Abstract
Bisphenol A (BPA) and di(2-ethylhexyl)phthalate (DEHP) are abundant endocrine disrupting chemicals (EDCs). In recent years, studies showed that EDCs may lead to neurodevelopmental diseases. The effects of prenatal exposure to these chemicals may have serious consequences. Moreover, exposure to EDCs as a mixture may have different effects than individual exposures. The present study aimed to determine the toxicity of BPA and/or DEHP on central nervous system (CNS) and neuroendocrine system in prenatal and lactational period in Sprague-Dawley rats. Pregnant rats were randomly divided into four groups: control (received vehicle); BPA group (received BPA at 50 mg/kg/day); DEHP group (received DEHP at 30 mg/kg/day); and combined exposure group (received both BPA at 50 mg/kg/day and DEHP at 30 mg/kg/day) during pregnancy and lactation by oral gavage. At the end of lactation, male offspring (n = 6) were randomly grouped. The alterations in the brain histopathology, neurotransmitter levels and enzyme activities in the cerebrum region, oxidative stress markers, and apoptotic effects in the hippocampus region were determined at adulthood. The results showed that exposure to EDCs at early stages of life caused significant changes in lipid peroxidation, total GSH and neurotransmitter levels, and activities of neurotransmitter-related enzymes. Moreover, BPA and/or DEHP led to apoptosis and histopathologic alterations in the hippocampus. Therefore, we can suggest that changes in oxidant/antioxidant status, as well as in neurotransmitters and related enzymes, can be considered as the underlying neurotoxicity mechanisms of BPA and DEHP. However, more mechanistic studies are needed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endocrine disrupting chemicals (EDCs) disrupt the normal endocrine functions and alter the endocrine phenotype of the exposed individual through various mechanisms (Diamanti-Kandarakis et al. 2009). Bisphenol A (BPA) and di (2-ethylhexyl) phthalate (DEHP) are EDCs that are widely present in the environment. BPA is widely used in production of polycarbonate plastics. DEHP is the one of the most widely used phthalic acid ester. Both of these compounds are used as plasticizers (Kabir et al. 2015).
In the last decades, studies were performed that show the exposure to EDCs might lead to cognitive deficiencies, disrupted neurodevelopment along with hyperactivity, attention deficit problems, aggression, and depression (Ipapo et al. 2017; Perera et al. 2016; Arbuckle et al. 2016; Harley et al. 2013). It was reported that EDCs can have a wide range of effects on brain development and many different mechanisms are proposed. Some of these effects are suggested to be through the neuroendocrine system (Colborn 2004). The neuroendocrine system is a complex system consisting of neurons, glands, non-endocrine tissues, humoral signals, hormones, and neurochemicals that function to regulate physiological and behavioral processes (Waye and Trudeau 2011). There is a growing evidence that antropogenic chemicals such as EDCs may act on the neuroendocrine system and they may affect peripheral organ systems and physiological processes (Adewale et al. 2011; Andrade et al. 2006; McCaffrey et al. 2013).
Functions of neuroendocrine system are largely related to the functions of neurotransmitters. Evidence suggest that brain neurotransmitter systems (i.e., dopaminergic, adrenergic, serotonergic, and cholinergic systems) play crucial roles in the activation and/or inhibition of neuroendocrine system. This relationship necessitates the examination of neurotransmitters for the evaluation of neuroendocrine system functions (Sullivan and Dufresne 2006; Bauer et al. 2002; Ohno et al. 1987). The regulation of both dopamine (DA) and serotonin (5-HT) production by estrogen-dependent mechanisms makes these systems vulnerable to the adverse effects of EDCs. Previous studies have reported that BPA and phthalate exposure might lead to imbalance in the levels of these neurotransmitters in various regions of the brain (Honma et al. 2006; Nakamura et al. 2010; Matsuda et al. 2010; Matsuda et al. 2012; Wang et al. 2016). Most EDCs can accumulate in the brain, may act directly on neuronal functions and affect normal brain development. They may also cause neurotoxicity through other mechanisms, like oxidative stress. Brain is vulnerable to oxidative damage and this might be one of the underlying factors of high incidence of different neurodegenerative diseases throughout the globe (Grandjean and Landrigan 2014).
The results of exposure to different EDCs can vary due to the chemical’s intrinsic properties, dose, and the timing of exposure. It is predicted that the exposure during the developmental stages (prenatal and neonatal) of the nervous system may have more serious consequences (Negri-Cesi et al. 2008). EDC exposure can be from various sources and exposure to multiple EDCs is inevitable in daily life. The effects of combined exposures are expected to be different from the effects of individual exposures (Nohynek et al. 2013).
Concerning all the available data, our aim was to evaluate neuroendocrine disrupting effects of exposure to BPA and/or DEHP in prenatal and lactational periods in Sprague-Dawley rats. We have investigated their effects on dopaminergic, serotonergic, and cholinergic systems and whether oxidative stress is an underlying factor on histopathological and apoptotic changes in hippocampus.
Experimental procedures
Chemicals, reagents and kits
BPA, DEHP, bovine serum albumin (BSA), and EDTA were purchased from Sigma-Aldrich (Mannheim, Germany). Digitonin was purchased from BDH Chemicals (Radnor, PA). 4-(2-Hydroxyethyl)piperazine-1-ethanesulfonic acid (HEPES) and Dulbecco’s phosphate buffer saline (DPBS) were purchased from Biowest (Riverside, MO). Hematoxylin crystals, hydrogen peroxide, and Tris were purchased from Merck (Kenilworth, NJ).
Thiobarbituric acid reactive substances (TBARS) assay kit and glutathione (GSH) assay kit were purchased from Cayman Chemical (Ann Arbor, MI). DA levels were measured by a specific rat ELISA kit of Cusabio (Hubei, China). 5-HT levels were determined by the specific rat ELISA kit of LifeSpan Biosciences (Seattle, WA). Monoamine Oxidase Assay Kit [Monoamine oxidase A (MAO-A) and monoamine oxidase B (MAO-B)] was obtained from OxiSelect™, Cell Biolabs, Inc. (San Diego, CA). Acetylcholine esterase (AChE) activity assay kit was purchased from from BioVision (San Francisco, CA). Rat dopamine transporter (DAT) assay kit was from Elabscience (Houston, TX). In Situ Cell Death Detection Kit, POD was from Sigma-Aldrich (Mannheim, Germany).
Animals and exposure
Pregnant Sprague-Dawley rats were supplied from Gazi University Laboratory of Animal Breeding and Experimental Research Center (GUDAM). The study was approved by Gazi University Animal Ethics Committee (G.U.ET–18.043).
The animals were randomly divided into 4 groups with 3 pregnant rats in each. Animals were housed in cages, at room temperature 20 ± 2 °C, relative humidity 40%, and 12 h light/dark cycle. Both water and food were given ad libitum. The study grouped as follows:
-
1.
Control (vehicle) group: Corn oil was administered by intra-gastric gavage (i.g)
-
2.
BPA group: BPA (dissolved in 1 ml of 96% ethanol and diluted with corn oil) was administered as 50 mg/kg/day by i.g.
-
3.
DEHP: DEHP (dissolved in corn oil) was administered as 30 mg/kg/day by i.g.
-
4.
BPA and DEHP group: BPA was administered as 50 mg/kg/day and DEHP was administered as 30 mg/kg/day.
Rats were dosed from gestational day (GD) 6 to postnatal day (PND) 21 (end of lactational period). Offspring were not dosed. The male offspring in all groups were weaned at 21 days. The study groups consisted of randomly selected male rats from each group (n = 6). At postnatal 12th week, animals were euthanized under the anesthesia and brain tissues were removed quickly. Cerebrum tissues were used for the measurement of antioxidant parameters and neurotransmitter, neurotransmitter transporter, and related enzyme levels/activities. All tissues were kept at – 80 οC until the tissue homogenates were prepared.
Tissue preparation
Brain tissues were washed in physiological and deionized water and divided into two hemispheres. One hemisphere (left) of the brain tissue was dissected into three parts (cerebrum, midbrain, and hindbrain). Cerebrum tissue was frozen into liquid nitrogen after dissection and kept at – 80 °C until analysis of oxidative stress and neuroendocrine parameters. The other hemisphere (right) was used for histopathological analysis and determination of apoptosis. DA and noradrenaline (NA) in the prefrontal cortex (PFC) modulate superior cognitive functions, and are involved in the etiology of depressive and psychotic symptoms. As PFC consists of the largest part of cerebrum, we choose cerebrum to determine the neurotransmitter, neurotransmitter transport, and related enzyme levels/activities (Devoto and Flore 2006). Neurons in the entorhinal cortex, hippocampus CA1 region, frontal cortex, and amygdala are the populations of neurons most sensitive to the oxidative stress and therefore to neurodegeneration. We have chosen cerebrum tissue as it contains the frontal cortex and the cerebrum is a bigger tissue that enables the researcher to perform more experiments. We did not chose the hippocampus to make these measurements, as only CA1 region is sensitive to neuropathological alterations (Wang and Michaelis 2010). Moreover, literature suggests that phthalates and BPA mainly cause cytoarchitectural alterations (i.e., condensed nuclei, vacuole formation and remarkable degeneration, shrinkage of pyramidal neurons in CA1 and CA3 regions; disorganized hilar cells and hyperplasia in dentate gyrus) and apoptosis in hippocampus of particularly male offspring. These changes might lead to cognitive impairment and endocrine dysfunction in immature animals. Therefore, for the measurement of apoptosis, we have chosen the hippocampal region (Mahaboob Basha and Radha 2020; Wang et al. 2020). For the experiments, 100–150 mg of cerebrum tissues were used.
Histopathological examination
For light microscopy, Bouin’s solution was used to fix the cerebrum tissue samples. Later, they were processed for paraffin sections (5 μm). For routine histological investigations, staining was made by using hematoxylin and eosin for hippocampus region. Leica DM6000B microscope (Wetzlar, Germany) was used for examinations and Leica DC490 digital camera (Wetzlar, Germany) was used to photograph the images.
Determination of glutathione
Cerebrum tissues were homogenized in Tris/DTPA/PMSF buffer, centrifuged in 4000 rpm for 10 min, and later deproteinated with equal volume of metaphosphoric acid (12.5 M). After deproteination, total GSH was measured using a glutathione assay kit, based on the reaction of the sulfhydryl group of GSH with 5,5′-dithio-bis-(2-nitrobenzoic) acid (DTNB) to produce a yellow colored 5-thio-2-nitrobenzoic acid (TNB), and in the same cycle, GSH was simultaneously converted to GS-TNB. The absorbance values of the samples were measured at 414 nm.
Results were expressed in nmol/mg protein (Habig et al. 1974).
Determination of lipid peroxidation
Cerebrum tissue was homogenized in Tris/diethylene triamine pentaacetic acid (DTPA)/ phenylmethylsulfonyl uoride (PMSF) buffer and centrifuged at 4000 rpm for 10 min. Malondialdehyde (MDA) levels, as a biomarker of lipid peroxidation, were measured using a TBARS Assay kit. The kit protocol was based on the colorimetric measurement of the MDA-thiobarbituric acid (TBA) complex, which forms under high temperature and acidic conditions. The color intensity of MDA-TBA complex was measured at 530 nm spectrophotometrically. The calculations of the amount of MDA were made by a standard curve and the results were expressed as nmol/mg protein. Results were expressed as nmol/mg protein (Richard et al. 1992).
Neurotransmitter levels
After cerebrum tissues were isolated from other parts of the brain, they were homogenized in 1X phosphate buffer saline (PBS). The samples were centrifuged in 4000 rpm for 10 min.
DA levels were detected by a specific rat sandwich ELISA kit using quantitative sandwich immunoassay. The plate was coated with specific DA antibodies. Samples and standards were added to each well. After other substances that did not bind to these antibodies were removed, specific biotin-conjugated antibodies were added to the wells. Later, avidin-horse radish peroxidase (HRP) conjugate was added. After incubation, wells were washed, wash buffer was removed, and the blue color was measured at 450 nm. Calculations were made by using standard curves. DA levels were expressed as ng/ml.
5-HT levels were measured by a specific rat kit based upon a competitive ELISA principle. The plate was coated with specific 5-HT antibody. Samples and standards were added to each well along with HRP-antigen conjugate. Later, TMB reagent was added and kit was incubated. After stop solution was added, color development was measured at 450 nm. The color development was inversely correlated to the amount of 5-HT in the samples. Calculations were made by using standard curves. 5-HT levels were expressed as ng/ml.
Monoamine oxidase A and B activities
Activities of MAO-A and MAO-B were measured in cerebrum mitochondria. The isolation of mitochondria was performed by using the differential centrifugation method (Amigo et al. 2016). Later, MAO-A and MAO-B activities in samples were measured by a spectrophotometric assay kit by using different substrates while hydrogen peroxide was being formed. If MAO-A was to be measured, an inhibitor for MAO-B was added or vice versa. Hydrogen peroxide later reacts with a prob to form a pink/reddish compound in the presence of HRP conjugate. The absorbance was measured at 540 nm. Results were expressed in mU/mg protein.
Acetylcholine esterase activity
For measurement of AChE activity, cerebrum tissues were homogenized in 1X phosphate buffer saline (PBS). Tissues were later centrifuged (4000 rpm, 10 min) and AChE activities of the samples were measured by a colorimetric AChE activity assay kit that employs the kinetic measurement of AChE activity. Briefly, the AChE in the samples converts acetylcholine substrate to choline, which was then oxidized by choline oxidase (CO) to produce an intermediate. The intermediate reacted with a highly specific probe to generate color. The color intensity was measured at 570 nm. Results were given as mU/mg protein.
Dopamine transporter levels
Cerebrum tissues were homogenized in 1X PBS. Later, homogenates were centrifuged (4000 rpm, 10 min). DAT levels were measured by a specific rat ELISA kit by using quantitative sandwich immunoassay. Briefly, standards or samples were added to the ELISA plate that was pre-coated with an antibody specific to rat DAT. Then, a biotinylated detection antibody specific for rat DAT and Avidin-HRP conjugate were added to each well and incubated. Free components were washed away and the substrate solution was added to each well. The enzyme-substrate reaction was terminated by the addition of stop solution. The absorbance (that was proportional to the concentration of rat DAT) of the final yellow product was measured spectrophotometrically at 450 nm. Then, the concentrations of DAT in the samples were calculated by a standard curve. Results were given as nmol/mg protein.
Determination of apoptosis
Apoptosis in the hippocampus was determined by terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling (TUNEL) method by using an “In Situ Cell Death Detection Kit, POD”. After staining, TUNEL positive neuron and glia cell counts in hippocampus CA1, CA2, and CA3 regions were counted by using a light microscope (Leica DM 6000) (Gavrieli et al. 1992; Gorczyca et al. 1992).
Statistical analysis
A statistics program (Statistical Package for Social Sciences Program, SPSS 17.0, Chicago, IL) was used for statistical analysis. Kruskal–Wallis one-way analysis of variance was used to determine the differences among the groups, and later, Mann–Whitney U test was used to identify the differences between the study groups. Results are expressed as mean ± standard deviation (SD). p values < 0.05 were considered as statistically significant.
Results
Relative brain weights
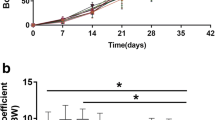
The relative brain weights (brain weight/body weight) are given in Fig. 1. The relative brain weight in the DEHP group increased by 18% (p < 0.05 vs. control). An insignificant elevation (15%) was determined in BPA + DEHP group vs. control group (p > 0.05).
Histopathological examination
Several degenerated neurons and microglia were observed between the pyramidal neurons in the hippocampus CA1 region of both BPA and DEHP groups. In addition, significant perivascular edema was observed in the CA1 region in the DEHP group. In the CA3 region of the hippocampus, we observed pyramidal neurons contracted in their cytoplasm and there were empty spaces around these neurons. In the CA1, CA2, and CA3 region of the hippocampus tissue of the BPA + DEHP group, shrunken and degenerated neurons with dark stained cytoplasm were determined and condensed nucleated glia cells were observed. In addition, unlike other study groups, neuron-free areas were detected. All findings indicate the presence of degeneration and histopathological changes in the hippocampal region, either by individual exposures or by combined exposure. Light micrographs of the hippocampus tissues are shown in Fig. 2.
Light photomicrographs of the hippocampus tissues in the study groups. In the CA1, CA2, and CA3 region of the hippocampus of the control group, pyramidal neurons and glia cells were observed. Degenerate neurons (white arrow) and microglia cells with flat nuclei (black arrow) were observed in study groups. (hematoxylin-eosin × 200)
Glutathione levels
Total GSH levels of brain tissue in the study groups are given in Fig. 3 a. Total GSH levels were markedly increased in all of the study groups when compared to control (BPA group, 146%; DEHP group, 92%; BPA + DEHP group, 144% vs. control; p < 0.05, all).
Total glutathione and malondialdehyde levels in the study groups. a Total glutathione levels; b Malondialdehyde levels. GSH, total glutathione; MDA, malondialdehyde. *p < 0.05, **p < 0.01. All of the study groups have significantly higher total GSH levels vs. control group. The difference between BPA and DEHP groups is statistically significant. The difference between DEHP and BPA + DEHP groups is also statistically significant. All of the study groups have higher MDA levels vs. control group. The difference between BPA and DEHP groups is statistically significant. The difference between DEHP and BPA + DEHP groups is also statistically significant
Lipid peroxidation
Lipid peroxidation levels in the study groups are shown in Fig. 3 b. There was a marked elevation in MDA levels in all study groups compared to the control group (BPA group, 122%; DEHP group, 52%; BPA + DEHP group, 99%, vs. control; p < 0.05, all).
Neurotransmitter levels
Neurotransmitter levels in the study groups are given in Fig. 4. In all study groups, we observed marked decreases in DA levels compared to the control group (BPA group, 41%; DEHP group, 35%; BPA + DEHP group, 47% vs. control; p < 0.05, all) (Fig. 4a). The decrease was even more significant in BPA + DEHP group. 5-HT levels were also significantly lower in all study groups vs. control (BPA and DEHP groups, 19%; BPA + DEHP group, 24%) (p < 0.05, all; Fig. 4b).
Neurotransmitter levels in the study groups. DA, dopamine; 5-HT, serotonin. *p < 0.05. All of the study groups have significantly lower DA levels vs. control group. The difference between other study groups is not significant. All of the study groups have lower 5-HT levels vs. control group. The difference between other study groups is not significant
Dopamine transporter levels
Dopamine transporter levels are given in Fig. 5. In the DEHP group, DAT levels decreased by 47.4% vs. control (p < 0.05). There was a ~ 16% insignificant decrease in DAT levels in both BPA and BPA + DEHP group (p > 0.05).
Monoamine oxidase A and B activities
MAO-A activity was unchanged in BPA group vs. control group, but markedly elevated in both DEHP and BPA + DEHP groups (53% and 26%, respectively vs. control) (Fig. 6a). While MAO-B activity did not markedly change in BPA and DEHP groups when compared to control, it was it was 6.7-fold higher in BPA + DEHP group vs. control (p < 0.05) (Fig. 6b).
MAO-A and MAO-B activities in the study groups. a MAO-A activity; b MAO-B activity. MAO-A, monoamine oxidase A; MAO-B, monoamine oxidase B. *p < 0.05. There are no significant differences in MAO-A activity between the groups. BPA + DEHP group has significantly higher MAO-B activity vs. all other study groups and control. The difference in MAO-B activity between BPA and DEHP groups is not statistically significant
Acetylcholine esterase activity
AChE activity of the study groups is shown in Fig. 7. AChE activity was higher in BPA (117%) and BPA + DEHP (106%) groups vs. control (p < 0.05, both). However, we observed a 19% increase in the DEHP group and it was not statistically significant (p > 0.05).
Apoptosis
Apoptotic cells were counted for three times on three consecutive days. Two researchers counted the apoptotic cells on the same day and the mean of the counting of these two researchers is given. The photomicrographs of TUNEL assay are shown in Fig. 8. The percentages of apoptotic neurons and glia in hippocampal sub-regions are given in Fig. 9.
Ratios of TUNEL positive cells to normal cells in hippocampus sub-regions (CA1, CA2, CA3) . *p < 0.05. In both CA1 and CA2 regions, all of the study groups have higher apoptotic neurons compared to control. The differences between the study groups are not significantly different. In CA3 region, only BPA + DEHP group show significantly higher apoptotic neurons vs. control. The differences between DEHP and BPA and DEHP and BPA + DEHP groups are not statistically significant. In CA1 region, BPA and BPA + DEHP groups have significantly higher apoptotic glial cells vs. control. The differences between DEHP and BPA and DEHP and BPA + DEHP groups are not statistically significant. In CA2 region, BPA group has significantly higher apoptotic glial cells vs. control. The differences between DEHP and BPA and BPA and BPA + DEHP groups are statistically significant. In CA3 region, none of the groups has statistically significant changes in apoptotic glia cells. The differences between DEHP and BPA and BPA and BPA + DEHP groups are not statistically significant
In TUNEL analysis, the mean percentage of apoptotic cells in neurons and glia cells (CA1, CA2, and CA3 regions) in the experimental groups was evaluated. In control, 49% of the neurons and 51% of the glia cells were found to be TUNEL positive in the CA1 region. In CA2 region, 49% of neurons and 53% of glia cells were found to be TUNEL positive. In CA3 region, 47% of neurons and 52% of glia cells were found to be TUNEL positive.
We observed that that 85% of neurons and 66% of glia cells were apoptotic in CA1 region in BPA group. In the CA2 region, 73% of the neurons and 78% of the glia cells were found to be TUNEL positive. In CA3 region, 69% of neurons and 67% of glia cells were TUNEL positive. The percentages of apoptotic neurons and glia increased in CA1, CA2, and CA3 regions in BPA group vs. control group and this increase was significant in CA1 and CA2 regions (p < 0.05, both).
In the DEHP group, 88% neurons in the CA1 region were apoptotic and 64% glia showed apoptosis. In CA2 region, 76% of neurons and 52% of glia cells were found to be TUNEL positive. It was observed that 74% of neurons and 54% of glia cells were TUNEL positive in CA3 region. The percentages of apoptotic neurons in CA1 and CA2 regions increased significantly in the DEHP group vs. control (p < 0.05).
In BPA + DEHP group, 91% neurons were apoptotic in the CA1 region and 69% glia cells showed apoptosis. In the CA2 region, 73% of neurons and 60% of glia cells were TUNEL positive. In the CA3 region, 73% of the neurons and 75% of the glia cells were TUNEL positive. In the BPA + DEHP group, apoptotic neuron percentage increased significantly in all three regions when compared to control (p < 0.05). There was a marked increase in apoptotic glia percentage in CA1 region (p < 0.05). Although the increase in CA3 region was higher than the control group, it was not statistically significant although there was a borderline significance (p = 0.051).
Discussion
The reprotoxicity of EDCs is well-documented in literature. However, little is known about their effects on CNS. It is emphasized that EDCs can affect brain development through many different mechanisms and some of these effects can be demonstrated through the neuroendocrine system, which provides integration between the brain and the peripheral endocrine system (Colborn 2004; Parent et al. 2011). The CNS and neuroendocrine system function in balance with all neurotransmitters, neuropeptides, and neuro-hormone systems. EDCs may disrupt this balance and cause various neurological damages. In recent years, the prevalence of neurodevelopmental diseases has increased along with a rise in the production of these toxic chemicals (Gore 2010).
The vulnerability of CNS to EDCs depends on doses and duration. In addition, the responses of different parts of the brain can vary (Negri-Cesi et al. 2008). The cerebrum is the central location where most brain functions are regulated (Meyer 2001). Therefore, we determined the effects of EDCs on brain functions by measuring neuroendocrine and oxidative stress parameters in this region in the present study.
Relative brain weights in DEHP group increased by 18% vs. control. Although there was an increase in BPA and combined exposure groups in comparison to control, this increase was insignificant. These findings indicate that prenatal exposure to these substances changes the brain-body weight ratio in adulthood. The effects of maternal BPA and/or DEHP exposure on brain weights may change due to the dose and duration of exposure (Tanida et al. 2009; Kabuto et al. 2004). Tanida et al. (2009) found that prenatal BPA and DEHP exposure reduced brain weight in male rats at 6 weeks; however, the same effect was not observed in combined exposure. In the same study, relative brain weight increased in all groups at 2 weeks of age compared to control while this effect disappeared after 4 and 6 weeks (Tanida et al. 2009). To our knowledge, there is no study in literature that evaluates the effects of prenatal EDC exposure on brain weight during adulthood (12 week in rats). It can be predicted that dose and timing of exposure may affect the findings.
In the present work, we observed the degeneration of pyramidal neurons in CA1 region of hippocampus in all experimental groups. In addition, there was degeneration in CA3 region in the combined exposure group. According to these results, we can suggest that exposure to BPA and DEHP in prenatal and early postnatal period may cause morphological alterations in hippocampus tissue. The neurons of the CA1 region of the hippocampus are important for spatial learning and memory. In addition, the strength of the connections between the CA1 region and the CA3 region is essential for long-term memory (Rubin et al. 2014). Research has shown that the development of synaptic plasticity and synaptogenesis in brain areas that affect memory development is estrogen-dependent. There are studies showing that EDCs impair memory development in animals (Eilam-Stock et al. 2012; MacLusky et al. 2005; Dai et al. 2015; Smith and Holahan 2014). Eilam-Stock et al. (2012) showed a decrease in memory test results and decreased dendrite density of 10–25% in CA1 region of hippocampus in adult male rats as a result of single dose BPA exposure (40 μg/kg) (Eilam-Stock et al. 2012). Dai et al. (2015) showed that memory decreased in 6-week-old male mice which were exposed to DEHP at 50 and 200 mg/kg/day during prenatal period and lactation (Dai et al. 2015). Our findings support the results of previous studies and we can suggest that combined exposure has more severe adverse effects in both CA1 and CA3 regions.
The mean percentage of apoptotic neuron and glia cells in CA1, CA2, and CA3 regions of hippocampus in experimental groups was evaluated by TUNEL analysis. In single exposure groups, as well as in the combined exposure group, the percentages of apoptotic neurons and glia cells were increased in CA1 and CA2 regions. In the BPA + DEHP group, apoptotic neuron percentage was also increased in CA3 region; however, no significant changes were observed in apoptotic neurons in single exposure groups. The findings are consistent with histopathological changes. In different in vivo and in vitro studies, researchers observed that exposure to EDCs may cause apoptosis in nerve cells. Li et al. (2013) showed that apoptotic cells and caspase-3 levels were increased in hippocampal neurons of rats which were exposed to dibutyl phthalate (500 mg/kg/day) during prenatal and lactation periods (Li et al. 2013). The results of the present work are consistent with the previous studies. Additionally, this is the first study evaluating combined exposure to BPA and DEHP. Our results suggest that combined exposure to BPA and DEHP may have more serious effects than single exposure. It is generally suggested that oxidative stress causes neuronal death, which in turn leads to alterations in brain. These alterations finally cause neurodegenerative diseases. Massive neuronal loss is observed particularly in Alzheimer’s disease. Though it is known that glial cells are more resistant to oxidative stress when compared to neurons, extensive oxidative stress may also lead to apoptotic glial cell death. In fact, signs of apoptosis are observed in both neurons and glial cells in the brains of patients with Alzheimer’s disease (Kitamura et al. 1999).
In the current study, DA and 5-HT levels decreased significantly in all study groups vs. control (p < 0.05). The data indicate that dopaminergic and serotonergic functions may change with EDC exposure during prenatal and lactation periods and these alterations may play a role in developmental and behavioral disorders that may occur in older age. In literature, there are only studies that investigate the effects of single exposure to EDCs on monoaminergic systems. Only one study focused on the combined exposure (Tanida et al. 2009). After single exposure to EDCs, the effects can vary due to the experimental design. In one study, the researchers observed that BPA exposure during prenatal and lactation periods increased 5-HT concentrations in dorsal rafe nuclei; DA in striatum and 3,4-dihydroxyphenylacetic acid (DOPAC, a metabolite of dopamine); homovanillic acid (HVA, a major catecholamine metabolite); and 5-hydroxyindoleacetic acid (5-HIAA, the primary metabolite of 5-HT) levels in forebrain. These findings suggests that BPA exposure increases DA release from nerve endings and the rate of conversion of monoamines to their metabolites (Honma et al. 2006; Nakamura et al. 2010; Matsuda et al. 2010; Matsuda et al. 2012).
We have also evaluated the metabolism of monoaminergic neurotransmission in the cerebrum region by separately measuring MAO-A and MAO-B activities. Significant increase in MAO-B activity in the DEHP+BPA group indicates that biotransformation rate of DA and 5-HT can increase after combined exposure DEHP and BPA. This finding is consistent with the decrease in DA and 5-HT levels in the combined exposure group. In the literature, there is only one study that showed the effect of BPA exposure on MAO enzymes. In this study, MAO levels were measured in three different parts of the brain (hippocampus, amygdala, and medulla oblongata) in male offspring born from pregnant mice receiving subcutaneously BPA (250 ng/kg/day) from the 10th day of pregnancy to the end of lactation period. MAO-B activity in medulla oblongata decreased compared to the control group. In other parts of the brain, no changes were observed (Matsuda et al. 2012). There are not any studies that determined the effects of phthalate and combined BPA + phthalate exposure on MAO enzyme activities. There are studies showing the association between exposure to EDCs and the development of anxiety (Panagiotidou et al. 2014; Poimenova et al. 2010; Chen et al. 2014). Experiments on rodents show that changes in the dopaminergic system are one of the mechanisms of anxiogenic effects and that DOPAC/DA ratio and MAO-A/B levels are related to anxiety behavior (Cox et al. 2010; Chen et al. 2004; Chiavegatto et al. 2009). In our study, it can be suggested that the increase in MAO-B activity in combined exposure group may be one of the anxiogenic mechanisms of EDCs.
By measuring DAT levels in the cerebrum region, we tried to evaluate the alterations in there-uptake mechanism of DA in the study groups. In the DEHP group, DAT levels decreased by 47% vs. control. Although BPA and BPA + DEHP groups showed a 16% decrease compared to the control group, this difference was not statistically significant. In one study, it was found that there was more than a 2-fold decrease in the expression of DAT genes in rats exposed to single dose of 20 μg/kg BPA in the neonatal period. Ishido et al. (2004) administered a single dose of dicyclohexyl phthalate (DCHP) (87 nmol/10 μl, by intracisternal route) to 5-day-old rats. When rats reach to the age of 4–5 weeks, DAT levels were found to be decreased in the middle brain region (Ishido et al. 2004). DAT plays a critical role in terminating the neurotransmission of DA after its re-uptake by neurons and maintaining DA homeostasis in CNS. Changes in dopaminergic neurotransmission due to aging are thought to be the result of decreased DAT expression. Moreover, DAT not only controls the magnitude and duration of the extracellular DA signal, but also maintains intracellular DA levels. There was a 95% reduction in stored DA levels despite an increase in DA synthesis in mice that have suppressed DAT expression (McHugh and Buckley 2015). In our study, the decrease in DAT levels observed in all exposure groups, especially in the DEHP group, may be due to a decrease in DAT expression and/or apoptosis occurring in dopaminergic neurons. The decrease in DAT levels together with the decrease in DA levels is a finding supporting the fact that DAT is an important transporter for the protection of DA levels within the cells.
In our study, AChE activity in the cerebrum region of rats in BPA and combined exposure groups showed significant increases vs. control. When similar studies with BPA are examined, it can be suggested that different dose and exposure periods may change the results. Luo et al. (2013) found that AChE activity in the hippocampus region of rats exposed to BPA at a dose of 50 mg/kg/day during puberty showed decreases, whereas activity in the frontal cortex, hypothalamus, and cerebellum regions did not change (Luo et al. 2013). However, in another study, 5-month-old rats receiving 25 mg/kg/day BPA for 6 weeks showed significant increases in AChE activity in the hippocampus area vs. control. It was suggested that chronic BPA exposure would cause a decrease in hippocampal ACh levels due to an increase in AChE activity and this could lead to memory impairment (Khadrawy et al. 2016). These two studies showed differences in exposure time and dose. When the results are evaluated, it is observed that the effects of exposure in adolescence and adulthood may be opposite. Our present study differs from previous studies in that it covers prenatal and lactation periods. AChE is one of the most crucial and effective enzymes for the response of the nerves. Cholinergic neurotransmission can have a number of different effects on behavior. Cortical ACh release is increased during performances requiring attention. Therefore, decreased cerebral ACh neurotransmission can be considered as one of the mechanisms of attention deficit development (Woolf and Butcher 2011). In addition, increased AChE activity has been shown to induce apoptosis in neurons through different mechanisms, particularly by caspase 3 and 9 activation (Toiber et al. 2008). In our study, apoptosis observed with an increase in AChE activity also supports this finding.
There is evidence in literature that supports the hypothesis that oxidative stress has an important role in neurodegenerative diseases. CNS is particularly vulnerable to oxidative stress. Neuronal membranes contain unsaturated fatty acids that are highly sensitive to free radicals, making them more susceptible to oxidative damage (Halliwell 2006). Oxidative stress can be considered as a mechanism of adverse effects of EDCs on neuroendocrine parameters. In the present work, the effects of EDC exposure on prenatal and lactation periods on oxidative stress biomarkers were investigated. Increased levels of cerebral LP indicate the presence of oxidative stress due to exposure to EDCs. The increase in GSH levels can be considered as a defense mechanism of the organism against oxidative stress induced by EDCs. There are limited studies in the literature evaluating LP and GSH levels in rat brain tissue after BPA and DEHP exposure. Khadrawy et al. (2016) found that LP and GSH levels increased in cortex and hippocampus regions of 5-month-old rats exposed to BPA at 10 mg/kg/day for 10 weeks and 25 mg/kg/day for 6 weeks (Khadrawy et al. 2016). In another study, male rats were exposed to BPA 50 mg/kg/day orally 3 times a week for 6 weeks and the researchers reported significant increases in LP levels in the cerebrum region (El-Missiry et al. 2014). In another study, MDA levels significantly increased in mice which were exposed to high doses of dibutyl phthalate for 28 days (Yan et al. 2016). However, there are not any studies that evaluate the oxidative stress-causing effects of combined exposure to EDCs in CNS.
In conclusion, all these results indicate that prenatal, early postnatal, and lactational EDC exposure may have adverse effects on CNS and neuroendocrine system, and these effects may persist even in later life. BPA seems to be responsible for the oxidative stress-causing effects in BPA + DEHP group. Similar changes in total GSH and MDA levels and also in AchE activity were observed in both BPA and BPA + DEHP groups. The adverse effects of EDCs on the neuroendocrine system were determined as decreased monoamine neurotransmitter (DA and 5-HT) levels in the cerebrum region, decreased DAT levels, increased MAO-B and AChE activity, and impaired oxidant/antioxidant balance. These data show that exposure to EDCs causes imbalance in neurotransmitter systems in the brain. Moreover, there is growing evidence that show the involvement of oxidative stress in the pathology of several neurological disorders, including Alzheimer’s disease, Parkinson’s disease, and stroke. In addition, the vulnerability of the central nervous system to oxidative stress mediated injury is well established as neurons consume large amounts of oxygen and the brain has many areas containing high iron content, and neuronal mitochondria generate large amounts of hydrogen peroxide. Furthermore, neuronal membranes contain polyunsaturated fatty acids that have particular susceptibility to ROS. Therefore, biomarkers of oxidative stress (mainly biomarkers of lipid peroxidation) can have practical clinical applications as biomarkers.
BPA and/or DEHP exposures can lead to apoptosis and histopathological changes in the hippocampus area. There are many studies in which exposure to EDCs is associated with neurodevelopmental diseases in humans (Ipapo et al. 2017; Perera et al. 2016; Arbuckle et al. 2016; Harley et al. 2013). We can suggest that neurochemical imbalances and oxidative stress may be the cause of these conditions. However, it is suggested that most of the neurological diseases may arise from both genetic predisposition and exposure to environmental chemicals. Exposure to EDCs prenatally and in early life can lead to genotoxic effects and perhaps mutations in genes responsible for neurodegenerative disorders (Matilla-Dueñas et al. 2017). Therefore, the presence of multiple mechanisms and cross-effects at all different parts of the neuroendocrine axis should be considered when studying the toxic effects of EDCs.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adewale HB, Todd KL, Mickens JA, Patisaul HB (2011) The impact of neonatal bisphenol-A exposure on sexually dimorphic hypothalamic nuclei in the female rat. Neurotoxicology 32:38–49
Amigo I, Traba J, Reuda CB (2016) Isolating brain mitochondria by differential centrifugation. Bio-protocol 6:1–7
Andrade AJM, Grande SW, Talsness CE, Grote K, Chahoud I (2006) A dose–response study following in utero and lactational exposure to di-(2-ethylhexyl)-phthalate (DEHP): non-monotonic dose–response and low dose effects on rat brain aromatase activity. Toxicology 227:185–192
Arbuckle TE, Davis K, Boylan K, Fisher M, Fu J (2016) Bisphenol A, phthalates and lead and learning and behavioral problems in Canadian children 6–11 years of age: CHMS 2007–2009. Neurotoxicology 54:89–98
Bauer M, Heinz A, Whybrow PC (2002) Thyroid hormones, serotonin and mood: of synergy and significance in the adult brain. Mol Psychiatry 7:140–156
Chen K, Holschneider DP, Wu W, Rebrin I, Shih JC (2004) A spontaneous point mutation produces monoamine oxidase A/B knock-out mice with greatly elevated monoamines and anxiety-like behavior. J Biol Chem 279:39645–39652
Chen F, Zhou L, Bai Y, Zhou R, Chen L (2014) Sex differences in the adult HPA axis and affective behaviors are altered by perinatal exposure to a low dose of bisphenol a. Brain Res 1571:12–24
Chiavegatto S, Izidio GS, Mendes-Lana A, Aneas I, Freitas TA, Torrao AS et al (2009) Expression of alpha-synuclein is increased in the hippocampus of rats with high levels of innate anxiety. Mol Psychiatry 14:894–905
Colborn T (2004) Neurodevelopment and endocrine disruption. Environ Health Perspect 112:944–949
Cox KH, Gatewood JD, Howeth C, Rissman EF (2010) Gestational exposure to bisphenol A and cross-fostering affect behaviors in juvenile mice. Horm Behav 58:754–761
Dai Y, Yang Y, Xu X, Hu Y (2015) Effects of uterine and lactational exposure to di-(2-ethylhexyl) phthalate on spatial memory and NMDA receptor of hippocampus in mice. Horm Behav 71:41–48
Devoto P, Flore G (2006) On the origin of cortical dopamine: is it a co-transmitter in noradrenergic neurons? Curr Neuropharmacol 4:115–125
Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, Zoeller RT, Gore AC (2009) Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev 30:293–342
Eilam-Stock T, Serrano P, Frankfurt M, Luine V (2012) Bisphenol-A impairs memory and reduces dendritic spine density in adult male rats. Behav Neurosci 126:175–185
El-Missiry MA, Othman AI, Al-Abdan MA, El-Sayed AA (2014) Melatonin ameliorates oxidative stress, modulates death receptor pathway proteins, and protects the rat cerebrum against bisphenol-A-induced apoptosis. J Neurol Sci 347:251–256
Gavrieli Y, Sherman Y, Ben-Sasson SA (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119:493–501
Gorczyca W, Bruno S, Darzynkiewicz RJ, Gong JP, Darzynkiewicz Z (1992) DNA strand breaks occurring during apoptosis—their early insitu detection by the terminal deoxynucleotidyl transferase and nick translation assays and prevention by serine protease inhibitors. Int J Oncol 1:639–648
Gore AC (2010) Neuroendocrine targets of endocrine disruptors. Hormones (Athens) 9:16–27
Grandjean P, Landrigan PJ (2014) Neurobehavioural effects of developmental toxicity. Lancet Neurol 13:330–338
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658
Harley KG, Gunier RB, Kogut K, Johnson C, Bradman A, Calafat AM, Eskenazi B (2013) Prenatal and early childhood bisphenol A concentrations and behavior in school-aged children. Environ Res 126:43–50
Honma T, Miyagawa M, Suda M, Wang RS, Kobayashi K, Sekiguchi S (2006) Effects of perinatal exposure to bisphenol A on brain neurotransmitters in female rat offspring. Ind Health 44:510–524
Ipapo KN, Factor-Litvak P, Whyatt RM, Calafat AM, Diaz D, Perera F, Rauh V, Herbstman JB (2017) Maternal prenatal urinary phthalate metabolite concentrations and visual recognition memory among infants at 27 weeks. Environ Res 155:7–14
Ishido M, Masuo Y, Suzuki JS, Oka S, Niki E, Morita M (2004) Dicyclohexylphthalate causes hyperactivity in the rat concomitantly with impairment of tyrosine hydroxylase immuno reactivity. J Neurochem 91:69–76
Kabir ER, Rahman MS, Rahman I (2015) A review on endocrine disruptors and their possible impacts on human health. Environ Toxicol Pharmacol 40:241–258
Kabuto H, Amakawa M, Shishibori T (2004) Exposure to bisphenol A during embryonic/fetal life and infancy increases oxidative injury and causes underdevelopment of the brain and testis in mice. Life Sci 74:2931–2940
Khadrawy YA, Noor NA, Mourad IM, Aboul Ezz HS (2016) Neurochemical impact of bisphenol A in the hippocampus and cortex of adult male albino rats. Toxicol Ind Health 32:1711–1719
Kitamura Y, Taniguchi T, Shimohama S (1999) Apoptotic cell death in neurons and glial cells: implications for Alzheimer’s disease. Jpn J Pharmacol 79:1–5
Li XJ, Jiang L, Chen L, Chen HS, Li X (2013) Neurotoxicity of dibutyl phthalate in brain development following perinatal exposure: a study in rats. Environ Toxicol Pharmacol 36:392–402
Luo G, Wei R, Niu R, Wang C, Wang J (2013) Pubertal exposure to bisphenol A increases anxiety-like behavior and decreases acetylcholinesterase activity of hippocampus in adult male mice. Food Chem Toxicol 60:177–180
MacLusky NJ, Hajszan T, Leranth C (2005) The environmental estrogen bisphenol A inhibits estradiol-induced hippocampal synaptogenesis. Environ Health Perspect 113:675–679
Mahaboob Basha P, Radha MJ (2020) Gestational and lactational exposition to di-n-butyl phthalate increases neurobehavioral perturbations in rats: a three generational comparative study. Toxicol Rep 7:480–491
Matilla-Dueñas A, Corral-Juan M, Rodríguez-Palmero Seuma A, Vilas D, Ispierto L, Morais S, Sequeiros J, Alonso I, Volpini V, Serrano-Munuera C, Pintos-Morell G, Álvarez R, Sánchez I (2017) Rare neurodegenerative diseases: clinical and genetic update. Adv Exp Med Biol 1031:443–496
Matsuda S, Saika S, Amano K, Shimizu E, Sajiki J (2010) Changes in brain monoamine levels in neonatal rats exposed to bisphenol A at low doses. Chemosphere 78:894–906
Matsuda S, Matsuzawa D, Ishii D, Tomizawa H, Sutoh C, Nakazawa K (2012) Effects of perinatal exposure to low dose of bisphenol A on anxiety like behavior and dopamine metabolites in brain. Prog Neuro-Psychopharmacol Biol Psychiatry 39:273–279
McCaffrey KA, Jones B, Mabrey N, Weiss B, Swan SH, Patisaul HB (2013) Sex specific impact of perinatal bisphenol A (BPA) exposure over a range of orally administered doses on rat hypothalamic sexual differentiation. Neurotoxicology 36:55–62
McHugh PC, Buckley DA (2015) The structure and function of the dopamine transporter and its role in CNS diseases. Vitam Horm 98:339–369
Meyer G (2001) Human neocortical development: the importance of embryonic and early fetal events. Neuroscientist 7:303–314
Nakamura K, Itoh K, Yoshimoto K, Sugimoto T, Fushiki S (2010) Prenatal and lactational exposure to low-doses of bisphenol A alters brain monoamine concentration in adult mice. Neurosci Lett 484:66–70
Negri-Cesi P, Colciago A, Pravettoni A, Casati L, Conti L, Celotti F (2008) Sexual differentiation of the rodent hypothalamus: hormonal and environmental influences. J Steroid Biochem Mol Biol 109:294–299
Nohynek GJ, Borgert CJ, Dietrich D, Rozman KK (2013) Endocrine disruption: fact or urban legend? Toxicol Lett 223:295–305
Ohno M, Hamada N, Yamakawa J, Noh J, Morri H, Ito K (1987) Myasthenia gravis associated with Graves’ disease in Japan. Jpn J Med 26:2–6
Panagiotidou E, Zerva S, Mitsiou DJ, Alexis MN, Kitraki E (2014) Perinatal exposure to low-dose bisphenol A affects the neuroendocrine stress response in rats. J Endocrinol 220:207–218
Parent AS, Naveau E, Gerard A, Bourguignon JP, Westbrook GL (2011) Early developmental actions of endocrine disruptors on the hypothalamus, hippocampus and cerebral cortex. J Toxicol Environ Health B Crit Rev 14:328–345
Perera F, Roen Nolte EL, Wang Y, Margolis AE, Calafat AM, Wang S et al (2016) Bisphenol A exposure and symptoms of anxiety and depression among inner city children at 10–12 years of age. Environ Res 151:195–202
Poimenova A, Markaki E, Rahiotis C, Kitraki E (2010) Corticosterone-regulated actions in the rat brain are affected by perinatal exposure to low dose of bisphenol a. Neuroscience 167:741–749
Richard MJ, Portal B, Meo J, Coudray C, Hadjian A, Favier A (1992) Malondialdehyde kit evaluated for determining plasma and lipoprotein fractions that react with thiobarbituric acid. Clin Chem 38:704–709
Rubin R, Watson PD, Duff MC, Coen NJ (2014) The role of the hippocampus in flexible cognition and social behavior. Front Hum Neurosci 742:1–15
Smith CA, Holahan MR (2014) Reduced hippocampal dendritic spine density and BDNF expression following acute postnatal exposure to Di(2-Ethylhexyl) phthalate in male long Evans rats. PLoS One 9:1–9
Sullivan RM, Dufresne MM (2006) Mesocortical dopamine and HPA axis regulation: role of laterality and early environment. Brain Res 1076:49–59
Tanida T, Warita K, Ishihara K, Fukui S, Mitsuhashi T, Sugawara T, Tabuchi Y, Nanmori T, Qi WM, Inamoto T, Yokoyama T, Kitagawa H, Hoshi N (2009) Fetal and neonatal exposure to three typical environmental chemicals with different mechanisms of action: mixed exposure to phenol, phthalate, and dioxin cancels the effects of sole exposure on mouse midbrain dopaminergic nuclei. Toxicol Lett 189:40–47
Toiber D, Berson A, Greenberg D, Melamed-Book N, Diamant S, Soreq H (2008) N-Acetylcholinesterase-induced apoptosis in Alzheimer’s disease. PLoS One 3:1–12
Wang X, Michaelis EK (2010) Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci 2:12
Wang R, Xu X, Weng H, Yan S, Sun Y (2016) Effects of early pubertal exposure to di-(2-ethylhexyl) phthalate on social behavior of mice. Horm Behav 80:117–124
Wang Y, Du X, Wang D, Wang J, Du J (2020) Effects of bisphenol A exposure during pregnancy and lactation on hippocampal function in newborn rats. Int J Med Sci 17:1751–1762
Waye A, Trudeau VL (2011) Neuroendocrıne dısruptıon: more than hormones are upset. J Toxicol Environ Health 14:270–291
Woolf NJ, Butcher LL (2011) Cholinergic systems mediate action from movement to higher consciousness. Behav Brain Res 221:488–498
Yan B, Guo J, Liu X, Li J, Yang X, Ma P, Wu Y (2016) Oxidative stress mediates dibutyl phthalate induced anxiety-like behavior in Kunming mice. Environ Toxicol Pharmacol 45:45–51
Funding
This study was supported by Hacettepe University Scientific Projects Coordination Unit [Project no: TYL-2018-17006].
Author information
Authors and Affiliations
Contributions
AY performed tissue preparation, measurement of neuroendocrine parameters and oxidative stress parameters, and TUNEL assay. GO performed dosing and euthanasia of animals, tissue preparation, and measurement of neuroendocrine parameters. AB performed dosing and euthanasia of animals and measurement of oxidative stress parameters. PE performed study conception and design, euthanasia of animals, tissue preparation, statistical analyses, and evaluation of results. NY performed histopathological examinations and TUNEL assay. NDZ performed histopathological examinations and TUNEL assay. BKG performed study conception and design, statistical analyses, evaluation of results, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
The study was approved by Gazi University Animal Ethics Committee (G.U.ET–18.043).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Responsible Editor: Ludek Blaha
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yirun, A., Ozkemahli, G., Balci, A. et al. Neuroendocrine disruption by bisphenol A and/or di(2-ethylhexyl) phthalate after prenatal, early postnatal and lactational exposure. Environ Sci Pollut Res 28, 26961–26974 (2021). https://doi.org/10.1007/s11356-021-12408-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12408-9