Abstract
Silicon (Si) is a beneficial element which was proven to enhance the tolerance of plants to excess metal in a given growth medium. However, the efficacy of Si in mitigating Cu toxicity in plants can vary between plant species and with the amount of copper (Cu) present in the soil/medium. An experiment was performed to investigate the role of Si in alleviating Cu toxicity in Tanzania guinea grass (Panicum maximum cv. Tanzania). The experimental design consisted on complete random blocks with tree replicates containing three Si rates (0, 1, and 3 mmol L−1) and four Cu rates (0.3, 250, 500, and 750 μmol L−1). The grass was grown for 62 days in a greenhouse under hydroponic conditions, with a total of 36 pots. Thirteen days after sowing, seedlings were transplanted to pots and grown for further 25 days, and then exposed to the set Cu rates for 7 days. The plants were also evaluated more for 30 days after the first harvesting. The results confirmed that the Si supply to Tanzania guinea grass can alleviate the effects of excessive Cu. Plant yield increased with Si supply and decreased with the increment of Cu rates in both growth periods. Copper concentration in diagnostic leaves (DL) and in roots, and Cu content in shoots and roots were higher in plants exposed to Cu of 750 μmol L−1 with no Si application than in other combinations. Besides reducing Cu concentration in plant tissues, the most important Si role was reducing the transport of Cu from roots to shoots, which allowed successive harvesting.

Graphical abstract
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Copper is an essential element for plants, being directly involved in the photosynthetic electron transport chain (Maksymiec 1997). Despite its importance, Cu in excess may cause irreversible damages to plants. This element has been widely used in the formulation of pesticides for the control of fungal diseases in crops around the world (Megateli et al. 2013), which has led to numerous cases of Cu toxicity in plants (Nogueirol et al. 2010; Girotto et al. 2016). Along with the increased Cu from pesticides, other forms of anthropogenic Cu contamination include (1) mining and smelting of other metals, (2) industrial activity, and (3) improper waste disposal in soil (Nogueirol et al. 2010). In order to remediate the contaminated environment, some plant species with phytoextraction potential are being studied.
Plants contain dozens of enzymes and proteins containing Cu, and this metal acts as an electron carrier in photosynthesis and as a receptor of chemical energy derived from proteins and oxidative enzymes (Epstein and Bloom 2004). Excess copper induces excessive production of reactive oxygen species, which damage the mitochondria, chloroplasts, and peroxisomes of the cells, causing the dysfunction of these organelles that are responsible for the main metabolic processes, including the production of chemical energy, photosynthesis, photorespiration, oxidative phosphorylation, β-oxidation, and tricarboxylic acid cycle (Chandna et al. 2012).
Phytoremediation is a sustainable and eco-friendly technique that can be applied in metal-contaminated soils. Besides having a considerable metal tolerance, the chosen species must have phytoextraction potential. Panicum maximum cv. Tanzania possesses these characteristics and studies are beginning to confirm its potential as a promising species for phytoextraction (Souza Junior et al. 2018; Leite and Monteiro 2019). Compared to hyperaccumulating species, Tanzania guinea grass has higher Cu, cadmium, and barium extraction potential because it is able to accumulate more of the metal due to its higher biomass production (Monteiro et al., 2011; Gilabel et al. 2014; Rabêlo et al. 2016). Also, this grass can regrow after the harvest of its biomass, thus alleviating the need for resowing, simplifying and accelerating the environment recovery.
Panicum maximum is a tropical forage grass species that is grown throughout ~ 20% of the 100 million hectares of cultivated pastures in Brazil (Torres et al. 2016). This species has a number of cultivars, including Tanzania guinea grass, which is characterized by its rusticity, high leaf to stem ratio, high seed production, high regrowth rate, and low seasonality (Jank et al. 2010; Jank et al. 2013). Besides the use of this grass for animal feeding and ground cover, its potential for phytoextraction of contaminant metals has been investigated in a number of studies (Monteiro et al. 2011; Gilabel et al. 2014; Rabêlo et al. 2016; Souza Junior et al. 2018; Leite and Monteiro 2019).
Studies that aim to enhance the phytoremediation potential of plants and to modulate the phytoextraction of metals have been conducted by adding chemical elements to the growth medium. Silicon (Si) is widely reported as a beneficial growth-promoting element, which promotes protection against biotic (Van Bockhaven et al. 2013; Vulavala et al. 2016) and abiotic (Kim et al. 2014; Abdula et al. 2016; Cooke and Leishman 2016) stresses. Plants well-nourished with Si are able to better deal with metal contamination due to the capacity of Si in holding the metal in the cell wall by providing binding sites (Li et al. 2008). Silicon also acts on lowering apoplastic efflux inducing less metal transport (Ma and Yamaji 2006) and metal compartmentalization in the cell vacuole (Wang et al. 2015). Silicon has presented satisfactory results on improving Cu tolerance in plants, but this has not yet been proven for tropical grasses such as Tanzania guinea grass.
It is believed that Si plays an important role on alleviating Cu toxicity in Tanzania guinea grass. Silicon may contribute to the increase in the tolerance of Tanzania guinea grass to Cu toxicity, especially in relation to the protection of the photosynthetic apparatus as a consequence of the reduction of Cu transport from roots to shoots (Adrees et al. 2015). The reduction of Cu transport in shoots may sound counterintuitive for the purpose of phytoextraction of this metal from the substrate, but reducing the rate of uptake prevents the plant from absorbing excessive concentrations of Cu which leads to a reduction in biomass production and therefore a reduction in overall phytoextraction from the plant as a whole (Gilabel et al. 2014; Souza Junior et al. 2018).
The aim of the present study was to evaluate the role of Si on the enhancement of phytoextraction potential of Tanzania guinea grass exposed to stressing Cu rates by measuring plant yield, chlorophyll contents, and concentrations and contents of Cu and Si.
Material and methods
Experimental conditions
The experiment was carried out in greenhouse conditions from January 7 to March 25, 2017, in the Soil Science Department of the “Luiz de Queiroz” College of Agriculture in the University of São Paulo, Piracicaba, Brazil. Seedlings of Tanzania guinea grass were obtained in sand washed with deionized water. Thirteen days after sowing, the seedlings reached 4–5-cm height and they were transplanted to 3.5-L pots filled with ground quartz as substrate in a hydroponic system. In the first 5 days after transplantation, the Hoagland and Arnon (1950) solution was used with 20% of ionic force in order to adapt the seedlings to the growth medium. Afterward, the nutrient solution was used with 100% of ionic strength (Table S1) and the seedlings were thinned to five per pot.
The substrate used was ground quartz because Tanzania guinea grass has an erect growth and also can grow up to 1 m. In other words, this grass requires a substrate wherein it can develop its roots and support itself. Despite the fact that ground quartz is composed by silica (silicon dioxide), it generally does not release significant amounts of Si into the solution at normal pH levels of water (Singh 2005). Since the nutrient solution pH level was kept around 6.0, there was no relevant release of Si.
The experiment was set on a complete random block design in a 3 × 4 factorial, with three replicates. Based on preceding research, three Si rates (0, 1, and 3 mmol L−1) and four Cu rates (0.3, 250, 500, and 750 μmol L−1) were adopted, which were introduced using Fertisil® (12% Si and 12% K2O) and CuCl2·2H2O. Thirty-six pots were used in this study with 1.5 L of nutrient solution each. Copper rates were applied at the 25th day after transplantation. Except for the 7 days of Cu exposure period, the concentration of Cu was unique, 0.3 μmol L−1, but plants in the experimental units were supplied with their respective Si rates. The harvest of the first growth was done 32 days after transplantation, when severe Cu toxicity was evidenced.
Aiming to evaluate the residual effects of Cu on the regrowth of Tanzania guinea grass, plants were evaluated in two growth periods. The harvesting time of both growths was determined by the onset of plant senescence (Schmidt and Monteiro, 2015). In order to allow the plants to regrow, 5 cm of the stalk was preserved. At the regrowth period, the harvest occurred 30 days after the first harvest.
Relative chlorophyll index (SPAD value)
Chlorophyll quantification was performed in the end of each growth period by indirect measurement obtained from the determination of SPAD units. For this determination, the portable device SPAD 502 was used and the procedure consisted of 10 measures in the middle third of the blade of the newly expanded leaves (diagnostic leaves), in all experimental units (Lavres Júnior et al. 2010).
Growth parameters and plant yield
The number of tillers and the number of leaves were counted at the end of each growth period. The shoots were collected in the two growth periods and the roots were collected just at the end of the second growth period. Each part of each replicate was stored in paper bags and dried in a forced-air oven at 65 °C for 72 h. After drying the collected plant material, the dry mass production of the plant parts was determined using an analytical scale. For the root surface measurement, it was separated approximately 20% of the fresh roots and immersed in 50 mg L−1 Gentian violet solution. After drying, this fraction of the root system was blushed, scanned, and transformed into a digital file in order to quantify the surface and root length by the software SIARCS 3.0 (Crestana et al. 1994).
Concentration and accumulation of Si and Cu
For the determination of Cu concentration in the plant tissue, the methodology described by Sarruge and Haag (1974) was used, which consists of nitric-perchloric digestion of the ground plant material and subsequent analysis of the extract in an atomic absorption spectrophotometer (PerkinElmer AAnalyst 400). Silicon concentration in the plant tissue was determined following the method described by Elliott and Snyder (1991), starting with the digestion of ground plant material using sodium hydroxide and hydrogen peroxide, after autoclaving and analyzing in a photocolorimeter with wavelength of 410 nm. Both Si and Cu concentrations were determined in the two most recently expanded leaves called diagnostic leaves (DL).
Silicon and Cu contents in the plant parts were calculated as the product of the concentration of these elements by the respective dry masses produced in each plant part. The copper transport factor was calculated by dividing the Cu content in the shoots from both growth periods by the Cu content in the roots (Kabata-Pendias and Pendias 2001).
Statistical analysis
All data were statistically analyzed using the software “Statistical Analysis System” (SAS Institute 2008). Analysis of variance was performed firstly by the GLM procedure. The F test was used to verify if there is significance in the interaction among Si rates × Cu rates. In the case with significant interaction, an outspread of this interaction was carried out. When there was no significance in the interaction, a comparison of means for Si rates and regression analysis by the REG procedure for Cu rates were performed. Comparison of means was done by the Tukey test at 5%.
Results
Plant yield, morphology, and chlorophyll content
The yield characteristics of the Tanzania guinea grass were influenced by the exposure rates of both Cu and Si. The number of tillers during the first growth was affected by both Si and Cu rates, without significant interaction (Fig. 1a, b). As Cu rates increased, the number of tillers decreased with the plants exposed to 750 μmol L−1 and 0.3 μmol L−1 of Cu differing by about 18% (Fig. 1a). Silicon supply increased the number of tillers by 14 to 23% compared with plants that were grown in the no-Si supply condition, but there was no significant difference between plants grown with 1 and 3 mmol L−1 of Si (Fig. 1b). In the second growth, the interaction Cu rates × Si rates was significant for the number of tillers (Fig. 1c). The number of tillers declined with increasing Cu rates. Silicon supplementation of 3 mmol L−1 decreased the number of tillers in plants under Cu rates of 0.3 and 250 μmol L−1; however, as the toxicity increased, Si supply resulted in higher values.
Number of tillers per pot in the first (a and b) and second growth period (c) of Panicum maximum cv. Tanzania as influenced by Cu and Si rates. In (b), different letters show significant difference (Tukey test, p < 0.05). In (c), different capital letters and different lowercase letters indicate significant difference (Tukey test, p < 0.05) among Cu and Si rates, respectively
The number of leaves was also affected by Si and Cu rates separately, without significant interaction in the first growth (Fig. 2a, b). The increasing rate of Cu resulted in lower leaf production (Fig. 2a). Plants treated with 750 μmol L−1 had 36% less leaves than those exposed to 0.3 μmol L−1 of Cu. Plants exposed to 3 mmol L−1 of Si showed the highest number of leaves and there was no significant difference between 0 and 1 mmol L−1 of Si (Fig. 2b). In the second growth, the interaction Cu rates × Si rates was significant (Fig. 2c). There was no significant difference between 0 and 1 mmol L−1 of Si regardless of the Cu rate applied. However, when the grass was exposed to Cu rates of 500 and 750 μmol L−1 and combined with Si of 3 mmol L−1, the number of leaves increased.
Number of leaves per pot in the first (a and b) and second growth period (c) of Panicum maximum cv. Tanzania as influenced by Cu and Si rates. In (b), different letters show significant difference (Tukey test, p < 0.05). In (c), different capital letters and different lowercase letters indicate significant difference (Tukey test, p < 0.05) among Cu and Si rates, respectively
The interaction Cu rates × Si rates was significant for the shoot dry weight of Tanzania guinea grass, in both growths (Fig. 3a, b). In the first growth period, the highest shoot dry weight was obtained with the combination of maximum Si rate and with the Cu rate of 0.3 μmol L−1 (Fig. 3a). Silicon supplementation of 3 mmol L−1 showed to significantly contribute to shoot dry weight in all Cu treatments. In the second growth period, the highest shoot dry weight was also achieved in the combinations with 0.3 μmol L−1 of Cu, however, with no significant difference between Si rates (Fig. 3b). The lowest values were acquired with the maximum Cu rate combined with no-Si supply. Except for the combinations under no Cu stress, silicon supply was responsible for the highest shoot dry weight in every Cu rate. Furthermore, plants exposed to the highest Cu rate did not regrow in the absence of Si.
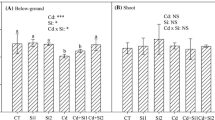
Root dry weight and root surface area of Tanzania guinea grass showed a similar performance with a significant interaction Cu rates × Si rates (Fig. 4a, b). Both parameters were improved by Si supply in the Cu rates of 500 and 750 μmol L−1, with the highest values for plants exposed to Cu of 0.3 μmol L−1 and the lowest values in plants treated with 750 μmol L−1 of Cu. When exposed to no Cu excess (Cu of 0.3 μmol L−1), the roots of the grass showed no benefit from the Si application. Root length was significantly affected by Cu rates but not by Si rates (Fig. 4c). Comparing the more contrasting Cu rates (0.3 and 750 μmol L−1), the Cu toxicity led to a reduction of 82% in root length.
The interaction Cu rates × Si rates was significant for SPAD in the first growth period (Fig. 5a). Tanzania guinea grass supplied with Si showed higher values for SPAD only in the Cu rates of 250 and 750 μmol L−1, but there was no significant difference between 0 and 1 mmol L−1 of Si. Plants supplied with Si of 3 mmol L−1 and exposed to Cu of 750 μmol L−1 had a SPAD value 36% higher than plants exposed to the same Cu rate without Si supply. There was no significant interaction Cu rates × Si rates in the second growth; however, both factors separately affected SPAD value (Fig. 5b, c). Copper rates positively influenced SPAD values up to 500 μmol L−1, with an increment of 36% compared to the Cu rate of 0.3 μmol L−1. Copper rates above 500 μmol L−1 reduced SPAD values by about 21% (Fig. 5b). Silicon supply also positively influenced SPAD values, even though there was no difference for Si between 1 and 3 mmol L−1 but there was significant difference between application and no supply of Si. Silicon application in the nutrient solution resulted in an increase of 15% in SPAD values (Fig. 5c).
SPAD values in diagnostic leaves in the first (a) and in the second growth period (b and c) of Panicum maximum cv. Tanzania as influenced by Cu and Si rates. In (a), different capital letters and different lowercase letters indicate significant difference (Tukey test, p < 0.05) among Cu and Si rates, respectively. In (b), different letters show significant difference (Tukey test, p < 0.05)
Cu and Si concentrations and contents
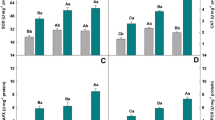
In the first growth period, the interaction Cu rates × Si rates was significant for Cu concentrations in diagnostic leaves (DL) and Cu content in shoots of Tanzania guinea grass. Plants supplied with Si showed less Cu in DL and accumulated less Cu in shoots in the first growth, when exposed to excessive Cu rates (Fig. 6a, c). In the second growth, Cu concentration in DL and Cu content in shoots were only affected by Cu rates (Fig. 6b, d). Plants exposed to Cu of 750 μmol L−1 concentrated 22 times more Cu in shoots than plants exposed to Cu of 0.3 μmol L−1. Copper content in shoots increased as the concentration of this metal in the nutrient solution increased up to 500 μmol L−1, after which, Cu content decreased. For Cu concentration and content in roots, the interaction Cu rates × Si rates was significant (Fig. 6e, f). In roots, the highest Cu concentration occurred in combinations with 0 and 1 mmol L−1 combined with Cu of 750 μmol L−1. The opposite was observed for Cu content in roots in plants exposed to Cu of 250 and 500 μmol L−1, where combinations with the highest Si rates showed the highest Cu content.
Copper concentration in diagnostic leaves (mg per kg of dry weight) (a, first growth; b, second growth) and in roots (e), and Cu content in shoots (c, first growth; d, second growth) and in roots (f) of Panicum maximum cv. Tanzania as influenced by Cu and Si rates. In (a, c, e, and f), different capital letters and different lowercase letters indicate significant difference (Tukey test, p < 0.05) among Cu and Si rates, respectively
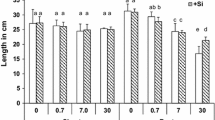
The interaction Cu rates × Si rates was significant to Si concentration in DL of Tanzania guinea grass during the first growth period (Fig. 7a). In the first growth, plants supplied with Si had higher Si concentration in DL in every Cu rate. In the second growth, Si concentration in DL increased with the Si supply and was only significantly affected by Si rates (Fig. 7b). In both the first and the second growth periods, the interaction Cu rates × Si rates was significant to silicon content in shoots (Fig. 7c, d). Silicon content in shoots in both growth periods increased with Si supply and decreased under Cu rates. In roots, the interaction Cu rates × Si rates was significant for Si concentration and content (Fig. 7e, f). Silicon concentration increased with the increasing Cu rates. The opposite occurred for Si content in roots, which decreased with the increase in Cu rates.
Silicon concentration in diagnostic leaves (g per kg of dry weight) (a, first growth; b, second growth) and roots (e), and Cu content in shoots (c, first growth; d, second growth) and roots (f) of Panicum maximum cv. Tanzania as influenced by Cu and Si rates. In (a, c, d, e, and f), different capital letters and different lowercase letters indicate significant difference (Tukey test, p < 0.05) among Cu and Si rates, respectively. In (b), different letters show significant difference by Tukey test (Tukey test, p < 0.05)
Accounting for both growth periods, the interaction Cu rates × Si rates was significant for Cu transport factor (Fig. 8). Tanzania guinea grass under excessive rates of Cu showed a lower Cu transport factor than when it was receiving the required Cu rate. Plants supplied with Si showed a lower Cu transport factor than plants not supplied with Si. It happened even when plants were under no Cu excess (0.3 μmol L−1 Cu). Silicon application did not significantly reduce the Cu transport factor just in the treatments with 250 μmol L−1 Cu. Furthermore, Si rates of 1 and 3 mmol L−1 were significantly different only in the combinations with 500 μmol L−1.
Discussion
Forage grasses are well known for their potential for high biomass production, characterized by a considerable number of leaves and tillers (Langer 1979). However, these characteristics are strongly affected when forages are exposed to metal toxicity. Tanzania guinea grass was greatly affected by Cu rates above 0.3 μmol L−1, which culminated in a low production of tillers, leaves, and shoot dry weight. Gilabel et al. (2014) also verified low production of aboveground biomass in Tanzania guinea grass as Cu rates increased above 0.3 μmol L−1. Nevertheless, the scenario changed when plants were supplied with Si. Plants that were exposed to various doses of Cu showed a higher number of tillers, leaves, and shoot dry weight when supplied with Si. Despite the fact that Si is not an essential element for higher plants, it has been already proven that Si can favor the growth and development of many plant species (Epstein 1994). Nowakowski and Nowakowska (1997) exposed wheat (Triticum aestivum) seedlings to Cu rates of 10, 20, and 40 μg cm−3 in combination with 500 μg cm−3 of Si and also reported greater increment in biomass production. In the current experiment, the Tanzania guinea grass also produced less belowground biomass when exposed to Cu excess in nutrient solution, but Si positively affected root dry weight and root area. Similar results were reported for cotton (Gossypium hirsutum) under Cu rates of 0, 25, and 50 μmol L−1 and Si rates of 0 and 1 mmol L−1 (Ali et al. 2016). The chlorophyll content of Tanzania guinea grass (indirectly quantified by SPAD values) during the first growth period dropped with the increment of Cu rates above 0.3 μmol L−1, but this reduction was less evident in plants supplied with Si. Rice (Oryza sativa) seedlings had reduced chlorophyll concentration when exposed to Cu toxicity with this reduction also being alleviated by the supply of Si (Kim et al. 2014). In the second growth period, the SPAD values of the grass increased with Cu rates. A likely explanation for this pattern is that the Cu toxicity that the plants were exposed to during the second growth period was lower than in the first growth period. Thus, these plants were able to readjust their metabolism and better deal with the disorders caused by excess Cu. This metabolism readjustment is denominated eustress, a stress that does not cause permanent damage but instead promotes plant health and productivity (Hideg et al. 2013). Plants exposed to residual Cu of 750 μmol L−1 were not able to readjust their metabolism because of the high rate of Cu that the grass was exposed to in the first growth period.
Even though Tanzania guinea grass produced less biomass as the Cu rates increased, Cu concentration in DL was also increased. Normal Cu concentrations in the younger leaves of Panicum maximum range from 4 to 14 mg kg−1 (Werner et al. 1996). In this study, the values reached over 600 mg kg−1 of Cu in the combinations of 750 μmol L−1 with no Si supply during the first growth. The low biomass production of Tanzania guinea grass intoxicated with Cu is strictly related to the concentration of this metal in plant tissues. The nutritional imbalance caused by excess Cu can induce destabilization of lipid components of the plasma membrane, causing increases in permeability, which induces an increase in Cu absorption itself (Maksymiec 1997). This membrane disruption can also reduce biomass production and chlorophyll contents in plants exposed to the highest Cu rate. The supply of Si reduced Cu concentrations in DL and in aboveground biomass in the first growth period. Collin et al. (2014) also found similar results when evaluating Cu concentration in aboveground biomass of bamboo (Phyllostachys fastuosa) exposed to Cu of 1.5 and 100 μmol L−1 and treated with 1.1 mmol L−1 of Si. Due to the reduction in Cu absorption when treated with Si, plants exposed to high Cu rates survived and still provided an appreciable biomass production in the second growth period. Plants exposed to 750 μmol L−1 and some combinations of 500 μmol L−1 of Cu did not survive in the absence of Si. In the second growth, the low Cu content in shoots of plants exposed to 750 μmol L−1 may be a consequence of their low aboveground biomass production. Copper concentration in roots also increased with the increment in Cu rates with no Si supply; however, Cu content in roots was higher in plants supplied with Si than those not supplied with Si. This can be explained by the substantially higher root biomass and reduction in the transport of Cu to the shoots in plants supplied with Si. Silicon supply similarly reduced Cu concentrations in plants of zinnia (Zinnia elegans) and snapdragon (Antirrhinum majus) exposed to Cu concentrations up to 150 μmol L−1 and supplied with Si of 1.7 and 3.4 mmol L−1 (Frantz et al. 2011).
Low copper uptake in the presence of Si is not yet fully understood. Previous studies have shown interesting results among the positive effect of Si on metal-intoxicated plants. A likely explanation for this is the reduction in apoplastic transport of metals by decreasing free metal concentration in the apoplast, which has already been confirmed in plants exposed to excess manganese (Iwasaki et al. 2002; Rogalla and Romheld 2002). According to Adrees et al. (2015), Si deposition may reduce metal concentration in the xylem as a consequence of the formation of a physical barrier in the endoderm proximity, thus reducing the cell wall porosity. Moreover, these authors stated that the concentrations of Cu in shoots are lower than those in the roots as Si decreases metal translocation. This decrease in metal translocation might be a result of alterations in the structure in shoots and roots, co-precipitation, chelation, and compartmentation of metals in plants. In the present study, once the Cu concentration and transport was low in the grass supplied with Si, these plants were exposed to a lower level of toxicity than those that did not receive Si supply. This was evidenced in a preliminary study, where we observed that ROS production was considerably lower in plants exposed to Cu rates of 500 and 750 μmol L−1 Cu and supplied with Si (unpublished results). Silicon supplementation also resulted in a better function of the antioxidant system enzymes of Tanzania guinea grass under Cu excess (unpublished results).
Silicon concentrations in DL were largely influenced by the increasing Cu rates in the first growth. Bamboo plants exposed to Cu rates up to 100 μmol L−1 and supplied with Si of 1.1 mmol L−1 also showed higher Si concentrations in shoots than those also supplied with Si in the absence of Cu. It suggests that this grass absorbs more silicon in an attempt of better deal with Cu toxicity. In the present study, concentrations of Si in roots showed a similar pattern to the shoots, the concentration rose as Cu rates increased. This goes against the findings of Mateos-Naranjo et al. (2015) that obtained lower Si concentration in roots of Spartina densiflora exposed to an excessive Cu rate (15 mmol L−1) than those exposed to the Cu rate of 0.5 μmol L−1. Kim et al. (2014) studied rice seedlings exposed to 100 μmol L−1 of Cu and found results that corroborate with the findings of the present study, wherein plants supplied with 1 mmol L−1 of Si had higher Si concentrations in their roots than the ones with the Cu rate of 0.5 μmol L−1. In both growth periods, Si content was increased with decreasing Cu rates. The accumulation of Si in belowground biomass also increased with Si supply and reduced by Cu rates. This is once more explained by the considerable large biomass of plants less intoxicated with Cu due to Si supply.
This study improves the current knowledge in regard to Panicum maximum cv. Tanzania and its potential for phytoextraction. Copper toxicity is becoming a worldwide problem and sustainable and eco-friendly solutions have to be investigated. Tanzania guinea grass performed well absorbing metal from the nutrient solution through phytoextraction when not supplied with Si. However, Si-supplied plants were able to control the absorption of Cu and promote regrowth after cutting. The grass exposed to the highest Cu rate showed the highest values of Cu concentration when not treated with Si and presented a lower biomass in both growth periods. In other words, under Cu toxicity, the main benefit of Si treatment to this grass is to maintain prolonged productivity and thus continued phytoextraction of Cu from the substrate.
Conclusion
Excess Cu negatively affected the biomass production and chlorophyll contents of Panicum maximum cv. Tanzania. Silicon supply improved biomass production and chlorophyll content by decreasing Cu concentration. Besides reducing Cu absorption, the most important role of Si was to reduce the transport of Cu from roots to shoots. Although Si supply did not increase Cu phytoextraction, it can allow successive harvesting of the aboveground biomass, which could be an interesting approach in phytoremediation programs.
References
Abdula SE, Lee HJ, Ryu H, Kang KK, Nou I, Sorrells ME, Cho YG (2016) Overexpression of BrCIPK1 gene enhances abiotic stress tolerance by increasing proline biosynthesis in rice. Plant Mol Biol Rep 34:501–511. https://doi.org/10.1007/s11105-015-0939-x
Adrees M, Ali S, Rizwan M, Zia-ur-Rehman M, Ibrahim M, Abbas F, Farid M, Qayyum MF, Irshad MK (2015) Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: a review. Ecotox Environ Safe 119:186–197. https://doi.org/10.1016/j.ecoenv.2015.05.011
Ali S, Rizwan M, Ullah N, Bharwana SA, Waseem M, Farooq MA, Abbasi GH, Farid M (2016) Physiological and biochemical mechanisms of silicon-induced copper stress tolerance in cotton (Gossypium hirsutum L.). Acta Physiol Plant 38:262. https://doi.org/10.1007/s11738-016-2279-3
Chandna R, Hakeem KU, Ahmad P (2012) Proteomic markers for oxidative stress: new tools for reactive oxygen species and photosynthesis research. In: Ahmad P, Prasad MNV (eds) Abiotic stress responses in plants: metabolism, productivity and sustainability. Springer, New York, pp 181–196
Collin B, Doelsch E, Keller C, Cazevieille P, Tella M, Chaurand P, Panfili F, Hazemann JL, Meunier JD (2014) Evidence of sulfur-bound reduced copper in bamboo exposed to high silicon and copper concentrations. Environ Pollut 187:22–30. https://doi.org/10.1016/j.envpol.2013.12.024
Cooke J, Leishman MR (2016) Consistent alleviation of abiotic stress with silicon addition: a meta-analysis. Funct Ecol 30:1340–1357. https://doi.org/10.1111/1365-2435.12713
Crestana S, Guimarães F, Jorge LAC, Ralish R, Tozzi CL, Torre A, Vaz CMP (1994) Avaliação da distribuição de raízes no solo por processamento de imagens digitais. Rev Bras Cienc Solo. 18:365–337
Elliott CL, Snyder GH (1991) Autoclave-induced digestion for the colorimetric determination of silicon in rice straw. J Agr Food Chem 39:1118–1119. https://doi.org/10.1021/jf00006a024
Epstein E (1994) The anomaly of silicon in plant biology. P Natl Acad Sci USA 91:11–17. https://doi.org/10.1073/pnas.91.1.11
Epstein E, Bloom A (2004) Mineral nutrition of plants: principles and perspectives. Sinauer Associates, Sunderland 380 p
Frantz JM, Khandekar S, Leisner S (2011) Silicon differentially influences copper toxicity response in silicon-accumulator and non-accumulator species. J Am Soc Hortic Sci 136:329–338
Gilabel AP, Nogueirol RC, Garbo AI, Monteiro FA (2014) The role of sulfur in increasing guinea grass tolerance of copper phytotoxicity. Water Air Soil Poll 225:1806–1816. https://doi.org/10.1007/s11270-013-1806-8
Girotto E, Ceretta CA, Rossato LV, Farias JG, Brunetto G, Miotto A, Tiecher TL, Conti L, Lourenzi CR, Schmatz R, Giachini A, Nicoloso FT (2016) Biochemical changes in black oat (Avena strigosa schreb) cultivated in vineyard soils contaminated with copper. Plant Physiol Biochem 103:199–207. https://doi.org/10.1016/j.plaphy.2016.02.030
Hideg E, Jansen MAK, Strid A (2013) UV-B exposure, ROS, and stress: inseparable companions or loosely linked associates? Trends Plant Sci 18:107–115. https://doi.org/10.1016/j.tplants.2012.09.003
Hoaghland D, Arnon DI (1950) The water culture method for growing plants without soil. California Agricultural Experiment Station, Berkeley, 31 p
Iwasaki K, Maier P, Fecht M, Horst WJ (2002) Leaf apoplastic silicon enhances manganese tolerance of cowpea (Vigna unguiculata). J Plant Physiol 159:167–173. https://doi.org/10.1078/0176-1617-00691
Jank L, Martuscello JA, Euclides VPB, Valle CB, Resende RMS (2010) Panicum maximum. In: Fonseca DM, Martuscello JA (eds) Forage plants. UFV, Viçosa, pp 166–196 (in Portuguese)
Jank L, Braz TGS, Martuscello JA (2013) Warm-season grasses. In: Reis RA, Bernardes TF, Siqueira GR (eds) Forage farming: science, technology and management of forage resources. Multipress, Jaboticabal, pp 109–125 (in Portuguese)
Kabata-Pendias A, Pendias H (2001) Trace elements in soils and plants, 3rd edn. CRC Press, Boca Raton
Kim YH, Khan AL, Kim DH, Lee SY, Kim KM, Waqas M, Jung HY, Shin JH, Kim JG, Lee IJ (2014) Silicon mitigates heavy metal stress by regulating P-type heavy metal ATPases, Oryza sativa low silicon genes, and endogenous phytohormones. BMC Plant Biol 14:13. https://doi.org/10.1186/1471-2229-14-13
Langer RHM (1979) How grasses grow. Eduard Arnold Ltd., London
Lavres Júnior J, Santos JDG, Monteiro FA (2010) Nitrate reductase activity and SPAD readings in leaf tissues of guinea grass submitted to nitrogen and potassium rates. Rev Bras Cienc Solo 34:801–809. https://doi.org/10.1590/S0100-06832010000300022
Leite TS, Monteiro FA (2019) Nitrogen form regulates cadmium uptake and accumulation in Tanzania guinea grass used for phytoextraction. Chemosphere 236:9. https://doi.org/10.1016/j.chemosphere.2019.07.055
Li J, Leisner SM, Frantz J (2008) Alleviation of copper toxicity in Arabidopsis thaliana by silicon addition to hydroponic solutions. J Am Soc Hortic Sci 133:670–677
Ma JF, Yamaji N (2006) Silicon uptake and accumulation in higher plants. Trends Plant Sci 11:392–397. https://doi.org/10.1016/j.tplants.2006.06.007
Maksymiec W (1997) Effect of copper on cellular processes in higher plants. Photosynthetica. 34:321–342. https://doi.org/10.1023/A:1006818815528
Mateos-Naranjo E, Galle A, Florez-Sarasa I, Perdomo JA, Galmes J, Ribas-Carbo M, Flexas J (2015) Assessment of the role of silicon in the Cu-tolerance of the C-4 grass Spartina densiflora. J Plant Physiol 178:74–83. https://doi.org/10.1016/j.jplph.2015.03.001
Megateli S, Dosnon-Olette R, Trotel-Aziz P, Geffard A, Semsari S, Couderchet M (2013) Simultaneous effects of two fungicides (copper and dimethomorph) on their phytoremediation using Lemna minor. Ecotoxicology. 22:683–692. https://doi.org/10.1007/s10646-013-1060-2
Monteiro FA, Nogueirol RC, Melo LCA, Artur AG, Rocha F (2011) Effect of barium on growth and macronutrient nutrition in Tanzania guinea grass grown in nutrient solution. Commun Soil Sci Plan 42:1510–1521. https://doi.org/10.1080/00103624.2011.581725
Nogueirol RC, Alleoni LRF, Nachtigall GR, Melo GW (2010) Sequential extraction and availability of copper in Cu fungicide-amended vineyard soils from Southern Brazil. J Hazard Mater 181:931–937. https://doi.org/10.1016/j.jhazmat.2010.05.102
Nowakowski W, Nowakowska J (1997) Silicon and copper interaction in the growth of spring wheat seedlings. Biol Plantarum 39:463–466. https://doi.org/10.1023/A:1001009100026
Rabêlo FHS, Azevedo RA, Monteiro FA (2016) Proper supply of S increases GSH synthesis in the establishment and reduces tiller mortality during the regrowth of Tanzania guinea grass used for Cd phytoextraction. J Soils Sediments 1:1–10. https://doi.org/10.1007/s11368-016-1429-y
Rogalla H, Romheld V (2002) Role of leaf apoplast in silicon-mediated manganese tolerance of Cucumis sativus L. Plant Cell Environ 25:549–555. https://doi.org/10.1046/j.1365-3040.2002.00835.x
Sarruge JR, Haag HP (1974) Análises químicas em plantas. ESALQ, Piracicaba
SAS Institute (2008) SAS/STAT: qualification tools user’s guide, version 9.2, Cary
Schmidt F, Monteiro FA (2015) Sulphur supply affects morphology, growth and nutritional status of Tanzania guinea grass and Mineirão stylo. Grass Forage Sci 70:439–450. https://doi.org/10.1111/gfs.12122
Singh R (2005) Water and membrane treatment. In: Singh R (ed) Hybrid membrane systems for water purification: technology, systems design and operation. Elsevier Science, Colorado Springs, pp 57–130
Souza Junior JC, Nogueirol RC, Monteiro FA (2018) NO3-/NH4+ ratios affect nutritional homeostasis and production of Tanzania guinea grass under Cu toxicity. Environ Sci Pollut R 25:14083–14096. https://doi.org/10.1007/s11356-018-1541-1
Torres FE, Teodoro PE, Benites SB, Oliveira EF, Correa CCG, Silva FA (2016) Number of cuts for estimating forage productivity in Panicum maximum. Biosci J 32:172–178. https://doi.org/10.14393/BJ-v32n1a2016-29307
Van Bockhaven J, De Vleesschauwer D, Hofte M (2013) Towards establishing broad-spectrum disease resistance in plants: silicon leads the way. J Exp Bot 64:1281–1293. https://doi.org/10.1093/jxb/ers329
Vulavala VKR, Elbaum R, Yermiyahu U, Fogelman E, Kumar A, Ginzberg I (2016) Silicon fertilization of potato: expression of putative transporters and tuber skin quality. Planta. 243:217–229. https://doi.org/10.1007/s00425-015-2401-6
Wang SH, Wang FY, Gao SC (2015) Foliar application with nano-silicon alleviates Cd toxicity in rice seedlings. Environ Sci Pollut R. 22:2837–2845. https://doi.org/10.1007/s11356-014-3525-0
Werner JC, Paulino VT, Cantarella H, Andrade NO, Quaggio JA (1996) Forrageiras. In: Van Raij B, Cantarella H, Quaggio JA, Furlani AMC (eds) Recomendações de adubação e calagem para o Estado de São Paulo. IAC, Campinas
Acknowledgments
Financial support to the study (grant no. 307030/2017-2) from the Brazilian National Research Council (CNPq) and fellowship granted to the first author during his whole Master’s program from Coordination for the Improvement of Higher Education Personnel (CAPES) are gratefully acknowledged. The authors also thank the participants of the Plant Mineral Nutrition Research Group at ESALQ-USP for the help in all phases of the research project.
Funding
Brazilian National Research Council (CNPq)—financial support to the study and fellowship to the second author (grant no. 307030/2017-2).
Coordination for the Improvement of Higher Education Personnel (CAPES) – Financial Code 001—fellowship granted to the first author during his whole Master’s program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Vieira Filho, L., Monteiro, F.A. Silicon modulates copper absorption and increases yield of Tanzania guinea grass under copper toxicity. Environ Sci Pollut Res 27, 31221–31232 (2020). https://doi.org/10.1007/s11356-020-09337-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09337-4