Abstract
This study proposes a comparison of different ex situ technologies aimed at the removal of polycyclic aromatic hydrocarbons from marine sediments in terms of performances, costs and energy balance. In accordance with the principles of water-energy nexus, anaerobic bioremediation, soil washing and thermal desorption were investigated under low liquid phase and temperature conditions using phenanthrene (PHE) as model compound. After 42 days of anaerobic bioremediation, the highest PHE biodegradation of 68 and 64% was observed under denitrifying and methanogenic conditions, respectively, accompanied by N2 and CH4 production and volatile fatty acid accumulation. During soil washing, more than 97% of PHE was removed after 60 min using a solid-to-liquid ratio of 1:3. Along the same treatment time, low-temperature thermal desorption (LTTD) allowed a PHE removal of 88% at 200 °C. The economic analysis indicated that LTTD resulted in a higher cost (i.e. 1782 € m−3) than bioremediation and soil washing (228 and 371 € m−3, respectively). The energy balance also suggested that bioremediation and soil washing are more sustainable technologies as a lower required energy (i.e. 16 and 14 kWh m−3, respectively) than LTTD (i.e. 417 kWh m−3) is needed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are a group of hazardous compounds that pose risks to human and ecological health (Muangchinda et al. 2017). Marine sediments are often contaminated by PAHs due to oil spillage and atmospheric deposition of particulate emissions produced by industrial activities (Nam et al. 2008; Mat-Shayuti et al. 2019). Moreover, PAHs could be generated by early diagenetic reactions when a high abundance of perylene is detected in sediments (Wakeham et al. 1980; Readman et al. 2002; Wakeham and Canuel 2016). A high quantity of contaminated marine sediments usually results in waste management problems after dredging (Sprovieri et al. 2007), due to the limited natural attenuation of PAH pollution (Louati et al. 2014). For this reason, PAH-contaminated sediments need to be remediated prior to sediment disposal or reuse, in accordance with the national regulation (Mattei et al. 2016). Bioremediation, soil washing and thermal desorption are available ex situ technologies that can be used for contaminated sediment remediation (Agarwal and Liu 2015; Kuppusamy et al. 2016).
Bioremediation is an eco-friendly process involving microorganisms to remove PAHs from contaminated sites (Yu et al. 2014; Cecotti et al. 2018). Bioremediation occurs through a biostimulation when nutrients or extra-organic sources are added to the soil to improve the metabolic activity of microorganisms (Oleszczuk and Baran 2005; Qiao et al. 2014; Masy et al. 2016). An alternative to biostimulation is bioaugmentation, which consists in the supplementation of enriched indigenous or exogenous microorganisms in the polluted soil to speed up the remediation efficiency (Haleyur et al. 2019). Bioremediation may be applied under anaerobic conditions employing methanogenic, sulfate-reducing and denitrifying conditions (Agarry and Owabor 2011; Yang et al. 2018; Bianco et al. 2020). However, the bioavailability of PAHs, meant as the available part of the contaminant for biological conversion (Guo et al. 2016), is often a limiting factor when maintaining a bioprocess in sediments or soils (Yu et al. 2014).
Physical-chemical remediation processes overcome the limiting factor of bioremediation, as they allow to also remove contaminants that are recalcitrant to microbial degradation. Among the physical-chemical techniques, soil washing is an attractive technology due to its simplicity, low costs and high removal efficiency (Lee and Hosomi 2000; Sun et al. 2009). Soil washing consists in the use of solvents or reagents to improve the mass transfer of hydrophobic pollutants from the soil matrix to the liquid phase (Yap et al. 2012; Ferraro et al. 2017). Previous studies have proven the efficiency of different washing agents such as ethanol, 2-propanol, acetone and 1-pentanol for PAH removal (Silva et al. 2005).
Thermal desorption is a remediation technology with scientific and practical interest due to its high removal efficiency and low gas emissions in the presence of gas purification units (Smith et al. 2001). Controlling the temperature of the thermal treatment enhances the desorption of organic pollutants from different matrices such as soils, sludge and sediments (Pope et al. 2000; Renoldi et al. 2003). At higher temperatures, the removal efficiency of high-boiling point organic contaminant increases (Bulmău et al. 2014).
For bioremediation, soil washing and thermal desorption, the water content plays a major role during the treatment. Wet conditions are widely applied during bioremediation to overcome the mass transfer limitations and provide a better environment for the microbial consortia involved (Sayara et al. 2010; Abbassi-Guendouz et al. 2012). However, a too high water content lowers the amount of soil or sediment to be treated in the unit of time. Regarding soil washing, the use of a high solid-to-liquid ratio (e.g. 1:5 or 1:10) generally results in higher removal efficiencies but also a considerable amount of spent washing solution that must be treated prior to discharge (Chattopadhyay and Chattopadhyay 2015; Trellu et al. 2016). During thermal desorption, an excessive presence of water increases the energy input to evaporate the contaminants as water sequesters part of the heat provided (Zhao et al. 2019).
In this study, the use of dry conditions, a low solid-to-liquid ratio and low temperature was proposed for anaerobic bioremediation, soil washing and thermal desorption, respectively. Nowadays, emerging concerns such as the saving of water resources, the production of energy in a sustainable way and the control of pollution are receiving more and more attention within a “water-energy nexus” perspective (Landa-Cansigno et al. 2019; Meireles and Sousa 2019). In this line, a techno-economic assessment of bioremediation, soil washing and thermal desorption as ex situ processes for the remediation of PAH-contaminated marine sediments as well as energy balance considerations were here provided. Phenanthrene (PHE), one of the most present PAHs in environment, was selected as representative pollutant due to its intermediate toxicity, hydrophobicity and environmental persistence (Louati et al. 2014).
In particular, the specific objectives of this study were to (1) evaluate the efficiency of biostimulation/bioaugmentation, soil washing and thermal desorption in removing PHE from marine sediments; (2) implement the experimental data on a commercial software in order to assess the remediation costs and (3) consider a balance of required and possibly recovered energy during each remediation approach to allow choosing the most appropriate and sustainable technology.
Materials and methods
Chemicals
Phenanthrene (grade ≥ 98%), potassium nitrate (grade ≥ 99%), sodium hydroxide (grade ≥ 97%) and sodium sulfate (grade ≥ 99%) were all purchased from Sigma-Aldrich (Germany). Acetone (grade 100%), acetonitrile (HPLC grade), butanol (grade ≥ 96%), sodium acetate (grade ≥ 99%) and sulfuric acid (grade ≥ 96%) were supplied by VWR (Italy). Ethanol (≥ 99.9%) was purchased from Merck (Germany). Deionized water with an electrical conductivity lower than 0.3 μS cm−1 was used to prepare all solutions.
Sediment sampling and spiking
Marine sediments were collected from Formia seaside (Lazio Region, Italy) and the physical-chemical properties are reported in Table 1. No PAH was detected in the uncontaminated sediment. After characterization, the sediment was dried and passed through a 2-mm mesh to remove the coarse fraction. PHE was used as model PAH due to its intermediate aqueous solubility (i.e. 0.823 mg L−1) and hydrophobicity (i.e. log KOW 4.57) (Smith et al. 2009; Yang et al. 2018). The uncontaminated sediment was spiked by dissolving PHE in acetone, according to literature procedure (Mueller and Shann 2006; Steffen et al. 2007). The selected value of initial PHE concentration was about 200 mg kg−1 dry sediment, as reported by Arienzo et al. (2017) for real PHE-contaminated marine sediments. Subsequently, the contaminated sediment was mixed, placed under a fume hood for 72 h to allow acetone evaporation and stored in glass containers in the dark (Brinch et al. 2002). Afterwards, an aging protocol was conducted for 37 days at room temperature (Lukić et al. 2016) in order to simulate an actual PHE-contaminated sediment.
Experimental design
Bioremediation
Bioremediation was conducted treating the contaminated sediment with combined biostimulation and bioaugmentation under denitrifying, methanogenic and sulfate-reducing conditions (Chang et al. 2003; Sayara et al. 2011). The selected sources of microorganisms were digestate and sewage sludge (Table 1), collected from a full-scale anaerobic digester located in Capaccio (Campania Region, Italy) and a wastewater treatment plant situated in Cassino (Lazio Region, Italy), respectively. Five operative conditions, i.e. contaminated sediment with (i) sewage sludge (S), (ii) digestate (D), (iii) sewage sludge + 2000 mg L−1 of nitrate (SN, with NO3− supplied as potassium nitrate), (iv) digestate + 2000 mg L−1 of sulfate (DS, with SO42− supplied as sodium sulfate), (v) digestate + 2000 mg L−1 of acetate (DA, with CH3COO− supplied as sodium acetate), were investigated. For each condition, 12 serum bottles (100 mL) were loaded with a dry portion of sediment (10 g) mixed with the inoculum at a ratio of 10:1 (dry w/dry w) and filled with water to reach a total moisture content of 60% (Namkoong et al. 2002; Frutos et al. 2010). Afterwards, all the bottles were flushed with argon to guarantee anaerobiosis, prior to being incubated in a water bath under mesophilic conditions (i.e. at 37 ± 1 °C) (Sayara et al. 2010) for 42 days and placed on a gyratory shaker at 160 rpm (Gielnik et al. 2019) to simulate a mechanical agitation. The samples were collected after 7, 14, 21 and 42 days by sacrificing three bottles at a time for each condition. In SN, DS and DA, denitrifying, sulfate-reducing and methanogenic conditions were ensured by restoring the initial content of nitrate, sulfate and acetate, respectively, when the observed NO3−, SO42− and CH3COO− concentrations were below 50 mg L−1.
Soil washing
Soil washing tests were performed in glass bottles (80 mL) at room temperature (Fabbricino et al. 2018), with a 1:3, 1:5 and 1:10 solid-to-liquid (S/L) ratio. A solution with 50% of ethanol (ETOH) and water (w/w) was used as washing agent (Yap et al. 2012). Sediment samples (6 g) were shaken at 200 rpm in a horizontal shaker for 24 h (Lee and Hosomi 2000), and PHE concentration was determined by sacrificing three bottles for each S/L ratio after 1, 2, 5, 15, 30, 60, 180, 360, 720 and 1440 min.
Thermal desorption
Thermal desorption, testing low temperatures in 1 h of experimentation (Smith et al. 2001), was carried out with ceramic crucibles containing 10 g of sediment each (Renoldi et al. 2003) and introduced in a muffle furnace at 100, 150 and 200 °C. PHE concentrations were measured after 5, 15, 30 and 60 min sacrificing three crucibles per time for each temperature.
Analytical methods
Total solids (TS) (Astm 1998), volatile solids (VS) (APHA, AWWA, WEF. 2012), pH (Gurung et al. 2018), electrical conductivity (EC) (Gupta and Hanks 2010), total organic carbon (TOC) (Chen et al. 2015), alkalinity (A) and Kjeldahl nitrogen (TKN) (APHA, AWWA, WEF 2005), sulfate and nitrate concentrations (Kiskira et al. 2017) were measured according to the methods reported elsewhere. The samples were centrifuged at 4000 rpm for 30 min to separate the liquid fraction from the solid phase (Sun et al. 2009). PHE was extracted from samples by ultra-sonication (Sun et al. 1998) and was quantified using a LC-20AD HPLC (Shimadzu, Japan) equipped with a Kinetex® 3.5 μm PAH (150 × 4.6 mm) column (Phenomenex, USA) and an SPD-20A UV detector (Shimadzu, Japan) set at 254 nm. PHE bioavailability was predicted using a butanol extraction (Ehlers and Luthy 2003; Yang et al. 2010). Biomethane, nitrogen gas (i.e. N2) and N2O production were measured with a water displacement method with an alkaline trap (12% of sodium hydroxide) to remove carbon dioxide and hydrogen sulfide (Budiyono et al. 2010; Mancini et al. 2016). The gas composition (i.e. %CH4, %N2, %N2O) was determined with a 3400 GC-TCD/ECD gas chromatograph (Varian-Agilent, USA) equipped with a Restek Packed column. Total VFAs, reported as equivalent acetic acid concentration (mg HAc·L−1), were analyzed according to Mancini et al. (2016), using the previously described HPLC equipped with a Rezex ROA-Organic Acid H+ column (Phenomenex, USA) and a SPD-20A UV detector set at 210 nm.
Economic analysis
The costs for sediment remediation (€ m−3) were estimated considering the most performing operating conditions in terms of PHE removal used during bioremediation, soil washing and thermal desorption. The retention time and temperature were implemented in the Remedial Action Cost Engineering and Requirements (RACER) software (11.5 version, AECOM, USA) as crucial operating parameters strongly affecting the remediation efficiencies and costs. Indeed, a higher temperature can enhance the microbial metabolism rate (Mohan et al. 2006), PAH dissolution and vaporization (Haritash and Kaushik 2009; Bulmău et al. 2014) as well as increase the energy cost. In contrast, a lower retention time results in a reduced capital cost (Bustillo-Lecompte et al. 2014) as smaller reactors would be required. The contamination area, the safety level of machining operation and mobilization distance were assumed as 7646 m3 (i.e. 10,000 CY) (Federal Remediation Technologies Roundtable n.d.), high (A) and 1.6 km (i.e. 1 mi), respectively. The assumption of a short mobilization distance was done to minimize the percentage of this cost in favor of the remediation cost. Moreover, the costs of dredging, water removal by leaking drums and disposal were not considered because common for all the processes considered in this study. Detailed information about input parameters in RACER are reported in Table 2 and in the supporting information accompanying the manuscript (S1. Parameter implementation on RACER). Note that the obtained costs were only indicative of real values, since costs were modeled without specific components of the site (e.g. treatment goals due to national legislation). The RACER software contains the latest assembly unit price information from the 2016 Government Cost Book (USACE TRACES, Huntsville).
Energy balance
A rough estimate of the energy balance was performed for the bioremediation, soil washing and thermal desorption experimental conditions implemented in RACER. All energy requests were reported as kWh m−3 of sediment.
The energy required for the maintenance of mesophilic conditions during the bioremediation process was estimated as a function of water heat exchanger, gas boiler and pump suggested by RACER. The energy necessary for the water heat exchanger was derived as follows (Esen and Yuksel 2013):
where \( \dot{Q} \) is the amount of heat per time unit (kW); \( \dot{m} \) is the mass flow rate of the water (kg s−1); Cp,w represents the heat capacity of water (kJ·(kg K)−1) and ∆T is the temperature difference of water between the outlet and inlet of the heat exchanger (i.e. 17 K). Hence, the energy consumption Ec (kWh) was obtained by multiplying the sum of Eq. (1), boiler and pump power (kW) with the retention time of the biological process (h). Moreover, the specific energy recovery \( \overline{{\mathrm{E}}_{\mathrm{r}}} \) (kWh kg VS−1) during the bioremediation process was estimated from the obtained biomethane yield, as reported by Mancini et al. (2016):
where BMP is the final cumulative biomethane yield (m3 CH4·kg VS−1); CHP represents a combined heat and power unit (with a 50% conversion efficiency) and Cf is the conversion factor (i.e. 10 kWh m−3). Hence, the energy recovered Er (kWh) was obtained by multiplying Eq. (2) and the content of VS (kg). The energy balance for bioremediation process was calculated by subtraction of Ec with Er.
The energy necessary for soil washing (kWh) was obtained with RACER, whereas the energy needed for thermal desorption was estimated as follows:
where Etd is the amount of energy per time unit (kWh); mw, mPHE and ms are the mass of water, PHE and sediment (kg), respectively; Cp,PHE and Cp,s are the heat capacity values of PHE (Steele et al. 1990) and sediment, respectively (kJ·(kg K)−1); Cl,w and Cl,PHE are the heat of vaporization of water and PHE (Roux et al. 2008) (kJ kg−1), respectively; and ∆T is the temperature difference between the desorption chamber and inlet (i.e. 180 K).
Statistical analyses
Data were analyzed by one-way analysis of variance (ANOVA) followed by the post hoc Tukey test to evaluate significant differences (p < 0.05) among the operating conditions investigated. All statistical analyses were performed with Minitab 19 Statistical Software (Minitab LLC, USA).
Results and discussion
Bioremediation
PHE biodegradation
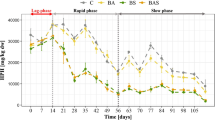
The remaining PHE (%) after 0, 7, 14, 21 and 42 days of bioremediation under all the operating conditions (i.e. SN, S, DS, DA and D) is shown in Fig. 1a. PHE removal slowly proceeded during the initial 7 days as PHE (%) at “t7” was not significantly different than PHE at “t0” (p > 0.05), except for D where a 17% PHE removal was observed at “t7” (p < 0.05). The low, common PHE removal was probably due to an initial acclimation period of the microbial consortia involved, whereas the difference between the D, DS and DA conditions was associated with the higher sulfate and acetate concentration, which likely resulted in a slight inhibition of the bacteria in digestate.
Ratio [%] between the PHE concentration at time “t” and that at time “t0” in the contaminated sediment (a); cumulative, specific biomethane yield [mL CH4·g VS−1] (b); and total VFA concentrations [mg HAc·L−1] during 42 days of anaerobic biostimulation/bioaugmentation (c). S = contaminated sediment + sewage sludge; D = contaminated sediment + digestate; SN = contaminated sediment + sewage sludge + 2000 mg L−1 of nitrate; DS = contaminated sediment + digestate + 2000 mg L−1 of sulfate; DA = contaminated sediment + digestate + 2000 mg L−1 of acetate; predicted = predicted biodegradability of PHE. The same letter (Tukey test) represents no significant differences (p > 0.05) among different treatments. Error bars indicate deviation standard values of analyses in triplicate
A significant PHE degradation (p < 0.05) of 59, 47, 41, 58 and 54% was observed after 14 days in SN, S, DS, DA and D (Fig. 1a), respectively. Moreover, the observed pH values among the experimental conditions (i.e. 7.3 ÷ 8.1, Table S1) were consistent with those shown as optimal for PAH degradation (i.e. 6 ÷ 8) (Tawari-Fufeyin et al. 2015). Considering sulfate, dioxide carbon and nitrate as major electron acceptors in PAH biodegradation (Maletić et al. 2018), and assuming that 1 μmol of CO2 may be used to produce 1 μmol of CH4, a reduction of 43 μmol of SO42−, 81 μmol of CO2 and 86 μmol of NO3− was obtained per μmol of PHE under sulfate-reducing, methanogenic and denitrifying conditions (Table S1), respectively. These results suggest that sulfate was a more efficient electron acceptor, as also reported by Chang et al. (2003). Nonetheless, after 14 days, PHE removal in DS was lower (p < 0.05) than that achieved in SN and DA, probably due to the presence of other forms of electron donors instead of PHE. A continuous reduction of sulfate (i.e. about 200 μmol of SO42− per week, Table S1), a significant consumption of acetic acid (p < 0.05) after 21 days (Fig. 1c) and no significant difference (p > 0.05) in biomethane yield after 7 days (Fig. 1b) were observed in DS, suggesting that acetate was presumably coupled to sulfate reduction. Similarly, Zhang and Lo (2015) observed that acetate may be preferentially used by bacteria to facilitate sulfate reduction in the absence of methane production.
During the conversion to methane by methanogens, acetate may donate electrons in favor to PAH degradation (Ambrosoli et al. 2005). However, no statistically differences (p > 0.05) in PHE removal between S, DA and D were observed after 14 days, despite a higher acetate reduction (p < 0.05) observed in S (Fig. 1a and c). Ebihara and Bishop (2003) reported that acetate supplementation for the biodegradation of recalcitrant organic contaminants may inhibit or have no effect. This suggests that in this study, only a small fraction of acetate was likely coupled to PHE considering an initial concentration of 2000 mg acetate·kg−1 TS (i.e. 199 μmol of acetate for each μmol of PHE), and a higher contamination would have instead been necessary to see a significant effect.
In the time lapse between 14 and 42 days, PHE degradation considerably slowed down and reached 68, 53, 45, 64 and 64% in SN, S, DS, DA and D, respectively, being only enhanced under denitrifying and methanogenic conditions (p < 0.05) probably because the predicted biodegradation (i.e. 58%) was achieved (Fig. 1a). The predicted biodegradation is representative of the bioavailable PAH amount (Cuypers et al. 2002), which is the bioaccessible fraction to microorganisms (Ortega-Calvo et al. 2013), and the observed value (Fig. 1a) was comparable to those observed in other PHE-contaminated marine sediments (i.e. 44 ÷ 70%) (Du et al. 2012; Ghosal et al. 2016). It is well known that limitations in the bioremediation of PAH-contaminated soils exist due to the low bioavailability of PAHs, as shown in the final phase of the process. On the other hand, a reduced bioavailability may be a positive aspect because it would lead to a reduced amount of highly accessible PAHs (Andersson et al. 2009), consequently mitigating the environmental risk.
Biogas composition and VFA evolution during PHE biodegradation
The cumulative biomethane yield obtained during 42 days of bioremediation under all the operating conditions (i.e. SD, S, DS, DA and D) is shown in Fig. 1b. After 7 days, a biomethane production of 40, 65, 11, 14 and 13 mL CH4·g VS−1 was observed in SN, S, DS, DA and D, respectively. The recorded pH values (Table S1) were optimal for methanogen growth (Ogejo and Li 2010) and, thus, did not represent a limiting factor for biomethane production.
Between 7 and 14 days, the biomethane yield significantly raised (p < 0.05) by about 50% in S reaching a cumulative production of 142 mL CH4·g VS−1 (Fig. 1b), accompanied by a significant consumption of acetate (Fig. 1c). In SN, a lower cumulative biomethane production (i.e. 80 mL CH4·g VS−1) than that obtained in S (p < 0.05) was achieved probably because of the occurrence of heterotrophic denitrification (Zou et al. 2014). Indeed, the observed N2 percentage in the gas phase raised from 15 to 69% in SN after 14 days (Fig. S1). N2O was below the detection limit (i.e. 0.1%) during denitrification likely due to the large availability of biodegradable organic carbon (Kampschreur et al. 2009). N2O is a greenhouse gas with a stronger impact than carbon dioxide. The absence of N2O formation confirms the sustainability of biological PHE removal under denitrifying conditions, not leading to the release of high-impact gas streams.
After 21 days (Fig. 1b), the cumulative biomethane production was lower (p < 0.05) in DS (i.e. 23 mL CH4·g VS−1) than DA and D (i.e. 61 and 39 mL CH4·g VS−1, respectively). The observed sulfate reduction of approximately 1000 mg SO42−·L−1 in DS (Table S1) suggests that sulfate-reducing bacteria likely outcompeted other anaerobic microorganisms, such as methanogenic archaea (Chen et al. 2008; Papirio et al. 2013). Therefore, the limited biomethane yield in DS coupled to the low PHE removal (Fig. 1a, b) probably indicated that methanogenesis promoted the anaerobic PAH degradation, as also reported by Chang et al. (2006).
After 42 days (Fig. 1b), S resulted in a higher (p < 0.05) cumulative biomethane yield (i.e. 273 mL CH4·g VS−1, respectively) than SN, DS, DA and D (i.e. 103, 29, 103 and 73 mL CH4·g VS−1, respectively), indicating a higher biomethane potential of sewage sludge than the digestate most likely due to a lower organic stability.
The total VFA evolution during 42 days of bioremediation under all operating conditions (i.e. SN, S, DS, DA and D) is shown in Fig. 1c, with the highest VFA concentrations being observed after 7 days. S showed a significantly higher (p < 0.05) VFA accumulation (i.e. 6173 mg HAc·L−1) than SN, DS, DA and D (i.e. 2467, 879, 3181 and 1064 mg HAc·L−1, respectively). The sewage sludge resulted in a higher VFA yield (i.e. 113 mg HAc·g VS−1) than digestate (i.e. 22 mg HAc·g VS−1), again demonstrating a higher biodegradability.
VFA concentrations significantly decreased (p < 0.05) from day 14 onwards (Fig. 1c) due to the activity of methanogens inoculated with both sewage sludge and digestate (Fig. 1b). In SN and DS, acetate consumption was probably also coupled to nitrate or sulfate reduction, as discussed above. Further information about specific VFAs are reported in the supporting information accompanying the manuscript (Table S2).
Soil washing
PHE removal after 1, 2, 5, 15, 30 and 60 min with ETOH washing at S/L ratios of 1:3, 1:5 and 1:10 is shown in Fig. 2. The removal of PHE from the contaminated sediment was regulated by the partition coefficient in the solution phase, in agreement with the general behavior of hydrophobic compounds (Kan et al. 1998; Thiele-Bruhn and Brümmer 2004). Hence, the partitioning process may be summarized in two phases (Fig. 2): (a) a first phase (the initial 5 min), where the labile fraction of PHE (i.e. about 70%) was readily desorbed; (b) a second phase (between 5 and 60 min), where the residual PHE fraction (i.e. about 30%) was slowly extracted due to the equilibrium between solid and liquid phase.
After 60 min (Fig. 2), no statistical differences (p > 0.05) were observed among the different S/L ratios and more than 97% of PHE was removed, suggesting that 1.5 g of ETOH was sufficient to treat efficiently 1 g of sediment, as observed for an S/L ratio of 1:3. These findings are highly important because the reduction of contact time and S/L ratio (i.e. 60 min and 1:3, respectively) during soil washing may involve a less capacity of soil washing plant. Moreover, one washing time was sufficient to extract more than 90% of PHE from the sediment, decreasing the operational costs (Lee and Hosomi 2000) and allowing ethanol to be recovered by distillation with a loss of about 10% (Silva et al. 2005).
In this study, the obtained PHE extraction (Fig. 2) was probably enhanced by a low organic matter content in the sediment (Table 1) being PAH lipophilic compounds. Sun et al. (2009), indeed, showed that PHE removal increased (i.e. 94, 71, 38 and 24%) with the decreasing of organic carbon in soil (i.e. 0.79, 1.36 1.88 and 2.33%, respectively). Similarly, Yap et al. (2012) obtained a soil washing efficiency of 80% with a co-solvent volumetric fraction and an S/L ratio of 0.8 and 1:2, respectively. In this work, the used co-solvent volumetric fraction (i.e. 0.6) enhanced PHE extraction because the micro-emulsion effect of alcohol molecules on the water structure was presumably improved, as also reported by Dougan et al. (2004) in a methanol/water system.
Thermal desorption
The residual PHE (%) during 60 min of thermal desorption at 100, 150 and 200 °C is illustrated in Fig. 3. After 5 min, no significant PHE removal (p > 0.05) was observed as PHE presumably remained in the sediment as melted (Smith et al. 2001). Thermal desorption started after 15 min (Fig. 3) and PHE removal was higher (p < 0.05) at 200 °C (i.e. 47%) than at 150 °C (i.e. 12%). At 100 °C, PHE was not desorbed (p > 0.05) probably because a higher temperature than the PHE melting point (i.e. 101 °C) was necessary to promote desorption (Bulmău et al. 2014). The desorption mechanism followed the thermodynamic principle, with the desorption of PHE increasing at higher temperatures, which allowed the breakdown of benzene rings (Pakpahan et al. 2009). After 60 min (Fig. 3), PHE was desorbed by 32 and 88% (p < 0.05) at 150 and 200 °C, respectively, following an exponential kinetics with a correlation coefficient of 0.97, as also reported by Smith et al. (2001).
The results obtained showed that PHE removal was efficiently accomplished at 200 °C, suggesting that the operating temperature and PHE vapor pressure (i.e. 10−2 Pa) positively influenced PHE desorption (Kim et al. 2016). A higher PHE desorption to nearly 100% could be obtained increasing the temperature to 250 °C, as reported by Renoldi et al. (2003). However, too high temperatures can result in the PAH isomerization, i.e. the transformation of a specific PAH into more reactive PAHs (Pope et al. 2000). PHE isomerization did not probably occur in this work since the chromatographic analysis did not show any other peak than PHE.
Techno-economic feasibility
The experimental conditions resulting in the highest PHE removal, which were SN (i.e. 68%), S/L ratio of 1:3 (i.e. 97%) and low temperature thermal desorption (i.e. LTTD) at 200 °C (i.e. 88%) for bioremediation, soil washing and thermal desorption, respectively, were selected for the economic analysis. The costs of remediation obtained by using in RACER the input data (Table 2) for each ex situ treatment investigated are reported in Table 3. Bioremediation had the lowest total cost (i.e. 274 € m−3), followed by soil washing and LTTD (i.e. 371 and 1782 € m−3, respectively).
Using bioremediation, the main costs (Table S3) were associated with the structural slab and bioreactor (i.e. 33 and 28% of the total cost, respectively). The highest costs of soil washing (Table S4) were related to the washing system and operation labor (i.e. 37 and 20% of the total cost, respectively), while the cost for LTTD (Table S5) was mostly due to the thermal desorption equipment and heating (i.e. 95%). The mobilization of the contaminated sediment accounted for less than 3% of total cost for all the remediation technologies due to the short selected distance (i.e. 1.6 km) between the treatment plants and the contaminated site. Moreover, the cost for liquid treatment prior to disposal/demobilization must not be neglected during soil washing (i.e. 2%), although RACER does not consider the distillation process for ETOH recovery and cost saving, as discussed in “Soil washing”. Compared to soil washing, bioremediation is more expensive in terms of materials consumed for bioreactor construction due to a longer retention time that requires the use of higher reaction volumes (Inoue and Katayama 2011). On the other hand, the labor cost is higher in soil washing likely due to more significant manpower for a continuous load of sediments, admixture of chemicals and plant maintenance. Regarding LTTD, the energy used for the heating furnace may justify the highest cost of equipment, as reported in “The water-energy-nexus”.
These results (Table 3) suggest that the use of bioremediation provides an economic benefit (Beškoski et al. 2011). Nevertheless, bioremediation is affected by PAH bioavailability (“PHE biodegradation”) that does not allow a complete PHE removal (Fig. 1a) and requires large bioreactor volumes due to a long PAH biodegradation time (i.e. 14 days). Soil washing leads to higher overall costs as a result of repeated charging and discharging of the washing system (Table 3), but also a higher PHE removal (Fig. 2). In each case, the reuse of the remediated sediments (e.g. for coastal replenishment and road-under surfaces) should be also taken into consideration in order to provide a more in-depth techno-economic analysis of the most suitable remediation technology. In contrast, the use of thermal desorption is not recommended due to the lower efficiency (Fig. 3) than soil washing at the same retention time (i.e. 1 h) and the higher cost of remediation (Table 3) that must be added to the costs of dredging (i.e. 1290 € m−3) (Chattopadhyay and Chattopadhyay 2015), always accounted for an ex-situ sediment treatment. For a further comprehension of the differences between the investigated ex situ processes, a list of advantages and disadvantages for each technology is reported in Table S6.
The water-energy-nexus
This study shows that decreasing the operating temperature (i.e. 200 °C or lower) and the amount of liquid phase used (i.e. 60% water content and S/L ratio of 1:3 for bioremediation and soil washing, respectively) is a more efficient approach to remediate PHE-contaminated sediments in terms of both removal percentages and costs. On top of this, the energy balance should also be considered during the selection of the most appropriate remediation technology. Indeed, the reduction of energy consumption and the rational use of water resources are two of the objectives of the water-energy nexus (Meireles and Sousa 2019).
The energy required for each remediation condition here considered, i.e. SN, S/L ratio of 1:3 and LTTD at 200 °C, and obtained by Eq. (1), RACER software and Eq. (3), respectively, is reported in Table 3. LTTD resulted in the highest energy consumption (i.e. 417 kWh m−3), followed by bioremediation and soil washing (i.e. 48 and 14 kWh m−3, respectively). Soil washing required a lower energy since the process was conducted under milder conditions (i.e. ambient temperature). The use of higher temperatures during bioremediation and LTTD led to an increase of the energy consumption. However, the biomethane produced during bioremediation allowed the recovery of 21 kWh m−3, with an overall energy balance of − 27 kWh m−3. For a further comparison, the condition resulting in the highest (p < 0.05) biomethane yield and similar (p > 0.05) PHE removal (i.e. S) was also considered. The energy recovered by using biomethane in a CHP unit raised to 32 kWh m−3 (net energy balance of − 16 kWh m−3, similar to that obtained with soil washing), and the overall costs decreased to 228 € m−3 as nitrate supplementation was not necessary.
These results (Table 3) show that the energy consumption was probably the main impact factor of total cost during thermal desorption, as discussed above. Soil washing suggests that the energy used for the process is not always a function of the total cost (Inoue and Katayama 2011), and other parameters such as operation labor are also important. During anaerobic bioremediation of PHE-contaminated sediment, the biomethane production may be an opportunity to heat the anaerobic bioreactor leading to cost and energy savings and a lower environmental impact (Esen and Yuksel 2013). However, the cost of collecting biomethane and burning it safely should be also considered for the scale up of the technology.
Conclusions
PHE is particularly widespread in marine environment, and the costs of common disposal methods for contaminated sediments have induced to find more eco-friendly and sustainable solutions. In this study, anaerobic bioremediation, soil washing and thermal desorption allowed to effectively remediate PHE-contaminated sediments with low organic content (i.e. 0.36 g TOC·kg TS−1) decreasing the PHE amount by 68, 97 and 88%, respectively, using a low liquid phase (i.e. 60% and S/L ratio of 1:3) and low temperature (i.e. 200 °C). A cost assessment, conducted by implementing the retention time and temperature of laboratory-scale experiments in the RACER software and accompanied by an energy balance, revealed that anaerobic bioremediation was the least demanding technology in terms of costs (i.e. 228 € m−3) and required a similar energy consumption (i.e. − 16 kWh m−3) to soil washing. On the other hand, soil washing was more effective in PHE removal and, therefore, the choice of the most sustainable technology would depend on the contamination threshold values imposed by the national legislation.
References
Abbassi-Guendouz A, Brockmann D, Trably E et al (2012) Total solids content drives high solid anaerobic digestion via mass transfer limitation. Bioresour Technol. https://doi.org/10.1016/j.biortech.2012.01.174
Agarry SE, Owabor CN (2011) Anaerobic bioremediation of marine sediment artificially contaminated with anthracene and naphthalene. Environ Technol. https://doi.org/10.1080/09593330.2010.536788
Agarwal A, Liu Y (2015) Remediation technologies for oil-contaminated sediments. Mar Pollut Bull. https://doi.org/10.1016/j.marpolbul.2015.09.010
Ambrosoli R, Petruzzelli L, Minati JL, Marsan FA (2005) Anaerobic PAH degradation in soil by a mixed bacterial consortium under denitrifying conditions. Chemosphere. https://doi.org/10.1016/j.chemosphere.2005.02.030
Andersson E, Rotander A, von Kronhelm T et al (2009) AhR agonist and genotoxicant bioavailability in a PAH-contaminated soil undergoing biological treatment. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-009-0121-9
APHA, AWWA, WEF (2005) Standard methods for the examination of water and wastewater, centinial addition. https://doi.org/10.1111/j.1467-9922.2010.00586.x
APHA, AWWA, WEF (2012) Fixed and volatile solids ignited at 550°C 2540 E. Stand. Methods Exam, Water Wastewater
Arienzo M, Donadio C, Mangoni O et al (2017) Characterization and source apportionment of polycyclic aromatic hydrocarbons (pahs) in the sediments of gulf of Pozzuoli (Campania, Italy). Mar Pollut Bull. https://doi.org/10.1016/j.marpolbul.2017.07.006
Astm-D-2216-98 (1998) Standard test method for laboratory determination of water (moisture) content of soil and rock by mass. ASTM Int. https://doi.org/10.1520/D4944-11.1.5
Beškoski VP, Gojgić-Cvijović G, Milić J et al (2011) Ex situ bioremediation of a soil contaminated by mazut (heavy residual fuel oil) - a field experiment. Chemosphere. https://doi.org/10.1016/j.chemosphere.2011.01.020
Bianco F, Race M, Papirio S, Esposito G (2020) Removal of polycyclic aromatic hydrocarbons during anaerobic biostimulation of marine sediments. Sci Total Environ 709:136141. https://doi.org/10.1016/j.scitotenv.2019.136141
Brinch UC, Ekelund F, Jacobsen CS (2002) Method for spiking soil samples with organic compounds. Appl Environ Microbiol. https://doi.org/10.1128/AEM.68.4.1808-1816.2002
Budiyono, Widiasa IN, Johari S, Sunarso (2010) The kinetic of biogas production rate from cattle manure in batch mode. Int J Chem Biomol Eng
Bulmău C, Mărculescu C, Lu S, Qi Z (2014) Analysis of thermal processing applied to contaminated soil for organic pollutants removal. J Geochemical Explor. https://doi.org/10.1016/j.gexplo.2014.08.005
Bustillo-Lecompte CF, Mehrvar M, Quiñones-Bolaños E (2014) Cost-effectiveness analysis of TOC removal from slaughterhouse wastewater using combined anaerobic-aerobic and UV/H2O2 processes. J Environ Manag. https://doi.org/10.1016/j.jenvman.2013.12.035
Cecotti M, Coppotelli BM, Mora VC et al (2018) Efficiency of surfactant-enhanced bioremediation of aged polycyclic aromatic hydrocarbon-contaminated soil: link with bioavailability and the dynamics of the bacterial community. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2018.03.303
Chang BV, Chang SW, Yuan SY (2003) Anaerobic degradation of polycyclic aromatic hydrocarbons in sludge. Adv Environ Res. https://doi.org/10.1016/S1093-0191(02)00047-3
Chang W, Um Y, Holoman TRP (2006) Polycyclic aromatic hydrocarbon (PAH) degradation coupled to methanogenesis. Biotechnol Lett. https://doi.org/10.1007/s10529-005-6073-3
Chattopadhyay S, Chattopadhyay D (2015) Remediation of DDT and its metabolites in contaminated sediment. Curr Pollut, Reports. https://doi.org/10.1007/s40726-015-0023-z
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Bioresour Technol. https://doi.org/10.1016/j.biortech.2007.01.057
Chen S, Zhang J, Wang X (2015) Effects of alkalinity sources on the stability of anaerobic digestion from food waste. Waste Manag Res. https://doi.org/10.1177/0734242X15602965
Cuypers C, Pancras T, Grotenhuis T, Rulkens W (2002) The estimation of PAH bioavailability in contaminated sediments using hydroxypropyl-β-cyclodextrin and Triton X-100 extraction techniques. Chemosphere. https://doi.org/10.1016/S0045-6535(01)00199-0
Dougan L, Bates SP, Hargreaves R et al (2004) Methanol-water solutions: a bi-percolating liquid mixture. J Chem Phys. https://doi.org/10.1063/1.1789951
Du J, Mehler WT, Lydy MJ, You J (2012) Toxicity of sediment-associated unresolved complex mixture and its impact on bioavailability of polycyclic aromatic hydrocarbons. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2011.11.099
Ebihara T, Bishop PL (2003) Effect of acetate on biofilms utilized in PAH bioremediation. Environ Eng Sci. https://doi.org/10.1089/10928750260418944
Ehlers LJ, Luthy RG (2003) Peer reviewed: contaminant bioavailability in soil and sediment. Environ Sci Technol. https://doi.org/10.1021/es032524f
Esen M, Yuksel T (2013) Experimental evaluation of using various renewable energy sources for heating a greenhouse. Energy Build. https://doi.org/10.1016/j.enbuild.2013.06.018
Fabbricino M, Ferraro A, Luongo V et al (2018) Soil washing optimization, recycling of the solution, and ecotoxicity assessment for the remediation of Pb-contaminated sites using EDDS. Sustain. https://doi.org/10.3390/su10030636
Federal Remediation Technologies Roundtable. https://frtr.gov/
Ferraro A, Fabbricino M, van Hullebusch ED, Esposito G (2017) Investigation of different ethylenediamine-N,N′-disuccinic acid-enhanced washing configurations for remediation of a Cu-contaminated soil: process kinetics and efficiency comparison between single-stage and multi-stage configurations. Environ Sci Pollut Res 24:21960–21972. https://doi.org/10.1007/s11356-017-9844-1
Frutos FJG, Escolano O, García S et al (2010) Bioventing remediation and ecotoxicity evaluation of phenanthrene-contaminated soil. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2010.07.098
Ghosal D, Ghosh S, Dutta TK, Ahn Y (2016) Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): a review. Front Microbiol. https://doi.org/10.3389/fmicb.2016.01369
Gielnik A, Pechaud Y, Huguenot D et al (2019) Bacterial seeding potential of digestate in bioremediation of diesel contaminated soil. Int Biodeterior Biodegradation 143:104715
Guo M, Gong Z, Allinson G et al (2016) Variations in the bioavailability of polycyclic aromatic hydrocarbons in industrial and agricultural soils after bioremediation. Chemosphere. https://doi.org/10.1016/j.chemosphere.2015.10.027
Gupta SC, Hanks RJ (2010) Influence of water content on electrical conductivity of the soil 1. Soil Sci Soc Am J. https://doi.org/10.2136/sssaj1972.03615995003600060011x
Gurung B, Race M, Fabbricino M et al (2018) Assessment of metal pollution in the Lambro Creek (Italy). Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2017.11.041
Haleyur N, Shahsavari E, Jain SS et al (2019) Influence of bioaugmentation and biostimulation on PAH degradation in aged contaminated soils: response and dynamics of the bacterial community. J Environ Manag. https://doi.org/10.1016/j.jenvman.2019.02.115
Haritash AK, Kaushik CP (2009) Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): a review. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2009.03.137
Inoue Y, Katayama A (2011) Two-scale evaluation of remediation technologies for a contaminated site by applying economic input-output life cycle assessment: risk-cost, risk-energy consumption and risk-CO 2 emission. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2011.06.029
Kampschreur MJ, Temmink H, Kleerebezem R et al (2009) Nitrous oxide emission during wastewater treatment. Water Res. https://doi.org/10.1016/j.watres.2009.03.001
Kan AT, Fu G, Hunter M et al (1998) Irreversible sorption of neutral hydrocarbons to sediments: experimental observations and model predictions. Environ Sci Technol. https://doi.org/10.1021/es9705809
Kim P, Lloyd J, Kim JW et al (2016) Recovery of creosote from used railroad ties by thermal desorption. Energy. https://doi.org/10.1016/j.energy.2016.05.117
Kiskira K, Papirio S, van Hullebusch ED, Esposito G (2017) Influence of pH, EDTA/Fe(II) ratio, and microbial culture on Fe(II)-mediated autotrophic denitrification. Environ Sci Pollut Res 24:21323–21333. https://doi.org/10.1007/s11356-017-9736-4
Kuppusamy S, Palanisami T, Megharaj M, et al (2016) Ex-situ remediation technologies for environmental pollutants: a critical perspective. In: Reviews of Environmental Contamination and Toxicology. https://doi.org/10.1007/978-3-319-20013-2_2
Landa-Cansigno O, Behzadian K, Davila-Cano DI, Campos LC (2019) Performance assessment of water reuse strategies using integrated framework of urban water metabolism and water-energy-pollution nexus. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-019-05465-8
Lee B-D, Hosomi M (2000) Ethanol washing of PAH-contaminated soil and Fenton oxidation of washing solution. J Mater 2:24–30. https://doi.org/10.1007/s10163-999-0012-7
Lukić B, Huguenot D, Panico A, et al (2016) Importance of organic amendment characteristics on bioremediation of PAH-contaminated soil. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-016-6635-z
Louati H, Ben SO, Soltani A et al (2014) Biostimulation as an attractive technique to reduce phenanthrene toxicity for meiofauna and bacteria in lagoon sediment. Environ Sci Pollut Res 21:3670–3679. https://doi.org/10.1007/s11356-013-2330-5
Maletić S, Murenji S, Agbaba J et al (2018) Potential for anaerobic treatment of polluted sediment. J Environ Manag. https://doi.org/10.1016/j.jenvman.2018.02.029
Mancini G, Papirio S, Lens PNL, Esposito G (2016) Effect of N-methylmorpholine-N-oxide pretreatment on biogas production from rice straw, cocoa shell, and hazelnut skin. Environ Eng Sci. https://doi.org/10.1089/ees.2016.0138
Masy T, Demanèche S, Tromme O et al (2016) Hydrocarbon biostimulation and bioaugmentation in organic carbon and clay-rich soils. Soil Biol Biochem. https://doi.org/10.1016/j.soilbio.2016.04.016
Mat-Shayuti MS, Tuan Ya TMYS, Abdullah MZ, Megat Khamaruddin PNF, Othman NH (2019) Progress in ultrasonic oil-contaminated sand cleaning: a fundamental review. Environ Sci Pollut Res 26:26419–26438. https://doi.org/10.1007/s11356-019-05954-w
Mattei P, Cincinelli A, Martellini T et al (2016) Reclamation of river dredged sediments polluted by PAHs by co-composting with green waste. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2016.05.140
Meireles I, Sousa V (2019) Assessing water, energy and emissions reduction from water conservation measures in buildings: a methodological approach. Environ Sci Pollut Res 27:4612–4629. https://doi.org/10.1007/s11356-019-06377-3
Mohan SV, Kisa T, Ohkuma T et al (2006) Bioremediation technologies for treatment of PAH-contaminated soil and strategies to enhance process efficiency. Rev Environ Sci Biotechnol. https://doi.org/10.1007/s11157-006-0004-1
Muangchinda C, Yamazoe A, Polrit D, Thoetkiattikul H, Mhuantong W, Champreda V, Pinyakong O (2017) Biodegradation of high concentrations of mixed polycyclic aromatic hydrocarbons by indigenous bacteria from a river sediment: a microcosm study and bacterial community analysis. Environ Sci Pollut Res 24:4591–4602. https://doi.org/10.1007/s11356-016-8185-9
Mueller KE, Shann JR (2006) PAH dissipation in spiked soil: impacts of bioavailability, microbial activity, and trees. Chemosphere. https://doi.org/10.1016/j.chemosphere.2005.12.051
Nam JJ, Thomas GO, Jaward FM et al (2008) PAHs in background soils from Western Europe: Influence of atmospheric deposition and soil organic matter. Chemosphere. https://doi.org/10.1016/j.chemosphere.2007.08.010
Namkoong W, Hwang EY, Park JS, Choi JY (2002) Bioremediation of diesel-contaminated soil with composting. Environ Pollut. https://doi.org/10.1016/S0269-7491(01)00328-1
Ogejo JA, Li L (2010) Enhancing biomethane production from flush dairy manure with turkey processing wastewater. Appl Energy. https://doi.org/10.1016/j.apenergy.2010.04.020
Oleszczuk P, Baran S (2005) Kinetics of PAHs losses and relationships between PAHs properties and properties of soil in sewage sludge-amended soil. Polycycl Aromat Compd. https://doi.org/10.1080/10406630591007170
Ortega-Calvo JJ, Tejeda-Agredano MC, Jimenez-Sanchez C et al (2013) Is it possible to increase bioavailability but not environmental risk of PAHs in bioremediation? J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2013.03.042
Pakpahan EN, Isa MH, Kutty SRM, Malakahmad A (2009) Effect of temperature on the formation and degradation of polycyclic aromatic hydrocarbons. https://eprints.utp.edu.my/1908/
Papirio S, Esposito G, Pirozzi F (2013) Biological inverse fluidized-bed reactors for the treatment of low pH- and sulphate-containing wastewaters under different COD/SO2-4 conditions. Environ Technol (United Kingdom). https://doi.org/10.1080/09593330.2012.737864
Pope CJ, Peters WA, Howard JB (2000) Thermodynamic driving forces for PAH isomerization and growth during thermal treatment of polluted soils. J Hazard Mater. https://doi.org/10.1016/S0304-3894(00)00267-3
Qiao J, Zhang C, Luo S, Chen W (2014) Bioremediation of highly contaminated oilfield soil: bioaugmentation for enhancing aromatic compounds removal. Front Environ Sci Eng 8:293–304. https://doi.org/10.1007/s11783-013-0561-9
Readman JW, Fillmann G, Tolosa I et al (2002) Petroleum and PAH contamination of the Black Sea. Mar Pollut Bull. https://doi.org/10.1016/S0025-326X(01)00189-8
Renoldi F, Lietti L, Saponaro S et al (2003) Thermal desorption of a PAH-contaminated soil: a case study. Trans Ecol Environ
Roux MV, Temprado M, Chickos JS, Nagano Y (2008) Critically evaluated thermochemical properties of polycyclic aromatic hydrocarbons. J Phys Chem Ref Data. https://doi.org/10.1063/1.2955570
Sayara T, Pognani M, Sarrà M, Sánchez A (2010) Anaerobic degradation of PAHs in soil: impacts of concentration and amendment stability on the PAHs degradation and biogas production. Int Biodeterior Biodegrad. https://doi.org/10.1016/j.ibiod.2010.02.005
Sayara T, Borràs E, Caminal G et al (2011) Bioremediation of PAHs-contaminated soil through composting: influence of bioaugmentation and biostimulation on contaminant biodegradation. Int Biodeterior Biodegrad. https://doi.org/10.1016/j.ibiod.2011.05.006
Silva A, Delerue-Matos C, Fiúza A (2005) Use of solvent extraction to remediate soils contaminated with hydrocarbons. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2005.05.022
Smith MT, Berruti F, Mehrotra AK (2001) Thermal desorption treatment of contaminated soils in a novel batch thermal reactor. In: Industrial and Engineering Chemistry Research. https://doi.org/10.1021/ie0100333
Smith KEC, Thullner M, Wick LY, Harms H (2009) Sorption to humic acids enhances polycyclic aromatic hydrocarbon biodegradation. Environ Sci Technol. https://doi.org/10.1021/es803661s
Sprovieri M, Feo ML, Prevedello L et al (2007) Heavy metals, polycyclic aromatic hydrocarbons and polychlorinated biphenyls in surface sediments of the Naples harbour (southern Italy). Chemosphere. https://doi.org/10.1016/j.chemosphere.2006.10.055
Steele WV, Chirico RD, Nguyen A, et al (1990) Determination of ideal-gas enthalpies of formation for key compounds. In: AIChE Symposium Series
Steffen KT, Schubert S, Tuomela M et al (2007) Enhancement of bioconversion of high-molecular mass polycyclic aromatic hydrocarbons in contaminated non-sterile soil by litter-decomposing fungi. Biodegradation. https://doi.org/10.1007/s10532-006-9070-x
Sun F, Littlejohn D, David Gibson M (1998) Ultrasonication extraction and solid phase extraction clean-up for determination of US EPA 16 priority pollutant polycyclic aromatic hydrocarbons in soils by reversed-phase liquid chromatography with ultraviolet absorption detection. Anal Chim Acta. https://doi.org/10.1016/S0003-2670(98)00186-X
Sun Y, Ji L, Wang W et al (2009) Simultaneous removal of polycyclic aromatic hydrocarbons and copper from soils using ethyl lactate-amended EDDS solution. J Environ Qual. https://doi.org/10.2134/jeq2008.0374
Tawari-Fufeyin P, Adams GO, Fufeyin PT et al (2015) Bioremediation, biostimulation and Bioaugmention: a review. Int J Environ Bioremediation Biodegrad. https://doi.org/10.12691/ijebb-3-1-5
Thiele-Bruhn S, Brümmer GW (2004) Fractionated extraction of polycyclic aromatic hydrocarbons (PAHs) from polluted soils: estimation of the PAH fraction degradable through bioremediation. Eur J Soil Sci. https://doi.org/10.1111/j.1365-2389.2004.00621.x
Trellu C, Ganzenko O, Papirio S et al (2016) Combination of anodic oxidation and biological treatment for the removal of phenanthrene and Tween 80 from soil washing solution. Chem Eng J. https://doi.org/10.1016/j.cej.2016.07.108
Wakeham SG, Canuel EA (2016) Biogenic polycyclic aromatic hydrocarbons in sediments of the San Joaquin River in California (USA), and current paradigms on their formation. Environ Sci Pollut Res 23:10426–10442. https://doi.org/10.1007/s11356-015-5402-x
Wakeham SG, Schaffner C, Giger W (1980) Poly cyclic aromatic hydrocarbons in recent lake sediments-II. Compounds derived from biogenic precursors during early diagenesis. Geochim Cosmochim Acta doi. https://doi.org/10.1016/0016-7037(80)90041-1
Yang Y, Zhang N, Xue M, Tao S (2010) Impact of soil organic matter on the distribution of polycyclic aromatic hydrocarbons (PAHs) in soils. Environ Pollut. https://doi.org/10.1016/j.envpol.2010.02.019
Yang X, Chen Z, Wu Q, Xu M (2018) Enhanced phenanthrene degradation in river sediments using a combination of biochar and nitrate. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2017.11.130
Yap CL, Gan S, Ng HK (2012) Evaluation of solubility of polycyclic aromatic hydrocarbons in ethyl lactate/water versus ethanol/water mixtures for contaminated soil remediation applications. J Environ Sci (China). https://doi.org/10.1016/S1001-0742(11)60873-5
Yu H, Huang GH, Xiao H, Wang L, Chen W (2014) Combined effects of DOM and biosurfactant enhanced biodegradation of polycylic armotic hydrocarbons (PAHs) in soil-water systems. Environ Sci Pollut Res 21:10536–10549. https://doi.org/10.1007/s11356-014-2958-9
Zhang Z, Lo IMC (2015) Biostimulation of petroleum-hydrocarbon-contaminated marine sediment with co-substrate: involved metabolic process and microbial community. Appl Microbiol Biotechnol 99:5683–5696. https://doi.org/10.1007/s00253-015-6420-9
Zhao C, Dong Y, Feng Y et al (2019) Thermal desorption for remediation of contaminated soil: a review. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.01.079
Zou G, Papirio S, Ylinen A et al (2014) Fluidized-bed denitrification for mine waters. Part II: Effects of Ni and Co. Biodegradation. https://doi.org/10.1007/s10532-013-9670-1
Funding
The Italian Ministry of Education, University and Research (MIUR) and the Erasmus Mundus Joint Doctorate Program ETeCoS3 (Environmental Technologies for Contaminated Solids, Soils, and Sediments) [grant agreement FPA number 2010-0009] co-funded this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Bingcai Pan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 51 kb)
Rights and permissions
About this article
Cite this article
Bianco, F., Monteverde, G., Race, M. et al. Comparing performances, costs and energy balance of ex situ remediation processes for PAH-contaminated marine sediments. Environ Sci Pollut Res 27, 19363–19374 (2020). https://doi.org/10.1007/s11356-020-08379-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08379-y