Abstract
Remediation of heavy metal and polycyclic aromatic hydrocarbon (PAH)–co-contaminated soils has drawn much more attention; phytoremediation is an often-used technique. Sudan grass (Sorghum sudanense (Piper) Stapf.) with developed root system and strong PAHs and heavy metal tolerance is a potential choice for phytoremediation. In this study, the application of tea saponin (TS) (1 g kg−1 soil) and nitrilotriacetic acid (NTA) (1 g kg−1 soil) was to improve the removal efficiency of Ni and pyrene. TS and NTA had no obvious effects on the growth and soluble proteins of Sudan grass. Ni concentration in root was higher than that in the shoot. The addition of TS and NTA increased the Ni concentration in the root by 25.98% in Ni-contaminated treatment. Pyrene was mainly accumulated in the shoot of Sudan grass. Pyrene concentration in shoot increased by 20.14% with TS-NTA in pyrene-contaminated treatment and increased by 31.97% in Ni-contaminated treatment. TS and NTA had significantly improved dissolved organic matter and soil microbial activity. Microbial activity increased by 16.75%, 18.07%, and 23.364% in pyrene-contaminated, Ni-contaminated, and pyrene and Ni–co-contaminated treatment, respectively. This study showed that phytoremediation of pyrene and Ni–co-contaminated soil by Sudan grass could be enhanced by the application of TS-NTA and the interaction between pyrene and Ni impacted the accumulation of Ni and pyrene in Sudan grass.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) and heavy metals are two major types of pollutants commonly found in soil, and they are often present together (Perez et al. 2010). Xu et al. (2018) reported that heavy metals (such as Ni and Cr) and PAHs were detected in rural soils. A similar finding was shown in the study of Kibblewhite (2018). The combination of PAHs and heavy metals has higher toxicity and remains in soil for a longer period of time (Perez et al. 2010). There is an urgent need to develop reasonable and effective methods for the remediation of pyrene and Ni–co-contaminated soil.
Phytoremediation, a cost-effective and environmentally friendly strategy, can remove both PAHs and heavy metals (Dai et al. 2020; Sarma et al. 2019). Sudan grass (Sorghum sudanense (Piper) Stapf.), a C4 plant, has the advantages of a developed root system and strong PAHs and heavy metal tolerance (Sivaram et al. 2018a). Sivaram et al. (2018b) had proved that Sudan grass could enhance the removal of PAHs, especially high molecular weight PAHs such as pyrene. Also, some studies had shown that Sudan grass could be applied to the phytoremediation of heavy metal–contaminated soil (Li et al. 2016; Shim et al. 2014).
Surfactants and chelating agents are able to enhance the phytoremediation efficiency by increasing the bioavailability of PAHs and heavy metals (Cheng et al. 2018; Yu et al. 2019). The effective and biodegradable enhancers should be selected in phytoremediation. Tea saponin (TS), a natural non-ionic surfactant which found in Camellia plants, could provide strong beneficial effects in soil remediation (Yu and He 2018). It had the ability to increase the accumulation of heavy metal in plants and promote the degradation of organic pollutants by microorganisms in soil (Cay 2016; Liu et al. 2017). Nitrilotriacetic acid (NTA), with a half-life of 2–7 days, has a strong chelating ability and low toxicity to microorganisms and plants (Yu et al. 2020). It had been proven to increase heavy metals removal from contaminated soil (Yan et al. 2017).
The purposes of this study were to (1) explore preliminarily the effect of TS and NTA on the phytoremediation efficiency of pyrene and Ni–co-contaminated soil; (2) investigate the effects of TS and NTA on plant soluble protein, soil microbial activity, and physicochemical properties; and (3) reveal the interaction of pyrene and Ni during phytoremediation. This study can provide a theoretical basis for phytoremediation in pyrene and Ni–co-contaminated soil.
Materials and methods
Soil treatment
Experimental soil for this study was collected from the campus of Shanghai University. Physicochemical properties of the control soil were as follows: pH 8.3, organic matter 19.6 g kg−1, total nitrogen 0.5 g kg−1, silt 60.4%, clay 7.4%, sand 32.2%, and Ni 24.4 mg kg−1; pyrene was not detected. Pyrene dissolved in acetone solution/NiCl2 solution was spiked into the soil as pyrene/Ni-contaminated soil. Co-contaminated soil was prepared by adding NiCl2 solution to the pyrene-contaminated soil. The contaminated soils were mechanically mixed and aged in darkness for about 3 months. The water holding capacity was kept at 75% during aging. The final concentrations of pyrene and Ni in single and combined contaminated soils were 50 and 200 mg kg−1, respectively. The setting of pollutant concentration was based on the environmental quality standards for soils of China.
Chemicals
Tea saponin (78%, purity) was bought from the Wuxi K-renown Environmental Protection Technology Co., Ltd. (China). Other reagents were all purchased from Sinopharm Chemical Reagent Co., Ltd. (China).
Experimental design
The experimental design was as follows: P (pyrene-contaminated soil), PE (pyrene-contaminated soil with enhancers), PS (pyrene-contaminated soil with Sudan grass), PES (pyrene-contaminated soil with enhancers and Sudan grass), N (Ni-contaminated soil), NE (Ni-contaminated soil with enhancers), NS (Ni-contaminated soil with Sudan grass), NES (Ni-contaminated soil with enhancers and Sudan grass), C (pyrene and Ni–co-contaminated soil), CE (pyrene and Ni–co-contaminated soil with enhances), CS (pyrene and Ni–co-contaminated soil with Sudan grass), and CES (pyrene and Ni–co-contaminated soil with enhancers and Sudan grass).
One kilogram of each spiked soil was placed in each pot and three pots for each treatment. Fifty seeds of Sudan grass (purchased from Jiangxi Agricultural Company, China) were initially sown in each pot, and they were subsequently adjusted to about 30 individual plants per pot. The concentrations of TS and NTA were both set to 1 g kg−1 soil. After 21 days of plant growth, the solution of TS and NTA (divided into seven times, once every 2 days) was added to the soil. In order not to change the soil pH, the pH of the adding solution was adjusted to 8.3. All pots were placed in the greenhouse (18–25 °C, with a 10:14 light/dark period and 60% relative humidity). After 42 days, the plants were harvested. All plant samples were placed in an − 80 °C refrigerator for testing. After drying and grinding, the plant samples were sieved (0.15-mm mesh) for analysis of Ni and pyrene.
Analysis of Ni in plants
Ni in plants was detected by acid digestion. Samples of the plant were digested with a mixture of HNO3 and HClO4 (3:5, v/v) at 220 °C. Ni concentration was determined by inductively coupled plasma optical emission spectrometry (ICPOES) after being filtered by microfiltration membrane (0.45 μm).
Analysis of water-soluble Ni in soil
There were four treatments: N (Ni-contaminated soil added with water), NE (Ni-contaminated soil added with enhancer solution), C (pyrene and Ni–co-contaminated soil added with water), and CE (pyrene and Ni–co-contaminated soil added with enhancer solution). Water or enhancer solution (1 g L−1) was added to the soil as 10 mL:1 g. After shaking for 24 h, the supernatant was filtered by microfiltration membrane (0.45 μm). The concentration of water-soluble Ni was analyzed by inductively coupled plasma (ICP).
Extraction and analysis of pyrene in plants
Total pyrene in plants was detected by the method referenced by Sun et al. (2010). The plant sample (1.00 g) was ultrasonically extracted in 10-mL elution of dichloromethane and acetone (v/v, 1:1) for 30 min. Then the supernatant was concentrated and followed by filtration through 4 g silica gel with 10 mL mixture of hexane and dichloromethane (v/v, 1:1). Finally, the resulting liquids were evaporated and redissolved in methanol to a final volume of 2 mL. Extracts were analyzed by HPLC (LC-20A) after passing through a 0.22-μm Teflon filter. The HPLC system was fitted with a UV detector and a 4.6 × 250-mm reverse phase C18 column, using methanol as the mobile phase, at a flow rate of 1 mL min−1 (40 °C). The wavelength of the UV detector was 245 nm for pyrene. The sample solution (10 μL) was injected into the HPLC system by an autosampler.
Analysis of physicochemical property in rhizosphere soil
The soil dissolved organic matter was extracted according to the method of Lin et al. (2019), with a soil to water ratio of 1:4 (w/v). The concentration of dissolved organic matter is obtained by multiplying the concentration of dissolved organic carbon by 1.724, and dissolved organic carbon was quantified by an automated TOC analyzer. Soil available nitrogen was analyzed by the method of alkaline hydrolysis diffusion; soil available phosphorus was determined by Mo-Sb colorimetry.
Determination of soluble protein concentration and soil microbial activity
The soluble protein concentration with different treatments was detected using Coomassie Brilliant Blue G-250 as a dye and bovine albumin (BSA) as a standard (Bradford 1976). Soil microbial activity was measured using the fluorescein diacetate (FDA) assay described by Chen et al. (2017). Phosphate buffer solution was added to the soil for 15 min, and then fluorescein diacetate (FDA) solution was added. After shaking for 2 h, the absorbance was read with OD490.
Statistical analysis
All data were expressed as means ± standard deviation (SD) and analyzed using the SPSS software program (version 22.0 for Windows). One-way analysis of variance (ANOVA) was used to analyze significant differences. Differences were statistically significant when P < 0.05 using the Duncan test. Drawing data results were using Origin 9.0
Results
The biomass of Sudan grass
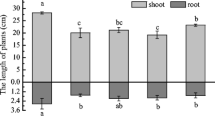
The effect of TS and NTA on the dry biomass of Sudan grass in contaminated soil is shown in Fig. 1. The shoot biomass of Sudan grass in Ni, pyrene, and co-contaminated treatments were 1.35 g pot−1, 1.82 g pot−1, and 1.73 g pot−1, respectively. After the addition of TS-NTA, the shoot biomass values of the three treatments were 1.39 pot−1, 1.81 g pot−1, and 1.74 g pot−1, respectively. The root biomass values of Sudan grass in Ni, pyrene, and co-contaminated treatments were 0.55 g pot−1, 0.66 g pot−1, and 0.65 g pot−1 and 0.44 g pot−1, 0.62 g pot−1, and 0.56 g pot−1 for the addition of TS-NTA, respectively. Basically, no significant differences between with and without the addition of TS-NTA were found in the biomass of Sudan grass (P < 0.05). The shoot biomass of Sudan grass in pyrene-contaminated treatment and pyrene and Ni–co-contaminated treatment was higher relative to that in Ni-contaminated treatment (P < 0.05).
Biomass of Sudan grass with different treatments (soil with Ni and plants (NS); soil with Ni, enhancers, and plants (NES); soil with pyrene and plants (PS); soil with pyrene, enhancers, and plants (PES); co-contaminated soil with plants (CS); co-contaminated soil with enhancers and plants (CES)). Different letters up the vertical bars are significantly different between treatments (P < 0.05)
Ni accumulation in Sudan grass
The effect of TS and NTA on the concentration of Ni in Sudan grass is shown in Fig. 2. Ni concentrations in shoot and root were ranged from 17.08 to 25.79 mg kg−1 and 94.02 to 135.20 mg kg−1, respectively. Root Ni concentration in Ni-contaminated treatment and shoot Ni concentration in pyrene and Ni–co-contaminated treatment were highly increased with the addition of TS and NTA (P < 0.05) (Fig. 2). The addition of TS-NTA increased Ni concentration in root by 25.98% in Ni-contaminated treatment. Ni concentration in shoot improved by 50.97% with the addition of TS-NTA in the presence of pyrene (P < 0.05). Ni concentration in root was obviously decreased with the presence of pyrene when TS-NTA was added (P < 0.05).
Ni concentrations in Sudan grass with different treatments (soil with Ni and plants (NS); soil with Ni, enhancers, and plants (NES); co-contaminated soil with plants (CS); co-contaminated soil with enhancers and plants (CES)). Different letters up the vertical bars are significantly different between treatments (P < 0.05)
Water-soluble Ni in soil
The effect of TS and NTA on the concentration of water-soluble Ni in soil is shown in Fig. 3. The concentration of water-soluble Ni in soil significantly increased with the addition of TS and NTA (P < 0.05). The water-soluble Ni concentration in Ni-contaminated treatment was increased by 985.24% with the addition of TS and NTA. And water-soluble Ni concentration was increased by 935.89% with the addition of TS and NTA in the presence of pyrene.
Water-soluble Ni concentrations in soil with different treatments (Ni-contaminated soil added with water (N); Ni-contaminated soil added with enhancer solution (NE); pyrene and Ni–co-contaminated soil added with water (C); pyrene and Ni–co-contaminated soil added with enhancer solution (CE)). Different letters up the vertical bars significantly are different between treatments (P < 0.05)
Pyrene accumulation in Sudan grass
The effect of TS and NTA on the concentration of pyrene in Sudan grass is shown in Fig. 4. The pyrene concentrations in shoot and root ranged from 279.92 to 369.42 mg kg−1 and 23.93 to 59.80 mg kg−1, respectively. The addition of TS and NTA significantly increased pyrene concentration in Sudan grass except for the root in pyrene-contaminated treatment (P < 0.05). Pyrene concentration in shoot increased by 20.14% with addition of TS and NTA in pyrene-contaminated treatment. Pyrene concentration in shoot and root increased by 31.98% and 149.92% with addition of TS and NTA in the presence of Ni.
Pyrene concentrations in Sudan grass with different treatments (soil with pyrene and plants (PS); soil with pyrene, enhancers, and plants (PES); co-contaminated soil with plants (CS); co-contaminated soil with enhancers and plants (CES)). Different letters up the vertical bars are significantly different between treatments (P < 0.05)
Soluble proteins
The effect of TS and NTA on soluble protein concentration in Sudan grass is shown in Fig. 5. The differences in soluble protein concentration with or without TS-NTA were not significant (P = 0.06). The results showed that soluble protein concentrations of Sudan grass in Ni, pyrene, and co-contaminated treatments without TS-NTA addition were 1.87 mg g−1, 2.58 mg g−1, and 2.24 mg g−1 and 1.68 mg g−1, 2.38 mg g−1, and 2.29 mg g−1 for the addition of TS-NTA, respectively. Similar with the results of Sudan grass biomass, soluble protein concentrations of Sudan grass in pyrene-contaminated treatment and pyrene-Ni co-contaminated treatment were higher relative to it in Ni-contaminated treatment (P < 0.05).
Soluble protein concentrations in Sudan grass with different treatments (soil with Ni and plants (NS); soil with Ni, enhancers, and plants (NES); soil with pyrene and plants (PS); soil with pyrene, enhancers, and plants (PES); co-contaminated soil with plants (CS); co-contaminated soil with enhancers and plants (CES)). Different letters up the vertical bars are significantly different between treatments (P < 0.05)
Physicochemical properties of rhizosphere soils
The impact of TS and NTA on the rhizosphere soil properties is presented in Table 1. The dissolved organic matter concentrations were ranged from 0.17 to 0.45 g kg−1. The addition of enhancers significantly increased dissolved organic matter concentration in rhizosphere soil (P < 0.05). Compared with the treatment with only adding TS-NTA, the concentration of dissolved organic matter in soil decreased slightly in the treatment with adding TS-NTA and planting Sudan grass. The available P and available N ranged from 3.94 to 5.48 mg kg−1 and 103.60 to 145.60 mg kg−1. Available P and available N of rhizosphere soil in enhancer-added treatments were increased to some different extent.
Microbial activity
Figure 6 shows the microbial activity in rhizosphere soil. Microbial activity was increased with addition of TS and NTA. After adding TS-NTA, soil microbial activities were increased by 16.75%, 18.07%, and 23.364%, respectively, in Ni-contaminated treatment, pyrene-contaminated treatment, and co-contaminated treatment (P < 0.05). The soil microbial activity in the presence of Ni and pyrene was lower than that in the presence of single Ni or single pyrene when TS-NTA was added (P < 0.05).
Microbial activities in soil with different treatments (soil with Ni (N); soil with Ni and enhancers (NE); soil with Ni and plants (NS); soil with Ni, enhancers, and plants (NES); soil with pyrene (P); soil with pyrene and enhancers (PE); soil with pyrene and plants (PS); soil with pyrene, enhancers, and plants (PES); soil with pyrene and Ni (C); co-contaminated soil with enhances (CE); co-contaminated soil with plants (CS); co-contaminated soil with enhancers and plants (CES)). Different letters up the vertical bars are significantly different between treatments (P < 0.05)
Discussion
Plant growth is an important indicator of phytoremediation. TS and NTA did not show a significant effect on the shoot biomass of Sudan grass, which was consistent with the researches of Fei et al. (2018) and Liu et al. (2017). This indicated that the addition of TS and NTA had no toxic effect on the growth of Sudan grass. Ni pollution would inhibit the growth of Sudan grass, but this inhibition would be relieved in the presence of pyrene. Ni exposure restricted the ability of plants to absorb minerals and water and reduced the photosynthesis of plants, which reduced overall plant growth (Fashola et al. 2016; Wiszniewska et al. 2018). However, some bacteria in soil had the ability to use PAHs as their source of carbon and energy, leading to the degradation of PAHs. And the degradation products would eventually be absorbed by plants as nutrients, thereby resulting in a promotion effect on plant growth (Anyasi et al. 2019).
Ni concentration in the root was higher than that in the shoot; this was consistent with many reports that heavy metals are mainly accumulated in root system (Pilipovic et al. 2019; Tauqeer et al. 2019). The concentration of water-soluble Ni in soil increased by TS and NTA; therefore, more Ni was absorbed by Sudan grass. Due to solubilization action by TS, desorption of pollutants in the soil matrix was increased (Maity et al. 2013). NTA, as a chelating agent, was confirmed to combine metal ions to form complexes and then increase bioaccessibility of metals in soil (Liu et al. 2018). Compared with the Ni concentration in Sudan grass in Ni-contaminated treatment, the Ni concentration in Sudan grass decreased in pyrene-Ni-contaminated treatment. The presence of pyrene had a negative impact on the active transport process which would be the way of the plant to uptake Ni, thus decreasing the concentration of Ni in Sudan grass (Zhang et al. 2019).
Many studies reported that PAHs were mainly accumulated in the roots of the plants (Liu et al. 2017; Wei et al. 2017). However, this study showed that pyrene was mainly accumulated in shoots. The lipid concentration of plant intracellular components determined the concentration of pyrene accumulation due to a higher lipophilicity of pyrene (Kang et al. 2010). The higher pyrene concentration in Sudan grass shoot could be explained by that the lipid concentration of shoot was higher than that of root (Liao et al. 2015). TS and NTA enhanced the pyrene concentration in Sudan grass which was ascribed to increased pyrene bioavailability. The addition of TS and NTA accelerated the transfer of pyrene from soil to the aqueous phase (Liang et al. 2017). Furthermore, bound and residual fractions of pyrene could be efficiently transformed into bioavailable fractions of pyrene because of the addition of TS and NTA (Lu et al. 2019).
Soluble proteins played an important role in protecting life matters and cellular membranes when plants exposed to contaminants; therefore, they often were used as resistant indexes (Pan et al. 2018). There were some stress proteins with anti-oxidation, and detoxification functions were used to protect plants from damage (Huang et al. 2017; Nie et al. 2016). Heavy metal ions at high concentrations could block protein synthesis by forming compounds or chelates with other metal ions (Huang et al. 2019). That could explain why the soluble protein concentrations in Ni-contaminated treatment were the lowest.
Organic matter is often used as a nutrient source for plants, and its increase can improve phytoremediation efficiency of contaminated soils (Bauddh and Singh 2015). Soil dissolved organic matter act as an active organic matter fraction in soil, and the dissolved organic carbon (DOC) in it can be directly utilized by soil microorganisms (Xu et al. 2020). Addition with TS and NTA could increase the concentrations of dissolved organic matter which would be utilized by microbes as their carbon source (Lopez-Gonzalez et al. 2013). The available P and available N in soil provide essential nutrients for plant growth (Wang et al. 2020). As a surfactant, TS could promote the conversion of total P to available P through its solubilization action. Adding NTA can effectively increase the available N concentration in the soil that could be utilized by plants and microorganisms (Liu et al. 2019).
The addition of TS and NTA significantly increased soil microbial activity. The addition of TS-NTA, as nutrients of microorganisms, increased both microbial activity and soil microbial biomass (Sun et al. 2016). The combination of pyrene and Ni had stronger toxicity on soil microorganism than that of single pyrene or Ni (Chen et al. 2017). PAHs in soil could increase the toxicity of heavy metals by facilitating their penetration through microbial membranes when PAHs and heavy metals coexist (Maliszewska-Kordybach and Smreczak 2003).
Conclusions
The addition of TS and NTA greatly promoted the accumulation of Ni and pyrene in Sudan grass by increasing the concentrations of Ni and pyrene in soil solution. In addition, TS and NTA significantly increased dissolved organic matter concentration and soil microbial activity, which was beneficial to the removal of pyrene in soil. Ni mainly accumulated in the root and pyrene mainly accumulated in the shoot. Ni accumulation in root was obviously decreased in the presence of pyrene when TS-NTA was added. The inhibition of Ni on the growth and soluble proteins of Sudan grass was relieved in the presence of pyrene. However, the toxicity of Ni on the soil microorganism increased in the presence of pyrene. The mechanisms of the accumulation of pyrene and the interaction between pyrene and Ni need further exploring.
References
Anyasi RO, Atagana HI, Sutherland R (2019) Identification and characterization of PAH-degrading endophytes isolated from plants growing around a sludge dam. Int J Phytoremediat 21:672–682
Bauddh K, Singh RP (2015) Effects of organic and inorganic amendments on bio-accumulation and partitioning of Cd in Brassica juncea and Ricinus communis. Ecol Eng 74:93–100
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cay S (2016) Enhancement of cadmium uptake by Amaranthus caudatus, an ornamental plant, using tea saponin. Environ Monit Assess 188:320
Chen X, Liu XY, Zhang XY, Cao LY, Hu XX (2017) Phytoremediation effect of Scirpus triqueter noculated plant-growth-promoting bacteria (PGPB) on different fractions of pyrene and Ni in co-contaminated soils. J Hazard Mater 325:319–326
Cheng M, Zeng GM, Huang DL, Yang CP, Lai C, Zhang C, Liu Y (2018) Tween 80 surfactant-enhanced bioremediation: toward a solution to the soil contamination by hydrophobic organic compounds. Crit Rev Biotechnol 38:17–30
Dai YY, Liu R, Zhou YM, Li N, Hou LQ, Ma Q, Gao B (2020) Fire Phoenix facilitates phytoremediation of PAH-Cd co-contaminated soil through promotion of beneficial rhizosphere bacterial communities. Environ Int 136:105421
Fashola MO, Ngole-Jeme VM, Babalola OO (2016) Heavy metal pollution from gold mines: environmental effects and bacterial strategies for resistance. Int J Environ Res Pub Health 13:20
Fei L, Xu PX, Dong Q, Mo Q, Wang ZL (2018) Young leaf protection from cadmium accumulation and regulation of nitrilotriacetic acid in tall fescue (Festuca arundinacea) and Kentucky bluegrass (Poa pratensis). Chemosphere 212:124–132
Huang XC, Tang KL, Xu XP, Cai CT (2017) Interaction of Fe-Mn plaque and Arthrobacter echigonensis MN1405 and uptake and translocation of Cd by Phytolacca acinosa Roxb. Chemosphere 174:585–592
Huang HM, Zhao YL, Xu ZG, Zhang W, Jiang KK (2019) Physiological responses of Broussonetia papyrifera to manganese stress, a candidate plant for phytoremediation. Ecotox Environ Safe 181:18–25
Kang FX, Chen DS, Gao YZ, Zhang Y (2010) Distribution of polycyclic aromatic hydrocarbons in subcellular root tissues of ryegrass (Lolium multiflorum Lam.). BMC Plant Biol 10:7
Kibblewhite MG (2018) Contamination of agricultural soil by urban and peri-urban highways: an overlooked priority? Environ Pollut 242:1331–1336
Li NY, Guo B, Li H, Fu QL, Feng RW, Ding YZ (2016) Effects of double harvesting on heavy metal uptake by six forage species and the potential for phytoextraction in field. Pedosphere 26:717–724
Liang XJ, Guo CL, Liao CJ, Liu SS, Wick LY, Peng D, Yi XY, Lu GN, Yin H, Lin Z, Dang Z (2017) Drivers and applications of integrated clean-up technologies for surfactant-enhanced remediation of environments contaminated with polycyclic aromatic hydrocarbons (PAHs). Environ Pollut 225:129–140
Liao CJ, Liang XJN, Lu G, Thai T, Xu WD, Dang Z (2015) Effect of surfactant amendment to PAHs-contaminated soil for phytoremediation by maize (Zea mays L). Ecotox Environ Safe 112:1–6
Lin YL, Zhang Y, Zhang FQ, Li R, Hu Y, Yu H, Tuyiringire D, Wang L (2019) Effects of bok choy on the dissipation of dibutyl phthalate (DBP) in mollisol and its possible mechanisms of biochemistry and microorganisms. Ecotox Environ Safe 181:284–291
Liu XY, Cao LY, Wang Q, Zhang XY, Hu XX (2017) Effect of tea saponin on phytoremediation of Cd and pyrene in contaminated soils by Lolium multiflorum. Environ Sci Pollut Res 24:18946–18952
Liu XY, Cao LY, Zhang XY, Chen J, Huo ZH, Mao Y (2018) Influence of alkyl polyglucoside, citric acid, and nitrilotriacetic acid on phytoremediation in pyrene-Pb co-contaminated soils. Int J Phytoremediat 20:1055–1061
Liu XY, Mao Y, Zhang XY, Gu PX, Niu YH, Chen XL (2019) Effects of PASP/NTA and TS on the phytoremediation of pyrene-nickel contaminated soil by Bidens pilosa L. Chemosphere 237:124502
Lopez-Gonzalez JA, Lopez MJ, Vargas-Garcia MC, Suarez-Estrella F, Jurado M, Moreno J (2013) Tracking organic matter and microbiota dynamics during the stages of lignocellulosic waste composting. Bioresour Technol 146:574–584
Lu HN, Wang W, Li F, Zhu LZ (2019) Mixed-surfactant-enhanced phytoremediation of PAHs in soil: bioavailability of PAHs and responses of microbial community structure. Sci Total Environ 653:658–666
Maity JP, Huang YM, Hsu CM, Wu CI, Chen CC, Li CY, Jean JS, Chang YF, Chen CY (2013) Removal of Cu, Pb and Zn by foam fractionation and a soil washing process from contaminated industrial soils using soapberry-derived saponin: a comparative effectiveness assessment. Chemosphere 92:1286–1293
Maliszewska-Kordybach B, Smreczak B (2003) Habitat function of agricultural soils as affected by heavy metals and polycyclic aromatic hydrocarbons contamination. Environ Int 28:719–728
Nie J, Liu YG, Zeng GM, Zheng BH, Tan XF, Liu H, Xie JL, Gan C, Liu W (2016) Cadmium accumulation and tolerance of Macleaya cordata: a newly potential plant for sustainable phytoremediation in Cd-contaminated soil. Environ Sci Pollut Res 23:10189–10199
Pan G, Liu WS, Zhang HP, Liu P (2018) Morphophysiological responses and tolerance mechanisms of Xanthium strumarium to manganese stress. Ecotox Environ Safe 165:654–661
Perez RM, Cabrera G, Gomez JM, Abalos A, Cantero D (2010) Combined strategy for the precipitation of heavy metals and biodegradation of petroleum in industrial wastewaters. J Hazard Mater 182:896–902
Pilipovic A, Zalesny RS, Roncevic S, Nikolic N, Orlovic S, Beljin J, Katanic M (2019) Growth, physiology, and phytoextraction potential of poplar and willow established in soils amended with heavy-metal contaminated, dredged river sediments. J Environ Manag 239:352–365
Sarma H, Sonowal S, Prasad MNV (2019) Plant-microbiome assisted and biochar-amended remediation of heavy metals and polyaromatic compounds - a microcosmic study. Ecotox Environ Safe 176:288–299
Shim J, Babu AG, Velmurugan P, Shea PJ, Oh BT (2014) Pseudomonas fluorescens JH 70-4 promotes Pb stabilization and early seedling growth of Sudan grass in contaminated mining site soil. Environ Technol 35:2589–2596
Sivaram AK, Logeshwaran P, Lockington R, Naidu R, Megharaj M (2018a) Impact of plant photosystems in the remediation of benzo a pyrene and pyrene spiked soils. Chemosphere 193:625–634
Sivaram AK, Logeshwaran P, Subashchandrabose SR, Lockington R, Naidu R, Megharaj M (2018b) Comparison of plants with C3 and C4 carbon fixation pathways for remediation of polycyclic aromatic hydrocarbon contaminated soils. Sci Rep 8:2100
Sun TR, Cang L, Wang QY, Zhou DM, Cheng JM, Xu H (2010) Roles of abiotic losses, microbes, plant roots, and root exudates on phytoremediation of PAHs in a barren soil. J Hazard Mater 176:919–925
Sun MM, Ye M, Shen FY, Xu JJ, Ye CL, Yu L, Hu F, Li HX, Jiang X, Kengara FO (2016) Function of nitrate ion and tea saponin application rates in anerobic PAH dissipation in paddy soil. Clean-Soil Air Water 44:667–676
Tauqeer HM, Mahmoodur R, Hussain S, Abbas F, Iqbal M (2019) The potential of an energy crop “Conocarpus erectus” for lead phytoextraction and phytostabilization of chromium, nickel, and cadmium: an excellent option for the management of multi-metal contaminated soils. Ecotox Environ Safe 173:273–284
Wang SX, Zhang L, Jiang ML, Wang J, Xia FZ, Shi L, Xia Y, Chen C, Shen ZG, Chen YH (2020) Cyclic and safety utilisation of Cu polluted biogas residue in saline-alkali soil. Sci Total Environ 704:135410
Wei R, Ni JZ, Li XY, Chen WF, Yang YS (2017) Dissipation and phytoremediation of polycyclic aromatic hydrocarbons in freshly spiked and long-term field-contaminated soils. Environ Sci Pollut Res 24:7994–8003
Wiszniewska A, Muszynska E, Hanus-Fajerska E, Dziurka K, Dziurka M (2018) Evaluation of the protective role of exogenous growth regulators against Ni toxicity in woody shrub Daphne jasminea. Planta 248:1365–1381
Xu YF, Dai SX, Meng K, Wang YT, Ren WJ, Zhao L, Christie P, Teng Y (2018) Occurrence and risk assessment of potentially toxic elements and typical organic pollutants in contaminated rural soils. Sci Total Environ 630:618–629
Xu ZK, Shao TY, Lv ZX, Yue Y, Liu AH, Long XH, Zhou ZS, Gao XM, Rengel Z (2020) The mechanisms of improving coastal saline soils by planting rice. Sci Total Environ 703:135529
Yan L, Li CL, Zhang JJ, Moodley O, Liu SR, Lan C, Gao Q, Zhang WJ (2017) Enhanced phytoextraction of lead from artificially contaminated soil by Mirabilis jalapa with chelating agents. Bull Environ Contam Toxicol 99:208–212
Yu XL, He Y (2018) Tea saponins: effective natural surfactants beneficial for soil remediation, from preparation to application. RSC Adv 8:24312–24321
Yu FM, Li Y, Li FR, Li CM, Liu KH (2019) The effects of EDTA on plant growth and manganese (Mn) accumulation in Polygonum pubescens Blume cultured in unexplored soil, mining soil and tailing soil from the Pingle Mn mine, China. Ecotox Environ Safe 173:235–242
Yu HY, Zhan J, Zhang QP, Huang HG, Zhang XZ, Wang YD, Li TX (2020) NTA-enhanced Pb remediation efficiency by the phytostabilizer Athyrium wardii (Hook.) and associated Pb leaching risk. Chemosphere 246:125815–125815
Zhang XY, Chen J, Liu XY, Gao MJ, Chen XP, Huang C (2019) Nickel uptake and distribution in Agropyron cristatum L. in the presence of pyrene. Ecotox Environ Safe 174:370–376
Funding
This work was supported by the project of the National Key Research and Development Program of China (No. 2018YFC1800600), the National Natural Science Foundation of China (Nos. 21677093, 21806100), and the Natural Science Foundation of Shanghai (No.18ZR1414100).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Elena Maestri
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, X., Shen, S., Zhang, X. et al. Effect of enhancers on the phytoremediation of soils polluted by pyrene and Ni using Sudan grass (Sorghum sudanense (Piper) Stapf.). Environ Sci Pollut Res 27, 41639–41646 (2020). https://doi.org/10.1007/s11356-020-09934-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09934-3