Abstract
The increasing role of chemistry in industrial production and its direct and indirect impacts in everyday life create the need for continuous search and efficiency improvement of new methods for decomposition/removal of different classes of waterborne anthropogenic pollutants. This review paper addresses a highly promising class of water treatment solutions, aimed at tackling the pressing problem of emerging contaminants in natural and drinking waters and wastewater discharges. Radiation processing, a technology originating from radiation chemistry studies, has shown encouraging results in the treatment of (mainly) organic water pollution. Radiation (“high energy”) processing is an additive-free technology using short-lived reactive species formed by the radiolysis of water, both oxidative and reducing, to carry out decomposition of organic pollutants. The paper illustrates the basic principles of radiolytic treatment of organic pollutants in water and wastewaters and specifically of one of its most practical implementations (electron beam processing). Application examples, highlighting the technology’s strong points and operational conditions are described, and a discussion on the possible future of this technology follows.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The increasing role of chemistry in industrial production and its direct and indirect impacts in everyday life create the need for continuous search and efficiency improvement of new methods for decomposition/removal of different classes of anthropogenic pollutants, constantly emitted to ambient air and water. This review paper addresses a highly promising water treatment solution, aimed at tackling the pressing problem of organic contaminants (CECs or contaminants of emerging concern in primis, but also applicable to other classes of pollutants) present in natural and drinking waters and wastewater discharges. CECs include pharmaceuticals and personal care products (PPCPs), some of which may, or are suspected to, act as endocrine disruptors (EDs) (EPA 2016). From a total of 564 chemicals suggested by various agencies, published papers or reports, as being suspected EDs, 147 were considered likely to be either persistent in the environment or produced at high enough volumes to be potential risks for public health. For these reasons, it is now a common regulatory and environmental priority to remove CEC compounds from drinking waters and discharge streams, as stated, for example, in the 2013 Berlaymont Declaration on Endocrine Disruptors (EurActive 2015).
Effective technologies for removal of these products from wastewater are not well established and are generally adopted on a preliminary trial basis: while removal of such compounds from drinking water and effluents has become a priority, no effective removal technologies have been discovered to date, capable to simultaneously remove all of the concerned contaminants (Table 1), even though some processes have demonstrated to remove certain contaminants, to some extent (Petrovic et al. 2003; Shi et al. 2012). Even some high removal rates of specific compounds observed in aqueous phase (by comparison of influent and effluent wastewater concentrations) do not imply full compound degradation to the same extent, as more detailed studies often reveal the presence of transformation by-products in the effluent (Petrovic et al. 2003) or their accumulation, either as original compounds or as by-products, in the excess sludge generated by the treatment process (Jelic et al. 2011), thereby only shifting environmental risk concerns to a different medium.
Even the most commonly used processes for degradation and removal of these pollutants, such as biodegradation and Advanced Oxidation Processes (AOPs), may result in the formation of degradation by-products, which, in turn, are not well studied for all primary compounds, and may also depend on the actual process (Tootchi 2010). It should be noted that radiolytic processes are actually a more intense subset of AOPs, with some distinguishing characteristics that will be discussed in detail in this paper. Typical screening approaches for known by-product compounds, typically carried out by low-resolution mass spectrometry using triple quadrupole technology, are not yet suitable for their online identification (Capodaglio 2016a), especially as biotransformation pathways are often unknown (Wu et al. 2012), and few analytical standards have been developed, for them (Helbling et al. 2010). Conventional AOP treatment in aqueous media is also likely to be a costly proposition, since extremely high conversion yields are needed, ideally to below detection limit, as these compounds retain their hazardous properties even at minute concentrations (Plumlee et al. 2014). Moreover, initial concentrations are very low, making specific treatment cost (cost per unit mass removed) quite high (Klavarioti et al. 2009).
Advanced Oxidation Processes for wastewater treatment
Advanced Oxidation Processes, generally defined as aqueous-phase oxidation methods, have been intensively investigated and developed since the 1970s. They are based on the generation in situ of highly reactive oxidative radicals for decomposition/degradation of organic pollutants in water and wastewater. AOPs include homogeneous and heterogeneous chemical and photochemical processes, such as ultraviolet (UV) photolysis, often in combination with chemical oxidizers (e.g., O3, H2O2) and/or external catalysts (e.g., TiO2), Fenton processes, electrochemical oxidation, sonochemical processes, supercritical water and wet air oxidation, and various combinations thereof (Ikehata et al. 2008; Biń and Sobera-Madej 2012). When applied to CECs, they address substances that are usually present in solution at trace levels (μg/L or even ng/L); thus, most analytical instruments for their quantification need pre-concentration steps such as solid-phase extraction (SPE), solid-phase microextraction (SPME), or accelerated solvent extraction (ASE) (Huerta-Fontela et al. 2010). These methods must be developed accurately for each individual class of compounds and suffer a limitation when they have to be applied for the simultaneous preconcentration of different contaminants (Bolong et al. 2009). There is still no available instrumentation for online, continuous determination of these compounds applicable in field conditions, in the drinking water industry (Capodaglio et al. 2016).
To date, research on AOPs has generated a roughly estimated ensemble of over 20,000 published works, summarized in recent books (Parsons 2004; Stefan 2016) and review articles on their application for removal of: pharmaceutical residues (Klavarioti et al. 2009; Esplugas et al. 2007), EDs (Esplugas et al. 2007), natural organic matter from drinking water (Matilainen and Sillanpaa 2010), biorecalcitrant organics (Ghatak 2014), pathogens (Tsydenova et al. 2015), water pollutants at large (Ribeiro et al. 2015), and on specific treatment of textile effluents (Buthiyappan et al. 2016).
It is conventionally agreed that CEC compounds’ complete removal (i.e., mineralization) from water, in order to eliminate any possible risk, would require treatment processes far more advanced than those currently available, even in facilities of last-generation design (Capodaglio 2016b). Among the “conventional” processes present in state-of-the-art wastewater treatment facilities (Bolong et al. 2009; Liu et al. 2009), physicochemical treatments such as coagulation–flocculation processes have generally been found unable to obtain significant removals, except when used conjunctly with other processes such as activated carbon powder (PAC) dosage (Callegari et al. 2017), or AOPs such as oxidation by chlorination and/or ozonation, and/or UV irradiation with peroxide (Westerhoff et al. 2005).
Applications and practical limitations of AOPs (and other commonly used processes) in water and wastewater treatment
Oxidation is considered a promising removal mechanism for some emerging organic compounds, especially when achieved using strong oxidants, such as chlorine or ozone. Table 2 shows reported removal ranges of some organic pollutants subject to selected AOPs (Snyder 2008). Ozone oxidizes substrates either directly or by generating hydroxyl radicals in water, which react with molecules in solution. Both are strongly reactive reagents and have been proposed (Huber et al. 2005) as promising options for the removal of CECs. Wang et al. (2011) investigated AOP removal efficiencies of eight pharmaceuticals (caffeine, acetaminophen, carbamazepine, sulfamethoxazole, trimethoprim, erythromycin, lincomycin, and codeine) as a function of treatment approaches and types of disinfections (free chlorine, ozone, monochloramine, and permanganate). Results showed that oxidative removal of these compounds varied significantly, according to the different oxidation pathways.
Overall, chlorination can be effective, but pH caused notable differences in its oxidation efficiency. Permanganate was also shown effective to decompose some PPCPs. Care is needed as oxidation of these chemicals has been shown to generate by-products, not fully studied, and which effects are mostly unknown (Mboula et al. 2013).
UV irradiation alone is the least effective in organic contaminants removal. Indication that UV alone cannot be practically considered as a feasible standalone removal option was shown in a study by Adams et al. (2002), proving that UV photolysis could remove 50–80% of the targeted compounds. However, in doing so, it required an absorbed dose 100 times higher than the one required for water disinfection and thus highly expensive, and unpractical, to deliver. Combination of photocatalysis and chemical irradiation for removal of CECs from urban wastewater, was reported, using TiO2 metal oxide semiconductor as catalyst to promote formation of free hydroxyl radicals, with irradiation by near-UV light (λ < 385 nm) (Belgiorno et al. 2007). Some endocrine disruptor compounds were removed, at ratios ranging from 12.5 to 99%, which required reaction times between 0.5 and 8 h, depending on the compound, at catalyst concentration in the reactor vessel between 0.2 and 2 g TiO2/L.
Bisphenol A (BPA) removal by photocatalysis under simulated solar light (Xenon arc lamps) was reported by Mboula et al. (2013): BPA conversion up to 99% was achieved with exposure times of up to 140 min. This corresponded, however, to a mineralization ratio of just 40% (the remaining 60% was transformed into intermediate by-products of unknown nature). By reducing exposure times to 20 min, overall conversion was limited to 35% (with just 10% mineralization).
Photocatalysis supplemented by ultrasonic irradiation has also been tested, where chemical degradation effects derive from acoustic cavitation, i.e., the formation, growth, and implosive collapse of cavitation bubbles in the solution. Under these conditions, extreme temperatures of several thousand degrees and pressures of several hundred atmospheres are developed locally within the bubbles, serving as “hot spots” for microreactions in an otherwise cold liquid. Destruction of chemicals can be achieved through a combination of pyrolytic reactions, occurring inside or near the bubbles, and hydroxyl-radical mediated reactions, occurring in the liquid bulk (Papadaki et al. 2004). Ultrasonic irradiation at power levels up to 125 W has also been applied, in addition to UV irradiation. The average extent of degradation was around 80% after 120 min of sonication; however, degradation intermediates proved difficult to oxidize further, with low mineralization rates. Only 20–25% of initial organic carbon was eventually transformed into CO2 (Belgiorno et al. 2007).
All of the processes illustrated above have some more or less significant drawbacks: almost all require extended contact times for the removal or destruction of contaminants to occur. These range from few to several hours to achieve transformations up to about 90% of the original contaminants’ mass. As an illustration, Table 3 reports literature reaction rate constants determined for a few organic compounds reacting with ozone and hydroxyl radicals: the latter shows reaction rates much faster (eight to nine orders of magnitude) than the former.
Furthermore, even when such transformations occur, they do not necessarily lead to full mineralization of contaminants, as the oxidation of these chemicals has often been found to generate by-products, with mostly unknown effects, which may also be as harmful as the original compounds, or even more (Huerta-Fontela et al. 2011). Full destruction of CECs by these technologies was shown to be heavily dependent on environmental and process conditions, with possible unpredictable cross-interferences among various factors. Since most of these processes require additives (PAC, nanomaterials, catalysts) to achieve notable removal rates, these may constitute extra cost factors, both as process coadjuvants, and by the fact that they may generate additional process residues (e.g., spent PAC and nanoparticles, process sludges) that should be properly treated or disposed of. Additionally, when strong oxidants are used, process and operator’s safety is always an issue. Some additives, either by improper use or by accident, could cause the generation of new pollutants (i.e., disinfection-by-products—DBPs, especially when using chlorine), potential precursors of carcinogenic compounds, or induce undesired contamination when released into the environment (Chang et al. 2009; Liu et al. 2009; Westerhoff et al. 2005; Wu et al. 2012).
Radiolysis and radiolysis-based processes
Radiolysis is defined as the dissociation of molecules by nuclear radiation, resulting in the cleavage of one or several chemical bonds by exposure to a high-energy flux. In this context, the radiation is generated by an ionizing source, and the resulting process is therefore different from, for instance, the photolysis of a chlorine molecule into two Cl radicals, where ultraviolet or visible-light source is used. The kinds of radiation employed in radiolytic environmental applications include γ-rays emitted by selected radionuclides (e.g., 60Co, 137Cs), X-rays, and electron beam (EB) from electron accelerators. They all have different properties, and therefore exhibit different technological benefits and disadvantages, but with very similar final effects. Research studies on radiolytic decomposition of organic pollutants initially employed γ-radiation, obtained mostly from 60Co sources (Cooper et al. 1998; Getoff 2002) and, more rarely, 137Cs sources, the latter being more difficult to handle (Gonzalez-Juarez et al. 2010; Abdel Daiem et al. 2013). Attempts on the use of spent nuclear reactor fuel as γ-source for degradation of polychlorinated biphenyls and pesticides were also reported (Mincher et al. 1991), without further known developments. Sparse applications can be found involving the use of X-rays (Trebše and Arčon, 2003), successfully employed for 4-chlorophenol degradation with simultaneous use of TiO2 (Gonzalez-Juarez et al. 2010).
In all these cases, none of these irradiation techniques could induce energy levels which may ultimately affect the radioactivity content of the irradiated solutions. While in laboratory studies γ-radiation may be frequently used (due to existing availability of sources), in technological-scale applications, the use of EB predominates (Cooper et al. 1998), since this process is particularly efficient concerning the irradiation dose/rate combination. This parameter, as shown in Table 4, is much more favorable for EB irradiation than for γs or X-rays (Farooq et al. 1993; Cooper et al. 2001; Gehringer and Eschweiler 2002; Getoff 2002). It should be noted from the table that 1 Ci (equivalent to 37 GBq in the SI system) corresponds to a very high value of radioactivity but generates a relatively low dose-rate compared to even small EB systems. Other practical considerations have contributed to shift technological advantage towards EB systems, including the very practical fact that EB (as well as X-rays) sources, can be simply switched off when the process is not needed. Furthermore, EB can be easily modulated according to treatment requirements. In pilot installations for water and wastewater treatment, therefore, electron accelerators are almost exclusively employed nowadays.
Although studies on wastewater treatment of ionizing radiation were initiated at MIT (Boston, USA) in the 1950s (Dunn 1953), they are still only marginally mentioned in some of the previously cited books and reviews concerning AOPs (Parsons 2004; Stefan 2016; Ribeiro et al. 2015). In a recently published, extensive review about treatment of priority water pollutants considered by the EU Water Framework Directive, radiolytic methods were included under “miscellaneous technologies,” with citation of just four original papers (Ribeiro et al. 2015), while the actual literature on radiolytic degradation of different classes of pollutants in wastewater treatment applications can be counted in hundreds of published works (Table 5, concerning just PPCPs).
After an initially high interest, radiolytic process applications in the water sector have largely fallen in desuetude, except for occasional applications, mainly due to limits of available technology and its operative limitations, rather than for lack of positive results. Significant progress in EB accelerator technology, promoted by important applications in other industrial realms, could contribute bringing the technology back to the water sector, thanks to lower equipment costs and energetic requirements and greater ease of operation combined with superior results. It should be pointed out that, with appropriate adaptation, all these processes can be employed both in gas- and liquid-phase treatments: applications of ionizing radiation in different industrial and environmental areas include nowadays very specific aspects, such as removal of toxic components from industrial flue gases (Chmielewski et al. 2003), sewage sludge processing (Wang and Wang 2007), soil and sediment treatment (Gray and Cleland 1998), recycling of automotive lubricants (Scapin et al. 2007), and many others (IAEA 2014), including water-related applications that are described in detail in this review. Only liquid-phase applications will be considered in this review: Table 5, showing recent applications to PPCPs removal from aqueous solutions, is an indication of current interest in this technology. Many applications shown in Table 5 led to complete mineralization of the original compounds (100% yield). It should be noted that the extent of mineralization, unlike simple compound’s removal, can be determined only by simultaneous monitoring of the specific compound and the solution’s total organ carbon, which shows whether target compound are also fully decomposed to mineral elements.
Fundamentals of radiolytic decomposition of pollutants
When a diluted aqueous solution (total solute concentration < 10%) is irradiated with ionizing radiation, practically all of the energy absorbed is used in the process of water radiolysis, without affecting the radioactivity of the solution itself. The main products of irradiation are both strongly oxidating, such as hydroxyl radicals (•OH), and strongly reducing species, like solvated electrons (e− aq), hydrogen radicals (•H), in addition to other, less reactive, species, such as H2, H2O2 and H3O+. Observed radical formation yields are constant in the pH range [3–11]. The yields of the reactive species formed in the process are expressed by the G value, a measure of chemo-radiation yield given by the number of molecules formed/consumed (μM) per joule of absorbed energy, for example, in the water radiolysis reaction
The G values of the generated species are 0.28, 0.28, and 0.06, respectively.
The possible reaction mechanisms of hydroxyl radicals (•OH) in radiolytic-driven processes are the same as in AOPs, i.e.,
In addition to these, however, water radiolysis offers the significant advantage, which could never be emphasized enough, of simultaneously conducting both oxidative and reductive processes of dissolved pollutants, due to the simultaneous formation of all those reactive species, following water irradiation. Radiolytic-dependent processes, therefore, commonly associated to AOPs, could therefore be better indicated as AORPs (Advanced Oxidation–Reduction Processes).
In the majority of specific radiolytic decomposition cases, it was shown that the highest oxidation reactivity towards organic compounds are exhibited by the hydroxyl radical (redox potential + 2.7 V in acidic solutions, + 1.8 V in neutral ones), followed (in reductive reactions) by solvated electrons (− 2.9 V) and hydrogen radicals (− 2.3 V). In oxygen-free conditions, solvated electrons and hydrogen radicals usually initiate reductive decomposition of organic pollutants, which is followed by oxidation due to hydroxyl radicals. All these species react with target pollutants depending on the structure and properties of the latters; their removal yield is different for different compounds, as shown by observed values reported in Table 6.
The efficiency of radiolytic decomposition of a given chemical depends on the chemical conditions of the solution during irradiation, which may favor reactions by a particular reactive species, on the absorbed radiation dose and, to a smaller extent, on the actual type of ionizing radiation (due to the mentioned dose-rate effect). An additional, important factor in case of natural media irradiation is the composition of the original water matrix, which may already contain chemical compounds acting as radicals scavengers. Some examples of such species, typically occurring in natural and wastewaters, and the rate constants of their reaction with radiolysis products are also summarized in Table 6.
It can be seen that, in some cases, reaction rates involving scavengers are of the same order of magnitude as those involving target organic pollutants. It may also happen that additional active species, depending on media matrix composition, can be formed by irradiation, enhancing pollutants decomposition during the process. As a result of both phenomena, pollutant decomposition yield in real wastewater may significantly differ from the one evaluated in pure, mono-component solutions.
In order to increase the intensity of radiolytic decomposition of target compounds, the process can be carried out in the presence of externally added substances, which interact with radiolysis products, generating additional amounts of hydroxyl radicals. This can be the case of N2O, which exhibits fast reactions with both e− aq and •H, converting the former to •OH radicals. Earlier studies on radiation treatment in the presence of different oxidants added to irradiated solutions, such as hypochlorite NaOCl (Craft and Eichholz, 1971) or ozone (Sakumoto and Miyata 1977), showed that these could significantly enhance radiation treatment yield.
When irradiation is carried out in air (oxygen) saturated solutions, •O2 − and HO• 2 radicals are also present, formed as the result of oxygen reaction with e− aq and •H. An additional source of •OH radicals can be the presence of H2O2, reacting with •H and e− aq, although an excessively high hydrogen peroxide concentration may scavenge •OH radicals from solution. Solvated electrons e− aq predominate when an irradiated neutral aqueous solution is saturated with N2 or Ar gas, and when the solution contains tert-butanol, also acting as scavenger of •OH radicals. When the pH of a solution is below 2, then the predominating reacting species are •H radicals. For technological reasons, the most appropriate process modifications to increase the concentration of reacting hydroxyl radicals are addition of ozone, or hydrogen peroxide, in limited quantities.
Participation, P, of •H and hydrated electrons e− aq in the radiolytic decomposition of a pollutant S in an aerated solution can be described by the following equations, where k is appropriate rate constants
It can be seen that, at a given absorbed dose, when oxygen concentration approaches 0, P values tend to 1.
The kinetics of radiolysis processes is determined by reaction order and values of rate constants for specific radical reactions, whereas the yield of the irradiation processes is commonly expressed by its G value, the value of dose constant, and the dose magnitude required for 50 or 90% decomposition of the solute (Mincher and Curry 2000).
The G value, as mentioned, represents a measure of chemo-radiation yield, i.e., the number of molecules of reactant consumed, or product formed, per 100 eVFootnote 1 of energy absorbed and can be calculated for a given absorbed dose using the following equation (Kurucz et al. 1991)
where
- C 0 :
-
Initial concentration of the analyte in the irradiated solution [M]
- C D :
-
Concentration of analyte [M] after absorbing dose D, expressed in Grays [Gy]Footnote 2
- N A :
-
Avogadro’s number (6.02 × 1023), and
- K f :
-
Conversion factor for G value units—from 100 eV/L to 6.24 × 1017 Gy
Although G values are commonly reported in applied radiolysis, they cannot be considered very useful for predicting the dose required to decompose a compound in practice, since this usually depends on the latter’s solute concentration, while (initial) G 0 values are often calculated for the smallest applied dose at the beginning of the irradiation, that is, from a linear kinetics approximation that is not fully representative of the actual reaction. The dose constant d, determines the decomposition rate as a function of the absorbed radiation dose and is calculated as the slope of the plot of ln(C D /C 0) against the absorbed dose [Gy]. It is usually considered a more reliable measure than G 0, because it uses all the data from the irradiation procedure. d values can be used to determine the absorbed dose required for 90% decomposition (D 0.90) of a compound, as follows (Cooper et al. 1993)
Large values of G 0 and d correspond to large pollutant decomposition yields (Mincher and Curry 2000). Conversely, increase of decomposition yield corresponds to a decrease of the dose required for required pollutant degradation to a given concentration level.
Experimental dose-constant values can be used as a first approximation estimate of the radiation dose required to remove a particular compound from a solution, where it represents the major contaminant (Kurucz et al. 1995a). From the previous considerations, it follows that the actual dose required will depend on the overall matrix composition and on the compounds actual decomposition pathway(s) in the presence of other contaminants. The obtained dose estimates can also be used to preliminarily evaluate treatment cost.
Effect of the liquid matrix on radiolytic decomposition yield of pollutants and its estimation
The literature on radical reactions, including reactions of many different compounds with radicals formed in water radiolysis, is quite vast (Buxton et al. 1987). Hundreds of works exist on the kinetics of radiolytic decomposition of various groups of environmental pollutants, however, studies have been carried out mostly on single-component synthetic solutions (Wojnarovitz and Takacs 2008).
The most troublesome factor in the kinetic modeling of radiolytic decomposition in real systems, for prediction of concentration changes of specific pollutants, is the influence of the actual solution matrix. Practically, the extent of this effect can be determined, to date, still only experimentally; therefore, application of radiolysis as a treatment process cannot be based solely on yield calculations but must be empirically tested in the most complex situations. On the other hand, the same statement is true for other current AOPs.
Knowledge of reaction rate constants can however be employed for preliminary radiolytic decomposition modeling, including determination of decomposition yields as a function of initial solute concentration, pH, and operational conditions. For example, decomposition of CCl4 at different pHs, initial concentration, and in the presence of methanol as scavenger was calculated and compared with results of EB irradiation of spiked treated groundwater, obtaining excellent correlation (Mak et al. 1997). In a similar work, decomposition of tri- and tetra-chloroethylene was modeled based on the known reactions kinetics with •OH radical, but further process representation improvement required additional specific determination of reaction rate constants with solvated electron and •H (Nickelsen et al. 2002).
In other examples concerning radiolytic decomposition of the herbicide MCPA by γ-irradiation (Bojanowska-Czajka et al. 2007), experimental data were compared with kinetic modeling at different pHs, and in the presence of varying H2O2 concentrations, to enhance •OH radical formation. While satisfactory results were obtained for MCPA concentrations changes under various irradiation conditions, the same model could not fully explain concentration changes of 4-chloro-2-methylphenol, the main by-product of the herbicide’s decomposition.
As an example of real wastewater matrix, industrial effluent from MCPA production can be considered: one of the main components of this waste is chloride (~ 90 g/L), which can act as scavenger of •OH radicals, as previously shown in Table 6. Yields of MCPA decomposition in pure aqueous synthetic solution, in oxidative conditions, were compared with those of the real industrial waste solutions containing chloride (Fig. 1). Comparison of the removal values observed for the synthetic solution and real wastewater, both containing the same chloride concentration, under equal irradiation and operating conditions, indicates an evident decomposition yield decrease in the complex matrix.
MCPA concentrations in 750 μM solutions after γ-irradiation with different doses and chemical conditions. a Synthetic aqueous solution. b Industrial wastewater from MCPA production, both with 90 g/L chloride. X-axis legend—A: pH 7, aerated; B: pH 0.5, aerated; C: pH 0.5, aerated, containing 4.8 mM H2O2; D: pH 7, aerated, containing 4.8 mM H2O2
Especially large yield differences, measured by d and D 0.9 values, separated even by an order of magnitude, have been obtained during irradiation of real industrial wastes. The most favorable conditions for radiolytic treatment of such wastes were generally found to be acidic conditions (as often observed in raw effluents) and stoichiometric addition of H2O2. In such cases, the decomposition yield can be just about half that of a synthetic solutions, but still close to the same order of magnitude. Explanation of the strong effect of pH on radiolysis in strongly oxidative conditions requires further study.
EB technology: state-of-the-art
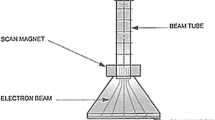
Electron beam is a process involving the use of electrons accelerated with high energy to treat an object, or medium, for a variety of purposes. The basic components of a typical electron beam device are schematized in Fig. 2: in a sealed device kept under high vacuum, a heated emitter (cathode) releases electrons that are then accelerated by a grid, using high-voltage power supply (DC) or radiofrequency (in the more compact designs). Electrostatic and/or magnetic fields control beam propagation (focusing and deflection) towards the exit window. Electrons emerge from the window with an energy proportional to the voltage applied to the anode and in quantity depending on cathodic current. By adjusting these parameters, it is possible to control, respectively, the penetration of the beam (proportional to energy) and the dose-rate (proportional to current). EB machines used in the water and wastewater treatment field are usually energetically rated between 600 keV and 1.5 MeV, although accelerators up to 10 MeV have been used (Wang et al. 2016; He et al. 2014). As a reference number, a 1-kGy dose, such as those commonly used to achieve wastewater disinfection, is a relatively low value, in the low range of those commonly used, for example, in food distribution industry applications (irradiation of fresh vegetables for fungi and bacteria control, in order to increase shelf life) (Cleland 2005). The generation of an electron beam with direct high voltage allows to achieve conversion of input AC power to beam power with efficiencies greater than 95%, compared to the approximate 30% efficiency of the equivalent transformation achieved in UV lamps, thus making EB irradiation a highly energy-efficient process.
In water-related applications, EB irradiation takes advantage of special characteristics not available in most other treatment technologies: absence of need for chemical additives (although some, such as O3 and H2O2, could be used in small quantities to further enhance radicals production), capability of generating at the same time both strong oxidants and reducers in the irradiated water (superoxides, a.k.a. hyperoxides, O2 −, hydroxyl radicals, •HO, hydrogen atoms, H, and solvated electrons, e− (aq)) that carry out the degradation of the pollutants, potentially until complete mineralization, or to a desired degree of decomposition, compatible with other conventional processes (Fig. 3). All the generated radical species are very reactive and very short-lived, with a half-life in the order of 10 μs at 10−4 M concentration, and have much stronger oxidation potentials, as reported in the “Fundamentals of radiolytic decomposition of pollutants” section, than O3 (+ 2.07 V), H2O2 (+ 1.77 V), and chlorine (+ 1.36 V). The reactions that occur with EB irradiation are quite similar to those occurring in AOPs, where oxidation is largely brought about by hydroxyl-radicals, but extremely more intense and rapid due to greater density of different radical species.
These reactions do not introduce any possibility whatsoever of an eventual secondary environmental contamination, since no residual radiation content will remain in the irradiated medium after treatment, nor the electron accelerator assembly will retain residual radioactivity in its components. All degradative processes are purely based on the high reactivity of all the above mentioned, short-lived species formed by radiolysis. Water-borne contaminants degradation reactions are practically instantaneous (occurring in the order of milliseconds), with intensity and end-point controlled by the given irradiation dose, therefore they do not require large reaction vessels to sustain prolonged exposure times, like traditional AOPs, where process contact times can be timed in the order of hours. Generated radical species will revert back very quickly (in the order of few milliseconds) to their original water state, if they do not react immediately with pollutant molecules; therefore, no residues or radioactivity are left in the water. Furthermore, EB accelerators are usually designed and supplied with built-in shielding to avoid escape of high-energy particles, and as such are commonly used in low-security industrial facilities dedicated to standard production processes, with occupational safety risks much lower than processes involving highly reactive liquid or gases.
For practical purposes, therefore, these processes can be considered a safe, highly efficient alternative to traditional AOPs with none (or few) of the potential shortcomings related to the latter.
Delivery of EB for water treatment
A practical limitation to overcome for the use of EB-based radiolytic processes is the limited penetration of EB in water, about 5 cm for 10 MeV electron accelerators (Getoff 2002), that may reduce to a centimeter or less for 1.5 MeV facilities (Kurucz et al. 1995b). This creates the need for design of special devices to allow irradiation of continuously flowing solutions. Some examples of possible irradiation chambers are shown in Fig. 4. These include the weir/cascade-delivery system, employed first in the Miami research facility (Fig. 4a) (Kurucz et al. 1995b), and subsequently adopted in many other contemporary facilities, the up-flow stream irradiation shown developed in Brazil (Fig. 4b) (Rela et al. 2000; Duarte et al. 2002), and the nozzle-type injector, initially developed to deliver wastewater sprays in purification facilities for molasses distillery slops (Fig. 4c) (Pikaev et al. 2001a, b).
These designs allow fast-rate wastewater streams treatment: in the mentioned Miami EB facility, extensive pilot scale treatment of water and wastewater was conducted at rates up to 500 L/min (Kurucz et al. 1995b). In case of the up-flow stream installation, continuous treatment was achieved at a flow rate of 700 m3/h using a 60-kW, 1.5 MeV electron beam delivering a dose of 2 kGy (Rela et al. 2000). It should be again remembered that, by comparison, commonly used AOPs demand much longer reaction times. Mobile EB systems have also being designed and built since the 1990s for special field applications in USA (Nickelsen et al. 1998; Cooper et al. 2002) and South Korea (Kim et al. 2012a, b). These consist of self-contained modular facilities that can be transported to a particular location for in situ remediation.
Case studies of ionizing radiation application for wastewater treatment
The earliest applications of radiolysis in wastewater treatment were primarily focused on the disinfection of effluents of different origin, particularly municipal wastes (Farooq et al. 1993; Woodbridge et al. 1972; Trump et al. 1977). Studies also emphasized changes in toxicity of effluents as a result of the treatment itself (Thompson and Blatchley 1999; Moraes et al. 2004). In a pilot study equipped with a 60Co irradiator, it was demonstrated that γ-irradiation was capable of achieving processing of secondary effluent to produce usable water from raw sewage (Woodbridge et al. 1972). The treatment proved to be efficient not only for killing viruses and bacteria but also for reducing both bio- and non-biodegradable detergent concentrations. It was also observed that effluent from the full-scale secondary treatment plant supported widespread algal growth, while the γ-irradiated effluent fraction remained algae-free.
In several works on disinfection of sewage wastewater, it was concluded that EB treatment is a valuable alternative to chlorination of municipal liquid wastes (Trump et al. 1977). Comparison of EB treatment at a large-scale plant and γ-irradiation at the laboratory scale for disinfection of raw and secondary wastewater effluent found that, while γ-irradiation resulted in a somewhat better inactivation of different microorganisms (Farooq et al. 1993), the difference between the two processes was less than one order of magnitude. γ-Irradiation of municipal wastewater and chlorination were also compared in terms of toxicity outcomes using the C. dubia chronic toxicity test (Thompson and Blatchley 1999). It was shown that γ-irradiation induced significantly less toxicity than chlorination. With EB irradiation of industrial effluents at 20 kGy absorbed dose, 70 and 73% toxicity removals were observed respectively for Vibrio fisheri and Daphnia similis, (Moraes et al. 2004), while 60–100% removal of anionic surfactants present in a 0.7- to 11.6-mg/L concentration range was observed.
Recently it was demonstrated that EB irradiation of both synthetic and real industrial effluents can also result in removal of numerous inorganic components, including heavy metal ions (Ribeiro et al. 2004). This, however requires rather high absorbed doses (Table 7): with absorbed dose of 20 kGy, more than 96% of Cr, Fe, Zn, and Co were removed, from original g/L concentration levels in a variety of effluents, by reduction to insoluble forms. Cadmium seems less susceptible to precipitation by this process, as in a case of 15 mg/L Cd content in simulated wastewater only 44% of the metal ion was removed, at a high 500 kGy dose. Removal of bromate (Wang et al. 2016) and nitrates (Guo et al. 2008a) from solution by irradiation processes was also reported. Additionally, Buxton et al. (1987) provide hundreds of rate constants for reactions of products of water radiolysis with inorganic species. This indicates how many different inorganic compounds may be also decomposed during irradiation of waster or wastewater.
Radiolysis treatment has been subsequently studied in an increasing number of applications to wastewaters of different origin. Studies conducted in Japan on the application of γ-irradiation to landfill leachates showed that radiolytic processing leads to considerable biodegradability improvement of main organic waste components (Sawai et al. 1981). Studies on radio-oxidation of phenol in petrochemical wastewater showed that in case of real wastewater, degradation of phenols content may be substantially lower than in synthetic solutions (Macesek et al. 1995). The advantage of EB treatment application to cellulosic wastewater was demonstrated by the acceleration of cellulose enzymatic hydrolysis rate after irradiation (Kumakura and Kaetsu 1984). When applied to pulp mill effluents, application of γ-irradiation essentially enhanced chlorinated organic compounds decomposition. These are formed during pulp bleaching with chlorine, or chlorine dioxide, and irradiation increased the removal of adsorbable organic halogen (AOX) from 50% (biological treatment only) to 96% (Taghipour and Evans 1996).
Application of electron beam to paper mill wastewater, in combination with conventional methods (coagulation, flocculation, and biological) and ozonation, showed removal of COD in the effluent to improved degrees, depending on configuration of the tertiary treatment system (Shin et al. 2002). Figure 5a shows an example of paper mill treatment system configuration including EB irradiation, with indication of BOD and COD values at specific stages. Such configuration not only results in > 98% COD removal (> 99% as BOD) but allows 70–80% effluent recirculation within the industrial process. Alternatives for different tertiary treatment setups, with achievable final TOC levels, are illustrated in Fig. 5b. The best results are obtained by those setups including either EB irradiation or irradiation with ozone addition (EB absorbed dose around 1 kGy).
Several applications of radiation processes for treatment of textile industry wastewaters are reported. Combined EB irradiation and biological treatment of dyeing, complex wastewater showed that the decrease in total pollutants content in the effluent could mainly be attributed to the instantaneous radiolytic conversion of terephthalic acid, a main organic pollutant contained in the specific wastewater (Han et al. 2002). Equal purification levels without irradiation could be obtained after 17 h of biodegradative processes, against 8 h of combined EB/biological treatment with preliminary irradiation at 10,000 m3/day (Han et al. 2012). Previous experiments in the same field involved application of γ-irradiation prior to coagulation in treatment of textile wastewater, essentially improving reduction of COD and TOC values (Perkowski and Kos 1988).
Figure 6 shows possible examples of combined EB/ozone installation for waste water treatment and EB/ozone installation for treatment carried in aerosol flow (Gehringer and Fiedler 1998; Pikaev et al. 1997).
The suitability of EB treatment to urban sewage treatment plant effluents was also examined, the main interest in this case was focused on the efficiency of radiolytic decomposition of multiclass surfactants (Petrovic et al. 2007). It was found that the application of relatively low irradiation doses (2–3 kGy) could decompose the whole spectrum of alkylphenolic compounds with potential estrogenic properties, simultaneously removing ionic and non-ionic surfactants from the effluent that may cause biological process impairments (Callegari and Capodaglio 2017). In EB irradiation of domestic wastewater for disinfection purposes, it has been reported that a low absorbed dose (1 kGy) is sufficient to eliminate all present pathogens (Trump et al. 1977).
Since radiolytic processes have the advantage of being easily implemented at a specific section of an existing treatment train, without requiring large storage vessels for the completion of the reactions (which are, as mentioned, practically instantaneous), a significant trend of combining radiolysis-based degradation processes for particularly resistant/refractory pollutants with other, more conventional treatments has been observed from the mid-1990s. This involved processes such as like coagulation and flocculation (Pikaev et al. 2001a, b; Bao et al. 2002), ozonation (Gehringer et al. 1995, 2006), biodegradation (Han et al. 2002, 2012; Rawat and Sarma 2013), UV-irradiation with suspended TiO2 catalyst (Zaki and El-Gendy 2014), and electro-oxidation supporting γ-irradiation (Barrera-Diaz et al. 2003).
In most cases, irradiation alone could very likely have achieved the complete mineralization of the target compounds; however, the main advantage of such combination systems is the possibility of working with low absorbed doses, sufficient for degradation of complex pollutants to species that can be then post-processed with conventional methods, resulting in reduction of the radiation treatment cost (smaller irradiation apparatus, lower energy requirements) and most often, of the overall treatment cost, even compared to conventional methods alone. Several reports on multistep treatment processes that include radiolytic step are presented in the literature (Shin et al. 2002; Thill et al. 2016).
Enhanced efficiency of EB irradiation combined with ozonation, leading to the formation of additional amounts of •OH radicals from the decomposition of ozone (Gehringer et al. 1993), was demonstrated in the treatment of high-load wastewater from molasses processing, where a preliminary treatment cost estimated was given as 3.04 USD/m3 to achieve an effluent level of BOD not exceeding 20 mg/L (Gehringer and Fiedler 1998). The same combination was applied also in an aerosol-flow facility for treatment of municipal wastewater, where the greater effectiveness was also attributed to the additional generation of hydroxyl radicals from ozone and only partly to direct pollutants reactions with ozone (Pikaev et al. 1997).
In disinfection applications, EB can be efficiently complemented by microwave irradiation, as demonstrated by irradiating sewage sludge from a food industry wastewater treatment plant (Martin et al. 2005). Very recently, a sequential EB irradiation, biotreatment, nanoscale zero-valent iron combined system was successfully employed for treatment of toxic recalcitrant metalworking wastewater, and was evaluated as a realistic, and potentially economic advantageous, option (Thill et al. 2016). As illustrated in Fig. 7, pre-treatment of pristine and exhausted metalworking fluids with EB irradiation allow for more efficient subsequent biological degradation, as monitored by COD removal.
In particular cases of environmental hazard, where wastewater or polluted groundwater must be treated onsite or transport of polluted liquids to stationary installations is overly hazardous or economically unfeasible, mobile-type accelerator has been built and employed for short-term EB irradiation treatment of limited amounts of liquid wastes (Cooper et al. 2002; Kim et al. 2012a, b). Table 8 summarizes recent data on EB technology applications for the treatment of wastewaters of different origin in various systems.
Economic aspects of EB treatment of water and wastewater
When considering the adoption of well established, widely used conventional methods, or other emerging technologies for wastewater treatment, one of the most relevant aspects in the evaluation of their application feasibility is their cost-effectiveness. The cost of currently established technologies is known to a fairly precise degree through empirical data, while costs of emerging technologies may be subject to unexpected or unaccounted factors. Although still at their industrial infancy, several attempts of estimating cost factors for water radiolytic technologies have been reported in the last two decades.
Perhaps the main issue concerning EB technology (at the moment, the most easily applicable among irradiation technologies) is associated with capital cost requirements for the accelerator itself, which are estimated somewhere between 1700 and 5000 kUSD for industrial scale applications including cost of accelerator, auxiliary equipment, transport, construction, and installation (Table 9). The cost of the accelerator needed for a specific application depends on both dose (D x [kGy]) necessary to degrade pollutants by the required (x) percentage, and flow rate (throughput capacity) of the wastewater (W [kg/h]), and increases with the increase of either, leading to the selection of accelerators with higher beam power (P [kW]) according to equation (Gehringer et al. 1995):
where η is the beam utilization factor, evaluated dosimetrically with value usually between 0.6 and 0.7.
An essential feature of EB accelerators is the high conversion efficiency of electric power to EB power, which for modern accelerators is in excess of 95%. In the case of UV lamps, for comparison, this is usually 20–30%, at most. An additional advantageous feature of EB devices is the extremely high dose-rate achievable, reaching the order of MGy/min, thus requiring very low irradiation times (in the order of fractions of a second). This eliminates the need for large reaction vessels and requires essentially a fast flow-through installation. Other essential factors are operational costs, including fixed costs of interest and depreciation, the variable costs of electricity, labor, maintenance, etc. (Han et al. 2012).
Some examples of cost-efficiency evaluation for different reported full-size installations, expressed as the cost of treatment per cubic meter of wastewater, are listed in Table 9. In case of installation for the treatment of textile dyeing wastewater, involving a three-window accelerator (1 MeV, 400 kW), with 1 kGy dose irradiation, followed by biodegradation, the overall cost of treatment was evaluated as 0.3 USD/m3 at a capacity of 10,000 m3/day (Han et al. 2012). This is comparable to the reported cost of 0.25 USD/m3 for groundwater treatment in an earlier installation with accelerator of 0.5 MeV and 40 kW beam power at a flow 146 m3/h (Gehringer et al. 1995). Application in the same facility of simultaneous ozonation of the irradiated groundwater solution at a treatment capacity increased to 1200 m3/h, resulted in significant cost reduction, down to 0.075 USD/m3.
An EB/O3 system, with beam power of 75 kW, was applied at a 50 m3/h facility for molasses processing wastewater treatment, with 70% COD reduction at absorbed dose of 2.7 kGy (Gehringer and Fiedler 1998). In this case, the cost of treatment was 3.17 USD/m3, comparable to conventional ozone/biological treatment for this type of waste. Shin et al. (2002) compared the overall cost of EB treatment of paper mill wastewater with other existing “traditional” technologies: estimated EB cost (1.03 USD/m3) was lower than those of the three other technologies considered, namely, use of activated carbon filter, including secondary coagulation (1.22 USD/m3), reverse osmosis (1.67 USD/m3), and evaporation (over 3.0 USD/m3). From these reported examples, it seems that EB could be in fact economically competitive in most cases to equivalent traditional processes. More specific current information about process costs, and especially about accelerator costs, should be necessary to draw more definitive conclusions. It should be noted that, in recent years, due to ongoing widespread application of EB technology in various industrial sectors (e.g., medical products, cables, new materials, food distribution and conservation), the market availability of these devices has improved, and their costs have diminished.
An indirect method that can be used for efficiency comparison of different wastewater treatment processing options is the electrical energy per order (EE/O) figure-of-merit, defined as the electrical energy (kWh) required to reduce the concentration of a pollutant by an order of magnitude in 1 m3 of water (Bolton et al. 1998). Although not directly related with process costs (it does not include investment costs, only O&M), it can provide a good idea about the possible feasibility of an alternative process approach.
Considering, for example, a process involving bleaching of methylene blue and phenol decay, EE/O values were calculated and compared for EB and UV/H2O2 and UV/TiO2 AOPs. Values < 3 were reported for EB and UV/H2O2, while for the UV/TiO2 process, the result was above 50. This can be attributed to the EB process efficiency of •OH radicals generation, which was estimated at 1.0 M/kWh, while process efficiency is 1.4 M/kWh for UV/H2O2 and 0.087 for UV/TiO2 (respectively, about + 40% and − 91%) (Bolton et al. 1998). Recently, similar results were reported considering the decomposition of two antibiotics in synthetic aqueous solutions, sulfamethazole (SMX), and chlortetracycline (CTCN) (Kim et al. 2012a, b). Energy consumption EE/O parameters were compared for an EB process carried out with 1 MeV, 40 kW accelerator, ozonation, and UV irradiation at 210 nm (UV/H2O2). Obtained values are summarized in Table 10, showing that among investigated AOPs, EB treatment is energetically much more efficient than either ozonation and UV irradiation. Table 11 summarizes characteristic reaction values in decomposition processes by ionizing radiation for carbamazepine (CBZ) present in synthetic solutions, spiked water, and wastewaters of different origin. CBZ is one of the most persistent and more difficult to degrade organics; therefore, the low values of the dose necessary for 90% destruction should be noted.
These results demonstrate that EB technology could be a competitive choice in AOP selection. It can be obviously concluded that, when choosing the optimal type of AOP, factors like matrix chemicals’ content and capital, construction, and maintenance costs should all be considered in the balance.
Discussion and conclusions
Inefficiency and inadequate management of water systems are nowadays fundamental issues for human water security and ecosystem sustainability (Grant et al. 2012; Capodaglio and Callegari 2015). Better management of available resources and more advanced technology to eliminate new classes of pollutants are needed (Shi et al. 2012). Among pollutant removal processes in water and wastewater purification systems, the last decades brought significant development of processes based on free radicals’ efficient reactions, Advanced Oxidation Processes, many of which are based on the reactivity properties of hydroxyl radicals •OH. Strangely enough, one of the most efficient ways for production of such radicals, water radiolysis by means of ionizing radiation, is often not even mentioned in extensive and comprehensive reviews of the field (e.g., Gligorovski et al. 2015) nor is much diffused in current practice. The effectiveness of aqueous radiolytic decomposition carried out through the use of ionizing radiation has been already and repeatedly demonstrated for numerous groups of organic compounds, including many hazardous anthropogenic environmental pollutants, in pilot and full-scale facilities for the treatment of wastes of different origin.
A unique advantage of radiolysis, compared to other AOP methods is the possibility of carrying out simultaneous decomposition both on oxidative and reductive pathways. Additionally, radiolysis does not require per se the use of additional reagents, although in some cases, the simultaneous presence of some chemicals, supporting increased production of active radicals, may enhance its yield and improve cost-efficiency. Another important advantage of EB irradiation, compared to other AOPs, is the kinetic rate of radiolytic processes (occurring in fractions of second), un-achievable with other methods, resulting from the elevated dose-rates achievable (e.g., for 1 MeV, 40 kW accelerator, the dose-rate is 40 kGy/s) (Kim et al. 2011). In addition, the technology does not exhibit any danger of inducing radioactivity in the irradiated media, and can be considered as a “clean technology,” as it can achieve complete mineralization of target compounds, without unexpected by-products, producing no organic sludge or air emissions.
Successful attempts on kinetic modeling of radiolytic reactions to obtain a quantitative prediction of the radiolytic removal yield of single compounds in pure aqueous solutions, as a function of their initial concentration and absorbed dose have been reported, and can be used to approximate the simulation of actual processes in real conditions. Applications indicate that when radiation processes are carried out in complex, heavy loaded wastewater matrices, they require only a few-folds increase of absorbed radiation dose due to the presence of radical scavengers and other interfering compounds, generating side reactions in the waste stream. Despite the demonstration of their numerous advantages, including cost efficiency, barriers remain for the adoption of this technology: the perceived high capital requirements in comparison to other instrumentally simpler AOP methods (although comprehensive cost analyses may prove otherwise) and often irrational fear of a nuclear-based technology (although these irradiation processes do not induce residual radioactivity and are often use on foodstuffs for human consumption).
As the recent two decades brought increased knowledge about the potential dangers of numerous classes of environmental pollutants (industrial, residues of pharmaceuticals, endocrine disruptors etc.), many of which are strongly resistant and are not eliminated completely by conventional methods from water and wastewater (Capodaglio et al. 2010; Kleywegt et al. 2011; Köck-Schulmeyer et al. 2013), there is an authentic need for especially efficient, non-selective purification methods. This may results in new development of hybrid installations, in which a particularly efficient additional step could consist in the use of EB irradiation, combined with conventional physicochemical and biological treatment, in order to achieve overall more effective pollutants removal. In such circumstances, radiolytic processes can occur at much lower doses than standalone processes, as shown in several applications. This may also encourage new future applications for the improvement of environmental protection, such as purpose-specific design of wastewater treatment process trains, enhancement of water reuse and reutilization possibilities, and better energy efficiency in the wastewater field. Appropriately selected radiation chemistry methods, and in particular EB technology, could constitute an important tassel of such a strategy.
Notes
Electron-volt (eV) (1 eV = 1.60217662e−19 J), by definition, is the amount of energy gained (or lost) by the charge of a single electron moving across an electric potential difference of 1 V. Not a SI unit, it is, however, commonly used in nuclear, radiation, and particle physics as a measure of energy, which follows the metric convention for magnitudes (e.g., 1 keV = 1000 eV).
The Gray [Gy] is a SI unit defined as the absorption of 1 J of radiation energy per kilogram of matter. It is used as a measure of absorbed dose and imparted specific energy. It is a physical quantity, does not take into account biological contexts, unlike its non-SI predecessor, the roentgen [R], measuring exposure, and the Sievert [Sv], measuring a dose equivalent. Its corresponding cgs unit, the rad (1 rad = 0.01 Gy), is still used occasionally in USA-originated literature.
References
Abdel Daiem MM, Rivera-Utrilla J, Ocampo-Perez R, Sanchez-Polo M, Lopez-Penalver JJ (2013) Treatment of water contaminated with diphenolic acid by gamma radiation in the presence of different compounds. Chem Eng 219:371–379
Abdou LAW, Hakeim OA, Mahmoud MS, El-Naggar AM (2011) Comparative study between the efficiency of electron beam and gamma irradiation for treatment of dye solutions. Chem Eng J 168:752–758
Adams C, Wang Y, Loftin K, Meyer M (2002) Removal of antibiotics from surface and distilled water in conventional water treatment process. J Environ Eng 128:253–260
Bae BU, Jung ES, Kim YK, Shin HS (1999) Treatment of landfill leachate using activated sludge process and electron-beam radiation. Water Res 33:2669–2673
Bao H, Liu Y, Jia H (2002) A study of irradiation in the treatment of wastewater. Radiat Phys Chem 63:633–636
Barrera-Diaz C, Urena-Nunuze F, Campos E, Palomar-Pardave M, Romero-Romo M (2003) A combined electrochemical-irradiation treatment of highly colored and polluted industrial wastewater. Radiat Phys Chem 67:657–663
Belgiorno V, Rizzo L, Fatta D, Della Rocca C, Lofrano G, Nikolaou A, Naddeo V, Meric S (2007) Review on endocrine disrupting-emerging compounds in urban wastewater: occurrence and removal by photocatalysis and ultrasonic irradiation for wastewater reuse. Desalination 215:166–176
Biń AK, Sobera-Madej S (2012) Comparison of the advanced oxidation processes (UV, UV/H2O2 and O3) for the removal of antibiotic substances during wastewater treatment. Ozone Sci Eng 34(2):136–139
Bojanowska-Czajka A, Drzewicz D, Kozyra C, Nałęcz-Jawecki G, Sawicki J, Szostek B, Trojanowicz M (2006) Radiolytic degradation of herbicide 4-chloro-2-methyl phenoxyacetic acid (MCPA) by γ-radiation for environmental protection. Ecotoxicol Environ Saf 65:265–277
Bojanowska-Czajka A, Drzewicz P, Zimek Z, Nichipor H, Nałęcz-Jawecki G, Sawicki J, Kozyra C, Trojanowicz M (2007) Radiolytic degradation of pesticide 4-chloro-2-methyl-phenoxyacetic acid (MCPA)—experimental data and kinetic modelling. Radiat Phys Chem 76:1806–1814
Bojanowska-Czajka A, Kciuk G, Gumiela M, Borowiecka S, Nałęcz-Jawecki G, Koc A, Garcia-Reyes JF, Solpan Ozbay D, Trojanowicz M (2015) Analytical, toxicological and kinetic investigation of decomposition of the drug diclofenac in waters and wastes using gamma radiation. Environ Sci Pollut Res 22:20255–20270
Bolong N, Ismail AF, Salim MR, Matsuura T (2009) A review of the effects of emerging contaminants in wastewater and options for their removal. Desalination 239:229–246
Bolton JR, Valladares JE, Zanin JP, Cooper WJ, Nickelson MG, Kajdi DC, Waite TD, Kurucz CN (1998) Figures-of-merit for advanced oxidation technologies: a comparison of homogeneous UV/H2O2, heterogeneous UV/TiO2 and electron beam processes. J Adv Oxid Technol 3:174–181
Borrely SI, Morais AV, Ros JM, Badaro-Perdroso X, Pereira MC, Higa MC (2016) Decoloration and detoxification of effluents by ionizing radiation. Radiat Phys Chem 124:198–202
Brusentseva SA, Makarenko ZN, Dolin PI (1978) Radiation decoloration of solutions of humic substances. High Energy Chem 12:189–192
Buxton GV, Greenstock CL, Helman WP, Ross AB (1987) Critical review of rate constants for reaction of hydrated electrons, hydrogen atoms and hydroxyl radical (•OH/H•-) in aqueous solution. J Phys Chem Ref Data 17:512–887
Callegari A, Capodaglio AG (2017) Effects of selected industrial pollutants on urban WWTPs activated sludge population, and possible mitigation strategies. Water Pract Technol 12(3). doi:10.2166/wpt.2017.064
Callegari A, Boguniewicz-Zablocka J, Capodaglio AG (2017) Experimental application of an advanced separation process for NOM removal from surface drinking water supply. (in press)
Cao DM, Zhang XH, Zhao SY, Guan Y, Zhang HQ (2011) Appropriate dose for degradation of levofloxacin lactate: gamma radiolysis and assessment of degradation product activity and cytotoxicity. Environ Eng Sci 28:183–189
Capodaglio AG (2016a) In-stream detection of waterborne priority pollutants, and applications in drinking water contaminant warning systems. Water Sci Technol Water Supply 17(3):707-725. doi:10.2166/ws.2016.168
Capodaglio AG (2016b) High energy radiation treatment for emerging and refractory contaminants in water and wastewater. Proceedings 13th IWA Leading Edge Conference on Water and Wastewater Technologies, June 13-16 Jerez de la Frontera, Spain
Capodaglio AG, Callegari A (2015) Water supply systems security: novel technologies for the online monitoring of unforeseeable events. WIT Trans Built Environ 151:251–263
Capodaglio AG, Suidan M, Venosa AD, Callegari A (2010) Efficient degradation of MtBE and other gasoline-originated compounds by means of a biological reactor of novel conception: two case studies in Italy and in the USA. Water Sci Technol 61(3):807–812
Capodaglio AG, Callegari A, Molognoni D (2016) Online monitoring of priority and dangerous pollutants in natural and urban waters: a state-of-the-art review. Manag Environ Qual 27(5):507–536
Chang HS, Choo KH, Lee B, Choi J (2009) The methods of identification, analysis, and removal of endocrine disrupting compounds (EDCs) in water. J Hazard Mater 172:1–12
Chmielewski AG, Sun Y-X, Licki J, Bulka S, Kubica K, Zimek Z (2003) NOx and PAHs removal from industrial flue gas by using electron beam technology with alcohol addition. Radiat Phys Chem 67:555–560
Cleland N (2005) Industrial application of electron accelerators, presented at the CERN accelerator school, small accelerator course, Zeegse, Netherlands, 24 May to 2 June. Available online https://cas.web.cern.ch/sites/cas.web.cern.ch/files/lectures/zeegse-2005/school-2.pdf
Cooper JW, Cadavid E, Nickelsen GM, Lin K, Kurucz NC, Waite DT (1993) Removing THMs from drinking water using high-energy electron-beam irradiation. J Am Water Works Assoc 85:106–112
Cooper WJ, Curry RD, O’Shea KE (eds) (1998) Environmental applications of ionizing radiation. Wiley, N Y. p 752
Cooper WJ, Nickelsen MG, Tobien T, Mincher BJ (2001) Radiation-induced oxidation. In: Oh CH (ed) Hazardous and radioactive waste treatment technologies handbook. CRC Press, Boca Raton
Cooper WJ, Nickelsen MG, Mezyk SP, Leslie G, Tornatore PM, Hardison W, Hajali PA (2002) MTBE and priority contaminant treatment with high energy electron beam injection. Radiat Phys Chem 65:451–460
Craft TF, Eichholz GG (1971) Synergistic treatment of textile dye wastes by irradiation and oxidation. Int J Appl Radiat Isot 22:543–547
Duarte CL, Sampa MHO, Rela PR, Oikawa H, Silveira CG, Azvedo AL (2002) Advanced oxidation process by electron-beam-irradiation-induced decomposition of pollutants in industrial effluents. Radiat Phys Chem 63:647–651
Duarte CL, Geraldo LL, Junior OAP, Borrely SI, Sato IM, Sampa MHO (2004) Treatment of effluents from petroleum production by electron beam irradiation. Radiat Phys Chem 71:443–447
Dunn CG (1953) Treatment of water and sewage by ionizing radiations. Sewage Ind Waste 25:1277–1281
EPA (2016) Contaminants of emerging concern including pharmaceuticals and personal care products. US EPA website https://www.epa.gov/wqc/contaminants-emerging-concern-including-pharmaceuticals-and-personal-care-products. Accessed May 2016
Esplugas S, Bila DM, Krause LGT, Dezotti M (2007) Ozonation and advanced oxidation technologies to remove endocrine disrupting chemicals (EDCs) and pharmaceuticals and personal care products (PPCPs) in water effluents. J Hazard Mater 149:631–642
EurActive (2015) The 2013 Berlaymont Declaration on endocrine disrupters. Website http://www.euractiv.com/health/top-scientists-call-eu-action-ho-news-519969
Farooq S, Kurucz CN, Waite TD, Cooper WJ (1993) Disinfection of wastewaters: high-energy electron vs. gamma irradiation. Water Res 27:1177–1184
Gehringer P, Eschweiler H (2002) The dose rate effect with radiation processing of water—an interpretative approach. Radiat Phys Chem 65:379–386
Gehringer P, Fiedler H (1998) Design of a combined ozone/electron beam process for waste water and economic feasibility of the process. Radiat Phys Chem 32:345–349
Gehringer P, Eschweiler H, Szinovatz W, Fiedler H, Steiner R, Sonneck G (1993) Radiation-induced OH radical generation and its use for groundwater remediation. Radiat Phys Chem 42:711–714
Gehringer P, Eschweiler H, Fiedler H (1995) Ozone-electron beam treatment for groundwater remediation. Radiat Phys Chem 46:1075–1078
Gehringer P, Eschweiler H, Weiss S, Reemtsma T (2006) Decomposition of aqueous naphtha-lene-1,5-disulfonic acid by means of oxidation processes. Ozone Sci Eng 28:437–443
Getoff N (2002) Factors influencing the efficiency of radiation-induced degradation of water pollutants. Radiat Phys Chem 65:437–446
Ghatak HR (2014) Advanced oxidation processes for the treatment of biorecalcitrant organics in wastewater. Crit Rev Environ Sci Technol 44:1167–1219
Gligorovski S, Strekovski R, Barbati S, Vione D (2015) Environmental implications of hydroxyl radicals (•OH). Chem Rev 115:13051–13092
Gonzalez-Juarez JC, Jimenez-Becerril J, Cejudo-Alvarez J (2010) Degradation of 4-chloro-phenol by gamma radiation with 137Cs and X-rays. J Mex Chem Soc 54:157–159
Grant SB, Saphores J-D, Feldman DL, Hamilton AJ, Fletcher TD, Cook PLM, Stewardson M, Sanders BF, Levin LA, Ambrose RF, Deletic A, Brown R, Jiang SC, Rosso D, Cooper WJ, Marusic I (2012) Taking the “waste” out of “wastewater” for human water security and ecosystem sustainability. Science 337:681–686
Gray KA, Cleland MB (1998) Environmental radiolysis for soil and sediment treatment: a review of chemistry, design, and economic issues. J Adv Oxid Technol 3:22–36
Guo Z, Tang D, Liu X, Zheng Z (2008a) Gamma irradiation-induced Cd2+ and Pb2+ removal from different kinds of water. Radiat Phys Chem 77:1021–1026
Guo Z, Zheng Z, Gu C, Zheng Y (2008b) Gamma irradiation-induced removal of low-concentration nitrite in aqueous solution. Radiat Phys Chem 77(2008):702–707
Guo ZB, Zhou F, Zhao YF, Zhang CZ, Liu FL, Bao CX, Lin MY (2012) Gamma irradiation-induced sulfadiazine degradation and its removal mechanisms. Chem Eng J 191:256–262
Han B, Ko J, Kim J, Kim Y, Chung W, Makarov IE, Ponomarev AV, Pikaev AK (2002) Combined electron-beam and biological treatment of dyeing complex wastewater. Pilot Plant Exp Radiat Phys Chem 64:53–59
Han B, Kim JK, Kim Y, Choi JS, Jeong KY (2012) Operation of industrial-scale electron beam wastewater treatment plant. Radiat Phys Chem 81:1475–1478
He S, Wang J, Ye L, Zhang Y, Yu J (2014) Removal of diclofenac from surface water by electron beam irradiation combined with a biological aerated filter. Radiat Phys Chem. doi:10.1016/j.radphyschem.2014.05.019
Helbling DE, Hollender J, Kohler H-PE, Singer H, Fenner K (2010) High-throughput identification of microbial transformation products of organic micropollutants. Environ Sci Technol 44:6621–6627
Huber MM, Gobel A, Joss A, Hermann N, Loffler D, Mcardell AR, Siegrist H, Ternes TA, Gunten U (2005) Oxidation of pharmaceuticals during ozonation of municipal wastewater effluents: a pilot study. Environ Sci Techol 39:4290–4299
Huerta-Fontela M, Ventura F, Galceran MT (2010) Fast liquid chromatography-quadrupole-linear ion trap mass spectrometry for the analysis of pharmaceuticals and hormones in water. J Chromatogr A 1217:4212
Huerta-Fontela M, Galceran MT, Ventura F (2011) Occurrence and removal of pharmaceuticals and hormones through drinking water treatment. Water Res 45:1432–1442
IAEA (2014) Nuclear technology review. International Atomic Energy Agency, Vienna
Ikehata K, Gamal E-DM, Snyder SA (2008) Ozonation and advanced oxidation treatment of emerging organic pollutants in water and wastewater. Ozone Sci Eng 30(1):21–26
Illes E, Takacs E, Dombi A, Gajda-Schranz K, Gonter K, Wojnarovits L (2012) Radiation induced degradation of ketoprofen in dilute aqueous solution. Radiat Phys Chem 81:1479–1483
Jeaong J, Song WH, Cooper WJ, Jung J, Greaves J (2010) Degradation of tetracycline antibiotics: mechanisms and kinetic studies for advanced oxidation/reduction processes. Chemosphere 78:533–540
Jelic A, Gros M, Ginebreda A, Cespedes-Sanchez R, Ventura F, Petrovic M, Barcelo D (2011) Occurrence, partition and removal of pharmaceuticals in sewage water and sludge during wastewater treatment. Water Res 45(3):1165–1176
Kantoglu O, Ergun E (2016) Radiation induced destruction of thebaine, papaverine and noscapine in methanol. Radiat Phys Chem 124:184–190
Kim Y, Kim J, Han B (2011) Application of an electron accelerator for the treatment of waste-water from textile dyeing industries. J Korean Phys Soc 59:3489–3493
Kim SM, Kang WG, Kim JK, Han BS (2012a) Mobile type electron accelerator. US patent 8,277,738 B2
Kim T, Kim SD, Kim HY, Lim SJ, Lee M, Yu S (2012b) Degradation and toxicity assessment of sulfamethoxazole and chlorteteracycline using electron beam, ozone and UV. J Hazard Mater 227-228:237–242
Kimura A, Osawa M, Taguchi M (2012) Decomposition of persitent pharmaceuticals in wastewater by ionizing radiation. Radiat Phys Chem 81:1508–1512
Klavarioti M, Mantzavinos D, Kassinos D (2009) Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ Int 35:402–417
Kleywegt S, Pileggi V, Yang P, Hao C, Zhao X, Rocks X, Thach S, Cheung P, White-head B (2011) Pharmaceuticals, hormones and bisphenol A in untreated source and finished drinking water in Ontario, Canada—occurrence and treatment efficiency. Sci Total Environ 409:1481–1488
Köck-Schulmeyer M, Villagrasa M, Lopez de Alda M, Cespedes-Sanchez R, Ventura F, Barcelo D (2013) Occurrence and behavior of pesticides in wastewater treatment plants and their environmental impact. Sci Total Env 458-460:466–476
Kuk SH, Kim SM, Kang WG, Han B (2011) High-power accelerator for environmental applications. J Korean Phys Soc 59:3485–3488
Kumakura M, Kaetsu I (1984) Effect of electron beam current on radiation pretreatment of cellulosic wastes with electron beam accelerator. Radiat Phys Chem 23:523–527
Kurucz CN, Waite TD, Cooper WJ, Nickelsen MG (1991) High energy electron beam irradiation of water, wastewater and sludge. In: Lewins J, Becker M (eds) Advances in nuclear science and technology, vol 23. Plenum Press, New York, pp 1–43
Kurucz CN, Waire TD, Cooper WJ, Nickelsen MG (1995a) Empirical models for estimating the destruction of toxic organic compounds utilizing electron beam irradiation at full scale. Radiat Phys Chem 45:805–816
Kurucz CN, Waite TD, Cooper WJ (1995b) The Miami electron beam research facility: a large scale wastewater treatment application. Radiat Phys Chem 45:299–308
Kwon M, Yoon Y, Cho E, Jung Y, Lee BC, Paeng KJ, Kang JW (2012) Removal of iopromide and degradation characteristics in electron beam irradiation process. J Hazard Mater 227:126–134
Liu YK, Wang JL (2013) Degradation of sulfamethazine by gamma irradiation in the presence of hydrogen peroxide. J Hazard Mater 250:99–105
Liu Z, Kanjo Y, Mizutani S (2009) Removal mechanisms for endocrine disrupting compounds (EDCs) in wastewater treatment—physical means, biodegradation, and chemical advanced oxidation: a review. Sci Total Environ 407:731–748
Liu N, Wang T, Zheng M, Lei J, Tang L, Hu G, Xu G, Wu M (2015) Radiation induced degradation of antiepileptic drug primidone in aqueous solution. Chem Eng J 270(2015):66–72
Macesek F, Mikulaj V, Rajec P, Cech R, Matel L (1995) Radiation oxidation of phenol in the presence of petrochemical wastewater components. J Radioanal Nucl Chem 191:129–143
Mak FT, Zele SR, Cooper WJ, Kurucz CN, Waite TD, Nickelsen MG (1997) Kinetic modeling of carbon tetrachloride, chloroform and methylene chloride removal from aqeous solution using the electron beam process. Water Res 31:219–228
Martin DI, Margaritescu I, Cristea E, Togoe I, Ighigeanu D, Nemtanu MR, Oproiu C, Iacob N (2005) Application of accelerated electron beam and microwave irradiation to biological waste treatment. Vacuum 77:501–506
Matilainen A, Sillanpää M (2010) Removal of natural organic matter from drinking water by advanced oxidation processes. Chemosphere 80:351–0365
Mboula VM, Hequet V, Andres Y, Pastrana-Martınez LM, Dona-Rodrıguez JM, Silva AMT, Falaras P (2013) Photocatalytic degradation of endocrine disruptor compounds under simulated solar light. Water Res 47:3997–4005
Mezyk SP, Neubauer TJ, Cooper WJ, Peller JR (2007) Free-radical-induced oxidative and reductive degradation of sulfa drugs in water: absolute kinetics and efficiencies of hydroxyl radical and hydrated electron reactions. J Phys Chem A 111:9019–9024
Mincher BJ, Curry RD (2000) Considerations for choice of a kinetic fig. of merit in process radiation chemistry for waste treatment. Appl Radiat Isotop 52:189–193
Mincher BJ, Meikrantz DH, Murphy RJ, Gresham GKL, Connolly MJ (1991) Gamma-ray induced degradation of PCBs and pesticides using spent reaction fuel. Appl Radiat Isot 42:1061–1066
Moraes MCF, Romanell IMF, Sena HC, da Silva GP, Sampa MHO, Borrely SI (2004) Whole acute toxicity removal; from industrial and domestic effluents treated by electron beam radiation: emphasis on anion surfactants. Radiat Phys Chem 71:461–463
Nickelsen MG, Kajdi DC, Cooper WJ, Kurucz CN, Waite TD, Gensel F, Lorenzl H, Sparka U (1998) Field application of a mobile 20kW electron beam treatment system on contaminated groundwater and industrial wastes. In: Cooper WJ, Curry RD, O’Shea KE (eds) Environmental application of ionizing radiation. Wiley, New York, pp 451-466
Nickelsen MG, Cooper WJ, Secker DA, Rosocha LA, Kurucz CN, Waite TD (2002) Kinetic modeling and simulation of PCE and TCE removal in aqueous solutions by electron-beam irradiation. Radiat Phys Chem 65:579–587
Ocampo-Perez R, Rivera-Utrilla J, Sanchez-Polo M, Lopez-Penalver JJ, Leyva-Ramos R (2011) Degradation of antineoplastic cytarabine in aqueous solution by gamma radiation. Chem Eng J 174:1–8
Papadaki M, Emery R, Abu-Hassan M, Diaz-Bustos A, Metcalfe I, Mantzavinos D (2004) Sonocatalytic oxidation processes for the removal of contaminants containing aromatic rings from aqueous effluents. Sep Purif Technol 34:35–42
Parsons S (ed) (2004) Advanced oxidation processes for water and wastewater. IWA Publishing, London, p 368
Perkowski J, Kos L (1988) Purification of textile wastes by radiation and coagulation method (in polish). Przeg Wlok 444-446
Petrovic M, Gonzalez S, Barcelo D (2003) Analysis and removal of emerging contaminants in wastewater and drinking water. Trends Anal Chem 22(10):685–696
Petrovic M, Gehringer P, Eschweiler H, Barcelo D (2007) Radiolytic decomposition of multi-class surfactants and their biotransformation products in sewage treatment plant effluents. Chemosphere 66:114–122
Pikaev AK, Podzorova EA, Bakhtin OM (1997) Combined electron beam and ozone treatment of wastewater in the aerosol flow. Radiat Phys Chem 49:155–157
Pikaev AK, Ponomarev AV, Bludenko AV, Mini VN, Elizar’eva LM (2001a) Combined electron-beam and coagulation purification of molasses slops. Features of the method, technical and economic evaluation of large-scale facility. Radiat Phys Chem 61:81–87
Pikaev AK, Ponomarev AV, Bludenko AV, Minin VN, Elizae’eva LM (2001b) Combined electron-beam and coagulation purification of molasses distillery slops. Features of the method, technical and economic evaluation of large-scale facility. Radiat Phys Chem 61:81–87
Plumlee MH, Stanford BD, Debroux JF, Cory Hopkins D, Snyder SA (2014) Costs of advanced treatment in water reclamation. Ozone Sci Eng 36(5):485–495
Rawat KP, Sarma KSS (2013) Enhanced biodegradation of wastewater with electron beam pretreatment. Appl Radiat Isot 74:6–8
Razavi B, Song WH, Cooper WJ, Greaves J, Jeong J (2009) Free-radical-induced oxidative and reductive degradation of fibrate pharmaceuticals: kinetic studies and degradation mechanisms. J Phys Chem A 113:1287–1294
Rela PR, Sampa MHO, Duarte CL, Costa FE, Sciani V (2000) Development of an up-flow irradiation device for electron beam wastewater treatment. Radiat Phys Chem 57:657–660
Ribeiro MA, Sato IM, Duarte CL, Sampa MHO, Salvador VLR, Scapin MA (2004) Application of the electron-beam treatment for Ca, Si, P, Al, Fe, Cr, Zn, Co, As, Se, Cd and Hg removal in the simulated and actual industrial effluents. Radiat Phys Chem 71:423–426
Ribeiro AR, Nunes OC, Pereira MFR, Silva AMT (2015) An overview on the Advanced Oxidation Processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU. Environ Int 75:33–51
Roshani B, Karpel Vel Leitner N (2011) The influence of persulfate addition for the degradation of micropollutants by ionizing radiation. Chem Eng J 168:784–789
Sagi G, Kovacs K, Bersenyi A, Csay T, Takacs E, Wojanrovits L (2016) Enhancing the biological degradability of sulfamethoxazole by ionizing radiation treatment in aqueous solution. Radiat Phys Chem 124:179–183
Sakumoto A, Miyata T (1977) Treatment of waste water by a combined technique of irradiation and conventional method. Radiat Phys Chem 24:99–115
Sanchez-Polo M, Lopez-Penalver J, Prados-Joya G, Ferro-Garcia MA, Rivera-Utrilla J (2009) Gamma irradiation of pharmaceutical compounds, nitroimidazoles, as a new alternative for water treatment. Water Res 43:4028–4036
Sawai T, Sawai T, Yamazaki M (1981) The effect of γ-irradiation on the biodegradability of landfill leachate. Bull Chem Soc Jpn 54:313–314
Scapin MA, Duarte C, Sampa MHO, Sato IM (2007) Recycling of the used automotive lubricating oil by ionizing radiation process. Radiat Phys Chem 76:1899–1902
Shi H, Cheng X, Wu Q, Mu R, Ma Y (2012) Assessment and removal of emerging water contaminants. J Environ Anal Toxicol S2:003. doi:10.4172/2161-0525.S2-003
Shin H, Kim Y, Han B, Makarov IE, Ponomarev AV, Pikaev AK (2002) Application of electron beam to treatment of wastewater from papermill. Radiat Phys Chem 65:539–547
Snyder SA (2008) Occurrence, treatment, and toxicological relevance of EDCs and pharmaceuticals in water. Ozone Sci Eng 30(1):65–69
Song WH, Cooper WJ, Mezyk SP, Greaves J, Peake BM (2008) Free radical destruction of beta-blockers in aqueous solution. Environ Sci Technol 42:1256–1261
Stefan MI (2016) Advanced oxidation processes for water treatment: fundamentals and applications. IWA Publishing
Szabo L, Toth T, Homlok R, Takacs E, Wojnarovits L (2012) Radiolysis of paracetamol in dilute aqueous solution. Radiat Phys Chem 81:1503–1507
L. Szabó, J. Szabó, E. Illés, A. Kovács, Á. Belák, Cs. Mohácsi-Farkas, E. Takács, L. Wojnárovits (2017) Electron beam treatment for tackling the escalating problems of antibiotic resistance: eliminating the antimicrobial activity of wastewater matrices originating from erythromycin. Chem. Eng. J. 321:314-324.
Taghipour F, Evans GJ (1996) Radiolytic elimination of organochlorine in pulp mill effluent. Environ Sci Technol 30:1558–1564
Thill PG, Ager DK, Vojnovic B, Tesh SJ, Scott TB, Thomson IP (2016) Hybrid biolo-gical, electron beam and zero-valent nano iron treatment of recalcitrant metalworking fluids. Water Res 93:214–231
Thompson JE, Blatchley ER (1999) Toxicity effects of γ-irradiated wastewater effluents. Water Res 33:2053–2058
Tootchi L (2010) By-product formation from select pharmaceuticals during drinking water ozonation treatment. Electronic theses and dissertations. Paper 96. University of Windsor. Accessible online at http://scholar.uwindsor.ca/etd
Torun M, Gultekin O, Solpan D, Guven O (2015) Mineralization of paracetamol in aqueous solution with advanced oxidation processes. Environ Technol 36:970–982
Trebše P, Arčon I (2003) Degradation of organophosphorus compounds by X-ray irradiation. Radiat Phys Chem 67:527–530
Trump JG, Merrill EW, Wright KA (1977) Disinfection of sewage wastewater and sludge by electron treatment. Radiat Phys Chem 24:55–55
Tsydenova O, Batoev V, Batoeva A (2015) Solar-enhanced advanced oxidation processes for water treatment: simultaneous removal of pathogens and chemical pollutants. Int. J. Environ. Res. Public Health 12:9542–9561
Wang J, Wang J (2007) Application of radiation technology to sewage sludge processing: a review. J Hazard Mater 143:2–7
Wang C, Shi H, Adams CD, Gamagedara S, Stayton I, Timmon T, Ma Y (2011) Investigation of pharmaceuticals in Missouri natural and drinking water using high performance liquid chromatography-tandem mass spectrometry. Water Res 45:1818–1828
Wang L, Batchelor B, Pillai SD, Botlaguduru VSV (2016) Electron beam treatment for potable water reuse: removal of bromate and perfluorooctanoic acid. Chem Eng J 302:58–68
Westerhoff P, Yoon Y, Snyder S, Wert E (2005) Fate of endocrine-disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. Environ Sci Technol 39(17):6649-6663
Wojnarovitz L, Takacs E (2008) Irradiation treatment of azo dye containing wastewater: an overview. Radiat Phys Chem 77:225–244
Woodbridge DD, Mann LA, Garrett WR (1972) Usable water from raw sewage. Bull Environ Contam Toxicol 7:80–86
Wu Q, Shi H, Adams CD, Timmons T, Ma Y (2012) Oxidative removal of selected endocrine-disruptors and pharmaceuticals in drinking water treatment systems, and identification of degradation products of triclosan. Sci Total Environ 439:18–25
Yu SH, Lee BJ, Lee MJ, Cho IH, Chang SW (2008) Decomposition and mineralization of cefaclor by ionizing radiation: kinetics and effects of the radical scavengers. Chemosphere 71:2106–2112
Zaki AA, El-Gendy NA (2014) Removal of metal ions from wastewater using EB irradiation in combination with HA/TiO2/UV treatment. J Hazard Mater 271:275–282
Zheng M, Xu G, Pei J, He X, Xu P (2014) EB-radiolysis of carbamazepine: in pure-water with different ions and in surface water. J Radioanal Nucl Chem 302:139–142
Zona R, Solar S, Sehested K (2012) OH-radical induced degradation of 2,4,5-trichloropheno-xyacetic acid (2,4,5-T) and 4-chloro-2-methylphenoxyacetic acid (MCPA): a pulse radiolysis and gamma-radiolysis study. Radiat Phys Chem 81:152–159
Acknowledgements
This work was partly supported by a grant from the Polish National Center of Science (NCN), project OPUS 8, number 2014/15/B/ST4/04601. The authors wish to thank Prof. Krzysztof Bobrowski (INCT, Warsaw) for valuable discussion and help in editing the final version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Vítor Pais Vilar
Rights and permissions
About this article
Cite this article
Trojanowicz, M., Bojanowska-Czajka, A. & Capodaglio, A.G. Can radiation chemistry supply a highly efficient AO(R)P process for organics removal from drinking and waste water? A review. Environ Sci Pollut Res 24, 20187–20208 (2017). https://doi.org/10.1007/s11356-017-9836-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9836-1