Abstract
Natural steroid hormones in the aquatic environment have attracted increasing attention because of their strong endocrine disrupting potency. Seven steroid hormones (estrone, 17α-estradiol, 17β-estradiol, estriol, testosterone, androstenedione, and progesterone) were analyzed from surface water and sediment sampled from Chaohu Lake, its upstream rivers (the Hangbu River, Nanfei River, Shiwuli River, and Pai River), drainage from the adjacent farmland, and treated and untreated municipal sewage. Concentrations of the seven target steroid hormones ranged from below the detection limit (ND) to 69.5 ng L−1 in the water of Chaohu Lake and the upstream rivers. Three steroids—estrone, estriol, and 17α-estradiol—were found in relatively high residual concentrations in the water, with maximum concentrations of 69.5 ng L−1, 51.5 ng L−1, and 23.3 ng L−1, respectively. All of the target steroid hormones except estriol were detected in the sediment in concentrations of ND–16344 ng kg−1. The dominant steroid hormone in the sediment of Chaohu Lake and the upstream rivers was 17α-estradiol. In the Shiwuli River and the Pai River, the dominant steroid hormones (estrone and estriol) were the same as those in the untreated municipal sewage. This confirmed the deduction that untreated municipal sewage was the major source of steroid hormone residues in these two rivers. The main steroid hormone in the water of the Hangbu River and Chaohu Lake was 17α-estradiol, the same as that in the farmland drainage. In addition, 17α-estradiol was verified as the major factor in the contribution of farmland drainage to the pollution in these rivers. The water in the Nanfei River had high concentrations of estriol and 17α-estradiol. This indicates that both untreated municipal sewage and farmland drainage were the major sources. The discharge of steroid hormones from the four rivers to Chaohu Lake was approximately 75.1 kg year−1, with the highest contributor being 17α-estradiol (24 kg year−1). Therefore, based on the results of this study, the farmland drainage should be controlled to reduce the steroid hormone pollution in Chaohu Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Natural steroid hormones (SHs), which originate from the excretion (feces and urine) of humans and livestock, have attracted worldwide attention because of their endocrine-disrupting effects on aquatic organisms and humans (Adeel et al. 2017; Yuan et al. 2018). An earlier study found that the steroid estrogen 17β-estradiol (βE2) could stimulate the production of a female protein in male fish at concentrations of 1–10 ng L−1 (Routledge et al. 1998). Subsequent studies have focused on the presence of steroid estrogens, such as estrone (E1), 17β-estradiol (βE2), and estriol (E3), in natural water bodies and the potential risks they pose (Gong et al. 2016; Jeon et al. 2017; Shi et al. 2014; Wang et al. 2015). A recent study found that the steroid progestin progesterone (PG) could affect the gene expression in zebrafish (Danio rerio) early in their development at 2 ng L−1 (Zucchi et al. 2012). Steroid androgens, such as testosterone (TS), can also suppress the antibody-forming cell responses in Chinook salmon (Slater and Schreck 1993). Animal manure contains more androgen and progestogen hormones than estrogen hormones (Shore and Shemesh 2003). Androgens and progestogens were still found as the dominant class, followed by estrogens, in sewage treatment plant effluents in Beijing, China (Chang et al. 2011).
Many studies have focused on the content and composition of steroid hormones in point sources, such as municipal sewage treatment plants and farms with lagoon waste disposal systems (Hutchins et al. 2007; Liu et al. 2012; Yarahmadi et al. 2018). Huang et al. (2013) investigated the incidence of estrogens in Dianchi Lake and upstream rivers only in relation to the wastewater released from the eight adjacent sewage treatment plants. Liu et al. (2012) reported on the concentrations of steroids in a typical swine farm and the release of such steroids into the adjacent stream. In reality, animal manure-borne hormones also enter the soils through agricultural applications and are transported to adjacent streams by runoff or farmland drainage (Gall et al. 2011; Lafrance and Caron 2013; Luo et al. 2013). These studies reported extensively on contamination from steroid estrogens. In contrast, few studies have focused on the residual concentrations of steroid androgens and progestogens, which can potentially carry similar risks and should therefore receive as much attention as estrogens. In addition, only a few studies have assessed the effects of the release of steroid hormones from the combination of point sources and agricultural non-point sources in environmentally sensitive zones, e.g., ecological preservation areas.
Chaohu Lake (CH), located in southeastern China and adjacent to Hefei city, is the fourth largest freshwater lake in China. It is the primary reserve domestic water source for Hefei’s 7.86 million residents. Chaohu Lake also supplies water to the growing industrial and agricultural sectors in and around the city. Chaohu Lake has abundant fishery resources, 54 fish species (Guo et al. 2007). Because it is affected by waste and wastewater from industry, agriculture, and daily life, Chaohu Lake has been one of the most hypereutrophic lakes in China since the 1980s. Recent studies have shown that Chaohu Lake also suffers from severe contamination by organic pollutants, such as polycyclic aromatic hydrocarbons, perfluorooctanoic acid, and perfluorooctane sulphonate (Kong et al. 2017; Kong et al. 2018; Liu et al. 2015). The lake was likely contaminated by steroid hormones transported from municipal sewage through the upstream rivers and agricultural drainage from the adjacent farmlands. The distribution of steroid hormones in this area is still largely unknown. In particular, data on the spatial distribution and the contributions of the sources of upstream rivers are not available.
Estrogens, such as estrone, estriol, and 17β-estradiol, were most frequently detected in rivers and lakes (Gong et al. 2016; Jeon et al. 2017; Wang et al. 2012). A previous study by the authors found higher concentrations of estrogen 17α-estradiol than other estrogens in the ditchwater adjacent to greenhouses where there was long-term use of animal manure compost (Zhang et al. 2015). Testosterone, androstenedione, and progesterone, typical androgens and progestogens in the wastewater from sewage treatment plants and animal-feeding operations, pose a risk to the environment (Chang et al. 2011; Liu et al. 2012). Therefore, seven steroid hormones including estrone (E1), 17β-estradiol (βE2), 17α-estradiol (αE2), estriol (E3), testosterone (TS), androstenedione (ADD), and progesterone (PG) were selected for this study. The goal was to determine the composition and content patterns of steroid hormones in Chaohu Lake and its upstream rivers. To ascertain the sources mainly responsible for steroid hormones contamination in Chaohu Lake, samples from the municipal sewage and adjacent agricultural drainage areas were also analyzed.
Materials and methods
Study area
Chaohu Lake (117.00° E–118.29° E, 30.56° N–32.02° N) is located in Hefei city, Anhui Province, in southeast China. It has an average depth of 2.7 m and a surface water area of 573 km2. Based on a line crossing Zhongmiao Temple and Mushan Island, Chaohu Lake is divided into two sections: west and east. Because of the growing pollutant load from the upstream rivers in recent decades, Chaohu Lake has suffered from unprecedented levels of pollution. There are 33 tributaries flowing into Chaohu Lake. The Nanfei River, Shiwuli River, Pai River, and Hangbu River are the major inflow rivers. They contribute approximately 80% of the total discharge into the Chaohu Lake. The pollutant load can be greatly affected by the land cover in the Chaohu Lake watershed.
The land cover pattern in the basin has changed a great deal because of the development of agriculture, industry, and urban areas. The land cover pattern has been significantly affected by the conversion of agricultural land to urban and industrial use in recent decades. The land cover in the Chaohu Lake watershed is shown in (Fig. S1). In the western part of the Chaohu Lake, urbanization has had a major effect on the Nanfei River and the Shiwuli River. In addition, farmland may be a source of the pollutants in the aquatic environment of the Pai River and the Hangbu River. In the eastern part of the Chaohu Lake, the surrounding farmland, grassland, and urban land are closely associated with the water quality of the rivers flowing into it. The water in these rivers is supplied mainly by natural rainfall. Treated domestic wastewater from municipal sewage treatment plants (STPs) in Hefei has been released directly into these rivers, and it has flowed into Chaohu Lake.
Sampling and analysis
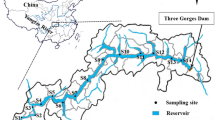
Samples were taken along a continuum from urban wastewater and agricultural drainage to the adjacent aquatic environment in May and January 2016. The sampling sites are shown in Fig. 1. Locations 1–9, 10–12, 13–15, and 16–19 were in the Nanfei River, Shiwuli River, Pai River, and Hangbu River. Locations 20–22 and 23–24 were in the western and eastern sections, respectively, of Chaohu Lake. Samples of surface water and sediment were collected from the rivers and the lake at the same time. Three drainage samples were taken from the ditches adjacent to the greenhouses. Surface water samples (1.5 L) were collected at 0–1 m of the overlying water depths.
For the steroid hormones analysis for each water sample, sodium azide was added at 200 mg L−1 to inhibit the microbial degradation of the steroid hormones. The surface sediment samples (5 cm depth) were collected with a grab sampler. In addition, 24-h flow proportional composite influents and effluents were collected from four sewage treatment plants located near the Nanfei River, Shiwuli River, Pai River, and Hangbu River. Farmland drainage was sampled from three ditch sites adjacent to greenhouses. All samples were transported to the laboratory within 7 h of collection. The water samples were analyzed immediately upon arrival at the laboratory, and the sediment samples were stored at − 20 °C for further extraction and analysis. Pretreatment and analysis of steroid hormones in environmental samples have been described in a previous study by the authors (Zhang et al. 2015). The concentrations of seven steroid hormones were analyzed using an Acquity UPLC-ESI/MS/MS (Waters Corporation, Milford, MA, USA). The detection limits, recoveries, and method detection limits (MDLs) are shown in Table 1 and Table 2, respectively.
For algae analysis in lake water, another portion of water samples (1 L) was collected and fixed with Lugol’s iodine solution in situ. The final concentration was limited to 15‰. The fixed quantitative samples were put into glass-measuring cylinders immediately upon arrival at the laboratory and concentrated to 30–100 ml based on different densities prior to counting on a microscope. The entire process lasted at least 3 days. Species were examined with an inverted microscope (Zeiss Axiovert 25, magnification × 400), and a 1-ml Sedgewick-Rafter counting frame was used for cell counting. For the error to be less than 10%, the total phytoplankton cell numbers counted each time must be more than 400.
Annual steroid hormone inflow to Chaohu Lake from four rivers
Based on the steroid hormones in the inlet section and the output water of each river, the flux of steroid hormones discharged into Chaohu Lake from the Nanfei River, Shiwuli River, Pai River, and Hangbu River was calculated by the following equation: AI = ΣSHs × I × T. ΣSHs is the sum of steroid hormone concentrations in nanograms per liter; I is the inflow to Chaohu Lake from each river in 2016 in cubic meters per second; AI is the annual inflow of ΣSHs to Chaohu Lake; and T is 365 days. The discharge of inflowing rivers was calculated according to the method proposed by Raymond et al. (2013). The procedures for calculating the discharge from the inflowing rivers are shown in supporting information.
Results and discussion
Steroid hormone residues and their distribution coefficients in Chaohu Lake and rivers
The amount of steroid hormones in the water and sediment of Chaohu Lake and its four upstream rivers is shown in Fig. 2. The values for the seven target steroid hormones in the river water ranged from below the detection limit (ND) to 69.5 ng L−1. In descending order, the maximum concentrations of estrogens were 69.5 ng L−1 (E3), 51.5 ng L−1 (αE2), 23.3 ng L−1 (E1), and 3.63 ng L−1 (βE2). The average concentrations of estrogens exhibited similar trends with regard to the maximum concentrations. E3 was found to have the highest mean concentration (7.07 ng L−1). This was followed by the mean concentrations for E1 (4.0 ng L−1) and αE2 (6.99 ng L−1). βE2 had the lowest average concentration, 1.36 ng L−1, of the estrogens. Estrogens were frequently detected in the rivers and lake. Previous studies found maximum concentrations of E1, βE2, and E3 in the water of the Yellow river, Pearl River, and the three Tianjin urban rivers of 8.0–55.8 ng L−1, 1.6–117 ng L−1, and 5.3–46.4 ng L−1, respectively (Lei et al. 2009; Wang et al. 2012; Yang et al. 2014). The maximum concentrations of E1 and βE2 in this study were comparable to those found in these bodies; however, the concentration of E3 was higher than that in Dianchi Lake, Taihu Lake, and the three rivers from Tianjin (Huang et al. 2013; Lei et al. 2009; Zhou et al. 2016).
For the androgens and progestogens, the concentration of PG was lower, with a maximum value of 3.5 ng L−1. The maximum concentrations of TS and ADD were below 2 ng L−1. The average concentrations were 0.39 ng L−1, 0.09 ng L−1, and 0.69 ng L−1 for ADD, TS, and PG, respectively. The residual concentrations in this study were much lower than those previously reported for the four rivers flowing from Beijing. These were 8.6 ng L−1, 99 ng L−1, and 26 ng L−1 for ADD, TS, and PG, respectively (Chang et al. 2009).
All of the target steroid hormones except E3 were detected in the sediment, and the concentrations of the steroids varied considerably, ND–16344 ng kg−1. Higher concentrations of αE2 were found in the sediment. The maximum concentration was 16,344 ng kg−1. The maximum concentrations of other detected steroid hormones were below 2000 ng kg−1. The average concentrations of αE2 in the sediment were 6897 ng kg−1, with corresponding concentrations of E1 and βE2 of 457 ng L−1 and 581 ng kg−1, respectively. For the androgens and progestins, the average concentration of ADD, TS, and PG were all below 300 ng kg−1. The occurrence of detected steroids in the surface water and sediment samples varied. E3 was detected in the water from the lake and the rivers, but no residue was found in the sediment.
The distribution coefficients (Kd and L kg−1) were calculated by the following equation: Kd = S/C, where S is the concentration of a steroid in the sediment (mg kg−1) and C is the concentration of a steroid in the water (mg L−1). Higher Kd values indicated lower transportability of the compounds. The distribution coefficients (Kd) of the steroids were calculated using their maximum concentrations in the sediment and water samples. The highest value was 2.0 × 103 L kg−1, which was found for αE2, followed, in descending order, by the values for TS, PG, ADD, βE2, E1, and E3 (Fig. 3). A similar sequence of Kd values for these steroids would be obtained if their mean concentrations were used for the calculation. Of the steroid hormones, E3 had the lowest Kd, and this was in accordance with the findings for the soil and groundwater (Zhang et al. 2015). A possible explanation is that E3 has a lower octanol–water partition coefficient (log Kow 2.81) than other steroid hormones (log Kow 3.43–4.01) (Sangster et al. 2015).
Steroid hormone spatial and temporal distribution in the Chaohu Lake system
The total concentrations of steroid hormones in the river water varied significantly, 27.4–100 ng L−1, at the sites (Fig. 4). Sites 6, 10, 12, 13, and 15 had higher total concentrations of steroid hormones. These are the sites located in the Nanfei River (site 6), Shiwuli River (sites 10, 12), and Pai River (sites 13, 15). The highest steroid hormone content, 100 ng·L−1, was found in the water sample from site 10. Sites 12, 13, and 15 were located downstream from the sewage treatment plants’ discharges (STP2 and STP3); thus, the content of the steroid hormones in these sites could be influenced greatly by the sewage treatment plants’ effluents. Surprisingly, of all the sampling locations, site 10, located upstream from the STP2 discharge, had the highest total concentration.
The results indicate that besides the sewage treatment plants’ discharge, other types of discharge were contributing to the presence of steroid hormones in the river water. The Shiwuli River, where site 10 is located, flows through Hefei, an urban center (Fig. S1), and a great deal of untreated municipal sewage is discharged directly into the rivers. Site 6, located on a tributary of the Nanfei River, might be affected by the runoff and drainage from the adjacent farmland. Concerning the concentration of specific steroid hormones, higher concentrations of E1 occurred at site 10 (23.3 ng L−1) and site 13 (22.2 ng L−1). The highest concentration of αE2 (51.5 ng L−1) was found at site 6. The highest concentrations of E3 and βE2, 69.4 ng L−1 and 3.6 ng L−1, respectively, were seen at site 10. The highest concentrations of TS and ADD, 0.16 ng L−1 and 2.3 ng L−1, respectively, were found at sites 12 and 13.
The total concentration of steroid hormones in Chaohu Lake, as shown in Fig. 5, was higher in the eastern part than that in the western part in both January and May, and this was negatively correlated with the number of algae that were found. Steroid hormones, such as ADD, PG, and βE2, can be degraded and absorbed by microalgae in fresh water (Abul-Hajj and Qian 1986; Peng et al. 2014; Wang et al. 2017). For example, the green microalgae Scenedesmus obliquus and Chlorella pyrenoidosa have strong biodegradation and sorption abilities in PG (Peng et al. 2014). The western half of the lake had lower total steroid hormone concentrations than the eastern half. This might be the result of the higher degradation and sorption capacity of the microalgae.
Steroid hormones in eastern and western Chaohu Lake during May and January. HBR, Hangbu River; NFR, Nanfei River; PR, Pai River; SWR, Shiwuli River; SHs, steroid hormones; E1, estrone; βE2, 17β-estradiol; E3, estriol; αE2, 17α-estradiol; PG, progesterone; TS, testosterone; ADD, androstenedione; EHL, eastern half lake; WHL, western half lake; Chl, chlorophyta; Cya, cyanobacteria; Bac, Bacillariophyta; Cry, Crypotophyta
Differences in biodegradation rates could also be the reasons for the seasonal variations in the steroid hormone concentrations in Chaohu Lake. As is illustrated in Fig. 5, the total concentration of steroid hormones in Chaohu Lake in May was lower than that in January. This could be the result of the mitigation of biodegradation under low temperatures in January. Similar seasonal variations have been found in the Mira River in Portugal and the Yangtze Estuary in China (Nie et al. 2015; Rocha et al. 2016).
Contribution of adjacent sources to the steroid hormone composition of Chaohu Lake
The steroid hormone composition of the various sources, including the untreated municipal sewage, treated municipal sewage, farmland drainage, Chaohu Lake, and rivers are illustrated in Fig. 6. All seven target steroid hormones were detected in the untreated municipal sewage, treated municipal sewage, and farmland drainage. In the treated municipal sewage, the sum of the average concentrations of steroid hormones was 199.3 ng L−1, with concentrations of E1 and E3 being 48.6 ng L−1 and 63.6, respectively. Relatively lower concentrations of αE2 and βE2 were found, 9.4 ng L−1 and 3.2 ng L−1, respectively. The concentration of ADD, 66.5 ng L−1, was the highest detected for the androgens in the untreated municipal sewage. This was significantly higher than that found for TS (3.0 ng L−1). A relatively low concentration of PG (4.93 ng L−1) was detected in untreated municipal sewage compared with the concentrations of E1, E3, and ADD. E1 and E3 were found to be the most abundant estrogens in the wastewater. This was similar to the results for the sewage treatment plants around Dianchi Lake, China (Huang et al. 2014). Humans usually do not excrete 17α-E2 but life stock, especially cows, do. The observed αE2 in the untreated sewage was not excreted by humans but possibly resulted from the biological conversion exhibited in that of E1 (Hutchins et al. 2007). Similar to the results of (Chang et al. 2011), ADD and PG were found to be the dominant compounds in the androgens and progestogens. After sewage treatment, the average concentrations of E1 and E3 in the treated municipal sewage reduced to 0.93 ng L−1 and 1.11 ng L−1, respectively. The concentrations of αE2 and βE2 were also reduced; they were 3.52 ng L−1 and 1.01 ng L−1, respectively. The average concentration of ADD in the treated municipal sewage was 1.84 ng L−1. For TS and PG, the concentrations were below 1 ng L−1.
The steroid hormones composition in different sources, adjacent rivers and Chaohu Lake. HBR, Hangbu River; NFR, Nanfei River; PR, Pai River; SWR, Shiwuli River; CH, Chaohu Lake; E1, estrone; βE2, 17β-estradiol; E3, estriol; αE2, 17α-estradiol; PG, progesterone; TS, testosterone; ADD, androstenedione; TMS, treated municipal sewage; UMS, untreated municipal sewage; FD, farmland drainage
For the farmland drainage, the sum of the average steroid hormone concentrations was 19.3 ng L−1, which was lower than that in the untreated municipal sewage but higher than that in the treated municipal sewage. The average concentration of αE2 (14.7 ng L−1) in the farmland drainage was found to be relatively high, followed by the concentrations of E1 (2.91 ng L−1), βE2 (0.88 ng L−1), and E3 (0.13 ng L−1). The average concentrations of PG, TS, and ADD were 0.16 ng L−1, 0.13 ng L−1, and 0.39 ng L−1, respectively.
Previous studies have found that the concentrations of steroid hormones in the surface water near agricultural fields increased after applications of animal manure (Lafrance and Caron 2013; Liu et al. 2018; Zhang et al. 2015). Manure type and treatment will affect the transport of steroid hormones to surface water (Biswas et al. 2017; DeLaune and Moore 2013). Zheng et al. (2008) found αE2 to be the largest percentage of the total hormone in fresh solid dairy manure; however, αE2 was seldom found in the excreta of poultry (Hanselman et al. 2003). A larger amount of cattle manure might have been applied to the farmland in this watershed, and this might have increased the αE2 residue in the farmland drainage. In this study, the total estrogen concentration, 19.3 ng L−1, found in the farmland drainage was higher than the 3.35 ng L−1 concentration in the ditchwater from adjacent greenhouses where animal manure compost had been applied (Zhang et al. 2015). It is clear that the use of animal waste that was not thoroughly treated resulted in the increase of manure-borne estrogens in the area under study.
The sum of the average steroid hormones in the treated municipal sewage was 4.49 ng L−1, which was lower than that in the Hangbu River (10.5 ng L−1), Shiwuli River (43.1 ng L−1), Pai River (43.8 ng L−1), and Nanfei River (20.1 ng L−1). Thus, the release of treated municipal sewage was not the main reason for the elevated concentrations of steroid hormones in these rivers. The untreated municipal sewage had higher concentrations of steroid hormones than those found in these four rivers. The main contributors were E1 and E3. In the Shiwuli River, E3 and E1 were present in higher average concentrations, 22.9 ng L−1 and 12.9 ng L−1, respectively, than other steroid hormones. Similarly, E3 and E1, with average concentrations of 21.9 ng L−1 and 13.3 ng L−1, respectively, were the main steroid hormone contributors in the Pai River. The Shiwuli River and Pai River contained the same dominant steroid hormones as those in the untreated municipal sewage, thus confirming the deduction that untreated municipal sewage was the major source of steroid hormone residuals in these two rivers.
The Hangbu River flows through the Hefei suburbs, where a large number of agricultural lands is adjacent. Manure-borne hormones can be transported through the runoff produced by rainfall or irrigation (Lafrance and Caron 2013; Lee et al. 2015). As shown in Fig. 6, αE2 was the main steroid hormones in the farmland drainage and the water in the Hangbu River. It was also verified as the major contribution of farmland drainage to Chaohu Lake. Unlike the Shiwuli River, Pai River, and Hangbu River, the water in the Nanfei River had high concentrations of E3 and αE2. This indicates that untreated municipal sewage and farmland drainage were the chief sources.
The major upstream rivers, including the Shiwuli River, Pai River, Nanfei River, and Hangbu River, discharge directly into Chaohu Lake. The results of the calculations for the annual inflow of steroid hormones to Chaohu Lake from each river are shown in Table 3. The discharge of steroid hormones from the four rivers was 75.1 kg year−1. The highest amount released from the upstream rivers was αE2 (24 kg year−1), followed by E3 (20.8 kg year−1), E1 (18.8 kg year−1), and βE2 (6.8 kg year−1). The quantities of androgens and progestogens were less than those of estrogens. Of the four major rivers investigated in this study, the discharge of steroid hormones from the Nanfei River and Hangbu River was more than that from the Pai River and Shiwuli River. Most of the αE2 released into Chaohu Lake came from the Hangbu River and Nanfei River.
Environmental implications
The maximum concentrations of E1, βE2, αE2, and E3 were 3.63–69.5 ng L−1. To date, little research has been done on αE2 in natural water. The maximum concentration of αE2 in this study could induce the gene expression of vitellogenin (VTG-I) in Japanese medaka (Oryzias latipes) (Huang et al. 2010; Shappell et al. 2010). E1, E3, and βE2 were accepted as the highest contributors to estrogenic activity in rivers and lakes (Gong et al. 2016; Nie et al. 2015; Wang et al. 2015). The maximum concentrations of βE2 in the Chaohu Lake watershed were found to have exceeded 1 ng L−1, which was higher than the lowest observed effective concentration for the βE2 induction of vitellogenin in male trout (Hansen et al. 1998). In addition, the data on steroid androgens and progestogens in natural water bodies were enriched, and in-depth knowledge was gained about the steroid hormone levels in the Chaohu Lake watershed. It is worth noting that, unlike the result for estrogens, the maximum concentrations of PG exceeded the lowest observed effective concentration (2 ng L−1) for larval fish (Zucchi et al. 2012).
This study confirmed that untreated municipal sewage and farmland drainage are the two major sources of steroid hormones in the Chaohu Lake watershed; therefore, more sewage treatment plants should be constructed in Hefei city to treat the wastewater to reduce the direct discharge to the rivers. The steroid hormones detected in the farmland drainage originated mainly from the animal manure applied to the farmland. Prior to its application to agricultural soils, heavy manure should be treated, i.e., composted, to reduce the hormone content.
Conclusions
Steroid hormones are continuously released into the environment through municipal sewage and the surface non-point runoff of manure used in agriculture. Some steroids, such as βE2, αE2, and PG, pose potential risks to aquatic organisms in the Chaohu Lake system. For the four upstream rivers and Chaohu Lake, dominant steroid hormones E1, E3 and αE2 came from untreated municipal sewage and farmland drainage. Untreated municipal sewage and farmland drainage should be controlled to reduce the steroid hormone pollution in Chaohu Lake. Furthermore, higher degradation and sorption capacity of the microalgae might lead to reduction of steroid hormones in the water of Chaohu Lake.
References
Abul-Hajj YJ, Qian X (1986) Transformation of steroids by algae. J Nat Prod 49:244–248
Adeel M, Song X, Wang Y, Francis D, Yang Y (2017) Environmental impact of estrogens on human, animal and plant life: a critical review. Environ Int 99:107–119
Biswas S, Kranz WL, Shapiro CA, Snow DD, Bartelt-Hunt SL, Mamo M, Tarkalson DD, Zhang TC, Shelton DP, van Donk SJ, Mader TL (2017) Effect of rainfall timing and tillage on the transport of steroid hormones in runoff from manure amended row crop fields. J Hazard Mater 324:436–447
Chang H, Wan Y, Hu J (2009) Determination and source apportionment of five classes of steroid hormones in urban rivers. Environ Sci Technol 43:7691–7698
Chang H, Wan Y, Wu S, Fan Z, Hu J (2011) Occurrence of androgens and progestogens in wastewater treatment plants and receiving river waters: comparison to estrogens. Water Res 45:732–740
DeLaune PB, Moore PA (2013) 17β-estradiol in runoff as affected by various poultry litter application strategies. Sci Total Environ 444:26–31
Gall HE, Sassman SA, Lee LS, Jafvert CT (2011) Hormone discharges from a midwest tile-drained agroecosystem receiving animal wastes. Environ Sci Technol 45:8755–8764
Gong J, Huang Y, Huang W, Ran Y, Chen D (2016) Multiphase partitioning and risk assessment of endocrine-disrupting chemicals in the Pearl River, China. Environ Toxicol Chem 35:2474–2482
Guo L, Xie P, Ni L, Hu W, Li H (2007) The status of fishery resources of Lake Chaohu and its response to eutrophication. Acta Hydrobiologica Sinica 31:700–705
Hanselman TA, Graetz DA, Wilkie AC (2003) Manure-borne estrogens as potential environmental contaminants: a review. Environ Sci Technol 37:5471–5478
Hansen PD, Dizer H, Hock B, Marx A, Sherry J, McMaster M, Blaise C (1998) Vitellogenin – a biomarker for endocrine disruptors. TrAC Trends Anal Chem 17:448–451
Huang B, Wang B, Ren D, Jin W, Liu J, Peng J, Pan X (2013) Occurrence, removal and bioaccumulation of steroid estrogens in Dianchi Lake catchment, China. Environ Int 59:262–273
Huang B, Li X, Sun W, Ren D, Li X, Li X, Liu Y, Li Q, Pan X (2014) Occurrence, removal, and fate of progestogens, androgens, estrogens, and phenols in six sewage treatment plants around Dianchi Lake in China. Environ Sci Pollut R 21:12898–12908
Huang C, Zhang Z, Wu S, Zhao Y, Hu J (2010) In vitro and in vivo estrogenic effects of 17α-estradiol in medaka (Oryzias latipes). Chemosphere 80:608–612
Hutchins SR, White MV, Hudson FM, Fine DD (2007) Analysis of lagoon samples from different concentrated animal feeding operations for estrogens and estrogen conjugates. Environ Sci Technol 41:738–744
Jeon S, Hong S, Kwon B-O, Park J, Song SJ, Giesy JP, Khim JS (2017) Assessment of potential biological activities and distributions of endocrine-disrupting chemicals in sediments of the west coast of South Korea. Chemosphere 168:441–449
Kong X, He W, Qin N, Liu W, Yang B, Yang C, Xu F, Mooij WM, Koelmans AA (2017) Integrated ecological and chemical food web accumulation modeling explains PAH temporal trends during regime shifts in a shallow lake. Water Res 119:73–82
Kong X, Liu W, He W, Xu F, Koelmans AA, Mooij WM (2018) Multimedia fate modeling of perfluorooctanoic acid (PFOA) and perfluorooctane sulphonate (PFOS) in the shallow lake Chaohu, China. Environ Pollut 237:339–347
Lafrance P, Caron E (2013) Impact of recent manure applications on natural estrogen concentrations in streams near agricultural fields. Environ Res 126:208–210
Lee B, Kullman SW, Yost EE, Meyer MT, Worley-Davis L, Williams CM, Reckhow KH (2015) Predicting characteristics of rainfall driven estrogen runoff and transport from swine AFO spray fields. Sci Total Environ 532:571–580
Lei B, Huang S, Zhou Y, Wang D, Wang Z (2009) Levels of six estrogens in water and sediment from three rivers in Tianjin area, China. Chemosphere 76:36–42
Liu S, Ying G-G, Zhou L-J, Zhang R-Q, Chen Z-F, Lai H-J (2012) Steroids in a typical swine farm and their release into the environment. Water Res 46:3754–3768
Liu W-X, He W, Qin N, Kong X-Z, He Q-S, Yang B, Yang C, Jorgensen SE, Xu F-L (2015) Temporal-spatial distributions and ecological risks of perfluoroalkyl acids (PFAAs) in the surface water from the fifth-largest freshwater lake in China (Lake Chaohu). Environ Pollut 200:24–34
Liu Y, Lin Y, Yen C, Miaw C, Chen T, Wu M, Hsieh C (2018) Identification, contribution, and estrogenic activity of potential EDCs in a river receiving concentrated livestock effluent in Southern Taiwan. Sci Total Environ 636:464–476
Luo Q, Adams P, Lu J, Cabrera M, Huang Q (2013) Influence of poultry litter land application on the concentrations of estrogens in water and sediment within a watershed. Environ Sci: Processes Impacts 15:1383–1390
Nie M, Yan C, Dong W, Liu M, Zhou J, Yang Y (2015) Occurrence, distribution and risk assessment of estrogens in surface water, suspended particulate matter, and sediments of the Yangtze Estuary. Chemosphere 127:109–116
Peng F-Q, Ying G-G, Yang B, Liu S, Lai H-J, Liu Y-S, Chen Z-F, Zhou G-J (2014) Biotransformation of progesterone and norgestrel by two freshwater microalgae (Scenedesmus obliquus and Chlorella pyrenoidosa): transformation kinetics and products identification. Chemosphere 95:581–588
Raymond PA, Hartmann J, Lauerwald R, Sobek S, McDonald C, Hoover M, Butman D, Striegl R, Mayorga E, Humborg C, Kortelainen P, Durr H, Meybeck M, Ciais P, Guth P (2013) Global carbon dioxide emissions from inland waters. Nature 503:355–359
Rocha MJ, Cruzeiro C, Reis M, Pardal MÂ, Rocha E (2016) Pollution by oestrogenic endocrine disruptors and β-sitosterol in a south-western European river (Mira, Portugal). Environ Monit Assess 188:240
Routledge EJ, Sheahan D, Desbrow C, Brighty GC, Waldock M, Sumpter JP (1998) Identification of estrogenic chemicals in STW effluent. 2. In vivo responses in trout and roach. Environ Sci Technol 32:1559–1565
Shappell NW, Hyndman KM, Bartell SE, Schoenfuss HL (2010) Comparative biological effects and potency of 17α- and 17β-estradiol in fathead minnows. Aquat Toxicol 100:1–8
Sangster JL, Oke H, Zhang Y, Bartelt-Hunt SL (2015) The effect of particle size on sorption of estrogens, androgens and progestagens in aquatic sediment. J Hazard Mater 299:112–121
Shi J, Liu X, Chen Q, Zhang H (2014) Spatial and seasonal distributions of estrogens and bisphenol A in the Yangtze River Estuary and the adjacent East China Sea. Chemosphere 111:336–343
Shore LS, Shemesh M (2003) Naturally produced steroid hormones and their release into the environment. Pure Appl Chem 75:1859–1871
Slater CH, Schreck CB (1993) Testosterone alters the immune response of chinook salmon, Oncorhynchus tshawytscha. Gen Comp Endocrinol 89:291–298
Wang L, Ying G-G, Chen F, Zhang L-J, Zhao J-L, Lai H-J, Chen Z-F, Tao R (2012) Monitoring of selected estrogenic compounds and estrogenic activity in surface water and sediment of the Yellow River in China using combined chemical and biological tools. Environ Pollut 165:241–249
Wang P, Wong Y-S, Tam NF-Y (2017) Green microalgae in removal and biotransformation of estradiol and ethinylestradiol. J Appl Phycol 29:263–273
Wang Y, Wang Q, Hu L, Lu G, Li Y (2015) Occurrence of estrogens in water, sediment and biota and their ecological risk in Northern Taihu Lake in China. Environ Geochem Health 37:147–156
Yang J, Li H, Ran Y, Chan K (2014) Distribution and bioconcentration of endocrine disrupting chemicals in surface water and fish bile of the Pearl River Delta, South China. Chemosphere 107:439–446
Yarahmadi H, Duy SV, Hachad M, Dorner S, Sauvé S, Prévost M (2018) Seasonal variations of steroid hormones released by wastewater treatment plants to river water and sediments: distribution between particulate and dissolved phases. Sci Total Environ 635:144–155
Yuan S, Huang C, Ji X, Ma M, Rao K, Wang Z (2018) Prediction of the combined effects of multiple estrogenic chemicals on MCF-7 human breast cancer cells and a preliminary molecular exploration of the estrogenic proliferative effects and related gene expression. Ecotox Environ Safe 160:1–9
Zhang F, Xie Y, Li X, Wang D, Yang L, Nie Z (2015) Accumulation of steroid hormones in soil and its adjacent aquatic environment from a typical intensive vegetable cultivation of North China. Sci Total Environ 538:423–430
Zheng W, Yates SR, Bradford SA (2008) Analysis of steroid hormones in a typical dairy waste disposal system. Environ Sci Technol 42:530–535
Zhou L, Zhang B, Zhao Y, Wu Q (2016) Occurrence, spatiotemporal distribution, and ecological risks of steroids in a large shallow Chinese lake, Lake Taihu. Sci Total Environ 557-558:68–79
Zucchi S, Castiglioni S, Fent K (2012) Progestins and antiprogestins affect gene expression in early development in zebrafish (Danio rerio) at environmental concentrations. Environ Sci Technol 46:5183–5192
Funding
This research was funded by the National Natural Science Foundation of China (No. 41201513) and the Environmental Protection Public Welfare Program of China (201509074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ester Heath
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 253 kb)
Rights and permissions
About this article
Cite this article
Zhang, F., Yu, Q. & Yang, L. Watershed scale patterns in steroid hormones composition and content characters at a typical eutrophic lake in southeastern China. Environ Sci Pollut Res 26, 6107–6115 (2019). https://doi.org/10.1007/s11356-018-04120-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-04120-y