphorus (TP), total organic carbon (TOC), and chemical oxygen demand (COD).
Macroinvertebrate sampling procedure
In each river, three samples of benthic macroinvertebrates in each studied river were sampled according to Chinese Technical Guidelines for Species Monitoring Freshwater-Benthic Macroinvertebrates (HJ 710.8—2014). Samples were collected from July 8 to August 15, 2018, in three sampling sites in each river using a 1 m × 1 m quadrat distributed randomly along a 50-m stretch. Within each quadrat, macroinvertebrates were sampled using a D-frame aquatic dip net (opening: 25.4 cm L × 30.5 cm W; mesh size: 500 μm) by disturbing vegetation and substrate; the samples were then preserved in 70% ethanol for storage and sorted and all macroinvertebrates then identified to family level using Merritt et al. (2008) and classified into groups according to their ability to water pollution using the Family Tolerance Value (Mandaville 2002).
Differences in the structure of benthic macroinvertebrate communities were then assessed by calculating total abundance, total richness, Shannon-Wiener diversity (H’), Pielou’s evenness (Shannon 1997), the abundance and richness of EPT (Ephemeroptera, Plecoptera, and Trichoptera), and richness of intolerant taxa for each river group. To further investigate specific community composition changes, indicator taxa for each group of river were selected using multilevel pattern analysis at significance level of p < 0.05.
Statistical analysis
Differences in habitat features, physio-chemical parameters, and macroinvertebrate alpha (α) diversity properties in three river groups were evaluated through analysis of variance with post hoc Tukey-Kramer test (Torres-Mellado et al. 2012). Environmental factors and α-diversity indexes were ln (x + 1) transformed if the residuals deviated from normality. The similarity in macroinvertebrate community among three river groups was then assessed by analysis of similarities using the “anosim” function in “vegan” in R statistical environment (R Core Team 2017). A p-value of 0.05 was used as the cutoff for significance.
To explore relationships between habitat characteristics, physio-chemical features, spatial factors, and α-diversity of macroinvertebrate, respectively, Spearman’s correlation coefficients were calculated, and explanatory variable that indicates significant multi-collinearity (Spearman correlation coefficient ≥0.70) was excluded from further analysis (Cai et al. 2017). The macroinvertebrate abundance matrices were Hellinger-transformed, and detrended correspondence analysis (DCA) was then carried out using “decorana” function in R package vegan to choose response model (linear or unimodal) for the macroinvertebrate community data. The length of the first DCA ordination axis was less than four, which indicated that RDA was suitable for taxonomic composition. Accordingly, RDA was performed, and the significance was tested using the “anova.cca” function in “vegan.” Explanatory variables were selected by performing forward selection using function “forward.sel” in the “packfor” R package. Monte Carlo permutation test was then applied to test the contribution significance of each variable. Finally, variation partitioning was performed to explore the pure contribution of each group (i.e., habitat, environmental data, and spatial factors) to the variation of macroinvertebrate community using the “varpart” function in the “vegan” R package (Borcard et al. 2018). Multivariate analyses including DCA, RDA, forward selection, and variation partitioning were performed according to Borcard et al. (2018).
Results
Habitat characteristics
Significant differences in water velocity (F2,6 = 6.661, p = 0.030) and substrate diversity (F2,6 = 71.18, p < 0.001) were detected between the three river groups; restored rivers had a higher water velocity than both degraded rivers and undisturbed rivers (Fig. 2e); the substrate diversity in the undisturbed and restored rivers was remarkably higher than degraded rivers (p < 0.001) (Fig. 2f). Four types of sediment sizes (boulder, cobble, peddle, granule) formed the riverbed of restored and undisturbed rivers, whereas degraded rivers have only one kind of particles (2–4 mm granule). The habitat diversity in undisturbed and restored rivers was also much higher than that in degraded rivers. Riffles, pools, and islands constituted the habitat structure of the undisturbed and restored rivers, whereas degraded rivers were formed by pools and a few islands. No significant difference was observed in canopy cover between the three river groups (F2,6 = 4.198, p = 0.072).
Box plots of the (a) turbidity, (b) total nitrogen (TN), (c) total organic carbon (TOC), (d) chemical oxygen demand (COD), (e) water velocity, and (f) substrate Shannon-Wiener diversity in three contrasting river types within Anji City Region, PRC. Mean values (± SE, n = 3) are presented; different lowercase letters indicate a significant difference observed at p = 0.05 level
Physico-chemical properties of surface water
Analysis of variance indicated no significant differences among three river groups in river width (F2,6 = 0.336) and mean river depth (F2,6 = 0.791) and no difference in water variables such as pH (F2,6 = 0.325), DO (F2,6 = 1.716), NH4-N (F2,6 = 2.619), NO3-N (F2,6 = 2.498), and TP (F2,6 = 1.609). However, variables exhibited significant differences in water turbidity (F2,6 = 11.75, p = 0.008), TN (F2,6 = 16.17, p = 0.004), and COD (F2,6 = 5.965, p = 0.038) in different river groups. Undisturbed rivers had significantly lower concentrations of TN, TOC, and COD and turbidity than the degraded rivers (p = 0.003, p = 0.047 p = 0.032, and p = 0.014, respectively; Fig. 2a–d). Restored rivers possessed a higher turbidity (p = 0.013) and a slightly increased TN concentration (p = 0.060) than undisturbed rivers (Fig. 2a, b), whereas a weak reduction in TN was found in restored rivers compared to degraded rivers (p = 0.073) (Fig. 2b).
Benthic macroinvertebrate community
In total, 9,990 specimens of macroinvertebrates were identified, 4,006 individuals in undisturbed rivers, 5,792 in restored rivers, and 192 in degraded rivers. Macroinvertebrate α-diversity values (Table 1, Table 2) showed that there were significant differences among river types for total abundance (F2,6 = 37.32, p < 0.001), total richness (F2,6 = 222.20, p < 0.001), EPT abundance (F2,6 = 90.40, p < 0.001), EPT richness (F2,6 = 67.41, p < 0.001), intolerant species richness (F2,6 = 122.10, p < 0.001), and Shannon-Wiener diversity (F2,6 = 49.00, p < 0.001). Both reference sites and restored sites had significantly higher total abundance, total richness, EPT abundance, EPT richness, Shannon-Wiener diversity, and intolerant taxa richness than degraded rivers (p < 0.001) (Table 1, Table 2, Fig. 3), whereas no significant difference of taxonomic diversity was detected between undisturbed rivers and restored rivers (p > 0.05). No difference was found among three river groups for the evenness of macroinvertebrates (F2,6 = 0.532).
Box plots of macroinvertebrate alpha-diversity (a) total abundance, (b) total richness, (c) EPT taxa abundance, (d) EPT taxa richness, (e) macroinvertebrate diversity (Shannon-Wiener diversity), and (f) intolerant taxa richness in undisturbed, restored, and degraded rivers within the Anji City Region, PRC. Mean values (± SE, n = 3) are presented; different lowercase letters indicate a significant difference observed at p = 0.05 level
The analysis of similarities (ANOSIM) based on the macroinvertebrate samples showed a significant difference of macroinvertebrate community compositions among the three river groups (R = 0.845, p = 0.001). Among the 46 families of macroinvertebrates identified in this survey, thirteen taxa were selected as indicator taxa (Table 3). Eight species were highly associated with undisturbed rivers, including dominant family Leptophlebiidae (22.35%), Perlidae (7.43%), and some other species like Dytiscidae, Scirtidae, Coenagrionidae, Hydrophilidae, Leptoceridae, and Tipulidae. Leptophlebiidae, Perlidae, Leptoceridae, Dytiscidae, and Coenagriidae were significantly more distributed in the reference sites than both urban river groups (p < 0.05 in all cases), and no difference of these taxa was found between restored rivers and urban degraded rivers (p > 0.05). Five indicator taxa (Corbiculidae, Glossiphoniidae, Erpobdellidae, Lymnaeidae, and Heptageniidae) were found in restored rivers, and dominant species were the Caenidae (31.21%), Chironomidae (14.95%), and Baetidae (12.39%). Of the EPT taxa sampled, Caenidae was the most dominant family in the restored sites and was significantly more abundant than that in degraded urban rivers (p = 0.05) and comparable to undisturbed rivers (p > 0.05), and Baetidae and Heptageniidae were also presented in the restored rivers in greater numbers than in degraded rivers (p = 0.088, p = 0.066, respectively), although these trends were not significant. Two of the tolerant taxa (Corbiculidae and Glossiphoniidae), however, were significantly greater in restored rivers compared to both degraded and undisturbed rivers (p < 0.05). No indicator taxon was allocated to degraded rivers, but degraded rivers had a higher abundance of Tubificidae (46.92%), Chironomidae (32.36%), and Viviparidae (12.26%) (Table 3).
Correlation between environmental variables and macroinvertebrate community
The correlation between macroinvertebrate α-diversity and environmental variables (i.e., habitat characteristics, and physico-chemical variables) are listed in Table 4. The relationship among environmental variables, spatial factors, and total macroinvertebrate community structure was examined by constrained redundancy analysis (RDA), and eigenvalues of 0.500 and 0.249, respectively, for axis one and two were generated (Fig. 4). The environmental variables including habitat characteristic, physico-chemical variables, and spatial variables explained 74.9% of the variance in macroinvertebrate community structure. Monte Carlo permutation tests revealed that substrate diversity, water velocity, COD, and longitude significantly affected the macroinvertebrate community (p < 0.05 in all cases). The macroinvertebrate assemblages of undisturbed rivers were mainly structured by diverse substrates (F2,6 = 3.472, p = 0.004) and low COD concentration (F2,6 = 2.285, p = 0.022). COD in the surface water (F2,6 = 25.599, p = 0.006) was also a major factor influencing macroinvertebrate community in degraded rivers. In restored rivers, the macroinvertebrate communities showed a strong correlation with water velocity (F2,6 = 3.801, p = 0.014), substrate diversity (F2,6 = 9.843, p = 0.018), and longitude (F2,6 = 5.687, p = 0.026).
Relative importance of environmental, spatial, and habitat factors
Variation partitioning showed that 44% of the community taxonomic composition was explained by three sets of environmental variables; habitat factors explained 22%, followed by physico-chemical variables (ENV, 5%) and spatial factors (4%); 12% of the variation was shared by all three sets, 4% between habitat and ENV and 2% between ENV and spatial factors (Fig. 5a). No shared effect was found between habitat and spatial factors (Fig. 5a). In terms of indicator taxa, 36% of the total variation was explained by the three explanatory sets of variables. Habitat feature was still the main factor explaining 10%, spatial factors explained 2%, and physico-chemical variables explained nothing; 4% of the variation was shared by all three sets, 11% between ENV and spatial factors, 9% between spatial factors and ENV, and 5% between habitat and ENV (Fig. 5b).
Venn diagrams illustrating the variation partitioning analysis for a taxonomic composition and b indicator taxa (taxa at family level). Habitat, ENV, and spatial factor are sets of variables representing habitat variables, physico-chemical variables, and spatial factors, respectively. Residuals are shown in the lower right corner. All fractions based on adjusted R2 are shown as percentages of total variation
Discussion
Taxonomic diversity of macroinvertebrate communities
Overall, there were significant differences in macroinvertebrate community composition between the restored and degraded rivers. The taxonomic diversity and composition of macroinvertebrate community in restored rivers were distinct from degraded rivers and strongly associate with habitat characteristic substrate diversity and water velocity, indicating that habitat restoration had impacted the structure of the communities. Compared with degraded rivers, there was a significant increase in macroinvertebrate diversity and total richness in restored rivers; meanwhile, EPT richness and intolerant taxa richness also increased under habitat restoration. These results are in accordance with the stated hypothesis and in line with previous studies in northern Poland and elsewhere (Matthaei and Diehl 2005; Miller et al. 2010; Obolewski et al. 2016), indicating that habitat heterogeneity had significant, positive effects on macroinvertebrate richness and diversity. In-stream habitat restoration enhanced the macroinvertebrate richness and diversity (Flores et al. 2017).
The difference in macroinvertebrate diversity reflects the variation of habitat characteristics and physico-chemical variables (Turley et al. 2016; Shi et al. 2019). As demonstrated previously, increased depth and frequency of pools should increase species richness through higher habitat heterogeneity (Brasher 2003). Obolewski et al. (2016) also suggested that restoration approach rehabilitation induced hydrological connectivity, improved water quality, and increased the diversity and abundance of macrozoobenthos. Here, substrate composition, organic carbon TOC, and nutrient TN were important in influencing macroinvertebrate diversity. Riverbed reconstruction and aquatic macrophyte re-introduction applied to the restored rivers enhanced the substrate diversity, diverse substrate, and large size particle (e.g., cobbles) and can enhance the stability of habitats and form abundant interstitial spaces for macroinvertebrates (Luo et al. 2017). Some macroinvertebrates are very sensitive to organic pollutants and water quality degradation (Kalyoncu and Gülboy 2009; Patang et al. 2018). The decline in organic carbon and nutrient level in restored rivers may improve the water quality and stimulate the development of macroinvertebrates of low tolerance value. This finding differs with many habitat restoration schemes which resulted in modest/unsuccessful ecological responses for the persist of constraints such as degraded hydrological regimes, rare food availability, and high pollutant loads (Tullos et al. 2009; Palmer et al. 2010). Jähnig and Lorenz (2008) declared that the diminish of diverse source populations under multiple-factor impairments and cumulative alterations of streams might be another reason for the failed response under ecological restoration.
Relative abundance of EPT and intolerant species also increased in restored rivers compared to degraded rivers. Many pollution-intolerant taxa belong to the EPT insect orders Ephemeroptera, Plectoptera, and Trichoptera. The observed increase in sensitive EPT taxa agrees with earlier observations in field studies and mesocosm experiments, suggesting that EPT taxa are sensitive to environmental degradation and habitat simplification (Cabria et al. 2011; Ilarri et al. 2018), EPT taxa often decline where there is a reduction in flow velocity accompanied by clearing of coarse substrates including coarse woody debris (CWD), and excess fine sediment deposition, which reduced food availability (Ryan 1991), ruined sheltering areas of specific macroinvertebrate taxa such as stonefly (Kärnä et al. 2018), and physically damages gills and filter-feeding apparatus by abrasion or clogging (Jones et al. 2012; Piggott et al. 2015).
Determinants of macroinvertebrate community composition
Distinct macroinvertebrate communities were found among river types. These differences were closely related to the changes in water velocity and substrate diversity, COD, and longitude of the rivers. These results support the hypothesis that macroinvertebrate community composition was driven by habitat characteristics, river discharge, physico-chemical variables, and spatial factors and in line with a summarized concept that benthic macroinvertebrate species are sensitive to both hydromorphology and water quality factors in their environment (Mandaville 2002; Shi et al. 2019). Habitat characteristics contributed to most of the variation of the entire macroinvertebrate community and the structure variation of indicator taxa, followed by ENV and spatial factors (Englund et al. 1997). This supports the view of Jähnig and Lorenz (2008) and Luo et al. 2017) that habitat-specific variables explained the major variation in macroinvertebrate community composition. Macroinvertebrate fauna can always be classified into flow exposure groups (obligate, facultative, and avoiders) and habit groups (clinger, burrowers, sprawlers, and swimmers) in accordance with their preference towards hydromorphology conditions that is guided by their flow exposure preferences and behavioral activities (Merritt et al. 2008). Rivers with diverse substrates can provide a high variability of micro-habitats and heterogeneous food resources for macroinvertebrates (Mandaville 2002), especially as water velocity varies at different seasons; hence a diverse species assemblage, adapted to various natural flows, can be maintained. In our study, the changes in substrate diversity and flow velocity induced by habitat restoration were important in shaping the macroinvertebrate communities in restored rivers compared to those in degraded rivers. The increase in substrate diversity and flow velocity in the restored rivers induced a more diverse habitat type, which sustain the development of macroinvertebrate taxa with preferences for each particular habitat and hydrology condition (Dewson et al. 2007; Elbrecht et al. 2016).
Differences in physico-chemical variables (e.g., TN and TOC) further contributed to the shifts in macroinvertebrate community composition among three river types, though the influence is not as strong as habitat characteristics. Given that water quality conditions are a product of catchment-wide processes which act as large-scale filter of the regional species pool (Poff 1997), but habitat-scale variation drives differences in macroinvertebrate communities within the species pool, which yield a greater statistical influence (White et al. 2019). In our study, heavy organic pollutants in the degraded rivers led to higher abundance of tolerant families Tubificidae, Chironomidae, and Viviparidae (Al-Shami et al. 2011; Arimoro 2009), whereas restored rivers improved habitat heterogeneity, declined the nutrient and organic pollutants, provided more favorable conditions for the development of sensitive EPT taxa (including abundant taxa Baetidae and indicator taxon Heptageniidae; Patang et al. 2018; Luo et al. 2017), and facilitated the establishment of some low organic pollutant tolerant taxa that live in specific habitats, such as indicator taxa Glossiphoniidae and Corbiculidae (Luo et al. 2017). These results are similar to those reported for the river Danube and Illinois streams (Heatherly et al. 2007; Rico et al. 2016) and an indoor experiment (Beermann et al. 2018). Implying that habitat restoration shifted the dominant pollution-tolerant macroinvertebrates to sensitive EPT taxa with the improvement of river habitat and water quality, facilitated the establishment of some low tolerant taxa that live in specific habitat such as sediment, riffle, pool, aquatic plant, and exist under low level of pollution in restored rivers, and this distinguishes the macroinvertebrate community in restored rivers from the community in the other two river types.
The shared effects of hydro-morphological and water chemical factors (ENV vs. habitat vs. spatial factor), however, had greater influences on macroinvertebrate communities than single effect of physico-chemical or spatial factors. Consistent with the idea of Rico et al. (2016), who indicated that chemical pollution had a lower contribution to invertebrate community than shared effect of habitat characteristics and physico-chemical conditions. Spatial factors have a lower contribution on the macroinvertebrate community variance than physico-chemical and habitat variables. The biological communities in rivers may change along the variation of spatial factors (Vannote et al. 1980). However, habitat and water quality conditions, rather than spatial factors, best explained the variance of invertebrate community and diversity (Rico et al. 2016).
Overall, the macroinvertebrate community clustered in the restored rivers possessed greater diversity and richness, the community composition was distinct from that in the degraded and undisturbed rivers, and these changes were caused mainly by improved habitat characteristics, followed by physico-chemical variables and lastly spatial factors. Habitat restoration recovered the macroinvertebrate community composition in urban rivers in a positive way, which is in line with a meta-analysis result performed by Miller et al. (2010), whereas some studies showed small or none ecological effect of improved habitat conditions (Jähnig et al. 2010; Palmer et al. 2010). Restoration response may be varied both spatially and temporally, and the restoration approaches applied also influence the variance. Further study and evaluation of the river restoration programs would help to form an integrated view of restoration progress and efficiency of different restoration approaches, which provides water managers and policy makers an integrated guidance for future planning of ecological restoration and management strategies.
Conclusions
In this study, we examined the effect of habitat restoration on macroinvertebrate community composition in the urban rivers with and without restoration by comparing them to undisturbed rivers. The results support our hypothesis that habitat restoration positively altered the benthic macroinvertebrate community structure in comparison to that in degraded rivers. Attributing to the increase in substrate diversity and flow velocity, and accompanying decline in total nitrogen and total organic chemical in the surface water, habitat restoration induced higher values in diversity, in richness, and in abundance of macroinvertebrate, and higher richness and abundance of less tolerant EPT taxa. This study supports the hypothesis that applying habitat restoration in river management enhances habitat heterogeneity and improves the water quality, which can in turn stimulate the shift of macroinvertebrate community composition in urban rivers. Accordingly, habitat restoration is an efficient approach to recover the aquatic biodiversity in degraded urban rivers and to enhance river ecosystem health for freshwater conservation and management.
Data availability
The data and materials used to support the findings of this study are shared by the requesting author.
References
Al-Shami SA, Rawi CSM, Ahmad AHS, Hamid A, Nor SAM (2011) Influence of agricultural, industrial, and anthropogenic stresses on the distribution and diversity of macroinvertebrates in Juru River Basin, Penang. Malaysia Ecotox Environ Safe 74:1195–1202. https://doi.org/10.1016/j.ecoenv.2011.02.022
Arimoro FO (2009) Impact of rubber effluent discharges on the water quality and macroinvertebrate community assemblages in a forest stream in the Niger Delta. Chemosphere 77:440–449. https://doi.org/10.1016/j.chemosphere.2009.06.031
Beechie T, Richardson JS, Gurnell AM, Negishi J (2012) Watershed processes, human impacts, and process-based restoration. In: Stream and watershed restoration: a guide to restoring riverine processes and habitats. John Wiley & Sons, Ltd. https://doi.org/10.1002/9781118406618.ch2
Beermann AJ, Elbrecht V, Karnatz S, Ma L, Matthaei CD, Piggott JJ, Leese F (2018) Multiple-stressor effects on stream macroinvertebrate communities: a mesocosm experiment manipulating salinity, fine sediment and flow velocity. Sci Total Environ 4:961–971. https://doi.org/10.1016/j.scitotenv.2017.08.084
Bernhardt ES, Sudduth EB, Palmer MA, Allan JD, Meyer JL, Alexander G, Follastad-Shah J, Hassett B, Jenkinson R, Lave R, Rumps J, Pagano L (2007) Restoring rivers one reach at a time: results from a survey of US river restoration practitioners. Restor Ecol 15:482–493. https://doi.org/10.1111/j.1526-100X.2007.00244.x
Besacier-Monbertrand AL, Paillex A, Castella E (2014) Short-term impacts of lateral hydrological connectivity restoration on aquatic macroinvertebrates. River Res Appl 30(5):557–570. https://doi.org/10.1002/rra.2597
Borcard D, Gillet F, Legendre P (2018) Numerical ecology with R, second ed. Springer International Publishing, Cham https://doi.org/10.1007/978-3-319-71404-2
Brasher AM (2003) Impacts of human disturbances on biotic communities in Hawaiian streams. BioScience 53:1052–1060. https://doi.org/10.1641/0006-35682003053[1052:IOHDOB]2.0.CO;2
Cabria MÁ, Barquín J, Juanes JA (2011) Micro distribution patterns of macroinvertebrate communities upstream and downstream of organic effluents. Water Res 45:1501–1511. https://doi.org/10.1016/j.watres.2010.11.028
Cai Y, Xu H, Vilmi A, Tolonen KT, Tang X, Qin B, Gong Z, Heino J (2017) Relative roles of spatial processes, natural factors and anthropogenic stressors in structuring a lake macroinvertebrate metacommunity. Sci Total Environ 601–602:1702–1711. https://doi.org/10.1016/j.scitotenv.2017.05.264
Core Team R (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Dewson ZS, James AB, Death RG (2007) A review of the consequences of decreased flow for instream habitat and macroinvertebrates. J North Am Benthol Soc 26:401–415. https://doi.org/10.1899/06-110.1
Dos Santos SD, Molineri C, Reynaga M, Basualdo C (2011) Which index is the best to assess stream health? Ecol Indic 11:582–589. https://doi.org/10.1016/j.ecolind.2010.08.004
Douglas M, Lake PS (1994) Species richness of stream stones - an investigation of the mechanisms generating the species-area relationship. Oikos 69:387–396. https://doi.org/10.2307/3545851
Duan XH, Wang ZY, Xu MZ (2010) Benthic macroinvertebrate and application in the assessment of stream ecology. Tsinghua University Press, Beijing, China In Chinese
Dudley TL, Cooper SD, Hemphill N (1986) Effects of macroalgae on a stream invertebrate community. J N Am Benthol Soc 52:93–106. https://doi.org/10.2307/1467864
Elbrecht V, Beermann AJ, Goessler G, Neumann J, Tollrian R, Wagner R, Wlecklik A, Piggott JJ, Matthaei CD, Leese F (2016) Multiple-stressor effects on stream invertebrates: a mesocosm experiment manipulating nutrients, fine sediment and flow velocity. Freshw Biol 61(4):362–375. https://doi.org/10.1111/fwb.12713
Englund G, Maimqvist B, Zhang YX (1997) Using predictive models to estimate effects of flow regulation on net-spinning caddis larvae in North Swedish rivers. Freshw Biol 37:687–697. https://doi.org/10.1046/j.1365-2427.1997.00178.x
Ernst AG, Warren DR, Baldigo BP (2012) Natural-channel-design restorations that changed geomorphology have little effect on macroinvertebrate communities in headwater streams. Restor Ecol 20:532–540. https://doi.org/10.1111/j.1526-100X.2011.00790.x
Flores L, Giorgi A, Gonzalez JM, Larranaga A, Diez JR, Elosegi A (2017) Effects of wood addition on stream benthic invertebrates differed among seasons at both habitat and reach scales. Ecol Eng 106:116–123. https://doi.org/10.1016/j.ecoleng.2017.05.036
Frainer A, Polvi LE, Jansson R, McKie BG (2018) Enhanced ecosystem functioning following stream restoration: the roles of habitat heterogeneity and invertebrate species traits. J Appl Ecol 55(1):377–85. https://doi.org/10.1111/1365-2664.12932
Garcia X-F, Schnauder I, Pusch MT (2012) Complex hydromorphology of meanders can support benthic invertebrate diversity in rivers. Hydrobiologia 685:49–68. https://doi.org/10.1007/s10750-011-0905-z
Geist J, Hawkins SJ (2016) Habitat recovery and restoration in aquatic ecosystems: current progress and future challenges. Aquat Conserv-Mar Freshw Ecosyst 26:942–962. https://doi.org/10.1002/aqc.2702
Guo K, Wu NC, Wang C, Yang DG, He YF, Luo JB, Chai Y, Duan M, Huang X, Riis T (2019) Trait dependent roles of environmental factors, spatial processes and grazing pressure on lake phytoplankton metacommunity. Ecol Indic 103:312–320. https://doi.org/10.1016/j.ecolind.2019.04.028
Heatherly T, Whiles MR, Royer TV, David MB (2007) Relationships between water quality, habitat quality, and macroinvertebrate assemblages in Illinois streams. J Environ Qual 36(6):1653–1660. https://doi.org/10.2134/jeq2006.0521
Hilsenhoff WL (1988) Rapid field assessment of organic pollution with a family-level biotic index. J N Am Benthol Soc 71:65–68. https://doi.org/10.2307/1467832
Ilarri MI, Amorim L, Souza AT, Sousa R (2018) Physical legacy of freshwater bivalves: effects of habitat complexity on the taxonomical and functional diversity of invertebrates. Sci Total Environ 634:1398–1405. https://doi.org/10.1016/j.scitotenv.2018.04.070
Jähnig SC, Lorenz AW (2008) Substrate-specific macroinvertebrate diversity patterns following stream restoration. Aquat Sci 703:292–303. https://doi.org/10.1007/s00027-008-8042-0
Jähnig SC, Brabec K, Buffagni A, Erba S, Lorenz AW, Ofenböck T, Verdonschot PFM, Hering D (2010) A comparative analysis of restoration measures and their effects on hydromorphology and benthic invertebrates in 26 central and southern European rivers. J Appl Ecol 47:671–680. https://doi.org/10.1111/j.1365-2664.2010.01807.x
Jones JI, Murphy JF, Collins AL, Sear DA, Naden PS, Armitage PD (2012) The impact of fine sediment on macro-invertebrates. River Res Appl 28:1055–1071. https://doi.org/10.1002/rra.1516
Kail J, Brabec K, Poppe M, Januschke K (2015) The effect of river restoration on fish, macroinvertebrates and aquatic macrophytes: a meta-analysis. Ecol Indic 58:311–321. https://doi.org/10.1016/j.ecolind.2015.06.011
Kail J, McKie B, Verdonschot PFM, Hering D (2016) Preface: Effects of hydromorphological river restoration—a comprehensive field investigation of 20 European projects. Hydrobiologia 769(1):1–2. https://doi.org/10.1007/s10750-016-2659-0
Kalyoncu H, Gülboy H (2009) Benthic macroinvertebrates from dari¨oren and isparta streams Isparta/Turkey-biotic indices and multivariate analysis. J Appl Biol Sci 31:79–86
Kärnä O-M, Heino J, Grönroos M, Hjort J (2018) The added value of geodiversity indices in explaining variation of stream macroinvertebrate diversity. Ecol Indic 94(1):420–429. https://doi.org/10.1016/j.ecolind.2018.06.034
Karr JR (1999) Defining and measuring river health. Freshw Biol 41:221–234. https://doi.org/10.1046/j.1365-2427.1999.00427.x
Knouft JH, Chu ML (2015) Using watershed-scale hydrological models to predict the impacts of increasing urbanization on freshwater fish assemblages. Ecohydrology 8:273–285. https://doi.org/10.1002/eco.1506
Kondolf GM (1997) Application of the pebble count: Notes on purpose, method, and variants. J Am Water Resour Assoc 33:79–87 https://doi.org/10.1111/j.1752-1688.1997.tb04084.x
Laasonen P, Muotka T, Kivijarvi I (1998) Recovery of macroinvertebrate communities from stream habitat restoration. Aquat Conserv-Mar Freshw Ecosyst 81:101–113. https://doi.org/10.1002/SICI1099-0755199801/028:1/3C101::AID-AQC251/3E3.0.CO;2-4
Lake PS, Palmer MA, Biro P, Cole J, Covich AP, Dahm C et al (2000) Global change and the biodiversity of freshwater ecosystems. Bioscience 50:1099–1107. https://doi.org/10.1641/0006-35682000050[1099:GCATBO]2.0.CO;2
Lepori F, Palm D, Brannas E, Malmqvist B (2005) Does restoration of structural heterogeneity in streams enhance fish and macroinvertebrate diversity? Ecol Appl 15:2060–2071. https://doi.org/10.1890/04-1372
Li K, He CG, Zhuang J, Zhang ZX, Xiang HY, Wang ZQ, Yang H, Sheng L (2015) Long-term changes in the water quality and macroinvertebrate communities of a subtropical river in south China. Water 7:63–80. https://doi.org/10.3390/w7010063
Lin QY, Sekar R, Marrs RH, Zhang YX (2019) Effect of river ecological restoration on biofilm microbial community composition. Water 11:6. https://doi.org/10.3390/w11061244
Lin QY, Zhang YX, Marrs RH, Sekar R, Luo X, Wu NC (2020) Evaluating ecosystem functioning following river restoration: the role of hydromorphology, bacteria, and macroinvertebrates. Sci Total Environ 743:140583. https://doi.org/10.1016/j.scitotenv.2020.140583
Lorenz S, Leszinski M, Graeber D (2016) Meander reconnection method determines restoration success for macroinvertebrate communities in a German lowland river. Int Rev Hydrobiol 101(3–4):123–131. https://doi.org/10.1002/iroh.201501823
Louhi P, Mykrä H, Paavola R, Huusko A, Vehanen T, Mäki-Petäys A, Muotka T (2011) Twenty years of stream restoration in Finland: little response by benthic macroinvertebrate communities. Ecol Appl 21(6):1950–1961. https://doi.org/10.1890/10-0591.1
Luo K, Hu X, He Q, Wu Z, Cheng H, Hu Z, Mazumder A (2017) Impacts of rapid urbanization on the water quality and macroinvertebrate communities of streams: a case study in Liangjiang New Area. China Sci Total Environ 621:1601–1614. https://doi.org/10.1016/j.scitotenv.2017.10.068
Mandaville SM (2002) Bioassessment of freshwaters using benthic macroinvertebrates-a primer. Soil Water Conservation Society of Metro Halifax. Dartmouth, NS, Canada
Martín EJ, Ryo M, Doering M, Robinson CT (2018) Evaluation of restoration and flow interactions on river structure and function: channel widening of the Thur River, Switzerland. Water 10(4):439. https://doi.org/10.3390/w10040439
Matthaei CD, Diehl S (2005) Large-scale river restoration enhances geomorphological diversity and benthic diversity. Environmental Science.
Merritt RW, Cummins KW, Berg MB (2008) An introduction to the aquatic insects of North America. Kendall Hunt Publishing Company, Dubuque, IA
Miller SW, Budy P, Schmidt JC (2010) Quantifying macroinvertebrate responses to in-stream habitat restoration: applications of meta-analysis to river restoration. Restor Ecol 18:8–19. https://doi.org/10.1111/j.1526-100X.2009.00605.x
Obolewski K, Glinskalewczuk K, Ozgo M, Astel A (2016) Connectivity restoration of floodplain lakes: an assessment based on macroinvertebrate communities. Hydrobiologia 7741:23–37. https://doi.org/10.1007/s10750-015-2530-8
Palmer MA, Menninger HL, Bernhardt ES (2010) River restoration, habitat heterogeneity and biodiversity: a failure of theory or practice? Freshw Biol 55:205–222. https://doi.org/10.1111/j.1365-2427.2009.02372.x
Palmer MA, Hondula KL, Koch BJ (2014) Ecological restoration of streams and rivers: shifting strategies and shifting goals. Annu Rev Ecol Evol Syst 45:247–269. https://doi.org/10.1146/annurev-ecolsys-120213-091935
Patang F, Soegianto A, Hariyanto S (2018) Benthic macroinvertebrates diversity as bioindicator of water quality of some rivers in east Kalimantan, Indonesia. Hindawi. Int J Ecol 1–11. https://doi.org/10.1155/2018/5129421
Piggott JJ, Townsend CR, Matthaei CD (2015) Climate warming and agricultural stressors interact to determine stream macroinvertebrate community dynamics. Glob Chang Biol 21:1887–1906. https://doi.org/10.1111/gcb.12861
Plafkin JL, Barbour JL, Porter MT, Gross KD, Hughes RM (1989) Rapid bioassessment protocols for use in streams and rivers: benthic macroinvertebrates and fish. United States Environmental Protection Agency. Washington, D.C.
Poff NL (1997) Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. J N Am Benthol Soc 16(2):391–409. https://doi.org/10.2307/1468026
Rico A, Van den Brink PJ, Leitner P, Graf W, Focks A (2016) Relative influence of chemical and non-chemical stressors on invertebrate communities: a case study in the Danube River. Sci Total Environ 571:1370–1382. https://doi.org/10.1016/j.scitotenv.2016.07.087
Ryan PA (1991) Environmental-effects of sediment on new-Zealand streams - a review N. Z. J. Mar. Freshw Res 25:207–221. https://doi.org/10.1080/00288330.1991.9516472
Schmutz S, Jurajda P, Kaufmann S, Lorenz AW, Muhar S, Paillex A et al (2016) Response of fish assemblages to hydromorphological restorationin central and northern European rivers. Hydrobiologia 769(1):67–78. https://doi.org/10.1016/j.geomorph.2020.107447
Shannon CE (1997) The mathematical theory of communication. M D Computing 14:306–317
Shao ML, Xie ZC, Ye L, Cai QH (2006) Monthly change of community structure of zoobenthos in Xiangxi Bay after impoundment of three gorges reservoir. Acta Hydrob Sin 30:64–69. In Chinese. https://doi.org/10.1007/s11515-007-0034-2
Shi X, Liu J, You X, Bao K, Meng B (2019) Shared effects of hydromorphological and physico-chemical factors on benthic macroinvertebrate integrity for substrate types. Ecol Indic 105:406–414. https://doi.org/10.1016/j.ecolind.2018.02.028
Smith M, Kay W, Edward D, Papas P, Richardson KSJ, Simpson J et al (1999) AusRivAS: using macroinvertebrates to assess ecological condition of rivers in Western Australia. Freshw Biol 41:269–282. https://doi.org/10.1046/j.1365-2427.1999.00430.x
Spänhoff B, Arle J (2007) Setting attainable goals of stream habitat restoration from a macroinvertebrate view. Restor Ecol 15:317–320. https://doi.org/10.1111/j.1526-100X.2007.00216.x
Strayer DL (2006) Challenges for freshwater invertebrate conservation. J N Am Benthol Soc 25:271–287. https://doi.org/10.1899/0887-3593200625[271:CFFIC]2.0.CO;2
Taniguchi H, Tokeshi M (2004) Effects of habitat complexity on benthic assemblages in a variable environment. Freshw Biol 49(9):1164–1178. https://doi.org/10.1111/j.1365-2427.2004.01257.x
Torres-Mellado GA, Escobar I, Palfner G, Casanova-Katny MA (2012) Mycotrophy in Gilliesieae, a threatened and poorly known tribe of Alliaceae from central Chile. Rev Chil Hist Nat 85:179–186. https://doi.org/10.4067/S0716-078X2012000200004
Tullos D, Penrose D, Jennings G, Cope W (2009) Analysis of functional traits in reconfigured channels: implications for the bioassessment and disturbance of river restoration. J N Am Benthol Soc 28:80–92. https://doi.org/10.1899/07-122.1
Turley MD, Bilotta GS, Chadd RP, Extence CA, Brazier RE, Burnside NG, Pickwell AGG (2016) A sediment-specific family-level biomonitoring tool to identify the impacts of fine sediment in temperate rivers and streams. Ecol Indic 70:151–165. https://doi.org/10.1016/j.ecolind.2016.05.040
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137. https://doi.org/10.1139/f80-017
White JC, Krajenbrink HJ, Hill MJ, Hannah DM, House A, Wood PJ (2019) Habitat-specific invertebrate responses to hydrological variability, anthropogenic flow alterations, and hydraulic conditions. Freshw Biol 64:555–576. https://doi.org/10.1111/fwb.13242
Zan RB, Kondolf GM, Riostouma B (2017) Evaluating stream restoration project: what do we learn from monitoring? Water 93. https://doi.org/10.3390/w9030174
Zhang YX, Richardson JS, Negishi JN (2004) Detritus processing, ecosystem engineering, and benthic diversity: a test of predator-omnivore interference. J Anim Ecol 73:756–766. https://doi.org/10.1111/j.0021-8790.2004.00849.x
Zhang YX, Richardson JS, Pinto X (2009) Catchment-scale effects of forestry practices on benthic invertebrate communities in Pacific coastal stream ecosystems. J Appl Ecol 46:1292–1303. https://doi.org/10.1111/j.1365-2664.2009.01718.x
Zhang YX, Dudgeon D, Cheng DS, Thoe W, Fok L, Wang ZY, Lee JHW (2010) Impacts of land use and water quality on macroinvertebrate communities in the Pearl River drainage basin, China. Hydrobiologia 652:71–88. https://doi.org/10.1007/s10750-010-0320-x
Zhang YX, Juvigny-Khenafou N, Xiang HY, Lin QY, Wu ZJ (2019) Multiple stressors in China’s freshwater ecoregions. In: Sabater S, Elosegi A, Ludwig R (eds). Multiple stress in river ecosystems: status, impacts and prospects for the future. 1st ed. Elsevier, 193–204. https://doi.org/10.1016/B978-0-12-811713-2.00011-X
Acknowledgements
We are grateful to Eduardo Medina-Roldan, Zheng Chen, and Yi Zou for their comments on this field research; Hongyong Xiang, Noel Juvigny-Khenafou, Qingsheng Zhu, and Hucheng Chang for their assistance with field work; and Kun Guo for his assistance in R processing.
Funding
This research was funded by Xi’an Jiaotong-Liverpool University (Grant number RDF-15-01-50), the Natural Science Foundation of Jiangsu Province (Grant number BK20171238), and the Foundation of Key Laboratory of Southwest China Wildlife Resources Conservation, China West Normal University (XNYB18-06).
Author information
Authors and Affiliations
Contributions
Conceptualization, Q.L. and Y.Z.; methodology, Q.L.; software, Q.L.; validation, Q.L. and Y.Z.; formal analysis, Q.L. and R.M.; investigation, Q.L.; resources, Y.Z.; data curation, Q.L.; original draft preparation, Q.L.; review and editing, Q.L., R.S., R.M., X.L., and Y.Z.; visualization, Q.L.; supervision, R.S., R.M., and Y.Z.; project administration, Y.Z.; and funding acquisition, Y.Z and N.W.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Thomas Hein
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Lin, Q., Zhang, Y., Marrs, R. et al. The effect of habitat restoration on macroinvertebrate communities in Shaoxi rivers, China. Environ Sci Pollut Res 29, 677–689 (2022). https://doi.org/10.1007/s11356-021-15559-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15559-x