Abstract
Advanced oxidation processes (AOPs) are based on the in situ production of hydroxyl radicals (•OH) and reactive oxygen species (ROS) in water upon irradiation of the sample by UV light, ultrasound, electromagnetic radiation, and/or the addition of ozone or a semiconductor. Diclofenac (DCF), one of the emerging organic contaminants (EOC), is of environmental concern due to its abundancy in water and is known to be subjected to AOPs. The current study uses density functional theory (DFT) to elucidate the mechanisms of the reactions between •OH and DCF leading to degradation by-products, P1-P9. The initial encounter of DCF with •OH is proposed to lead to either the abstraction of a hydrogen or the addition of the hydroxyl radical to the molecule. The results showed that OH addition radicals (R add) are both kinetically and thermodynamically favored over H abstraction radicals (R abs). The intermediate radicals give degradation by-products by subsequent reactions. The by-products P7 and P8 are easily formed in agreement with experimental findings. Finally, acute toxicities at three trophic levels are estimated with the Ecological Structure Activity Relationships program. DCF and most of the by-products were found to be harmful to aquatic organisms, P9 being the only by-product that is not harmful at all three trophic levels.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diclofenac, (2-[2′,6′-dichlorophenyl)amino] phenylacetic acid) (DCF) (Scheme 1), is widely used as a non-steroidal anti-inflammatory drug and pain-relieving analgesic in humans and domestic animals. Owing to the fact that DCF exhibits low biodegradability and is readily excreted with urine, it is frequently detected in sewage treatment plant effluents and fresh water resources with moderate levels of aquatic toxicity (Ziylan and Ince 2011). Hence, DCF has been classified as an “emerging water pollutant” due to its potential for long-term unpredictable health effects in humans. As such, a lot of research has been lately devoted to the degradation of the parent compound and its metabolites in water and advanced oxidation processes (AOPs) have been demonstrated to be viable solutions to the problem (Naddeo et al. 2009; Ravina et al. 2002; Ziylan et al. 2013). AOPs are based on the in situ production of hydroxyl radicals (•OH) and reactive oxygen species (ROS) in water upon irradiation of the sample by UV light, ultrasound, electromagnetic radiation, and/or the addition of ozone or a semiconductor. The advantages of •OH and ROS are that they are non-selective and highly reactive with a large range of organic compounds.
Reaction mechanisms for the decomposition of DCF by •OH-mediated oxidation have been proposed (Hartmann et al. 2008; Martinez et al. 2011; Perez-Estrada et al. 2005; Vogna et al. 2004; Ziylan et al. 2014), but need to be further explored. The results of previous studies show that the most common degradation intermediates are hydroxylated compounds and compounds that form upon cleavage of the C-N bond, such as dichloroaniline, dichlorophenol, and 2-indolinone. Subsequent oxidative ring cleavage was found to lead to carboxylic acid fragments via classic degradation pathways (Hartmann et al. 2008; Vogna et al. 2004). Further reaction of dichloroaniline with •OH was found to produce 4-amino-3,5-dichlorophenol, 2,6-dichloro-p-hydroquinone, and dichlorobenzene (Hartmann et al. 2008). In addition, a cyclic intermediate (1-(2,6-dichlorophenyl)-indolin-2-one) has been detected by some researchers at highly acidic conditions (Reddersen and Heberer 2003). The production of chloroanilines such as N-(2,6-dichlorophenyl-)2-aminotoluene has been reported as part of the reduction pathway during the degradation of chlorobenzene (Drijvers et al. 2000; Stavarache et al. 2002). Final products such as oxalic acid, ammonium, nitric acid, and hydrochloric acid were attributed to the mineralization process via complete cleavage of the NH bridge (Hofmann et al. 2007). More recently, it has been demonstrated that during •OH-mediated oxidation, the toxicity of a DCF sample follows an up-and-down pattern during the early stages of the reaction, but stabilizes to a non-toxic level at prolonged reaction times (Ziylan et al. 2014).
In recent years, photodegradation mechanisms, kinetics (An et al. 2015; Gao et al. 2014a; Gao et al. 2014b; Gao et al. 2016a; Gao et al. 2016b; Ren et al. 2013; Ren et al. 2012; Zeng et al. 2016), and toxicology assessments (Gao et al. 2015; Li et al. 2016) of several pollutants were explored by theoretical calculations. Sonochemical degradation of diclofenac has been investigated by several experimental methods; however, there is a little information on its degradation mechanisms (Hartmann et al. 2008; Sein et al. 2008; Vogna et al. 2004). Understanding the degradation pathway of diclofenac and the toxicological consequences of its degradation by-products is crucial for environmental studies.
The present work aims to extend and enrich our previous study (Ziylan et al. 2014) with a reaction mechanism proposed to the initial reactions of diclofenac with •OH to elucidate the mechanism of subsequent reactions yielding the most common degradation by-products reported in the literature for the sonochemical decay of the compound. The study also encompasses estimation of the acute toxicities of the intermediates to assess the relative risk of the parent compound and its oxidation by-products to the aquatic environment.
Computational methodology
Quantum chemical calculations
All computations were carried out with the Gaussian 09 program package (Frisch et al. 2009). The stationary points were located using the B3LYP/6-31+G(d) methodology (Becke 1993a; b; Domingo et al. 2007; Lee et al. 1988; Stephens et al. 1994) and characterized by frequency analysis, from which thermal corrections were obtained at 298 K. Local minima and first order saddle points were identified by the number of imaginary vibrational frequencies. Intrinsic reaction coordinate (IRC) (Gonzalez and Schlegel 1990) calculations were performed on the transition state structures to determine the connectivity of the transition state structure to the corresponding product and reactant. Conformational searches for the structures corresponding to reactants, transition states, and products were carried out with the same methodology. Energies were further refined with the MPWB1K/6-311+G(3df,2p) level of theory (Adamo and Barone 1998; Zhao and Truhlar 2004). The effect of the solvent environment was taken into account utilizing the integral equation formalism polarizable continuum model (IEFPCM) (Mennucci et al. 1997; Mennucci and Tomasi 1997), where water (ε = 78.36) was used as solvent. The stability of the radicals generated as a result of H abstraction and OH addition was calculated using Eq. (1) and Eq. (2) (An et al. 2014; Galano and Alvarez-Idaboy 2009) as:
The standard radical stabilization energies (RSE) of the generated radicals were also calculated using Eq. (3) (Coote et al. 2010; Degirmenci and Coote 2016), where ΔH is the enthalpy change of the following isodesmic reaction and R• is a carbon centered radical:
Note that when RSE is a positive number, R• is supposed to be more stable than •CH3 and if it is negative, it is less stable.
The Gibbs free energies of the fragments are utilized in order to find the most possible fragmentation sites on DCF/DCF−. The Gibbs free energies of the reactions were calculated by Eq. (4):
The basis set superposition error (BSSE) approach according to the Boys counterpoise method (Boys and Bernardi 1970; Kobko and Dannenberg 2001) was also utilized to calculate the energies for transition state structures of the initial reactions of diclofenac with •OH. The BSSE did not alter the general reaction profiles; the barriers have only increased slightly (1–2 kcal/mol). The correction being within the error margin of DFT, the BSSE-corrected results have not been included in the overall discussion of the mechanisms (Table S1).
Toxicity assessment
The acute toxicity of DCF and its oxidation by-products to green algae, daphnia, and fish were estimated in terms of EC50 (concentration of tested compound that inhibits the growth of 50% of green algae after 96 h of exposure) and LC50 (concentration of tested pollutant for 50% dead fish and daphnia after 96 and 48 h of exposures, respectively) using the Ecological Structure Activity Relationships (ECOSAR) software (ECOSAR 2014).
Results and discussion
Initial reactions of diclofenac with •OH
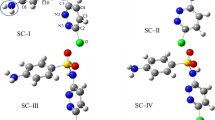
Diclofenac has two aromatic rings connected with an amine group. The previous experimental work of some of the authors show that during degradation, the solution pH decreases from 6.50 to 5.23 in 30 min and even lower as the degradation proceeds, indicating the formation of acidic intermediates (Ziylan et al. 2014). Because the medium is changing from a neutral to an acidic environment, the degradation of DCF is modeled in both its anionic and neutral forms (pKa of diclofenac is 4.1–4.5 (Scheytt et al. 2005)); their best conformers are depicted in Fig. 1. In both forms, there are strong intramolecular H-bonding interactions between the carboxylic oxygen and the amine hydrogen. As expected, these interactions are stronger in the anionic form (1.86 Å) than in the neutral form (2.08 Å) as a result of the excess negative charge on the oxygens in DCF−. Both ring systems in DCF (DCF−) are slightly twisted away from each other with a dihedral of −161.8° (164.3°) to avoid steric clashes, as seen in Fig. 1.
AOPs produce highly reactive •OH, which are the main oxidants in the degradation processes (Glaze and Kang 1989; Glaze et al. 1987). In aqueous medium, •OH can either add to diclofenac or abstract a hydrogen from it to give either addition or abstraction intermediates, respectively. In Schemes 2 and 3, the reaction sites of DCF are labeled with numbers (1–7) and letters (a, b) in order to characterize their reactivities upon H abstraction and OH addition reactions. •OH can abstract a hydrogen from the aromatic (1–6) or benzylic positions (7), leading to the formation of a radical carbon center on the DCF molecule and the elimination of water (Scheme 2).
Relative stabilities of the free radicals formed via H abstraction (R abs) during oxidation of diclofenac are summarized in Table 1. The most stable radical is the one corresponding to R abs7, which is almost 30 kcal/mol more stable than those on the benzene rings. H abstraction from the benzylic position is found to be more favorable—both thermodynamically and kinetically—when compared to H abstraction from the rings; this is expected based on the stabilization generated by conjugation of the benzylic radical in the former case. The preference of H abstraction from benzylic carbon over aromatic carbons is also reported by Sehested et al. (Sehested et al. 1975). Radicals on the ring are thermodynamically less favored than R abs7; however, their generation is still kinetically feasible in terms of their activation barriers (ΔG ‡ ~10 kcal/mol vs ~3 kcal/mol). Radicals on the second ring are more stable (RSE ~−8 kcal/mol) than those on the first ring (RSE ~−6 kcal/mol). When the two forms of diclofenac are compared, it is observed that DCF− is more prone to H abstraction than DCF with slightly lower activation barriers and higher exergonicities.
In aqueous medium, •OH can readily add to aromatic molecules producing hydroxycyclohexadienyl-type radicals (Mohan and Mittal 1999). Hence, •OH could add to DCF forming R add intermediates as shown in Scheme 3. As opposed to H abstraction, there are two additional reactive sites (a and b) for hydroxyl radical addition.
The stabilities of the R add intermediates (arenium radicals) as calculated by Eq. (2) are listed in Table 2 for DCF and DCF−. Unlike the H abstraction route, the radicals formed on the first ring are more exergonic than those on the second ring, but have slightly higher activation barriers. The addition of •OH to positions a and b is slightly less favorable than addition to the rings and the reaction is not likely since re-aromatization is not probable. However, those intermediates could lead to C-N bond cleavage and fragmentation (Gao et al. 2014a). Furthermore, similar to the H abstraction route, we found that OH addition to the anionic form of diclofenac is thermodynamically and kinetically more favorable than the neutral one. The complete reaction profiles for these two reactions are presented in Scheme 4.
The reactivity of DCF and DCF− show the same trends, nonetheless with lower energy barriers and higher exergonicities for DCF−. It is known that OH addition to benzene ring is kinetically more favored than H abstraction from the ring carbons (Song et al. 2009). Similarly, based on RSE results, one can see that OH addition radicals are more stable than •CH3; note also that the radicals generated by H abstraction on the ring carbons are less stable than the ones formed by OH addition. When the whole reaction path is examined (Scheme 4 for DCF− and Scheme S1 for DCF), the major H abstraction intermediate is found to be R abs7 and this may lead to the fragmentation of DCF/DCF−. R add2 and R add3 are the most easily formed radicals among others.
Fragmentation of DCF/DCF−
The fragmentation of DCF/DCF− is modeled in order to understand the most probable degradation paths and be able to predict the possible fates of the generated radical intermediates. All fragments are optimized and their reaction energies are calculated with Eq. (4) (Table 3). The fragmentation energies are displayed in an increasing order. Based on Hammond’s postulate, ring cleavages from either side of the nitrogen (entries 2 and 3) seem to be harder than decarboxylation (entry 1). The rupture of the C-Cl bond (entries 4 and 6) is almost equally difficult in both cases (Cl (1) and Cl (2)). On the other hand, the losses of OH (entry 7) in the neutral form and O in the anionic form are more endergonic than the others; decarboxylation is less endergonic for DCF− (50.6 kcal/mol) than for DCF (66.2 kcal/mol).
Analysis of the degradation by-products
Following the generation of radicals through H abstraction and OH addition, subsequent reactions are examined to understand the formation of the mostly encountered degradation by-products of DCF by sonolysis, namely P1-P9 (Scheme 5). The degradation mechanisms that follow are based on the outcomes of “Initial reactions of diclofenac with •OH”. Hydroxylated derivatives lead to the degradation by-products, P5 (Hartmann et al. 2008; Michael et al. 2014; Perez-Estrada et al. 2005), P6 (Hartmann et al. 2008; Vogna et al. 2004), P7, and P8 (Hartmann et al. 2008; Homlok et al. 2011; Michael et al. 2014; Perez-Estrada et al. 2005; Sein et al. 2008; Vogna et al. 2004). H abstraction intermediate R abs5 gives P4 (Encinas et al. 1998a; Encinas et al. 1998b; Lekkerkerker-Teunissen et al. 2012; Martinez et al. 2011; Poiger et al. 2001; Salgado et al. 2013). Decarboxylated fragment of DCF results in P3 (Homlok et al. 2011) by ring closure. Ring cleavage fragmentation produces P1 (Bae et al. 2013; Hartmann et al. 2008; Homlok et al. 2011; Perez-Estrada et al. 2005; Sarasidis et al. 2014; Sein et al. 2008; Vogna et al. 2004), P5, P6, and P9 (Hartmann et al. 2008; Vogna et al. 2004). The rearrangement gives cyclization product P2 (Bae et al. 2013; Hartmann et al. 2008). The mechanisms and the energetics of these products (P1-P9) are examined and displayed in Scheme 5. The detailed reaction mechanisms (Scheme S2-S10) and the reaction profiles (Fig. S1-S9) are reported in the supporting information.
Subsequent degradation mechanism of DCF involves •OH almost in every step because it is well known that high •OH concentrations are produced under extreme conditions in advanced oxidation processes (AOP) (An et al. 2010a; An et al. 2010b; Fang et al. 2013; Shu et al. 2013). Despite the high concentration of •OH, P2 and P3 are formed presumably by rearrangement of DCF before the attack of •OH (Scheme 5-I). P2 is the result of an intramolecular cyclization leading to water loss (Fig. S2, Scheme S3). Even though the activation barrier for P2 is very high (ΔG ‡ = 47.0 kcal/mol), its overall formation is exergonic (ΔG rxn = −12.7 kcal/mol). The mechanism suggested for the formation of P2 starts with cyclization and is followed by water elimination. A cyclic tetrahedral gemdiol intermediate would form by attack of nitrogen to the carbonyl carbon, and then the intermediate loses a water molecule aided by the solvent, yielding the amide P2.
Another degradation by-product that forms without •OH in its subsequent degradation mechanism is P3 (Scheme S4). The decarboxylation of DCF/DCF− is relatively facile as shown in “Fragmentation of DCF/DCF− ”. After decarboxylation, DCFdecarb is formed; the exocyclic methylene radical attacks the chlorinated carbon, ejecting a Cl• and yielding P3. The barrier for this reaction is easily overcome (ΔG ‡ = 16.7 kcal/mol) and the reaction is highly exergonic (ΔG rxn = −32.2 kcal/mol) due to the formation of the stable cyclic by-product, P3 (Fig. S3).
Besides P2 and P3, there is another cyclic degradation by-product P4, which is also encountered frequently in the degradation experiments of DCF. P4 is produced in two steps: in the first step, a radical center is generated by a H abstraction from position 3 (on ring II) and in the second step, R abs3 attacks the chlorinated carbon to form a five-membered cyclic product similar to the formation of P3 where a six-membered ring was formed (Scheme S5). The barrier to form P4 from R abs3 is 13.4 kcal/mol with very high exergonicity (ΔG rxn = −45.3 kcal/mol) (Fig. S4).
P1 was found experimentally to be formed at early stages of the reaction; furthermore, it is the precursor of the subsequent oxidative products, e.g., P5 and P6 (Ziylan et al. 2014). The C-N bond cleavage in DCF is a very common fragmentation site (Hartmann et al. 2008; Perez-Estrada et al. 2005; Vogna et al. 2004). P1 is the first by-product that forms upon cleavage of the bond between C of ring II and N (Scheme S2). This cleavage is initiated with •OH addition to position a (formation of R adda with 9.3 kcal/mol) and disruption of the aromaticity of ring II (22.6 kcal/mol) (Fig. S1). After formation of R adda, the C-N bond is broken with a proton transfer from OH to NH giving two products (ΔG rxn = −36.9). This step requires 22.6 kcal/mol energy for the anionic form. The same transition state cannot be located for neutral form of diclofenac. The final products are P1 and a radical. These first cleavage products of P1 (Fig. S1) give other degradation by-products (P5 and P6) by further reacting with •OH. •OH addition to P1 produces an arenium radical on the ring and this radical captures another •OH resulting in a diol with broken aromaticity in the ring (Scheme S6). In order to rearomatize, a water molecule is lost and hydroxylated aniline is generated, which is found experimentally as one of the degradation by-products of diclofenac, P5 with very high exergonicity (−105.9 kcal/mol relative to P1, Fig. S5). Another attack of an •OH to P5 results in substitution of OH with NH2 to give P6. •OH attacks to the carbon where NH2 is attached and forms easily (4.5 kcal/mol) an arenium radical on the ring (Scheme S7); the latter rearomatizes as the NH2 radical leaves (11.8 kcal/mol). P6 is found to be 6.7 kcal/mol more stable than P5 (Fig. S6).
P7 and P8 are considered as the main degradation by-products of DCF/DCF−. •OH adds to the ring (ΔG ‡ = 7.2 kcal/mol for P7 and ΔG ‡ = 5.7 kcal/mol for P8) forming an arenium radical which is followed by a barrierless addition of •OH (Scheme S8 and S9). The latter intermediate is expected to lose a water and yield the final product in an exergonic path (ΔG rxn = −108.4 kcal/mol for P7 and ΔG rxn = −108.5 kcal/mol for P8) (Fig. S7 and S8). Another •OH addition to position b in P8 results in the C-N bond breakage and formation of P9 (ΔG rxn = −33.6 kcal/mol). The mechanism is similar to the formation of P1 but with a lower activation barrier (ΔG ‡ = 6.8 kcal/mol) (Scheme S10, Fig. S9).
Comparison of the suggested reaction mechanisms with experimental sonolytic findings
As reported by Hartmann et al., the attack of •OH immediately leads to the formation of the hydroxylated DCF derivatives P7 and P8 (compounds 5 and 6 in Hartmann et al. (2008)). In the proposed pathway, •OH addition is followed by H2O elimination and the whole process is highly exergonic (Fig. S7 and S8). We also propose the formation of P9 (compound 20 in Hartmann et al. (2008)) by further hydroxylation of P8 followed by C-N bond cleavage.
Dichloroaniline (P1) is the main by-product observed within 30 min of sonication by Hartmann et al. (2008). The attack of hydroxyl radical leads to the formation of the hydroxylated diclofenac derivative which by attack of another •OH and water elimination leads to the formation of an intermediate (R addb). The latter leads to P1 by C-N bond cleavage. P1 is also the major by-product characterized by Ziylan et al. (compound D1) after 90 min (Ziylan et al. 2014). Note that the Gibbs free energy of activation of P1 is as low as 22.6 kcal/mol justifying its detection as the major product in both studies (Fig. S1). Hartmann et al. have claimed that the small degradation rate for P1 at low pH values (<4) is in good agreement with the sonolysis of aniline which is stable under acidic conditions (Jiang et al. 2002). The mechanism suggested in this study corroborates this finding since the formation of P1 is exothermic by 36.9 kcal/mol (Fig. S1).
The reaction of hydroxyl radical with 2,4-dichloroaniline (P1) can easily yield P5 in which the NH2 group can be further replaced by OH to yield P6. Both reactions occur easily as P1 is already formed (compounds 16 and 17 in Hartmann et al. (2008)). Ziylan et al. have identified the same species (D8, D9) after 90 min of irradiation (Ziylan et al. 2014).
The cyclic intermediate 1-(2,6-dichlorophenyl)-indolin-2-one (P2) was previously observed during sample preparation of diclofenac under acidic conditions (Reddersen and Heberer 2003). P2 was classified as an immediate by-product in the sonolysis of Hartmann et al. (compound 2) (Hartmann et al. 2008) We propose the ring closure to take place with the interference of an explicit water molecule. The activation Gibbs free energy is higher than the ones required in the other pathways (47.0 kcal/mol). However, this barrier could have been overcome under reaction conditions. The formation of the other cyclic by-products (P3 and P4) is less demanding. P3 which occurs by decarboxylation followed by cyclization has also been detected by Ziylan et al. (compound D4) (Ziylan et al. 2014).
2,6-dichloro-4-hydroxy aniline (P5) has also been reported as the by-product in the transformation pathway of DCF degradation by photocatalysis under UV-A and simulated solar irradiation and sonophotocatalyis (Ziylan et al. 2014).
In light of the experimental progression of the pH mentioned earlier (decrease from 6.50 to 5.23 in 30 min), in the late stages of the reaction when neutral DCF is the prevalent form, it is likely that energy demanding processes, e.g., fragmentation and cyclization rates, will increase. At the beginning of the AOP when pH is close to neutral and DCF− is predominant, H abstraction and OH addition are more likely to occur, leading to subsequent degradation reactions yielding by-products P1 and P4–P9.
Toxicity
The acute and chronic toxicity of diclofenac and its by-products to aquatic organisms was predicted using the ECOSAR software at three trophic levels (green algae, daphnia, and fish). The octanol–water partitioning coefficient (log K ow) was predicted as 4.02 for diclofenac, and the value is in good agreement with the experimental finding, 4.51 (Avdeef et al. 1998). As such, DCF is potentially toxic to aquatic organisms based on the literature classifying chemicals as toxic with log K ow < 6.0 (Konemann 1981; Mayer and Reichenberg 2006). The toxicity classification based on ranges of LC50 and EC50 (Table S2) was used to rationalize the acute toxicity of DCF and its by-products (Table 4). The estimated value of EC50 (41.41) is slightly higher than that found experimentally (33.26) (Ziylan et al. 2014). LC50 of diclofenac for daphnia and fish was found as 25.75 and 37.66 mg/L, respectively. The finding is in agreement with the European Union criteria (Table S2), which classify diclofenac as a harmful compound for the aquatic environment at all three trophic levels.
The aquatic toxicities upon degradation of diclofenac were tracked for three trophic levels (Table 4). As •OH-mediated degradation proceeds, toxicities fluctuate between harmful and toxic levels. Aquatic toxicities seem to be dependent on the presence of chlorine: P9, the only by-product that is classified as not harmful for all three organisms, is free of chlorine. P4, P7, and P8, which possess an acidic group, are also classified as not harmful for green algae. The toxicities of P1, P2, P3, P5, and P6 are higher than DCF/DCF− in regard to all three organisms. However, P5 is less toxic to fish (LC50 = 53.61 mg/L) and P6 is less toxic to daphnid (LC50 = 42.53 mg/L). Overall, the assessment of EC50 and LC50 shows that DCF and most of its oxidation by-products are harmful for aquatic organisms.
Conclusions
The study has highlighted a computational means of proposing the •OH-mediated reaction mechanism for the degradation of the emerging water pollutant, diclofenac (DCF/DCF−) in water. It was found that the reactions by OH addition and H abstraction occurred almost readily; the methylene hydrogens were more readily abstractable than the others, and the outcome is most likely the decarboxylation of the ring. The radical stabilization energies of the arenium radicals are higher than those of the radicals formed by H abstraction, indicating their longer lifetimes. The by-products (P1-P9) identified experimentally for the sonochemical degradation of DCF/DCF− were confirmed in this computational work. As reported previously, the intermediates, P5, P6, and P9, formed at a later stage from their precursors, P1 and P8. P1, P7, and P8 are the most readily formed by-products (Ziylan et al. 2014).
ECOSAR toxicity analysis revealed that DCF is toxic, while some of the oxidation by-products (P4, P7, P8, and P9) are less toxic, such that elimination of the compound by AOPs is a safe method as it provides less harmful products than the parent compound. This study is the first to provide a detailed mechanistic account of the degradation of diclofenac in aqueous medium by •OH-mediated reactions, complementing our previous work (Ziylan et al. 2014).
References
Adamo C, Barone V (1998) Exchange functionals with improved long-range behavior and adiabatic connection methods without adjustable parameters: the mPW and mPW1PW models. J Chem Phys 108:664–675. doi:10.1063/1.475428
An T, Gao Y, Li G, Kamat PV, Peller J, Joyce MV (2014) Kinetics and mechanism of (OH)-O-center dot mediated degradation of dimethyl phthalate in aqueous solution: experimental and theoretical studies. Environ Sci Technol 48:641–648. doi:10.1021/es404453v
An T, Yang H, Li G, Song W, Cooper WJ, Nie X (2010a) Kinetics and mechanism of advanced oxidation processes (AOPs) in degradation of ciprofloxacin in water. Appl Catal B Environ 94:288–294. doi:10.1016/j.apcatb.2009.12.002
An T, Yang H, Song W, Li G, Luo H, Cooper WJ (2010b) Mechanistic considerations for the advanced oxidation treatment of fluoroquinolone pharmaceutical compounds using TiO2 heterogeneous catalysis. J Phys Chem A 114:2569–2575. doi:10.1021/jp911349y
An TC, An JB, Gao YP, Li GY, Fang HS, Song WH (2015) Photocatalytic degradation and mineralization mechanism and toxicity assessment of antivirus drug acyclovir: Experimental and theoretical studies. Appl Catal B Environ 164:279–287. doi:10.1016/j.apcatb.2014.09.009
Avdeef A, Box KJ, Comer JEA, Hibbert C, Tam KY (1998) pH-metric logP 10. Determination of liposomal membrane-water partition coefficients of ionizable drugs. Pharm Res 15:209–215. doi:10.1023/a:1011954332221
Bae S, Kim D, Lee W (2013) Degradation of diclofenac by pyrite catalyzed Fenton oxidation. Appl Catal B Environ 134:93–102. doi:10.1016/j.apcatb.2012.12.031
Becke AD (1993a) Density-functional thermochemistry. 3. The role of exact exchange. J Chem Phys 98:5648–5652. doi:10.1063/1.464913
Becke AD (1993b) A new mixing of Hartree-Fock and local density-functional theories. J Chem Phys 98:1372–1377. doi:10.1063/1.464304
Boys SF, Bernardi F (1970) Calculation of small molecular interactions by differences of separate total energies—some procedures with reduced errors. Mol Phys 19:553. doi:10.1080/00268977000101561
Coote ML, Lin CY, Beckwith ALJ, Zavitsas AA (2010) A comparison of methods for measuring relative radical stabilities of carbon-centred radicals. Phys Chem Chem Phys 12:9597–9610. doi:10.1039/c003880f
Degirmenci I, Coote ML (2016) Comparison of thiyl, alkoxyl, and alkyl radical addition to double bonds: the unusual contrasting behavior of sulfur and oxygen radical chemistry. J Phys Chem A 120:1750–1755. doi:10.1021/acs.jpca.6b00538
Domingo LR, Aurell MJ, Arno M, Saez JA (2007) Toward an understanding of the acceleration of diels-alder reactions by a pseudo-intramolecular process achieved by molecular recognition. A DFT study. J Org Chem 72:4220–4227. doi:10.1021/jo070373j
Drijvers D, Van Langenhove H, Herrygers V (2000) Sonolysis of fluoro-, chloro-, bromo- and iodobenzene: a comparative study. Ultrason Sonochem 7:87–95. doi:10.1016/s1350-4177(99)00028-0
ECOSAR, (2014), http://www.epa.gov/oppt/newchems/tools/21ecosar.htm,
Encinas S, Bosca F, Miranda MA (1998a) Photochemistry of 2,6-dichlorodiphenylamine and 1-chlorocarbazole, the photoactive chromophores of diclofenac, meclofenamic acid and their major photoproducts. Photochem Photobiol 68:640–645. doi:10.1111/j.1751-1097.1998.tb02523.x
Encinas S, Bosca F, Miranda MA (1998b) Phototoxicity associated with diclofenac: a photophysical, photochemical, and photobiological study on the drug and its photoproducts. Chem Res Toxicol 11:946–952. doi:10.1021/tx9800708
Fang H et al (2013) Advanced oxidation kinetics and mechanism of preservative propylparaben degradation in aqueous suspension of TiO2 and risk assessment of its degradation products. Environ Sci Technol 47:2704–2712. doi:10.1021/es304898r
Frisch MJ et al (2009) Gaussian 09. Gaussian Inc., Wallingford
Galano A, Alvarez-Idaboy JR (2009) Guanosine plus OH radical reaction in aqueous solution: a reinterpretation of the UV-vis data based on thermodynamic and kinetic calculations. Org Lett 11:5114–5117. doi:10.1021/ol901862h
Gao Y, Ji Y, Li G, An T (2014a) Mechanism, kinetics and toxicity assessment of OH-initiated transformation of triclosan in aquatic environments. Water Res 49:360–370. doi:10.1016/j.watres.2013.10.027
Gao YP, An TC, Fang HS, Ji YM, Li GY (2014b) Computational consideration on advanced oxidation degradation of phenolic preservative, methylparaben, in water: mechanisms, kinetics, and toxicity assessments. J Hazard Mater 278:417–425. doi:10.1016/j.jhazmat.2014.05.081
Gao YP, An TC, Ji YM, Li GY, Zhao CY (2015) Eco-toxicity and human estrogenic exposure risks from (OH)-O-center dot-initiated photochemical transformation of four phthalates in water: a computational study. Environ Pollut 206:510–517. doi:10.1016/j.envpol.2015.08.006
Gao YP, Ji YM, Li GY, An TC (2016a) Theoretical investigation on the kinetics and mechanisms of hydroxyl radical-induced transformation of parabens and its consequences for toxicity: influence of alkyl-chain length. Water Res 91:77–85. doi:10.1016/j.watres.2015.12.056
Gao YP, Ji YM, Li GY, Mai BX, An TC (2016b) Bioaccumulation and ecotoxicity increase during indirect photochemical transformation of polycyclic musk tonalide: a modeling study. Water Res 105:47–55. doi:10.1016/j.watres.2016.08.055
Glaze WH, Kang JW (1989) Advanced oxidation processes—test of a kinetic-model for the oxidation of organic-compounds with ozone and hydrogen-peroxide in a semibatch reactor. Ind Eng Chem Res 28:1580–1587. doi:10.1021/ie00095a002
Glaze WH, Kang JW, Chapin DH (1987) The chemistry of water-treatment processes involving ozone, hydrogen-peroxide and ultraviolet-radiation. Ozone Sci Eng 9:335–352
Gonzalez C, Schlegel HB (1990) Reaction-path following in mass-weighted internal coordinates. J Phys Chem 94:5523–5527. doi:10.1021/j100377a021
Hartmann J, Bartels P, Mau U, Witter M, von Tumpling W, Hofmann J, Nietzschmann E (2008) Degradation of the drug diclofenac in water by sonolysis in presence of catalysts. Chemosphere 70:453–461. doi:10.1016/j.chemosphere.2007.06.063
Hofmann J, Freier U, Wecks M, Hohmann S (2007) Degradation of diclofenac in water by heterogeneous catalytic oxidation with H2O2. Appl Catal B Environ 70:447–451. doi:10.1016/j.apcatb.2005.11.023
Homlok R, Takacs E, Wojnarovits L (2011) Elimination of diclofenac from water using irradiation technology. Chemosphere 85:603–608. doi:10.1016/j.chemosphere.2011.06.101
Jiang Y, Petrier C, Waite TD (2002) Effect of pH on the ultrasonic degradation of ionic aromatic compounds in aqueous solution. Ultrason Sonochem 9:163–168. doi:10.1016/s1350-4177(01)00114-6
Kobko N, Dannenberg JJ (2001) Effect of basis set superposition error (BSSE) upon ab initio calculations of organic transition states. J Phys Chem A 105:1944–1950. doi:10.1021/jp001970b
Konemann H (1981) Quantitative structure-activity-relationships in fish toxicity studies .1. Relationship for 50 industrial pollutants. Toxicology 19:209–221. doi:10.1016/0300-483x(81)90130-x
Lee CT, Yang WT, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron-density. Phys Rev B 37:785–789. doi:10.1103/PhysRevB.37.785
Lekkerkerker-Teunissen K, Benotti MJ, Snyder SA, van Dijk HC (2012) Transformation of atrazine, carbamazepine, diclofenac and sulfamethoxazole by low and medium pressure UV and UV/H2O2 treatment. Sep Purif Technol 96:33–43. doi:10.1016/j.seppur.2012.04.018
Li GY, Nie X, Gao YP, An TC (2016) Can environmental pharmaceuticals be photocatalytically degraded and completely mineralized in water using g-C3N4/TiO2 under visible light irradiation?-implications of persistent toxic intermediates. Appl Catal B Environ 180:726–732. doi:10.1016/j.apcatb.2015.07.014
Martinez C, Canle M, Fernandez MI, Santaballa JA, Faria J (2011) Aqueous degradation of diclofenac by heterogeneous photocatalysis using nanostructured materials. Appl Catal B Environ 107:110–118. doi:10.1016/j.apcatb.2011.07.003
Mayer P, Reichenberg F (2006) Can highly hydrophobic organic substances cause aquatic baseline toxicity and can they contribute to mixture toxicity? Environ Toxicol Chem 25:2639–2644. doi:10.1897/06-142r.1
Mennucci B, Cances E, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics and in ionic solutions with a unified integral equation method: theoretical bases, computational implementation, and numerical applications. J Phys Chem B 101:10506–10517. doi:10.1021/jp971959k
Mennucci B, Tomasi J (1997) Continuum solvation models: a new approach to the problem of solute's charge distribution and cavity boundaries. J Chem Phys 106:5151–5158. doi:10.1063/1.473558
Michael I et al (2014) Proposed transformation pathway and evolution profile of diclofenac and ibuprofen transformation products during (sono)photocatalysis. Appl Catal B Environ 147:1015–1027. doi:10.1016/j.apcatb.2013.10.035
Mohan H, Mittal JP (1999) Pulse radiolysis investigations on acidic aqueous solutions of benzene: formation of radical cations. J Phys Chem A 103:379–383. doi:10.1021/jp983255w
Naddeo V, Belgiorno V, Ricco D, Kassinos D (2009) Degradation of diclofenac during sonolysis, ozonation and their simultaneous application. Ultrason Sonochem 16:790–794. doi:10.1016/j.ultsonch.2009.03.003
Perez-Estrada LA, Malato S, Gernjak W, Aguera A, Thurman EM, Ferrer I, Fernandez-Alba AR (2005) Photo-fenton degradation of diclofenac: identification of main intermediates and degradation pathway. Environ Sci Technol 39:8300–8306. doi:10.1021/es050794n
Poiger T, Buser HR, Muller MD (2001) Photodegradation of the pharmaceutical drug diclofenac in a lake: pathway, field measurements, and mathematical modeling. Environ Toxicol Chem 20:256–263. doi:10.1897/1551-5028(2001)020<0256:potpdd>2.0.co;2
Ravina M, Campanella L, Kiwi J (2002) Accelerated mineralization of the drug diclofenac via Fenton reactions in a concentric photo-reactor. Water Res 36:3553–3560. doi:10.1016/s0043-1354(02)00075-1
Reddersen K, Heberer T (2003) Formation of an artifact of diclofenac during acidic extraction of environmental water samples. J Chromatogr A 1011:221–226. doi:10.1016/s0021-9673(03)01173-7
Ren XH, Sun YM, Fu XW, Zhu L, Cui ZJ (2013) DFT comparison of the OH-initiated degradation mechanisms for five chlorophenoxy herbicides. J Mol Model 19:2249–2263. doi:10.1007/s00894-013-1760-9
Ren XH, Sun YM, Wu ZF, Meng FL, Cui ZJ (2012) The OH-induced degradation mechanism of 4-chloro-2-methylphenoxyacetic acid (MCPA) with two forms in the water: a DFT comparison. Chemosphere 88:39–48. doi:10.1016/j.chemosphere.2012.02.045
Salgado R et al (2013) Photodegradation kinetics and transformation products of ketoprofen, diclofenac and atenolol in pure water and treated wastewater. J Hazard Mater 244:516–527. doi:10.1016/j.jhazmat.2012.10.039
Sarasidis VC, Plakas KV, Patsios SI, Karabelas AJ (2014) Investigation of diclofenac degradation in a continuous photo-catalytic membrane reactor influence of operating parameters. Chem Eng J 239:299–311. doi:10.1016/j.cej.2013.11.026
Scheytt T, Mersmann P, Lindstadt R, Heberer T (2005) 1-octanol/water partition coefficients of 5 pharmaceuticals from human medical care: carbamazepine, clofibric acid, diclofenac, ibuprofen, and propyphenazone. Water Air Soil Pollut 165:3–11. doi:10.1007/s11270-005-3539-9
Sehested K, Christensen HC, Hart EJ, Corfitzen H (1975) Rates of reaction of O-, OH, and H with methylated benzenes in aqueous-solution—optical-spectra of radicals. J Phys Chem 79:310–315. doi:10.1021/j100571a005
Sein MM, Zedda M, Tuerk J, Schmidt TC, Golloch A, von Sonntao C (2008) Oxidation of diclofenac with ozone in aqueous solution. Environ Sci Technol 42:6656–6662. doi:10.1021/es8008612
Shu Z, Bolton JR, Belosevic M, El Din MG (2013) Photodegradation of emerging micropollutants using the medium-pressure UV/H2O2 advanced oxidation process. Water Res 47:2881–2889. doi:10.1016/j.watres.2013.02.045
Song W, Xu T, Cooper WJ, Dionysiou DD, De La Cruz AA, O'Shea KE (2009) Radiolysis studies on the destruction of microcystin-LR in aqueous solution by hydroxyl radicals. Environ Sci Technol 43:1487–1492. doi:10.1021/es802282n
Stavarache C, Yim B, Vinatoru M, Maeda Y (2002) Sonolysis of chlorobenzene in Fenton-type aqueous systems. Ultrason Sonochem 9:291–296. doi:10.1016/s1350-4177(02)00088-3
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch MJ (1994) AB-initio calculation of vibrational absorption and circular-dichroism spectra using density-functional force-fields. J Phys Chem 98:11623–11627. doi:10.1021/j100096a001
Vogna D, Marotta R, Napolitano A, Andreozzi R, d'Ischia M (2004) Advanced oxidation of the pharmaceutical drug diclofenac with UV/H2O2 and ozone. Water Res 38:414–422. doi:10.1016/j.watres.2003.09.028
Zeng XL, Zhang XL, Wang ZY (2016) Theoretical study on the OH-initiated oxidation mechanism of polyfluorinated dibenzo-p-dioxins under the atmospheric conditions. Chemosphere 144:2036–2043. doi:10.1016/j.chemosphere.2015.10.106
Zhao Y, Truhlar DG (2004) Hybrid meta density functional theory methods for thermochemistry, thermochemical kinetics, and noncovalent interactions: the MPW1B95 and MPWB1K models and comparative assessments for hydrogen bonding and van der Waals interactions. J Phys Chem A 108:6908–6918. doi:10.1021/jp048147q
Ziylan A, Dogan S, Agopcan S, Kidak R, Aviyente V, Ince NH (2014) Sonochemical degradation of diclofenac: byproduct assessment, reaction mechanisms and environmental considerations. Environ Sci Pollut Res 21:5929–5939. doi:10.1007/s11356-014-2514-7
Ziylan A, Ince NH (2011) The occurrence and fate of anti-inflammatory and analgesic pharmaceuticals in sewage and fresh water: treatability by conventional and non-conventional processes. J Hazard Mater 187:24–36. doi:10.1016/j.jhazmat.2011.01.057
Ziylan A, Koltypin Y, Gedanken A, Ince NH (2013) More on sonolytic and sonocatalytic decomposition of diclofenac using zero-valent iron. Ultrason Sonochem 20:580–586. doi:10.1016/j.ultsonch.2012.05.005
Acknowledgements
This work was supported by a grant from Tübitak (214Z198) and by the State Planning Organization of Turkey (DPT-2009K120520). S.A.Ç. acknowledges the TUBITAK BIDEB graduate student scholarship. The computational resources of the High Performance Computing Center and the National Center for High Performance Computing of Turkey (UHeM) under the grant number 5004052016 are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
ESM 1
(DOCX 5868 kb)
Rights and permissions
About this article
Cite this article
Agopcan Cinar, S., Ziylan-Yavaş, A., Catak, S. et al. Hydroxyl radical-mediated degradation of diclofenac revisited: a computational approach to assessment of reaction mechanisms and by-products. Environ Sci Pollut Res 24, 18458–18469 (2017). https://doi.org/10.1007/s11356-017-9482-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9482-7