Abstract
Water pollution by heavy metals is a great health concern worldwide. Lead and cadmium are among the most toxic heavy metals because they are dangerous for the human and aquatic lives. In this work, the removal of lead and cadmium from aqueous solutions has been studied using electrosynthesized 4-amino-3-hydroxynaphthalene-1-sulfonic acid-doped polypyrrole (AHNSA-PPy) films as a new adsorbent. Two distinct methods, including the immersion method, based on the Pb2+ and Cd2+ spontaneous removal by impregnation of the polymer in the solution, and the electro-elimination method, consisting of removal of Pb2+ and Cd2+ ions from the solution by applying a small electrical current (5 mA) to the polymer film, were developed: the evolution of Pb2+ and Cd2+ concentrations with time was monitored by inductively coupled plasma optical emission spectrometry (ICP-OES). The effect of pH on the adsorption and electro-elimination of Pb2+ and Cd2+ using the AHNSA-PPy film was investigated and optimized, showing that the ionic adsorption and electro-elimination processes were highly pH-dependent. The kinetics of Pb2+ and Cd2+ adsorption and electro-elimination were found to follow second-order curves. The maximum adsorption capacity values of the AHNSA-PPy film were 64.0 and 50.4 mg/g, respectively, for Pb2+ and Cd2+. The removal efficiency values were, respectively, for Pb2+ and Cd2+, 80 and 63% by the immersion method, and 93 and 85% by the electro-elimination method. Application of both methods to Senegal natural waters, fortified with Pb2+ and Cd2+, led to removal efficiency values of, respectively for Pb2+ and Cd2+, 76–77 and 58–59% by the immersion method, and of 82–90 and 80–83%, by the electro-elimination method.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nowadays, decontamination of polluted waters containing toxic, heavy metals constitutes a major health and environmental issue. Water contamination has many origins, such as the burning of fossil fuels, the exhaust gases of vehicles, mining, agriculture, and incineration of solid and liquid wastes. Indeed, most heavy metals are very toxic, even at very low concentrations, and are often resistant to the conventional treatments. Therefore, they can degrade the quality of drinking water resources and cause serious diseases (Nriagu and Pacyna 1988; Kimbrough et al. 1999; Poey and Philibert 2000; Goullé 1998). Among heavy metals, lead and cadmium are considered as the most toxic. Indeed, lead poisoning can provoke a decrease in fertility and an increase of fetus mortality (resulting in miscarriages) and of neurological, cardiovascular, or gastrointestinal diseases in the infants. Moreover, lead is known to yield mutagenic and carcinogenic effects (Goullé 1998; Lyn 2006) and to induce oxidative stress and DNA damage in mice (Jin et al. 2008). Excessive exposure to cadmium can cause death when it enters in mammalian cells and accumulates at high concentrations in the cytoplasmic and nuclear space (Detmar and Stefan, 1997; Satoh et al. 2003).
For these reasons, it is necessary to find effective techniques able to remove these heavy metals, toxic for humans and harmful for the environment. Therefore, a number of methods, like chemical precipitation (Macchi et al., 1996), adsorption (Sharma et al. 2013), ion-exchange (Dabrowski et al. 2004), electro-coagulation (Vasudevan et al. 2012), and membrane separation (Mortaheb et al. 2010), have been developed to trap heavy metallic ions in aqueous solutions. However, these methods suffer from various drawbacks, such as membrane fouling by metal hydroxides and carbonates, poor effectiveness for the removal of heavy metal at low concentrations, and high operating costs (Heidmann and Calmano, 2008).
In recent years, there has been a growing interest for applying bio/conducting polymeric materials to the removal of heavy metals, such as divalent cadmium and lead, because of their remarkable adsorption properties, low cost, ability to be functionalized with various metal chelating groups, and environmental compatibility (Mansour et al. 2011; Hasani and Eisazadeh, 2013; Karthik and Meenakshi 2015; Jiang et al. 2012). For example, Mansour et al. (2011) have showed that polyaniline coated on sawdust (PANI/SD) could be used to adsorb Cd2+ ions and to remove them from wastewater. It was demonstrated that the PANI/SD sorption capacity strongly depended on the concentration of heavy metals and on the solution initial pH. The adsorption of Cd2+ ions from aqueous solution was found to increase with pH, an optimum pH value of about 6 being obtained for maximum adsorption. Also, Hasani and Eisazadeh (2013) have utilized polypyrrole (PPy) and its nanocomposites, synthesized in the presence of sodium dodecylhydrogensulfonate (SDS) and other surfactants, for the removal of Cd2+ from aqueous solution by means of a batch method. The optimum conditions of sorption of PPy nanocomposites for the removal of Cd2+ were found to be a contact time of 45 min and a pH 5 value. In another study, Karthik and Meenakshi (2015) have synthesized a PANI-grafted chitosan (PGC) that was used as an adsorbent for the removal of Pb2+ and Cd2+ ions from aqueous solution. The adsorption of these ions on PGC was confirmed by FTIR, SEM, and EDX analysis. The authors showed that the pH of the working solution played a significant role in the adsorption process, the PGC sorption capacity being increased with the pH increase, and that the elimination of Pb2+ and Cd2+ by PGC was significantly affected in the presence of other metal ions. Also, Jiang et al. (2012) have assembled PANI nanofibers on micro- or millimeter-scale calcium alginate (CA) beads by a “competitive adsorption-restricted polymerization” approach for the removal of Pb2+ and Cu2+ in aqueous solutions. In this PANI/CA approach, which overcame the serious aggregation problem of PANI nanofibers, the Pb2+ removal was found to surpass 90% in a wide pH range from 3 to 7. In other studies, Heidari et al. (2009) used amino-functionalized mesoporous and nano mesoporous silica for the removal of Cd2+, Pb2+, and Ni2+ from a ternary aqueous solution; Maliyekkal et al. (2010) developed a novel hybrid material, cellulose-nanoscale-manganese oxide composite (C-NMOC), and applied it to Pb2+ removal from aqueous solutions; and Nassar (2010) employed Fe(3)O(4) magnetic nanoadsorbents for the rapid removal and recovery of Pb2+ ions from aqueous solutions by a batch adsorption technique. Moreover, Shameem et al. (2006) prepared chitosan-coated perlite beads for the adsorption of Cd2+ from pH 6.0 aqueous solutions, obtaining a very high adsorption capacity of 558 mg/g of chitosan, and Chauhan et al. (2006) used thiocarbamoyl chitosan (TC-chitosan) as adsorbent to simultaneously remove Cd2+ and hexavalent chromium from electroplating waste waters.
However, most of these important studies were performed by using polymeric materials that generally required time-consuming and multi-step chemical synthesis procedures. In many cases, a chelating substance had to be grafted on the polymer or a salt had to be inserted into the polymer structure to trap the metal ions. In a very recent paper, we have described the use of a new adsorbent, i.e., electrosynthesized 4-amino-3-hydroxynaphthalene-1-sulfonic acid-doped polypyrrole (AHNSA-PPy) films, for the efficient removal of chromium ions (Cr6+) from aqueous solutions (Sall et al. 2017). Hence, in the present work, we decided to develop and optimize two adsorption methods, based on AHNSA-PPy films as adsorbent, for removing Pb2+ and Cd2+ ions from aqueous solutions. The immersion method was based on the simple impregnation of an AHNSA-PPy film by the metallic salt aqueous solutions, while the newly developed electro-elimination method required the application of a 5-mA small current and the use of an AHNSA-PPy-coated electrode plunged in the metallic salt aqueous solution. AHNSA-PPy films were characterized by FTIR spectrometry, and the evolution of the Pb2+ and Cd2+ ion concentrations in aqueous solution was followed by ICP-OES. Kinetics of the Pb2+ and Cd2+ adsorption process were investigated. Also, both methods were applied to Senegal natural waters, fortified with Pb2+ and Cd2+.
Experimental
Chemicals
Pyrrole (Py—99%), 4-amino-3-hydroxynaphthalene-1-sulfonic acid (AHNSA), sodium hydroxide (NaOH), nitric acid (HNO3), sulfuric acid (H2SO4), cadmium sulfate (CdSO4), and lead nitrate [Pb(NO3)2] were obtained from Sigma-Aldrich and used as received.
Electrochemical experiments
The electrochemical experiments were carried out in a single-compartment three-electrode cell using an EG & G Princeton Applied Research PAR 362 Model potentiostat/galvanostat, equipped with a Kipp & Zonen X-Y recorder. A steel plate was used as working electrode, whereas a stainless steel grid and a saturated calomel electrode (SCE) were employed, respectively, as counter-electrode and reference electrode.
The electro-elimination method was performed by using a DC power supply (HAMEG Instruments, HM 8040-3) and two electrodes. The anode was a polymer-covered steel plate, and the cathode was a naked steel plate. The inter-electrode distance was 3.0 cm, and the submerged surface area of each electrode plate was 12 cm2.
Spectral characterization and analysis
Fourier-transform infrared (FTIR) spectra were measured using a Nicolet S-60 (FTIR) spectrometer (Nicolet Instrument Corp., Madison, WI), at a 2-cm−1 spectral resolution. The FTIR spectra of the AHNSA-PPy polymer films were recorded on Pt-coated glass plates by infrared-reflection absorption spectroscopy (IR-RAS), with a 30° incidence angle accessory. All spectra were baseline corrected, using OMNIC internal software, and were unsmoothed.
The Pb2+and Cd2+ concentrations in aqueous solution were determined by inductively coupled plasma-optical emission spectroscopy (ICP-OES), using a Perkin-Elmer Optima 8300 ICP-OES instrument. The natural water samples were analyzed after filtration through 0.7-μm glass fiber filters. The samples were injected through a Meinhard nebulizer (glass) K1, and all experiments were performed in triplicate. Pb 220 and Cd 226 wavelengths were selected for the ICP-OES analysis. The calibration curves ranged between concentrations of 10 μg L−1 and 10 mg L−1, with correlation coefficients of 0.999 for Pb2+ and Cd2+.
Electrosynthesis of AHNSA-PPy
The electrosynthesis of AHNSA-PPy was performed in an aqueous solution constituted of 0.01 M AHNSA + 0.007 M NaOH + 0.1 M Py (Sall et al. 2017). In order to completely dissolve poorly hydro-soluble AHNSA, the 0.01-M AHNSA + 0.007-M NaOH aqueous medium was stirred in an ultrasound bath for 5 min. Finally, a clear solution of pH = 5.1 was obtained, and an amount of Py, corresponding to a 0.1-M concentration, was added. Cyclic voltammetry (CV) was carried out during 20 cycles in the above-described medium, and a homogeneous AHNSA-PPy film was obtained on the working electrode (steel plate) by repetitively scanning the potential between − 0.8 and 1.0 V/SCE at a scan rate of 100 mV/s. After weighing the steel plate, it was found that a mass of polymer of about 30 mg was electrodeposited on the working electrode.
Procedures for the determination of lead and cadmium removal
For the immersion method, a steel plate covered by an AHNSA-PPy film was dove either in a 60-mg-L−1 Pb(NO3)2 or in a 60-mg-L−1 CdSO4 aqueous solution. Then, the Pb2+ or the Cd2+ concentration was monitored as a function of the polymer impregnation time and solution pH. The effect of the Pb(NO3)2 or CdSO4 solution pH changes on the polymer adsorption capacity was examined between pH values of 1 and 6 by measuring the Pb2+ or Cd2+ solution ICP-OES intensity. The solution pH values were adjusted by adding small amounts of H2SO4 or HNO3.
In the case of the electro-elimination method, an optimized electrical current of constant intensity (I = 5 mA) was applied between two electrodes, the anode being an AHNSA-PPy polymer-covered steel plate and the cathode being a naked steel plate. Both electrodes were plunged either in a 60-mg-L−1 Pb(NO3)2 or in a 60-mg-L−1 CdSO4 aqueous solution at an optimized pH value. Then, the Pb2+ or the Cd2+ concentration was followed by ICP-OES as a function of the electrolysis time.
We estimated the Pb2+ and Cd2+ removal efficiency (% R) of the polymer by means of Eq. (1):
where C0 and C t are, respectively, the Pb2+ or Cd2+ concentrations (mg/L) at times t = 0 and t.
The adsorption capacity of the polymer at the time t (q t ) was determined by Eq. (2):
where C0 and C t are, respectively, the Pb2+ [or Cd2+] concentrations (mg/L) at times t = 0 and t; m, the polymer mass (g); and V, the Pb(NO3)2 [or CdSO4] solution volume (L).
Application studies
For the application studies to Senegal natural waters, the different natural water samples were first filtered using 0.7-μm glass fiber filters. Then, the filtered samples were fortified by dissolving 60 mg of Pb(NO3)2 or CdSO4 in a 1-L volume of these samples, in order to reach an initial concentration = 60 mg L−1, and adjusted at the optimum pH value. The % R and q t values were determined for all natural water samples at impregnation time and electrolysis time optimum values.
Results and discussion
Effect of pH
The influence of pH on the adsorption process of Pb2+ and Cd2+ ions on the polymer-covered plate was investigated in the pH range of 1–6 by the immersion method. We followed by ICP-OES the evolution of the removed Pb2+ and Cd2+ percentages with the pH of the metal solution (Fig. 1). As can be seen, the Pb2+ and Cd2+ adsorption onto AHNSA-PPy films increased with pH up to a pH 6 optimum value for which the best removal efficiency was obtained. A similar pH trend was reported by Ramesh et al. (2013) in the case of the sorption of Pb2+onto hydroxyapatite surfaces.
Beyond this pH value of 6, especially in alkaline conditions, the removal efficiency remained practically constant. This behavior can be partly attributed to the appearance of precipitates resulting from the formation of very poorly hydro-soluble lead and cadmium hydroxides with different charges, such as Pb(OH)+ and Pb(OH)2 (Altin et al. 1999; Sari et al. 2007; Ramesh et al. 2013). In these conditions, an important part of lead and cadmium was precipitated, and another smaller part was adsorbed by AHNSA-PPy.
Upon going from pH 1 to 6, the observed increase of Pb2+ and Cd2+ removal efficiency with pH value is probably also due to the fact that the doping AHNSA molecules in their cationic (ammonium) form were gradually transformed into the zwitterionic form. It is in this later form that the sulfonic acid group (one of the AHNSA chelating sites) is dissociated and, therefore, because of its negative charge, can attract the Pb2+ or Cd2+ cations, which would partly explain the Pb2+ and Cd2+ sorption maximum value onto AHNSA-PPy found around pH 6. In contrast, in strongly acidic solution, the protonation of the AHNSA amino group would slow down the adsorption of metallic cations, leading to a lower removal efficiency at pH 1.
Since the Pb2+ and Cd2+ maximum removal efficiency values were observed in a pH 6 aqueous solution, we selected this optimal pH value of 6 for all our kinetic studies of the Pb2+ and Cd2+ removal, using the immersion method as well as the electro-elimination methods.
Kinetic study of the Pb2+ and Cd2+ removal by the immersion method
In preliminary experiments, we determined the effect of varying the initial concentration of Pb2+ and Cd2+ (20, 40, and 60 mg L−1) on the polymer removal efficiency of the metal ions and on the Pb2+ and Cd2+ adsorption kinetics. Since we found an increase in the AHNSA-PPy removal efficiency and the adsorption rate constant with the initial metal ion concentration, we decided to choose an optimal, initial concentration value of 60 mg L−1 for all our kinetic studies of the Pb2+ and Cd2+ removal, using the immersion method as well as the electro-elimination method.
Pb2+ removal by the immersion method
Our kinetic study was carried out at a room temperature in a pH 6 Pb(NO3)2 aqueous solution (60 mg L−1), in which a AHNSA-PPy polymer film-covered steel plate was plunged. The evolution of Pb2+ ion concentration was followed by ICP-OES as a function of the polymer impregnation time (Fig. 2). As can be seen, a progressive decrease of the Pb2+ concentration occurred with time. We noted a significant reduction of the Pb2+ concentration which diminished within 8 h from 60.0 to 11.5 mg L−1. This decrease corresponded to 81% Pb2+ removal efficiency.
Evolution of the Pb2+ and Cd2+ concentration in a pH 6 Pb(NO3)2 and CdSO4 aqueous solution (initial concentration = 60 mg L−1) as a function of the AHNSA-PPy polymer impregnation time (immersion method), followed by ICP-OES. Insert: evolution of the polymer adsorption capacity for (a) Pb2+ and (b) Cd2+. The quantity of metal ion (q t , in mg g−1 of polymer) adsorbed by AHNSA-PPy polymer (m = 30 mg) was measured by ICP-OES as a function of impregnation time. All measurements were carried out in 40 mL of pH 6 Pb(NO3)2 and CdSO4 aqueous solutions
The analysis of the adsorption kinetic of Pb2+ on a polymer-covered anode (m [polymer] = 30 mg) under optimized experimental conditions led to a second-order kinetic curve, since the curve 1/C t − 1/C0 as function of time was linear: y = 0.88 t with a correlation coefficient r2 = 0.991. We obtained a second-order rate constant value k2 = 0.88 ± 0.07 L mol−1 s−1.
Cd 2+ removal by the immersion method
We performed our kinetic study at a room temperature in a pH 6 CdSO4 aqueous solution (60 mg L−1), in which an AHNSA-PPy polymer film-covered steel plate was plunged. Then, the evolution of Cd2+ concentration was monitored by ICP-OES as a function of the polymer impregnation time (Fig. 2). It can be seen that the concentration of Cd2+ in solution progressively decreased with time. Our ICP-OES results showed that the Cd2+ concentration diminished within 8 h from 60.0 to 15.2 mg L−1, which indicated a 74.6% Cd2+ removal efficiency. We can conclude that, by using the immersion method, the AHNSA-PPy polymer was able to remove the majority of Cd2+ ions in aqueous solution. However, the removal efficiency, using the same method, was better in the case of Pb2+ ions.
The study of the adsorption kinetic of Cd2+ on a polymer-covered anode (m [polymer] = 30 mg) under optimized experimental conditions yielded a second-order kinetic curve, since the curve 1/C t − 1/C0 as function of time was linear: y = 0.36 t (correlation coefficient r2 = 0.988), from which we determined a second-order rate constant value k2 = 0.36 ± 0.01 L mol−1 s−1.
Kinetic study of the Pb2+ and Cd2+ removal by the electro-elimination method
We developed a novel electro-elimination method for the removal of Pb2+ and Cd2+ from aqueous solutions. This method consisted, first, of electro-deposition of the polymer (AHNSA-PPy film) on a steel anode by CV, as described in the experimental part. Then, two steel electrodes, i.e., a polymer-covered anode and an auxiliary bare steel cathode, were immersed in a pH 6 lead nitrate or cadmium sulfate aqueous solution (60 mg L−1) and submitted to an applied current of 5 mA.
Pb2+ removal by the electro-elimination method
We followed by ICP-OES the evolution of the Pb2+ concentration with time. As can be seen in Fig. 3, our kinetic results were characterized by an important and progressive decrease in the Pb2+ concentration with time. Indeed, within 6 h, the Pb2+ concentration decreased from 60.0 to 3.7 mg L−1, which corresponded to a 94% Pb2+ removal efficiency. It is worthwhile to note that, compared to the simple immersion method, our electro-elimination method yielded a significant augmentation of the Pb2+ removal efficiency from 81 to 94% within a 2-h shorter time period. These results demonstrated that the electro-elimination method was more efficient and more rapid than the immersion method for lead removal from aqueous solutions.
The kinetic analysis of Pb2+ electro-elimination, using a AHNSA-PPy polymer-covered anode (m [polymer] = 30 mg) under optimized experimental conditions, led, like for the immersion method, to a second-order kinetic curve, since the curve 1/C t − 1/C0 as function of time was linear: y = 3.9 t with a correlation coefficient r2 = 0.987. Therefore, we obtained a second-order rate constant value: k2 = 2.20 ± 0.07 L mol−1 s−1.
Compared to the k2 value found for Pb2+ adsorption by the simple immersion method, this k2 value was 2.5 times higher, which confirmed that the electro-elimination method was significantly more rapid than the immersion method for Pb2+ removal. This important rate constant increase can be explained by the acceleration effect of the applied current on the Pb(II) adsorption process.
Cd2+ removal by the electro-elimination method
We monitored by ICP-OES the change of Cd2+ concentration with time (Fig. 3). As can be seen, an important and progressive decrease in the Cd2+ concentration took place with time. Indeed, within 6 h, the Cd2+ concentration decreased from 60.0 to 6.0 mg L−1, which represented a 90% Cd2+ removal efficiency by the electro-elimination method. Therefore, compared to the simple immersion method, our electro-elimination method led to a very large increase of the Cd2+ removal efficiency from 74.6 to 90%, within a 2-h shorter time period. Our results showed that the electro-elimination method is much more efficient and more rapid than the immersion method for cadmium removal from aqueous solution, like in the case of lead removal.
The kinetic study of the Cd2+ electro-elimination, using a AHNSA-PPy polymer-covered anode (m [polymer] = 30 mg) under optimized experimental conditions, gave a second-order kinetic curve, since the curve 1/C t − 1/C0 as function of time was linear: y = 1.65 t with a correlation coefficient r2 = 0.997. We obtained a second-order rate constant value: k2 = 1.65 ± 0.10 L mol−1 s−1. Compared to the k2 value obtained for Cd2+ adsorption by the simple immersion method, this k2 value was more than four times higher, which confirmed that the electro-elimination method was much more rapid than the immersion method for Cd2+ removal. Like for the Pb2+ kinetic study, we attributed this very large rate constant increase to the acceleration effect of the applied current on the Cd2+ adsorption process.
Evolution of the polymer adsorption capacity for Pb2+ and Cd2+
Using Eq. (2), we calculated the adsorption capacity of the AHNSA-PPy polymer at different times (q t ) for Pb2+ and Cd2+, i.e., the quantity of lead and cadmium adsorbed per gram of polymer, which was monitored as function of impregnation time (Fig. 2—insert). As can be seen, the adsorbed quantity of Pb2+ and Cd2+ significantly increased with the polymer impregnation time and then reached a plateau region. The curves of Fig. 2 (insert) showed that, after 8 h of polymer impregnation time, maximum adsorption capacity values of 64.0 mg of Pb2+ and 50.4 mg of Cd2+ per gram of polymer, respectively, were found. In agreement with the kinetic study, our results indicated that the polymer adsorption capacity was better for lead than for cadmium. These adsorption capacity values were much greater than the maximum monolayer adsorption capacity values of 16.07 and 14.33 mg g−1, respectively, for Pb2+ and Cd2+ ions, obtained by Karthik and Meenakshi (2015), using polyaniline-grafted chitosan (PGC) as adsorbent, and of 18.72 mg g−1 for Cd2+ ions, found by Ramesh et al. (2011), using coir pith as adsorbent. Therefore, we can conclude that our AHNSA-PPy polymer is a relatively good adsorbent for the removal of Pb2+ and Cd2+ ions from aqueous solution.
Influence of Pb2+ and Cd2+ adsorption on the AHNSA-PPy FTIR spectra
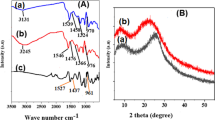
We compared the FTIR spectra of AHNSA-PPy polymer films on Pt-coated glass plates, before and after immersion in pH 6 Pb(NO3)2 and CdSO4 aqueous solutions (initial concentrations = 60 mg L−1) during 8 h (Fig. 4). The assignment of the main IR bands of AHNSA-PPy polymer was carried out on the basis of literature FTIR spectral data of analogous compounds (Bhandari et al. 2011; Ćirić-Marjanović et al. 2006; Just et al. 1999; Just et al. 2001; Sall et al. 2017; Socrates 2004).
The AHNSA-PPy FTIR spectrum (Fig. 4(a)) exhibited two bands located at 712 cm−1 (weak intensity) and 770 cm−1 (medium intensity), attributed to γC–H bending vibrations of the AHNSA naphthalene cycle (un-substituted and tri-substituted phenyl groups). Also, one strong band, located at 1035 cm−1, was assigned to the symmetric νS–O stretching vibration of the SO3H substituent. Two other bands situated at 1210 (strong intensity) and 1312 cm−1 (medium intensity) corresponded, respectively, to the C–O stretching vibration and to the O–H bending vibration of the naphthol group. Moreover, one strong band which appeared at 1555 cm−1 was assigned to the naphthalene cycle νC=C stretching vibrations. In addition, it was observed that there are several bands due to various vibrations of Py rings. For example, a strong band occurring at 910 cm−1corresponded to the γC–H oop bending vibrations of two adjacent hydrogens of 2,5 disubstituted Py rings. Also, two weak bands, appearing at 1090 and 1475 cm−1, were attributed, respectively, to the Py νC–N and νC=C stretching vibrations. Moreover, a strong and very wide band, with two components located at about 3100 and 3400 cm−1, was characteristic of the overlapping of the hydrogen-bonded stretching vibrations, νO–H of the naphthalene cycle (AHNSA OH substituent) and νN–H of the naphthalene cycle (AHNSA NH2 substituent) and of the Py cycle (N–H group). The presence of all these bands confirmed that the AHNSA amphoteric salt was inserted between the PPy polymer chains (Sall et al. 2017).
Several notable changes occurred in the polymer spectra after immersion in Pb2+ and Cd2+ solutions (Fig. 4(b, c)). The AHNSA absorption band located at 1035 cm−1, attributed to the SO3H substituent νS–O stretching vibrations, and other bands at 1210 and 1312 cm−1, corresponding to naphthol group vibrations, were slightly shifted and their intensity strongly diminished. Also, the wide band located in the 3100–3400-cm−1 region, corresponding to the stretching vibrations, νO–H of the naphthalene cycle, and νN–H of naphthalene and of Py, nearly disappeared after immersion in Pb2+ and Cd2+ solutions. The later result can be explained by strong electrostatic interactions of OH, NH2 (naphthalene), and NH (Py) groups with Pb2+ and Cd2+ ions, producing an important decrease of the intensity of the νO–H and νN–H stretching vibrations. Moreover, a weak band appeared at 1710 cm−1, which was ascribed to the overoxidation of PPy. This overoxidation reaction probably resulted from the attack of doped PPy chains by nucleophilic NO3−, SO42−, or OH species, used during the Pb2+ or Cd2+ adsorption process on the polymer (Lewis 1998; Novak et al. 1991). All these FTIR spectral changes clearly demonstrated the existence of interactions between Pb2+ or Cd2+ ions and N–H, O–H, and S–O chelating sites of AHNSA-PPy films, indicating that the nitrogen, oxygen, and sulfur atoms played an important role in the adsorption of Pb2+ and Cd2+ species on AHNSA-PPy polymer films, as already shown in the case of the interactions of Pb2+ and Cd2+ ions with other adsorbents (Chauhan et al. 2006; Karthik and Meenakshi 2015).
Application to natural waters
We investigated samples of different Senegal natural waters, fortified with 60 mg L−1 of Pb2+ or Cd2+ ion by means of both adsorption methods, under the above-described, optimized conditions (polymer mass = 30 mg, solution pH 6.0, I = 5 mA). The removal of Pb2+ and Cd2+ from these fortified natural water samples was studied by comparing the application of the simple polymer immersion method and of the newly developed, electro-elimination method.
Application of the immersion method
Using the immersion method, we dived a polymer-covered electrode into the natural water samples fortified with Pb(NO3)2 or CdSO4 solutions. Then, we followed the evolution of the lead or cadmium ICP-OES intensity as a function of the polymer impregnation time. For all fortified natural water samples, ICP-OES analysis indicated a progressive diminution of the Pb2+ or Cd2+ concentration with the impregnation time.
Figure 5 shows the gradual decrease of Pb2+ and Cd2+ concentrations in the case of a fortified river water sample. The Pb2+ and Cd2+ concentrations decreased, respectively, from 60.0 to about 13.8 and 24.6 mg L−1, within 8 h, which corresponded to respective metal ion removal percentages of 77 and 59%. In this river water sample, the adsorbed metal ion quantities (q t ) were, respectively for Pb2+ and Cd2+ ions, around 61.5 and 47.0 mg g−1 of polymer. These q t values are very close to those obtained in pure water (respectively, 64.0 mg of Pb2 and 50.4 mg of Cd2+ per gram of polymer).
In Table 1, we have given the values of lead and cadmium removal efficiency (R) and of adsorbed Pb2+ and Cd2 quantity (q t ) for all Senegal natural water samples under study.
As can be seen, the R values, measured in the natural water samples after an impregnation time of 8 h, were practically constant, being comprised, respectively, between 76.0 and 77.0% for Pb2+, and between 58.5 and 59.0% for Cd2+. They were lower than the R values of 80.0 and 74.7%, determined, respectively, for Pb2+ and Cd2+ in pure water. These relatively high R values indicated that the AHNSA-PPy polymer was able to remove the majority of Pb2+ and Cd2+ from fortified natural waters.
For all natural waters under study, the q t values were almost constant, ranging, respectively, for Pb2+ and for Cd2+, from 60.5 to 61.6 mg g−1 and from 46.8 to 47.0 mg/g of polymer. They were slightly smaller than the qt values of 64.0 and 50.4 mg g−1, obtained in pure water, respectively for Pb2+ and Cd2+, like in the case of the R values. Our results demonstrated a rather satisfactory adsorption efficiency of the AHNSA-PPy polymer. As already indicated, these q t values were greater than the maximum monolayer adsorption capacity values obtained by Karthik and Meenakshi (2015), using PGC as adsorbent.
The decreases of the lead and cadmium ion removal efficiency and adsorbed quantity values observed between pure water and the various fortified natural waters are probably due to the influence of other interfering metal ions, such as Ni2+, Co2+, Cu2+, and Zn2+, generally present in most natural waters, or Na+ and Mg2+ ions in sea water. These decreases could be explained by a kinetic competition between the various metallic ions, depending on their charge, size, and concentration, for the sorption sites on the adsorbent surface (Karthik and Meenakshi 2015; Nassar, 2010).
Overall, our results demonstrated the rather satisfactory removal efficiency and adsorption capacity of AHNSA-PPy, and the interest of applying our simple immersion method to the removal of Pb2+ and Cd2+ in natural waters.
Application of the electro-elimination method
In the case of newly developed, electro-elimination method, we put two steel electrodes, including a plate coated with AHNSA-PPy as anode and a bare plate as cathode, in various natural waters of Senegal, that were fortified with Pb(NO3)2 or CdSO4 solutions. Then, a maximum I value of 5 mA was applied at times = 0, 60, 120, 240, and 360 min. Samples were collected at these electrolysis times, and we followed the evolution of the lead and cadmium ICP-OES intensity. For all fortified natural water samples, there was a gradual decrease in the Pb2+ and Cd2+ concentrations with time.
We have presented in Fig. 6 a histogram showing the gradual decrease of lead and cadmium concentrations for a Senegal fortified river water sample. The Pb2+ and Cd2+ concentrations decreased from 60.0 to about 6.0, and 10.2 mg L−1, respectively, within an electro-elimination time of 6 h, which corresponded to respective removal percentages (R) of 90 and 83%. These R values obtained by the electro-elimination method were larger by, respectively, for Pb2+ and Cd2+ ions, 13 and 24% than those found for a fortified river water sample, using the simple immersion method. As expected, our results confirmed that the electro-elimination method was significantly more efficient than the immersion method for the removal of Pb2+ and Cd2+ from fortified river water samples.
Table 2 displays the values of Pb2+ and Cd2+ removal efficiency (R) and of adsorbed Pb2+ and Cd2+ quantity (q t ) for all Senegal natural water samples under study.
As can be seen, the R values, measured in the natural water samples after an electro-elimination time of 6 h, were fairly high, ranging, respectively, between 82 and 90% for Pb2+, and between 80 and 83% for Cd2+, according to the natural water sample. They were lower than the R values of 93 and 85%, determined, respectively, for Pb2+ and Cd2+ in pure water. Also, it is worthwhile to note that, compared to the results obtained for the immersion method, the R values measured for the electro-elimination method increased, importantly by 6 to 17% for Pb2+, and by 21 to 26% for Cd2+, depending on the natural water sample.
The q t values were comprised, respectively, for Pb2+ and for Cd2+, between 63.0 and 72.0 mg/g, and between 63.8 and 65.5 mg/g of polymer, according to the natural water sample (Table 2). They were slightly smaller than the q t values of 74.4 and 66.9 mg/g, obtained in pure water under optimized conditions, respectively for Pb2+ and Cd2+. Relatively to the results of the immersion method, the q t values measured by the electro-elimination method significantly increased for Pb2+ and for Cd2+, depending on the natural water sample.
Also, the electro-elimination method confirmed the occurrence of significant decreases of the lead and cadmium ion removal efficiency and adsorbed quantity values between pure water and the fortified natural waters, already revealed by the immersion method. As noted above, these decreases probably result from the influence of other interfering metal ions, such as Ni2+, Co2+, Cu2+, and Zn2+, generally present in most natural waters (Chauhan et al., 2012; Karthik and Meenakshi, 2015), or Na+ and Mg2+ ions in sea water.
Our results showed that, for all the fortified natural waters under study, the electro-elimination method, applied within a shorter time, considerably improved the lead and cadmium removal efficiency and adsorption capacity of AHNSA-PPy, relative to the immersion method. Therefore, it is preferable to use the electro-elimination method rather than the immersion one for removing efficiently and rapidly Pb2+ and Cd2+ from natural waters.
Conclusion
In this work, we have been able to utilize the electrosynthesized AHNSA-PPy polymer as a good adsorbent for the removal of Pb2+ and Cd2+ ions from aqueous solutions, either by the simple immersion method or by the newly developed electro-elimination method. For both methods, the efficiency of Pb2+ and Cd2+ removal was highly pH-dependent, the maximum removal efficiency being obtained at pH 6. The removal of Pb2+ and Cd2+ ions adsorbed on the AHNSA-PPy polymer was followed by ICP-OES. From our kinetic results, we can conclude that the second-order kinetic model is suitable to describe the adsorption process of Pb2+ and Cd2+ ions on AHNSA-PPy. Moreover, the removal efficiency of Pb2+ and Cd2+ increased, respectively, from about 80 and 75% within 8 h by the immersion method to about 93 and 85% within 6 h by the electro-elimination method, whereas the maximum adsorption capacity of Pb2+ and Cd2+ also increased, respectively, from about 64 and 50 mg g−1 by the immersion method to about 74 and 67 mg g−1 of polymer by the electro-elimination method. Therefore, we can deduce from these results that the electro-elimination method was much more efficient and more rapid than the immersion method for Pb2+ and Cd2+ removal from aqueous solution. Moreover, the application of both methods to different Senegal natural water samples fortified with Pb2+ or Cd2+ led to much higher removal efficiency and adsorption capacity values for the electro-elimination method than for the immersion method. We can also conclude from these application studies that the electro-elimination method was more efficient for Pb2+ and Cd2+ removal from fortified natural waters than the immersion method, and that the former method should be chosen for field studies.
References
Altin O, Ozbelge OH, Dogu T (1999) Effect of pH, flow rate and concentration on the sorption of Pb and Cd on montmorillonite. I. Experimental. J Chem Technol Biotechnol 74(12):1131–1138. https://doi.org/10.1002/(SICI)1097-4660(199912)74:12<1131::AID-JCTB158>3.0.CO;2-0
Bhandari H, Choudhary V, Dhawan SK (2011) Influence of self-doped poly(aniline-co-4-amino-3-hydroxy-naphthalene-1-sulfonic acid) on corrosion inhibition behavior of iron in acidic medium. Synth Met 161(9-10):753–762. https://doi.org/10.1016/j.synthmet.2011.01.026
Chauhan D, Jaiswal M, Sankararamakrishnan N (2012) Removal of cadmium and hexavalent chromium from electroplating waste water using thiocarbamoyl chitosan. Carbohydr Polym 88(2):670–675. https://doi.org/10.1016/j.carbpol.2012.01.014
Ćirić-Marjanović G, Trchová M, Matĕjka P, Holler P, Marjanović B, Juranić I (2006) Electrochemical oxidative polymerization of sodium 4-amino-3-hydroxynaphthalene-1-sulfonate and structural characterization of polymeric products. React Funct Polym 66(12):1670–1683. https://doi.org/10.1016/j.reactfunctpolym.2006.07.002
Dabrowski A, Hubicki Z, Podkoscielny P, Robens E (2004) Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere 56(2):91–106. https://doi.org/10.1016/j.chemosphere.2004.03.006
Detmar B, Stefan H (1997) Cadmium, gene regulation, and cellular signalling in mammalian cells. Toxicol Appl Pharmacol 144:247–261
Goullé J-P (1998) Métaux. In: Kintz P (ed) Toxicologie et pharmacologie médico-légales. Elsevier, Paris, pp 189–232
Hasani T, Eisazadeh H (2013) Removal of Cd (II) by using polypyrrole and its nanocomposites. Synth Met 175:15–20. https://doi.org/10.1016/j.synthmet.2013.04.026
Heidari A, Younesi H, Mehraban Z (2009) Removal of Ni(II), Cd(II), and Pb(II) from a ternary aqueous solution by amino functionalized mesoporous and nano mesoporous silica. Chem Eng J 153(1-3):70–79. https://doi.org/10.1016/j.cej.2009.06.016
Heidmann I, Calmano W (2008) Removal of Zn(II), Cu(II), Ni(II), Ag(I) and Cr(VI) present in aqueous solutions by aluminum electrolytic treatment. J Hazard Mater 152(3):934–941. https://doi.org/10.1016/j.jhazmat.2007.07.068
Jiang N, Xu Y, Dai LW, Dai L (2012) Polyaniline nanofibers assembled on alginate microsphere for Cu2+ and Pb2+ uptake. J Hazard Mater 215–216:17–24
Jin X, Ling-jun L, Chen W, Xiao-feng W, Wen-yu F, Li-hong X (2008) Lead induces oxidative stress, DNA damage and alteration of p 53, Bax and Bel-2 expressions in mice. Food and Chem Toxicology 46:1488–1494
Just P-E, Chane-Ching KI, Lacroix JC, Lacaze PC (1999) Anodic oxidation of dipyrrolyls linked with conjugated spacers: study of electronic interactions between the polypyrrole chain and the spacers. J Electroanal Chem 479(1):3–11. https://doi.org/10.1016/S0022-0728(99)00420-9
Just P-E, Chane-Ching KI, Lacroix JC, Lacaze PC (2001) Electrochemical oxidation of dipyrrolyl derivatives: application to the formation of reticulated conducting polymers with conjugated spacers. Electrochim Acta 46(20-21):3279–3285. https://doi.org/10.1016/S0013-4686(01)00620-X
Karthik R, Meenakshi S (2015) Removal of Pb (II) and Cd (II) ions from aqueous solution using polyaniline grafted chitosan. Chem Engin J 263:168–177
Kimbrough DE, Cohen Y, Winer AM, Creelman L, Mabuni C (1999) A critical assessment of chromium in the environment. Crit Rev Environ Sci Technol 29(1):1–46. https://doi.org/10.1080/10643389991259164
Lewis TW (1998) A study of the overoxidation of the conducting polymer polypyrrole. Ph D Thesis, University of Wollongong (Australia)
Lyn P (2006) Lead toxicity part II: the role of free radical damage and the use of antioxidants in the pathology and treatment of lead toxicity. Altern Med Rev 11:114–127.
Macchi G, Marani D, Pagano M, Bagnuolo G (1996) A bench study on lead removal from battery manufacturing wastewater by carbonate precipitation. Water Res 30(12):3032–3036. https://doi.org/10.1016/S0043-1354(96)00089-9
Maliyekkal SM, Lisha KP, Pradeep T (2010) A novel cellulose–manganese oxide hybrid material by in situ soft chemical synthesis and its application for the removal of Pb(II) from water. J Hazard Mater 181(1-3):986–995. https://doi.org/10.1016/j.jhazmat.2010.05.112
Mansour MS, Ossman ME, Farag HA (2011) Removal of Cd (II) ion from waste water by adsorption onto polyaniline coated on sawdust. Desalination 272(1-3):301–305. https://doi.org/10.1016/j.desal.2011.01.037
Mortaheb HR, Zolfaghari A, Mokhtarani B, Amini MH, Mandanipour V (2010) Study on removal of cadmium by hybrid liquid membrane process. J Hazard Mater 177(1-3):660–667. https://doi.org/10.1016/j.jhazmat.2009.12.082
Nassar NN (2010) Rapid removal and recovery of Pb(II) from wastewater by magnetic nanoadsorbents. J Hazard Mater 184(1-3):538–546. https://doi.org/10.1016/j.jhazmat.2010.08.069
Novák P, Rasch B, Vielstich W (1991) Overoxidation of polypyrrole in propylene Carbonate. An in situ FTIR Study. J Electrochem Soc 138:3300–3304
Nriagu JO, Pacyna JM (1988) Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 333(6169):134–139. https://doi.org/10.1038/333134a0
Poey J, Philibert C (2000) Toxicité des métaux. Rev Fr Lab 323:35–43
Ramesh ST, Gandhimathi R, Badabhagni N, Nidheesh PV (2011) Removal of Cd (II) from aqueous solution by adsorption onto coir pith, an agricultural solid waste: batch experimental study. Environ Engin Manag J 10:1667–1673
Ramesh ST, Rameshbabu N, Gandhimathi R, Srikanth Kumar M, Nidheesh PV (2013) Adsorptive removal of Pb(II) from aqueous solution using nano-sized hydroxyapatite. Appl Water Sci 3(1):105–113. https://doi.org/10.1007/s13201-012-0064-z
Sall ML, Diaw AKD, Gningue-Sall D, Chevillot-Biraud A, Oturan N, Oturan MA, Aaron J-J (2017) Removal of Cr (VI) from aqueous solution using electrosynthesized 4-amino-3-hydroxynaphthalene-1-sulfonic acid doped polypyrrole as adsorbent. Environ Sci Pollut Res 24(26):21111–21127. https://doi.org/10.1007/s11356-017-9713-y
Sari A, Tuzen M, Citak D, Soylak M (2007) Equilibrium, kinetic and thermodynamic studies of adsorption of Pb(II) from aqueous solution onto Turkish kaolinite clay. J Hazard Mater 149(2):283–291. https://doi.org/10.1016/j.jhazmat.2007.03.078
Satoh M, Kaji T, Tohyama C (2003) Low dose exposure to cadmium and its health effects. Part 3. Toxicity in laboratory animals and cultured cells. Nippon Eiseigaka Zasshi 57(4):615–623. https://doi.org/10.1265/jjh.57.615
Shameem H, Abburi K, Tushar KG, Dabir SV, Veera MB, Edgar DS (2006) Adsorption of divalent cadmium (Cd(II)) from aqueous solutions onto chitosan coated perlite beads. Ind Eng Chem Res 45:5066–5077
Sharma S, Pathania D, Singh P (2013) Preparation, characterization and Cr(VI) adsorption behavior study of poly(acrylic acid) grafted Ficus carica bast fiber. Adv Mater Lett 4(4):271–276. https://doi.org/10.5185/amlett.2012.8409
Socrates G (2004) Infrared and Raman characteristic group frequencies: tables and charts, 3rd edn. Wiley, New York
Vasudevan S, Lakshmi J, Sozhan G (2012) Simultaneous removal of Co, Cu, and Cr from water by electrocoagulation. Toxicol Environ Chem 94(10):1930–1940. https://doi.org/10.1080/02772248.2012.742898
Acknowledgements
One of us (M.L.SALL) gratefully thanks the French Embassy in Dakar (Senegal) for a Ph.D. grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Sall, M.L., Diaw, A.K.D., Gningue-Sall, D. et al. Removal of lead and cadmium from aqueous solutions by using 4-amino-3-hydroxynaphthalene sulfonic acid-doped polypyrrole films. Environ Sci Pollut Res 25, 8581–8591 (2018). https://doi.org/10.1007/s11356-017-1111-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-1111-y