Abstract
Several epidemiological studies have investigated the adverse health effects of air pollution, but studies reporting its effects on allergic rhinitis (AR) are limited, especially in developing countries having the most severe pollution. Limited studies have been conducted in China, but their results were inconsistent. So, we conducted a time-series study to evaluate the acute effect of six air pollutants (fine particulate matter [PM2.5], particulate matter with diameter less than 10 μm [PM10], sulfur dioxide [SO2], nitrogen dioxide [NO2], ozone [O3], and carbon monoxide [CO]) on hospital outpatient visits for AR in Xinxiang, China from January 1, 2015, to December 31, 2018. An over-dispersed Poisson generalized additive model adjusting for weather conditions, long-term trends, and day of the week was used. In total, 14,965 AR outpatient records were collected during the study period. Results found that each 10 μg/m3 increase in PM2.5, PM10, SO2, NO2, O3, and CO corresponded to 0.70% (95% confidence interval 0.00–1.41%), 0.79% (0.35–1.23%), 3.43% (1.47–5.39%), 4.54% (3.01–6.08%), 0.97% (− 0.11–2.05%), and 0.07% (0.02–0.12%) increments in AR outpatients on the current day, respectively. In the stratification analyses, statistically stronger associations were observed with PM2.5, PM10, SO2, NO2, and CO for AR outpatients < 15 years of age than in those 15–65 and ≥ 65 years of age, whereas the opposite result was found with O3. Associations between PM10, SO2, NO2, O3, and AR outpatients were higher in the warm season than those in the cool season. This study suggests that exposure to PM2.5, PM10, SO2, NO2, and CO was associated with increased AR risk and children younger than 15 years might be more vulnerable.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Air pollution has become a highly important health risk factor. As the largest developing country worldwide, China faces the most severe air pollution problems, especially in city clusters, including the Jing-Jin-Ji, Yangtze River Delta, and Pearl River Delta areas (Meng et al. 2012; van Donkelaar et al. 2010). The gross domestic product increased from 10,028 billion in 2000 to 90,031 billion in 2018, and the number of automobiles raised from 1.6 million in 2000 to 327 million in 2018 (Statistics 2000–2018). Rapid economic growth over the last decade has led to a continuous increase in energy consumption, which is the largest contributor to ambient air pollution (Maji et al. 2019). Furthermore, the sharp increase in vehicles also discharges great amounts of air pollutants (Dong et al. 2013; Jaakkola et al. 2006; Lehmann et al. 2001). Accumulating evidence has proposed that air pollution exposure increases the risk of morbidity and mortality, particularly in respiratory and cardiovascular diseases (BF and YL 2010; He et al. 2011; Li et al. 2015; Mihălţan et al. 2016; Shang et al. 2013).
AR is a very common disease that affects people of all ages, with about 30–40% of the population affected globally (Zou et al. 2018). Usually, AR is not a serious illness, but it has a considerable effect on quality of life and can have significant consequences if left untreated (Greiner et al. 2011; Nathan 2007). Sometimes, it can cause major illness and disability (Bousquet et al. 2008; Brozek et al. 2010). Previous evidence showed that air pollution may play important roles in the etiology of AR (Penard-Morand et al. 2005). Individuals with AR have nasal hyperreactivity, rendering them more responsive to air pollutant irritants (Dunlop et al. 2016). Compared to other respiratory diseases such as pneumonia and asthma, relatively few studies have analyzed the adverse effect of air pollution on AR (Chen et al. 2016; Deng et al. 2016; Hu et al. 2017), especially in developing countries. Huang et al. found that high concentrations of fine particulate matter (PM2.5) and particulate matter with diameter less than 10 μm (PM10) corresponded to aggravated subjective symptoms in children with AR (Huang et al. 2017). One study conducted in Beijing, China, observed strong associations between sulfur dioxide (SO2), nitrogen dioxide (NO2), PM10, and AR outpatients (Zhang et al. 2011). A study in Taiwan found that PM10, NO2, ozone (O3), and carbon monoxide (CO) were associated with AR morbidity (Chen et al. 2016). Other studies found that PM2.5 and PM10 were positively associated with AR outpatients in Changchun and Nanjing, respectively (Chu et al. 2019; Teng et al. 2017). Chen et al. also found a positive relationship between PM2.5 and AR in six cities (Changsha, Chongqing, Nanjing, Shanghai, Taiyuan, and Urumqi) (Chen et al. 2018). However, contradictory results have been found for PM10 and O3 in Taiwanese schoolchildren (Hwang et al. 2006).
With air pollution levels from industrial and motor vehicle emissions rising rapidly in quickly industrializing and urbanizing countries, respiratory disease prevalence has also been increasing (Zhang et al. 2015). Xinxiang, one of the oldest cities in China, is experiencing the worst air pollution problems. Annual concentrations of PM2.5, PM10, and NO2 in 2015 were approximately 2.7, 2.3, and 1.3 times higher than the Chinese National Ambient Air Quality Standard (PM2.5, 35 μg/m3; PM10, 70 μg/m3; NO2, 40 μg/m3), respectively. However, few studies have examined the adverse health effects of air pollution in Xinxiang. Thus, we designed this study to investigate the acute effects of six ambient air pollutants (PM2.5, PM10, SO2, NO2, O3, and CO) on hospital outpatient visits for AR.
Methods
Xinxiang, located in the center of the North China Plain, is about 340 km south of Beijing and 60 km north of Zhengzhou (Fig. 1). Xinxiang has over 1400 years of history and comprises four urban districts, with a total area of 8249 km2 and a population of 5.77 million at the end of 2017. The study area was limited to the four traditional urban districts (422 km2). Xinxiang has a warm temperate continental climate. The warm season is defined from April to September, while the cool season lasts from October to March.
Health data
There are six hospitals in Xinxiang, of which the PLA 371 Hospital is a military hospital and the Xinxiang Third People’s Hospital lacks an electronic health information system. Therefore, AR outpatient records were collected from the other four hospitals: Xinxiang First People’s Hospital, Xinxiang Second People’s Hospital, Xinxiang Central Hospital, and the Third Affiliated Hospital of Xinxiang Medical University from January 1, 2015, to December 31, 2018. These four hospitals are distributed in four municipal administrative districts of Xinxiang (Fig. 1) and cover all residents of Xinxiang. Computerized records were retrieved from the health information system of each hospital. Several covariates such as birth date, outpatient date, age, gender, address, department of visit, doctor, and diseases were extracted for each record. Disease names were diagnosed by clinical doctors, and all hospitals have the same diagnosis guideline. The International Classification of Diseases (ICD, 10th revision) codes were re-matched, and data cleaning and quality control procedures were conducted as previously described (Song et al. 2018). The number of daily hospital visits was summarized according to ICD10 codes (J30 for AR), and the data were also classified by season, sex, and age (< 15, 15–64, and ≥ 65 years).
The Institutional Review Board of Xinxiang Medical University approved the study protocol (NO. 2018-1207-0203).
Air pollution and meteorological data
In this study, daily concentrations of PM2.5, PM10, SO2, NO2, O3, and CO in Xinxiang were collected from the website of China’s National Urban Air Quality Real-time Publishing Platform (http//106.37.208.235.20035/). The daily average concentrations of these six ambient air pollutants were based on four fixed-site monitors operated under the Xinxiang Ministry of Ecology and Environment, which are distributed in urban districts (Fig. 1). Daily concentrations of PM2.5, PM10, SO2, NO2, and CO were calculated as the 24-h mean value, and the concentration of O3 was calculated as the maximal 8-h average. To calculate the mean concentrations of each pollutant, more than 75% of the hourly values must have been available on that day, otherwise, data for that day were excluded (Huang et al. 2017).
In addition, we collected two meteorological parameters in Xinxiang, the daily mean relative humidity and mean temperature, from China’s Meteorological Data Sharing Service System (http://data.cma.cn/). The geographic description of the meteorological monitor is also indicated in Fig. 1.
Statistical analyses
To better scrutinize our research, an over-dispersed generalized additive model (GAM) was used to examine the relationships between air pollutants and AR outpatients. Several time-varying confounders were adjusted based on previous studies (Liang et al. 2018; Qiu et al. 2012; Tian et al. 2016): (1) a natural smooth function of the calendar day with 7 degrees of freedom (df) per year was incorporated into the core model to exclude unmeasured long-term and seasonal trends longer than 2 months (7 df/year was selected as it could produce the smallest Akaike Information Criterion value); (2) a natural smooth function of daily mean temperature (6 df) and mean relative humidity (3 df) were structured to control their nonlinear confounding effects (Li et al. 2015); (3) a binary variable of public holidays and indicator variable of the day of the week were also calculated.
The exposure-response (E-R) curves of air pollutant effects on AR outpatients were plotted using a natural spline function with 3 df (Chen et al. 2018).
To explore the temporal pattern of six air pollutants on AR outpatients, several lag structures including single-day lags (lag 0 to lag 7) and moving average lag days (lag 0–1, 0–2, 0–3, 0–4, 0–5, 0–6, 0–7) were analyzed. Furthermore, two sensitivity analyses of two-pollutant models and df transformation studies (wherein the df value of time was transformed from 4 to 10 per year) were conducted to examine the robustness of our models.
Finally, three more stratification analyses were constructed to explore the potential modifying effect of gender, age, and season. The statistical significance between each stratification was calculated by 95% confidence intervals (CI) as before, in brief: (\( {\hat{Q}}_1-{\hat{Q}}_2 \))\( \pm 1.96\sqrt{{\left(\mathrm{S}\hat{\mathrm{E}}1\right)}^2+{\left(\mathrm{S}\hat{\mathrm{E}}2\right)}^2} \),where \( {\hat{Q}}_1 \) and \( {\hat{Q}}_2 \) are the estimates and SÊ1 and SÊ2 are their standard errors (Lin et al. 2016).
All statistical analyses were conducted in the R software (Version 3.3.3) using the mgcv package. Results with P < 0.05 were considered statistically significant. The effects are described as the percent changes and 95% CI in AR outpatients per 10 μg/m3 increase in air pollutants.
Results
Table 1 summarizes the descriptive statistics of daily AR outpatients, daily concentrations of six ambient air pollutants (PM10, PM2.5, SO2, NO2, O3, and CO), and daily meteorological conditions (mean relative humidity and temperature) from January 1, 2015, to December 31, 2018. In total, 14,965 AR outpatient records were collected from four hospitals, among which, there were 7912 male and 7050 female outpatients (3 more records with missing message). Additionally, of all AR outpatients, 2159 were < 15; 11,945 were 15–64; and 861 were ≥ 65 years of age. The annual mean concentrations of PM2.5, PM10, SO2, NO2, O3, and CO were 75.7, 132, 33.2, 48.4, 59.4, and 1377 μg/m3, respectively. These values for PM2.5, PM10, SO2, and NO2 were about 1.1, 3.8, 0.6, and 1.2 times higher than the Chinese National Ambient Air Quality Standards (PM2.5, 35 μg/m3; PM10, 70 μg/m3; SO2, 60 μg/m3; NO2, 40 μg/m3), and only 15 and 34 days of O3 and CO concentrations exceeded the daily limit (O3, 160 μg/m3; CO, 4000 μg/m3). The daily mean relative humidity was 60%, and the mean temperature was 16 °C in Xinxiang during our study period. Except for O3, the other five pollutants were strongly correlated with each other, with Spearman’s coefficients ranging from 0.44 to 0.90, and negatively correlated with daily mean temperature and mean relative humidity.
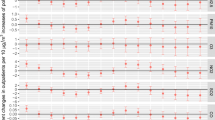
As shown in Fig. 2, the estimated effects of single-pollutant models are presented using different lag structures. Significant results were identified between air pollutants, except for O3, and outpatient visits for AR. Specifically, lag0 and lag01–lag07 for PM10; lag0, lag1, and lag01–lag06 for SO2; lag0, lag1, and lag01–lag07 for NO2; and lag0 for CO were statistically significant. The estimate between PM2.5 and AR outpatients on lag0 was marginal. Based on the model fit statistics, the current day (lag0) for PM2.5, PM10, SO2, NO2, and CO, and lag1 for O3 were selected as the best lag structures as they could produce the smallest generalized cross-validation value. This means that each 10 μg/m3 increase in concentrations of PM2.5, PM10, SO2, NO2, and CO corresponded to a 0.70% (95% CI 0.00–1.41%), 0.79% (0.35–1.23%), 3.43% (1.47–5.39%), 4.54% (3.01–6.08%), and 0.07% (0.02–0.12%) increase in daily hospital outpatients for AR on the current day, respectively. Meanwhile, the associations between O3 and AR outpatients were positive but not statistically significant with an estimate of 0.97% (95% CI − 0.11–2.05%).
The E-R curves for the relationships between six ambient air pollutants and daily AR outpatients are shown in Fig. 3. In general, the six curves were obviously positive. The E-R curves of O3 and CO were nearly S-shaped, rising sharply for concentrations ≥ 50 and ≥ 250 μg/m3 and flattening for concentrations ≥ 110 and ≥ 4000 μg/m3, respectively. The E-R curves of NO2, PM2.5, and PM10 showed a steep slope at concentrations < 1000, < 300, and < 350 μg/m3 and then drop down. The E-R curves of SO2 showed a flat slope at low concentrations and then a slight increase at concentrations ≥ 150 μg/m3.
Stratified analysis results are shown in Table 2. The estimated effects of PM2.5, PM10, SO2, NO2, and O3 on AR outpatients were a little higher in males than in females, although no significant differences were found. Significant differences were observed with age stratification, and the effects were strongest in patients < 15 years of age (except for O3). For the season modification, the effects of PM2.5 and CO were stronger in the cool season than in the warm season, but this difference was not significant; however, the effects of PM10, SO2, NO2, and O3 were stronger in the warm season than in the cool season, and statistical significance was determined for SO2 and O3.
Co-pollutants with a correlation coefficient < 0.7 were added to two-pollutant models. The associations of PM10, NO2, and O3 with AR outpatients remained robust after adjusting for co-pollutants (Table 3). However, when adjusting for SO2, a non-significant association was identified for PM2.5. For O3, all estimates were significant in the two-pollutant models. Meanwhile, the estimated effects of CO were insignificant when adjusting for PM10, NO2, and SO2. Altogether, these results indicate that PM10, NO2, and SO2 might play a more independent role in AR outpatient risk. Another sensitivity analysis showed similar results when the df for time were transformed from 4 to 10 (Fig. 4), suggesting that our results were likely not attributed to chance.
Discussion
We conducted a time-series study to investigate the acute association between six air pollutants (PM2.5, PM10, SO2, NO2, O3, CO) and hospital outpatient visits for AR. Significant associations were observed between PM2.5, PM10, SO2, NO2, CO, and AR outpatients. The associations were strongest in patients < 15 years of age and weakened as age increased. The acute effects of SO2 and O3 on AR outpatients were stronger in the warm season than in the cool season.
Compared to other respiratory diseases, few studies have focused on the association between ambient air pollution and AR risk. In this study, we found significant associations between PM10, SO2, NO2, CO, and AR outpatient visits. These findings are consistent with previous studies (Jo et al. 2017; Teng et al. 2017). One study conducted in Hobart, Australia, found that AR symptoms are associated with PM (IRR 1.06, 95% CI 1.04–1.08) (Jones et al. 2020). Min et al. observed a marginal association between PM2.5 and AR in children in Seoul, Korea (Min et al. 2020). Analyzed from 646,975 children records in Shanghai, Hu et al. showed that most of the pollutants (PM2.5, PM10, SO2, NO2, and O3) were significantly associated with AR (Hu et al. 2020). Using generalized additive Poisson model, Wang et al. also found significant association between PM2.5 and AR outpatient (Wang et al. 2020). A case-crossover study conducted in Taipei found positive associations between PM10, NO2, CO, O3, and AR morbidity on all days, the associations between SO2 and AR was significant in warm days (> 23 °C) but insignificant in cool days (< 23 °C) (Chen et al. 2016). A nationwide study covering 11 major Chinese cities showed that SO2 was significantly associated with adult AR prevalence (Zhang et al. 2009). One recently published research proved that PM2.5 and PM10 would induce increased serum-specific IgE of house dust mites (allergen of AR) (Ye et al. 2019). However, a 17 years follow-up study in Toronto did not find significant association between PM2.5, NO2, and O3 and AR (To et al. 2020). A recently published meta-analysis study also found no associations between PM10, O3, and childhood AR prevalence in Asia (Zou et al. 2018). Variations in study design, geographic distribution, study populations, and ethnicity may partially explain the inconsistencies between these results.
The exact mechanisms that underlie the associations between air pollutants and AR are not well established. Several pathways have been proposed, mainly inflammation exacerbation, oxidative stress promotion, signal transduction interference, enzyme inhibition, immunosuppression, and epigenetic dysregulation (Bayram 2017; Bloemsma et al. 2016; Chen et al. 2018; Heinrich and Schikowski 2018). For example, air pollutants such as PM, SO2, NO2, and O3 may damage the nasal mucosa and impair mucociliary clearance, thereby facilitating the introduction of inhaled pollutants to immune system cells (Dunlop et al. 2016). PM may enhance allergic inflammation and induce the development of allergic immune responses; NO2 is known for its adjuvant effect on airway hypersensitivity; and O3 could induce an influx of neutrophils and eosinophils in the nasal mucosa and further stimulate immunoglobulin E (IgE) synthesis (Chen et al. 2016). Furthermore, inhalable PM could carry high quantities of toxic pollutants, such as organic compounds and transition metals, which have the ability to generate pro-inflammatory responses by activating reactive oxygen species (Brook et al. 2004), thus potentially contributing to IgE overproduction and increased permeability of nasal mucosa in susceptible individuals (Diaz-Sanchez et al. 1999; Diaz-Sanchez et al. 1996). It has also been reported that PM2.5 may modify viability, apoptosis, cytokine release, and proliferation as well as apoptosis-regulating proteins of alveolar epithelial cells (Bayram 2017). There are no apparent plausible mechanisms by which CO influences the airway and enhances AR prevalence (Chen et al. 2016). Future studies are needed to investigate the mechanisms of CO-triggered AR.
Previous studies have demonstrated that the season might play an important role in the relationships between air pollutants and adverse health effects (Chou et al. 2017; Chu et al. 2019; Gu et al. 2017). Significantly stronger effects of SO2 and O3 on AR outpatients were found in the warm season in our study, which was consistent with previous studies (Chu et al. 2019; Zhang et al. 2013).
Generally, the highest association effects were found in outpatients under 15 years old, followed by those between 15 and 64 and ≥ 65 years of age. Previous studies speculated that children were most sensitive to air pollutants (Esposito et al. 2014; Zou et al. 2018). First, the respiratory system development is not complete at children. During the childhood, respiratory system is especially susceptible to toxicants (Goldizen et al. 2016; McBride 2015). Second, children have smaller airways than adults do, irritation caused by air pollution that would produce only a slight response in adult can result in potentially significant obstruction in a child (McBride 2015; Etzel 2007). Third, children have a higher rate of breathing, increasing the dose received (Goldizen et al. 2016). Fourth, children are shorter in height, thus their breath nearer the ground, and exposing them to higher concentrations of air pollutants (Goldizen et al. 2016; Etzel 2007). Fifth, children spend more time outside engaged in physical activity than adult does. Therefore, espousing them to higher dose of air pollutants (Goldizen et al. 2016). Finally, the detoxification systems of children are underdeveloped, which might cause serious health effects of exposure to environmental toxic substances (Goldizen et al. 2016). However, Wang et al. found higher sensitivity in the eldest outpatients (≥ 65) than in those 15–64 years of age (Wang and Chau 2013). In fact, several other studies observed no associations between PM and AR among children (Chu et al. 2019; Lin et al. 2016). These inconsistent results may be partially explained by economic statistics and the course of AR. Many AR episodes do not result in a hospital visit (Chen et al. 2016). In developing countries, especially in North China where the economy is poor, elderly patients did not visit the hospital until the symptoms were serious.
When patients were stratified by sex, we found higher associations in males, although the differences between sexes were insignificant. We speculate that this result might be due to occupational factors, as men may be engaged in more outdoor work, resulting in exposure to higher concentrations of air pollution. Esposito et al. found that boys are more affected than girls (Esposito et al. 2014), whereas the Nanjing study observed the opposite result (Chu et al. 2019). The exact reason for these differences between males and females must be elucidated in future studies.
Several limitations should be considered in our study. First, as our exposure data were obtained from fixed-site monitors, exposure measurement errors are inevitable. This could result in a misclassified exposure level. Previous studies have shown that this could produce a bias toward the null and underestimate the estimated effects (Zeger et al. 2000). Second, individual lifestyles (such as smoking and air conditioning use) and other exposure factors (such as plant pollen) can influence AR incidence (Samitas et al. 2018), which might affect the magnitude of the calculated estimates. Third, family history of AR and other allergy diseases is significantly associated with AR hospital outpatients (Dunlop et al. 2016). Due to the fact that the health data were collected from hospital outpatient records, the diseases history dates mentioned above were not collected. Finally, the study data were only collected from one highly polluted city, so the generalizability of our results may be limited.
Conclusions
This time-series study suggested that air pollutants, especially NO2, SO2, and PM10, could significantly increase AR outpatient visits in Xinxiang, China. Children < 15 years old are the most vulnerable. This analysis provides a better understanding of the health effects of air pollution in developing countries. Although the air pollution level has been sharply reduced in 2018, the implementation of policies that seek to improve air quality should remain a priority.
Abbreviations
- AR:
-
Allergic rhinitis
- PM2.5 :
-
Fine particulate matter
- PM10 :
-
Particulate matter with diameter less than 10 μm
- SO2 :
-
Sulfur dioxide
- NO2 :
-
Nitrogen dioxide
- O3 :
-
Ozone
- CO:
-
Carbon monoxide
- GAM:
-
Generalized additive model
- df:
-
Degrees of freedom
- IgE:
-
Immunoglobulin E
References
Bayram H (2017) Impact of air pollution on COPD; underlying mechanisms. Tanaffos 16:S10–S10
BF H, YL L (2010) Air pollution and prevalence of bronchitic symptoms among children in Taiwan. Chest 138:956–964
Bloemsma LD, Hoek G, Smit LAM (2016) Panel studies of air pollution in patients with COPD: systematic review and meta-analysis. Environ Res 151:458–468
Bousquet J et al (2008) Allergic rhinitis and its impact on asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2) LEN and AllerGen). Allergy 63(Suppl 86):8–160
Brook RD, Franklin B, Cascio W, Hong Y, Howard G, Lipsett M, Luepker R, Mittleman M, Samet J, Smith SC Jr, Tager I, Expert Panel on P, Prevention Science of the American Heart A (2004) Air pollution and cardiovascular disease: a statement for healthcare professionals from the expert panel on population and prevention science of the American Heart Association. Circulation 109:2655–2671
Brozek JL, Bousquet J, Baena-Cagnani CE, Bonini S, Canonica GW, Casale TB, van Wijk RG, Ohta K, Zuberbier T, Schunemann HJ, Global A, Asthma European N, Grading of Recommendations Assessment D, Evaluation Working G (2010) Allergic rhinitis and its impact on asthma (ARIA) guidelines: 2010 revision. J Allergy Clin Immunol 126:466–476
Chen CC, Chiu HF, Yang CY (2016) Air pollution exposure and daily clinical visits for allergic rhinitis in a subtropical city: Taipei, Taiwan. J Toxicol Environ Health Part A 79:494–501
Chen F, Lin Z, Chen R, Norback D, Liu C, Kan H, Deng Q, Huang C, Hu Y, Zou Z, Liu W, Wang J, Lu C, Qian H, Yang X, Zhang X, Qu F, Sundell J, Zhang Y, Li B, Sun Y, Zhao Z (2018) The effects of PM2.5 on asthmatic and allergic diseases or symptoms in preschool children of six Chinese cities, based on China, Children, Homes and Health (CCHH) project. Environ Pollut 232:329–337
Chou WC, Hsu CY, Ho CC, Hsieh JH, Chiang HC, Tsou TC, Chen YC, Lin P (2017) Development of an in vitro-based risk assessment framework for predicting ambient particulate matter-bound polycyclic aromatic hydrocarbon-activated toxicity pathways. Environ Sci Technol 51:14262–14272
Chu H, Xin J, Yuan Q, Wang M, Cheng L, Zhang Z, Lu M (2019) The effects of particulate matters on allergic rhinitis in Nanjing, China. Environ Sci Pollut Res Int 26:11452–11457
Deng Q, Lu C, Yu Y, Li Y, Sundell J, Norback D (2016) Early life exposure to traffic-related air pollution and allergic rhinitis in preschool children. Respir Med 121:67–73
Diaz-Sanchez D, Tsien A, Casillas A, Dotson AR, Saxon A (1996) Enhanced nasal cytokine production in human beings after in vivo challenge with diesel exhaust particles. J Allergy Clin Immunol 98:114–123
Diaz-Sanchez D, Garcia MP, Wang M, Jyrala M, Saxon A (1999) Nasal challenge with diesel exhaust particles can induce sensitization to a neoallergen in the human mucosa. J Allergy Clin Immunol 104:1183–1188
Dong GH, Qian ZM, Wang J, Trevathan E, Ma W, Chen W, Xaverius PK, Buckner-Petty S, Ray A, Liu MM, Wang D, Ren WH, Emo B, Chang JJ (2013) Residential characteristics and household risk factors and respiratory diseases in Chinese women: the Seven Northeast Cities (SNEC) study. Sci Total Environ 463-464:389–394
Dunlop J, Matsui E, Sharma HP (2016) Allergic rhinitis: environmental determinants. Immunol Allergy Clin N Am 36:367–377
Esposito S, Tenconi R, Lelii M, Preti V, Nazzari E, Consolo S, Patria MF (2014) Possible molecular mechanisms linking air pollution and asthma in children. BMC Pulm Med 14:31
Etzel RA (2007) Indoor and outdoor air pollution: tobacco smoke, moulds and diseases in infants and children. Int J Hyg Environ Health 210:611–616
Goldizen FC, Sly PD, Knibbs LD (2016) Respiratory effects of air pollution on children. Pediatr Pulmonol 51:94–108
Greiner AN, Hellings PW, Rotiroti G, Scadding GK (2011) Allergic rhinitis. Lancet 378:2112–2122
Gu S, Yang J, Woodward A, Li M, He T, Wang A, Lu B, Liu X, Xu G, Liu Q (2017) The short-term effects of visibility and haze on mortality in a Coastal City of China: a time-series study. Int J Environ Res Public Health 14
He F, Shaffer ML, Rodriguez-Colon S, Yanosky JD, Bixler E, Cascio WE, Liao D (2011) Acute effects of fine particulate air pollution on cardiac arrhythmia: the APACR study. Environ Health Perspect 119:927–932
Heinrich J, Schikowski T (2018) COPD patients as vulnerable subpopulation for exposure to ambient air pollution. Curr Environ Health Rep 5:70–76
Hu LW, Lawrence WR, Liu Y, Yang BY, Zeng XW, Chen W, Dong GH (2017) Ambient air pollution and morbidity in Chinese. Adv Exp Med Biol 1017:123–151
Hu Y, Xu Z, Jiang F, Li S, Liu S, Wu M, Yan C, Tan J, Yu G, Hu Y, Yin Y, Tong S (2020) Relative impact of meteorological factors and air pollutants on childhood allergic diseases in Shanghai, China. Sci Total Environ 706:135975
Huang C, Wang Q, Wang S, Ren M, Ma R, He Y (2017) Air pollution prevention and control policy in China. Adv Exp Med Biol 1017:243–261
Hwang BF, Jaakkola JJ, Lee YL, Lin YC, Guo YL (2006) Relation between air pollution and allergic rhinitis in Taiwanese schoolchildren. Respir Res 7:23
Jaakkola JJ, Ieromnimon A, Jaakkola MS (2006) Interior surface materials and asthma in adults: a population-based incident case-control study. Am J Epidemiol 164:742–749
Jo EJ, Lee WS, Jo HY, Kim CH, Eom JS, Mok JH, Kim MH, Lee K, Kim KU, Lee MK, Park HK (2017) Effects of particulate matter on respiratory disease and the impact of meteorological factors in Busan, Korea. Respir Med 124:79–87
Jones PJ, Koolhof IS, Wheeler AJ, Williamson GJ, Lucani C, Campbell SL, Bowman DMJS, Johnston FH (2020) Can smartphone data identify the local environmental drivers of respiratory disease? Environ Res 182:109118
Lehmann I, Rehwagen M, Diez U, Seiffart A, Rolle-Kampczyk U, Richter M, Wetzig H, Borte M, Herbarth O, Leipzig Allergy Risk Children S (2001) Enhanced in vivo IgE production and T cell polarization toward the type 2 phenotype in association with indoor exposure to VOC: results of the LARS study. Int J Hyg Environ Health 204:211–221
Li H, Chen R, Meng X, Zhao Z, Cai J, Wang C, Yang C, Kan H (2015) Short-term exposure to ambient air pollution and coronary heart disease mortality in 8 Chinese cities. Int J Cardiol 197:265–270
Liang H, Qiu H, Tian L (2018) Short-term effects of fine particulate matter on acute myocardial infraction mortality and years of life lost: a time series study in Hong Kong. Sci Total Environ 615:558–563
Lin H, Liu T, Xiao J, Zeng W, Li X, Guo L, Zhang Y, Xu Y, Tao J, Xian H, Syberg KM, Qian ZM, Ma W (2016) Mortality burden of ambient fine particulate air pollution in six Chinese cities: results from the Pearl River Delta study. Environ Int 96:91–97
Maji KJ, Ye WF, Arora M, Nagendra SMS (2019) Ozone pollution in Chinese cities: assessment of seasonal variation, health effects and economic burden. Environ Pollut 247:792–801
McBride DL (2015) Children’s lung health improves with reduced air pollution. J Pediatr Nurs 30:624–625
Meng X, Zhang Y, Zhao Z, Duan X, Xu X, Kan H (2012) Temperature modifies the acute effect of particulate air pollution on mortality in eight Chinese cities. Sci Total Environ 435-436:215–221
Mihălţan F, Deleanu O, Nemeș R, Ulmeanu R (2016) Air pollution and respiratory diseases – a problematic risk factor. Pneumologia (Bucharest, Romania) 65:122–125
Min KD, Yi SJ, Kim HC, Leem JH, Kwon HJ, Hong S, Kim KS, Kim SY (2020) Association between exposure to traffic-related air pollution and pediatric allergic diseases based on modeled air pollution concentrations and traffic measures in Seoul, Korea: a comparative analysis. Environ Health 19:6
Nathan RA (2007) The burden of allergic rhinitis. Allergy Asthma Proc 28:3–9
Penard-Morand C, Charpin D, Raherison C, Kopferschmitt C, Caillaud D, Lavaud F, Annesi-Maesano I (2005) Long-term exposure to background air pollution related to respiratory and allergic health in schoolchildren. Clin Exp Allergy 35:1279–1287
Qiu H, Yu IT, Tian L, Wang X, Tse LA, Tam W, Wong TW (2012) Effects of coarse particulate matter on emergency hospital admissions for respiratory diseases: a time-series analysis in Hong Kong. Environ Health Perspect 120:572–576
Samitas K, Carter A, Kariyawasam HH, Xanthou G (2018) Upper and lower airway remodelling mechanisms in asthma, allergic rhinitis and chronic rhinosinusitis: the one airway concept revisited. Allergy 73:993–1002
Shang Y, Sun Z, Cao J, Wang X, Zhong L, Bi X, Li H, Liu W, Zhu T, Huang W (2013) Systematic review of Chinese studies of short-term exposure to air pollution and daily mortality. Environ Int 54:100–111
Song J, Lu M, Zheng L, Liu Y, Xu P, Li Y, Xu D, Wu W (2018) Acute effects of ambient air pollution on outpatient children with respiratory diseases in Shijiazhuang, China. BMC Pulm Med 18:150
Statistics NBo (2000–2018): China Energy Statistical Yearbooks
Teng B, Zhang X, Yi C, Zhang Y, Ye S, Wang Y, Tong DQ, Lu B (2017) The association between ambient air pollution and allergic rhinitis: further epidemiological evidence from Changchun, northeastern China. Int J Environ Res Public Health 14:226
Tian L, Qiu H, Sun S, Lin H (2016) Emergency cardiovascular hospitalization risk attributable to cold temperatures in Hong Kong. Circ Cardiovasc Qual Outcomes 9:135–142
To T, Zhu J, Stieb D, Gray N, Fong I, Pinault L, Jerrett M, Robichaud A, Ménard R, van Donkelaar A, Martin RV, Hystad P, Brook JR, Dell S (2020) Early life exposure to air pollution and incidence of childhood asthma, allergic rhinitis and eczema. Eur Respir J 55
van Donkelaar A, Martin RV, Brauer M, Kahn R, Levy R, Verduzco C, Villeneuve PJ (2010) Global estimates of ambient fine particulate matter concentrations from satellite-based aerosol optical depth: development and application. Environ Health Perspect 118:847–855
Wang M, Wang S, Wang X, Tian Y, Wu Y, Cao Y, Song J, Wu T, Hu Y (2020) The association between PM2.5 exposure and daily outpatient visits for allergic rhinitis: evidence from a seriously air-polluted environment. Int J Biometeorol 64:139–144
Wang KY, Chau TT (2013) An association between air pollution and daily outpatient visits for respiratory disease in a heavy industry area. PLoS One 8:e75220
Ye Q, Zhang T, Mao JH (2019) Haze facilitates sensitization to house dust mites in children. Environ Geochem Health:1–9. https://doi.org/10.1007/s10653-019-00481-6
Zeger SL, Thomas D, Dominici F, Samet JM, Schwartz J, Dockery D, Cohen A (2000) Exposure measurement error in time-series studies of air pollution: concepts and consequences. Environ Health Perspect 108:419–426
Zhang F, Wang W, Lv J, Krafft T, Xu J (2011) Time-series studies on air pollution and daily outpatient visits for allergic rhinitis in Beijing, China. Sci Total Environ 409:2486–2492
Zhang F, Krafft T, Ye B, Zhang F, Zhang J, Luo H, Li M, Zhang Y, Zhou L, Xu J, Wang W (2013) The lag effects and seasonal differences of air pollutants on allergic rhinitis in Beijing. Sci Total Environ 442:172–176
Zhang L, Han D, Huang D, Wu Y, Dong Z, Xu G, Kong W, Bachert C (2009) Prevalence of self-reported allergic rhinitis in eleven major cities in China. Int Arch Allergy Immunol 149:47–57
Zhang Q, Qiu M, Lai K, Zhong N (2015) Cough and environmental air pollution in China. Pulm Pharmacol Ther 35:132–136
Zou QY, Shen Y, Ke X, Hong SL, Kang HY (2018) Exposure to air pollution and risk of prevalence of childhood allergic rhinitis: a meta-analysis. Int J Pediatr Otorhinolaryngol 112:82–90
Acknowledgements
The study was supported by the PhD Research Project of Xinxiang Medical University (XYBSKYZZ201804), Key Scientific Research Projects in Universities of Henan (19B330004) and Peak Subject Project of Public Health in Xinxiang Medical University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, J., Lu, M., An, Z. et al. Associations between air pollution and outpatient visits for allergic rhinitis in Xinxiang, China. Environ Sci Pollut Res 27, 23565–23574 (2020). https://doi.org/10.1007/s11356-020-08709-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08709-0