Abstract
Constructed wetlands are ecosystems that use plants and microorganisms to remediate pollution in soil and water. In this study, two parallel pilot-scale vertical flow wetland and horizontal flow wetland (VF-HF) systems were implemented to investigate the treatment performance and microorganism community structure in the secondary effluent of an industrial park wastewater treatment plant (WWTP) with a loading rate of 100 mm/day near the Yangtze River in Suzhou City, East China. Removal efficiencies of 82.3, 69.8, 77.8, and 32.3 were achieved by the VF-HF systems for ammonium nitrogen (NH4 +-N), total nitrogen (TN), total phosphorus (TP), and chemical oxygen demand (COD), respectively. The VF system specialized in COD and NH4 +-N removal (73.6 and 79.2 %), whereas the HF system mainly contributed to TN removal (63.5 %). The effluents in all seasons are capable of achieving the “surface water environmental quality standard” (GB3838-2002) grade IV. In the VF system, the 16S gene and nirK gene were significantly correlated with depth, with the 16S gene showing significant correlations with the dissolved oxygen (DO) level (r = 0.954, p < 0.05), which was determined by real-time PCR and high-throughput sequencing. Many types of bacteria capable of biodegradation, including nitrifiers, denitrifiers, and polyaromatic hydrocarbon (PAH) degraders (improvement of the BOD5/COD ratio), were observed, and they contributed to approximately 90 % of the nitrogen removal in the VF-HF system.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Constructed wetlands (CWs) are artificial ecosystems (Saeed and Sun 2012; Vymazal 2013; Zhang et al. 2012) which include a microbial community, substrate, and plants to remediate pollution in soil and water (De Jong 1976; Brix 1994a, b; Vymazal 2005a, 2009). CWs can be employed to treat a variety of wastewaters with success, such as municipal or domestic wastewater (Hamouri et al. 2007; Trang et al. 2010), storm water, and agricultural wastewaters (Greenway 1997; Merlin et al. 2002). Over the past two decades, the application of CWs has also extended to industrial wastewaters (Vymazal 2014) generated by the petrochemical industry (Chapple et al. 2002; Kadlec and Wallace 2009), pulp and paper industry (Abira et al. 2005; Choudhary et al. 2010), tannery industry (Calheiros et al. 2012; Chen et al. 2006; Hadad et al. 2006), textile industry (Dos Santos et al. 2007; Sharma et al. 2007; Davies et al. 2005), alcohol fermentation industry (Anastasiou et al. 2009; Rochard et al. 2002), and food processing industry (Gasiunas et al. 2005).
There are four types of CWs for wastewater treatment: free-floating plants (FFP), free water surface and emergent macrophytes (FWS), horizontal subsurface flow (HSSF, HF), and vertical subsurface flow (VSSF, VF) (Vymazal 2001; Vymazal 2005b). FWS systems are not used as frequently as HF or VF systems despite being one of the oldest designs in Europe (Brix 1994a; Vymazal 2001). VF systems provide good conditions for nitrification but not for denitrification. HF systems, however, can perform denitrification because of their limited capacity to transfer oxygen (Vymazal 2005b). In combined systems, the advantages of the HF and VF systems can be combined to complement each other, especially in VF-HF hybrid CWs, which can produce an effluent that is low in BOD5, fully nitrified and partly denitrified, thus resulting in much lower total nitrogen (TN) concentrations (Cooper 1999).
Currently, industrial parks play a significant role in the development of China’s economy and society (Bai et al. 2014; Shi and Yu 2014). The secondary effluent treatment of industrial park wastewater treatment plants (WWTPs) cannot achieve the receiving water body standard (“surface water environmental quality standard” (GB3838-2002) grade IV) because of a lack of effective advanced treatment technology. Different advanced treatment technologies have been investigated, including activated carbon adsorption (Pramanik et al. 2014, 2015), advanced oxidation processes (AOPs) (Zhao et al. 2014; James et al. 2014), ultrafiltration (UF) (Urtiaga et al. 2013), nanofiltration membranes (NF) (Aryal et al. 2015), and reverse osmosis (RO) (Yang et al. 2015; Meng et al. 2014); however, secondary effluent treatment for industrial park WWTPs requires high investment and maintenance costs. Among wastewater treatment technologies, CWs exhibit great advantages in terms of their low required investments and operation costs, high removal efficiency of N and P, and limited secondary pollution. The secondary effluent treatment of industrial park WWTPs is usually characterized by low biological degradability, high refractory organic matter, and variable water quality and quantity, which are difficult to effectively treat with single CWs. Nitrogen removal from the secondary effluent of industrial park wastewater becomes more difficult under severe conditions (such as a low COD/TN ratio). Therefore, a more effective integrated process should be constructed to accumulate an enhanced microbial community for better performance.
Microbial activities are recognized as a major contributor to the removal of wastewater nutrients, especially nitrogen. However, little is known of the critical roles played by various microbial communities, especially for the spatial evolution of microbial information in the combined processes (Caselles-Osorio and García 2006; Petitjean et al. 2012). Real-time PCR can effectively investigate functional gene density in the CWs but cannot reveal the associated microbial communities. High-throughput sequencing (HTS) techniques have shown great potential for analyzing microbial communities because of their unprecedented sequencing depth (Breitbart et al. 2009; DeLong et al. 2006); however, quantifying and determining correlations with other factors, such as dissolved oxygen (DO) and organic loads, are difficult with this technology. Thus, only a combination of multiple technologies can reveal detailed microbial information on CWs.
Therefore, we implemented two parallel pilot-scale VF-HF systems to investigate the treatment performance and microorganism mechanisms of CWs used to treat the secondary effluent from an industrial park WWTP near the Yangtze River in Suzhou City, China. Our objectives were to (i) determine the removal efficiencies of the VF-HF systems for the secondary effluent treatment of the industrial park WWTP and (ii) provide a better understanding of the spatial evolution of microbial community and the biological mechanisms of spatial nitrogen reduction in VF-HF systems.
Methods and materials
Experimental design
The experiment was set up at the industrial park WWTP (N 31°47′26.23″, E 120°47′50.97″) near the Yangtze River in Suzhou City, East China. The study site has a typical subtropical monsoon climate characterized by four distinct seasons (spring from March to May; summer from June to August; autumn from September to November; winter from December to February), a mild climate (average annual temperature of 15.4 °C), and abundant rainfall (average annual rainfall of 1054 mm).
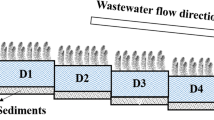
Outdoor mesocosms were set up as VF-HF systems. Polymethyl methacrylate VF tanks (0.49 m3: 70 cm × 70 cm × 100 cm) were filled with three layers of gravel (particle size: 0.5–3.0 mm; hydraulic conductivity: 0.068 cm/s; layer thickness: 75 cm) at the top and rock (particle size 8–16 mm; hydraulic conductivity: 15.77 cm/s; layer thickness: 15 cm) placed at the bottom (Fig. 1). Polymethyl methacrylate HF tanks (0.35 m3: 50 cm × 100 cm × 70 cm) were filled with one layer of gravel (particle size: 0.5–3.0 mm; hydraulic conductivity: 0.068 cm/s; thickness: 40 cm) (Fig. 1). Young (1 year old) reed plants (P. australis) were planted as a mono-species during spring in both the VF and HF tanks at a density of 16 strains/m2, and there were 8 strains in each tank. Immediately after the transplantations, the VF-HF systems were filled with the secondary effluent from the industrial park WWTP (Table 1). Both mesocosms were monitored from March 2013 to July 2014.
The secondary effluent from the industrial park WWTP was induced intermittently into the VF-HF systems each day to yield a hydraulic loading rate (HLR) of 100 mm/day during the start-up stage and operation period. The pH of secondary effluent ranged from 7.0 to 9.0, and the DO was about 6.0 mg/L.
Sample collection
Twelve sampling points along the VF-HF systems (Fig. 1) were used to investigate the spatial evolution of the pollutants, and these points were labeled Vinf, V1, V2, V3, V4, and Vefl in the VF system and Hinf, H1, H2, H3, H4, and Hefl in the HF system. The VF-HF systems influent and effluent were sampled approximately three times a week. Microorganism samples were collected from sampling points V1, V2, V3, V4, H1, H2, H3, and H4, and the biomass attached to the gravel was subjected to microbial community analysis and gene quantification.
The water samples were stored at −20 °C and transported in an ice box. The microorganism samples (biofilm attached to the substrates) were collected at the specified sites in the VF-HF systems and stored at −80 °C.
Water quality analysis
The chemical oxygen demand (COD), biological oxygen demand (BOD5), total nitrogen (TN), ammonium nitrogen (NH4 +-N), nitrate nitrogen (NO3 −-N), nitrite nitrogen (NO2 −-N), and total phosphorus (TP) were determined according to standard methods (APHA 2005).
The DO, pH, and total dissolved solids were recorded using oxygen meters (HQ30d, HACH, Loveland, Colorado, USA), pH meters (PHS-3D, Shanghai Precision & Scientific Instrument Co., Ltd., Shanghai, China), and total dissolved solids (TDS) meters (SX650, Shanghai Sanxin Instrument Factory, Shanghai, China).
Real-time PCR assays
The denitrifying bacteria were quantified by nirK gene-targeted real-time PCR, which codes a type of nitrite reductase with copper. Two primers, nirK876 (ATYGGCGGVAYGGCGA) and nirK1040 (GCCTCGATCAGRTTRTGGTT), were used. The conditions for nirK real-time PCR assays were 120 s at 50 °C and 900 s at 95 °C for enzyme activation. Six touchdown cycles were performed: 15 s at 95 °C, 30 s at 63 °C, and 30 s at 72 °C (also for the data acquisition step). The annealing temperature was progressively reduced by 1 °C. Finally, a cycle with an annealing temperature of 58 °C was repeated 40 times (Henry et al. 2004). Quantification of the AOB (nitrifying bacteria) was characterized by amoA genes targeted real-time PCR using the primers amoA-1 F (GGGGTTTCTACTGGTGGT) and amoA-2R (CCCCTCKGSAAAGCCTTCTTC) (Peralta et al. 2010). The experimental conditions were 900 s at 95 °C, 40 cycles of 15 s at 95 °C, 30 s at 63 °C, and 30 s at 72°. One last step from 60 to 95 °C with an increase of 0.2 °C/s was added to obtain a specific denaturation curve for all of the experiment.
Plasmids (pEASY-T1 Cloning Kit, Transtaq) containing cloned 16S genes (V3 region, 338f: CCTACGGGAGGCAGCAG,518r: ATTACCGCGGCTGCTGG), nirK genes, or amoA genes (which were cloned to DH5α before plasmid extraction) were used to draw standard curves (r 2 > 0.99 for all genes; amplification efficiencies were 100.4, 99.1, and 97.1 % for the 16S genes, nirK genes, and amoA genes, respectively).
High-throughput sequencing of the 16S rRNA gene
To better understand the spatial evolution of the bacterial communities along the VF-HF, MiSeq (HTS of the 16S rRNA gene) was conducted. After PCR amplification over 20 cycles, the sequencing and data analysis were performed by the Jiangsu Zhongyijinda Analytical & Testing Co., Ltd. The assemblage of species in each functional group was selected based on previous reports (Seviour and Nielsen 2010). Heat maps and cluster analyses were obtained using the R programming language (R 3.1.0). Typical genera that have known associations with denitrification were a focus of the calculations and analysis.
Statistical analysis
The statistical analysis was performed using the SPSS 17.0 software package for Windows. A two-sided t test was used to detect any significant differences in the mean influent and effluent values between different parameters as well as the pollutant removal efficiencies of the two wetland microcosms, with p < 0.05 defined as a significant difference according to the Pearson correlation analysis.
Results
Pollutant removal and nitrogen transformation
The VF-HF systems were implemented in June 2013 and became stable in August 2013 (approximately 3 months). During the operational period, the pollutant removal efficiency exhibited a positive correlation with temperature (seasonal variation) and presented a trend of summer > autumn > spring > winter. However, the effluents in all seasons could reach the “surface water environmental quality standard” (GB3838-2002) grade IV (COD, BOD5, NH3-N were 30 mg/L, 6 mg/L, 1.5 mg/L).
-
(1) COD removal
The mean influent concentration of COD was 35.9 mg/L, the mean effluent concentration of COD was 24.1 mg/L, and the mean removal rate of COD was 32.3 %. From the overall trend, COD removal was the first parameter to decrease, with the lowest values in winter and the highest values in summer (Fig. 2).
-
(2) Nitrogen removal and transformation
The mean influent concentration of NH4 +-N was 2.46 mg/L, the mean effluent concentration of NH4 +-N was 0.42 mg/L, and the mean removal rate of NH4 +-N was 82.3 % (Fig. 3), which is higher than the rates previously reported by Zhai et al. (2011) (71.7 %).
The mean TN removal efficiency was 69.8 %, which is higher than that previously reported by Ong et al. (2009) (60–67 %). Although different nature of wastewater as well as the CW configuration can influence removal performances, the VF-HF systems applied in this study exhibited satisfying nitrogen removal ability. Throughout the experimental period, the average effluent TN concentration was 1.34 mg/L (Fig. 3), the denitrifier density was much higher in the HF than in the VF, and the HF was responsible for 63.5 % of the TN removal.
-
(3) TP removal
As shown in Fig. 4, the mean removal efficiency of TP was 77.8 % and the mean effluent concentration was 0.02 mg/L. The most effect removal of P occurred with the quartz sand matrix, which is consistent with previous reports (Saeed and Sun 2011).
Spatial reduction of pollutants and distribution of functional genes
The spatial reduction of nitrogen and organics was further investigated by analyzing the removal efficiencies at different spatial sites of the VF-HF systems. Additionally, the microorganism abundance of different functional genes that play key roles in pollutant removal (Peralta et al. 2010) was also analyzed to reveal any interrelationships.
The best performance in summer for the entire VF-HF system was analyzed, and Fig. 5 presents the spatial reduction of COD, NH4 +-N, and TN and the functional gene abundance of the 16S, amoA, and nirK genes, which were used to approximate the total eubacteria, nitrifiers, and denitrifiers, respectively.
A reduction in the COD mainly occurred in the VF system, which accounted for 73.6 % of the total COD reduction. Moreover, the VF system contributed to 79.2 % of the NH4 +-N removal. This indicates that the VF system (especial for V1 and V2 parts) was significant in COD and NH4 +-N removal in the integrated CWs. Correspondingly, the HF system was significant in TN removal and accounted for 63.5 % of the TN removed. The removal of these pollutants was primarily related to microorganism effects, including the enrichment of total eubacteria, nitrifiers, and denitrifiers.
Furthermore, Fig. 5 indicates that correlations occurred among the abundance of different functional genes, the distance (along the water direction) and concentration of DO. In the VF, the eubacteria and nitrifiers were mainly distributed at the surface layer (V1 site), and this was most likely caused by the oxygen concentration in the CWs. The nitrifiers showed an especially high correlation with the DO concentration. However, the denitrifiers were mainly enriched in the HF, and the detailed correlation analysis is presented in discussion part.
Spatial evolution of the microbial community
In addition to the functional gene analysis, determining the spatial distribution of microbial communities is an effective method of revealing CW biodegradation mechanisms, which include the removal of nitrogen and organics. These spatially distributed samples were further analyzed by HTS of the 16S rRNA gene, which helped produce a greater understanding of the bacterial diversity in these systems compared with the results of previous studies that have utilized conventional molecular technologies (Ramond et al. 2012).
Figure S1 showed that the Proteobacteria, Bacteroidetes, and Actinobacteria were dominant category, which accounted for over 80 % in V1-V4. And the abundance of Proteobacteria was increased from V1 (36.19 %) to V4 (64.35 %). For HF, these three phyla also took up over 80 %, while the Proteobacteria abundance variation from H1 to H4 was not obvious.
Figure 6 shows the microbial community structures (heat map) in the VF-HF systems. Haliscomenobacter and Pseudomonas were the most dominant genera of the VF and HF systems and featured abundances of 5–10 % and 8–12 %, respectively. Obvious variation trends in the abundance of certain functional microbial communities were observed with distance, especially in the VF system. According to the cluster analysis, V1 and V2 were similar but exhibited great differences relative to V3 and V4 (similar results were observed in the HF system). The microbial community investigation and cluster analysis can help to distinguish function modules and present more coherent microbial information compared with the analysis of the entire system (Adrados et al. 2014).
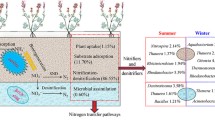
Although recent reports have applied HTS technology to reveal the microorganisms of CWs and produce accurate accounts of the dominant communities (Arroyo et al. 2015), analyses of the functional community groups in CW are lacking, and additional investigations must be performed to provide detailed information on the relationships of these groups with pollutant removal. In this study, the functional microorganisms were further classified into functional groups (Fig. 7), and their variation trends (abundance) were analyzed along with the influent. The MiSeq analysis identified a number of functional microorganisms (at the genus level) related to pollutant removal, including Aminobacter (related to ammoniation); Nitrosococcus, Nitrobacter, and Nitrospira (related to nitration); Acidovorax, Azoarcus, Rhodobacter, Thauera, etc. (related to denitrification); Rhizobium, Bradyrhizobium, etc. (related to fixation of nitrogen); Thiobacillus and Sulfurimonas (related to desulfuration); and Novosphingobium (related to polyaromatic hydrocarbon (PAH) degradation).
The concentration of PAH-degrading bacteria in the effluent (Fig. 7a, abundance as high as 1.4 %) indicated that PAH pollutants still remained in the tail water, which was because the water originated from an industrial park. Moreover, the PAH-degrading bacteria were distributed as follows: V1 < V2 < < V3 < V4. This order demonstrates that the PAHs were most likely degraded at the bottom of the VF system. Figure 7b shows the distribution of the Aminobacter community, and the highest abundance of 1.0 % was observed at site V4.
The nitrifiers were distributed as follows: V1 > V2 > V3 > V4 (Fig. 7c). The highest abundance of nitrifiers was 4.4 % at V1, which represents the oxygen-rich surface of the VF system (DO = 3.7 mg/L). The variation trend was similar to that observed by Wang et al. (2012); however, differences in the dominant nitrifiers were observed, with Nitrosomonas observed by Wang et al. and Nitrosococcus observed in this study. In addition, a higher biodiversity of nitrifiers was obtained in this study compared with that of Adrados et al. (2014), and this diversity indicated a higher potential for NH4 +-N removal. The higher abundance of nitrifiers in the system was partially caused by the optimal pH (7.41 ± 0.36) of the influent because nitrifiers are most active in a pH range of 7.0–7.8 (Liang et al., 2003).
The highest denitrifier abundance was approximately 18 % (Fig. 7d), which was much higher than the abundance observed in natural wetlands (Peralta et al. 2010). This result also indicated that the VF-HF systems achieved enhanced nitrogen removal over long periods of microorganism enrichment.
Discussion
Correlation analysis between pollutant reduction and microorganism communities in the VF-HF system
Microorganisms play a key role in reducing pollutants in CWs, and correlation analyses between microorganisms and other factors within CWs may help evaluate and estimate the potential of these environments, which has recently earned increased attention. Höfferle et al. (2010) demonstrated that nitrifying genes exhibited a significant correlation with CW depth. Peralta et al. (2010) confirmed that the microbial communities of CWs exhibited a great correlation to the C/N ratio. However, a more thorough analysis of the relationship among the CW spatial location, environmental factors, and microorganisms has not been performed.
Figure 5 shows the variation of functional genes along with certain environmental factors, such as distance (water direction) and DO level.
The Pearson correlation analysis showed that in the VF system, the 16S gene and nirK gene were significantly correlated with depth (r = 0.995, p < 0.01 for 16S, and r = 0.97, p < 0.05 for nirK gene), which indicates that the ability to remove COD and TN varies gradually with the increasing depth. However, the amoA gene did not show a significant correlation with the depth, which is inconsistent with the results of a previous study (Höfferle et al. 2010). Instead, the abundance of the amoA gene, which indicated an enhanced NH4 +-N removal ability, varied sharply from the surface to deeper sites in the VF system, which is shown in Fig. 5c.
In addition, the VF system was typically characterized by a good oxygen supply (Saeed and Sun 2012), which was significantly correlated to microorganism activity as well as the removal of organics and nitrogen. The 16S gene exhibited a highly significant correlation with the DO concentrations (r = 0.999, p < 0.001), which indicates that the DO most likely determined the total growth rate of eubacteria and removal of COD. The nitrogen removal-related genes did not show significant correlations with the DO concentration, which indicates that nitrogen removal in the VF system was determined by factors other than DO, such as the NH4 +-N concentration (Sims et al. 2012).
For the HF system, the amoA gene abundance exhibited a more significant correlation with distance (along the water direction, r = 0.954, p < 0.05) relative to the nirK gene, which had a greater likelihood of being affected by the location of plants (Chen et al. 2014). Moreover, the 16S gene showed a more significant correlation to the DO (r = 0.976, p < 0.05) than did the amoA and nirK genes, which is similar to the results for the VF system. This result also confirmed that the nitrogen removal rate was reliant on the DO level as well as other factors, such as the rate of nitrogen loading and effect of plant roots.
Therefore, nitrogen removal, which was enhanced in the VF-HF systems, was dependent on DO concentrations as well as mechanisms that are more complex and require further discussion.
Enhanced nitrogen removal in the VF-HF system
The combination of “VF-HF” systems has been characterized by a high nitrogen removal ability (Saeed and Sun 2012). Our integrated VF-HF system showed a much higher nitrogen removal efficiency relative to other studies on industrial wastewater (Table 2), which indicates that the VF-HF system in this study was enhanced. The lower COD of the influent and the C/N ratio caused low COD removal efficiencies in this study (Table 2); however, this result confirmed the high nitrogen removal rate when presented with a low carbon source.
According to the performance difference between the start-up period (no microorganism participation) and stable operational period (with microorganisms participation, Fig. 3), the microorganisms play the key role in pollutants removal, especially for the enhanced nitrogen removal, and the four functional groups in Fig. 7 are related to nitrogen removal in the VF-HF systems, and they all have relatively high abundance.
It can be observed that the nitrifier mainly distributed at the surface of the system, and its abundance gradually decreased in the VF system but slightly decreased in the HF system, which was mainly caused by DO variations in the systems (Fig. 5). However, the abundance of denitrifiers gradually increased in both the VF and HF systems, which mainly resulted from the changes in the DO and organics concentrations (produced by hydrolysis bacteria).
Saeed and Sun (2012) reviewed the biological nitrogen removal mechanisms of CWs, including ammonification, nitrification, denitrification, and other recently discovered effective nitrogen removal routes, such as partial nitrification-denitrification and anammox. In the study presented here, a limited community was observed for the newly effective routes, although this community improved the nitrogen removal efficiency because it contained PAH-degrading bacteria. Figure 7a shows that the abundance of Novosphingobium (related to PAH degradation) was as high as 1.4 % at the bottom of the VF system. These types of bacteria are seldom found in CWs (Zhao et al. 2011) but were especially enriched in the VF system in this study (Fig. 7a), which indicates that certain PAHs remained in the tail wastewater and suggests that our VF-HF systems became adapted to the wastewater after a long period of operation. These bacteria degraded the nonbiodegradable pollutants into smaller compounds that were more easily utilized by microorganisms. Therefore, based on the VF component, the BOD5/COD ratio (B/C) was elevated from 0.12 to 0.27, which was not only convenient for COD removal but also enhanced nitrogen removal because a superior carbon resource was provided for denitrification.
Figure 5b, c shows that the VF system was critical to nitrification, and the HF system was primarily responsible for denitrification. According to the cluster analysis and the denitrifying bacteria abundance, the entire system can be divided into two functional modules. V1 and V2 can spatially be considered the “nitrifying modules,” and they promote the B/C ratio for improved denitrification, whereas V3, V4, and H1-4 can be regarded as the “denitrifying modules.” Each module performs its own functions with higher efficiency. Divisions based on the community structure are more scientific than divisions based on the spatial structure because specific types of microorganisms played important roles in removing pollutants from the VF and HF systems.
Conclusions
Mean removal efficiencies of 82.3, 69.8, 77.8, and 32.3 % were achieved for NH4 +-N, TN, TP, and COD, respectively, by the VF-HF system, which treated the secondary effluent of an industrial park WWTP at a loading rate of 100 mm/day.
The spatial distribution of the functional gene abundance was correlated with the distance and DO level of the VF-HF systems. In the VF system, the 16S gene and the nirK gene were significantly correlated with depth, and the 16S gene exhibited a highly significant correlation with the DO level (r = 0.954, p < 0.05).
The microbial community analysis at different spatial locations showed a high abundance of nitrifiers and denitrifiers and certain B/C improvement bacteria, and these communities accounted for approximately 90 % of the nitrogen removal in the VF-HF system.
References
Abira MA, van Bruggen JJA, Denny P (2005) Potential of a tropical subsurface constructed wetland to remove phenol from pre-treated pulp and paper mill wastewater. Water Sci Technol 51:173–176
Adrados B, Sánchez O, Arias CA, Becares E, Garrido L, Mas J, Brix H, Morató J (2014) Microbial communities from different types of natural wastewater treatment systems: vertical and horizontal flow constructed wetlands and biofilters. Water Res 55:304–312
Anastasiou N, Monou M, Mantzavinos D, Kassinos D (2009) Monitoring of the quality of winery influents/effluents and polishing of partially treated winery flows by homogenous Fe(II) photo-oxidation. Desalination 248:836–842
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington, DC
Arroyo P, de Miera LES, Ansola G (2015) Influence of environmental variables on the structure and composition of soil bacterial communities in natural and constructed wetlands. Sci Total Environ 506:380–390
Aryal A, Sathasivan A, Heitz A, Zheng G, Nikraz H, Ginige MP (2015) Combined BAC and MIEX pre-treatment of secondary wastewater effluent to reduce fouling of nanofiltration membranes. Water Res 70:214–223
Bai L, Qiao Q, Yao Y, Guo J, Xie MH (2014) Insights on the development progress of National Demonstration eco-industrial parks in China. J Clean Prod 70:4–14
Breitbart M, Hoare A, Nitti A, Siefert J, Haynes M, Dinsdale E, Edwards R, Souza V, Rohwer F, Hollander D (2009) Metagenomic and stable isotopic analyses of modern freshwater microbialites in cuatro CiEnegas, Mexico. Environ Microbiol 11(1):16–34
Brix H (1994a) Use of constructed wetlands in water pollution control: historical development, present status, and future perspectives. Water Sci Technol 30:209–223
Brix H (1994b) Functions of macrophytes in constructed wetlands. Water Sci Technol 29:71–78
Bulc TG, Ojstršek A, Vrhovšek D (2006) The use of constructed wetland for textile wastewater treatment. MAOTDR 2006, Lisbon
Calheiros CSC, Quitério PVB, Silva G, Crispim LFC, Brix H, Moura SC, Castro PML (2012) Use of constructed wetland systems with Arundo and Sarcocornia for polishing high salinity tannery wastewater. J Environ Manag 95:66–71
Caselles-Osorio A, García J (2006) Performance of experimental horizontal subsurface flow constructed wetlands fed with dissolved or particulate organic matter. Water Res 40(19):3603–3611
Chapple M, Cooper P, Cooper D, Revitt M (2002) Pilot trials of a constructed wetland system for reducing the dissolved hydrocarbon in the runoff from a decommissioned refinery. University of Dar-es-Salaam, Tanzania
Chen TY, Kao CM, Yeh TY, Chien HY, Chao AC (2006) Application of a constructed wetland for industrial wastewater treatment: a pilot study. Chemosphere 64:497–502
Chen Y, Wen Y, Zhou Q, Vymazal J (2014) Effects of plant biomass on denitrifying genes in subsurface-flow constructed wetlands. Bioresour Technol 157:341–345
Choudhary AK, Kumar S, Sharma C (2010) Removal of chlorinated resin and fatty acids from paper mill wastewater through constructed wetland. World Acad Sci Eng Technol 80:67–71
Comino E, Riggio V, Rosso M (2011) Mountain cheese factory wastewater treatment with the use of a hybrid constructed wetland. Ecol Eng 37:1673–1680
Cooper PF (1999) A review of the design and performance of vertical-flow and hybrid reed bed treatment systems. Water Sci Technol 40(3):1–9
Davies LC, Carias CC, Novais JM, Martins-Dias S (2005) Phytoremediation of textile effluents containing azo dye by using Phragmites australis in a vertical flow intermittent feeding constructed wetland. Ecol Eng 25:594–605
De Jong J (1976) In: Tourbier J, Pierson RW (eds) Biological control of water pollution. Pennsylvania University Press, Philadelphia
DeLong EF, Preston CM, Mincer T, Rich V, Hallam SJ, Frigaard NU, Martinez A, Sullivan MB, Edwards R, Brito BR, Chisholm SW, Karl DM (2006) Community genomics among stratified microbial assemblages in the ocean’s interior. Science 311(5760):496–503
Dos Santos AB, Cervantes FJ, van Lier JB (2007) Review paper on current technologies for decolourisation of textile wastewater: perspectives for anaerobic biotechnology. Bioresour Technol 98:2369–2385
Gasiunas V, Strusevicius Z, Struseviciene MS (2005) Pollutant removal by horizontal subsurface flow constructed wetlands in Lithuania. J Environ Sci Health 40A:1467–1478
Greenway M (1997) Nutrient content of wetland plants in constructed wetlands receiving municipal effluent in tropical australia. Water Sci Technol 35(5):135–142
Hadad HR, Maine MA, Bonetto CA (2006) Macrophyte growth in a pilot scale constructed wetland for industrial wastewater treatment. Chemosphere 63:1744–1753
Hamouri BE, Nazih J, Lahjouj J (2007) Subsurface-horizontal flow constructed wetland for sewage treatment under Moroccan climate conditions. Desalination 215(13):153–158
Henry S, Baudoin E, Lopez-Gutierre JC (2004) Quantification of denitrifying bacteria in soils by nirK gene targeted real-time PCR. J Microbiol Methods 59:327–335
Höfferle Š, Nicol GW, Pal L, Hacin J, Prosser JI (2010) Mandić-Mulec I. Ammonium supply rate influences archaeal and bacterial ammonia oxidizers in a wetland soil vertical profile. FEMS Microbiol Ecol 74:302–315
James CP, Germain E, Judd S (2014) Micropollutant removal by advanced oxidation of microfiltered secondary effluent for water reuse. Sep Purif Technol 127:77–83
Kadlec RH, Wallace SD (2009) Treatment wetlands, 2nd edn. CRC Press, Boca Raton
Liang W, Wu ZB, Cheng SP, Zhou QH, Hu HY (2003) Roles of substrate microorganisms and urease activities in wastewater purification in a constructed wetland system. Ecol Eng 21:191–195
Meng M, Pellizzari F, Boukari SO, Leitner NKV, Teychene B (2014) Impact of e-beam irradiation of municipal secondary effluent on MF and RO membranes performances. J Membr Sci 471:1–8
Merlin G, Pajean JL, Lissolo T (2002) Performances of constructed wetlands for municipal wastewater treatment in rural mountainous area. Hydrobiologia 469(1/3):87–98
Ong SA, Uchiyama K, Inadama D (2009) Simultaneous removal of color, organic compounds and nutrients in azo dye-containing wastewater using up-flow constructed wetland. J Hazard Mater 165:696–703
Peralta AL, Matthews JW, Kent AD (2010) Microbial community structure and denitrification in a wetland mitigation bank. Appl Environ Microbiol 76:4207–4215
Petitjean A, Forquet N, Wanko A, Laurent J, Molle P, Mosé R (2012) Modelling aerobic biodegradation in vertical flow sand filters: impact of operational considerations on oxygen transfer and bacterial activity. Water Res 46(7):2270–2280
Pramanik BK, Roddick FA, Fan L (2014) Effect of biological activated carbon pre-treatment to control organic fouling in the microfiltration of biologically treated secondary effluent. Water Res 63:147–157
Pramanik BK, Roddick FA, Fan L, Jeong S, Vigneswaran S (2015) Assessment of biological activated carbon treatment to control membrane fouling in reverse osmosis of secondary effluent for reuse in irrigation. Desalination 364:90–95
Ramond JB, Welz PJ, Cowan DA, Burton SG (2012) Microbial community structure stability, a key parameter in monitoring the development of constructed wetland mesocosms during start-up. Res Microbiol 163:28–35
Rochard J, Ferrier VM, Kaiser A, Salomon N (2002) The application of constructed wetlands in the viticultural sector: experimentation on a winery effluent treatment device. University of Dar-es-Salaam, Tanzania
Saeed T, Sun GZ (2011) A comparative study on the removal of nutrients and organic matter in wetland reactors employing organic media. Chem Eng J 171:439–447
Saeed T, Sun GZ (2012) A review on nitrogen and organics removal mechanisms in subsurface flow constructed wetlands: dependency on environmental parameters, operating conditions and supporting media. J Environ Manag 112:429–448
Seviour RJ, Nielsen PH (2010) Microbial communities in activated sludge plants. Microbial Ecology of Activated Sludge. IWA Publishing, London, pp 95–126
Sharma KP, Sharma S, Sharma S, Singh PK, Kumar S, Grover R, Sharma PK (2007) A comparative study on characterization of textile wastewaters (untreated and treated) toxicity by chemical and biological tests. Chemosphere 69:48–54
Shi L, Yu B (2014) Eco-industrial parks from strategic niches to development mainstream: the cases of China. Sustain Sci 6:6325–6331
Sims A, Horton J, Gajaraj S, McIntosh S, Miles RJ, Mueller R, Hu Z (2012) Temporal and spatial distributions of ammonia-oxidizing archaea and bacteria and their ratio as an indicator of oligotrophic conditions in natural wetlands. Water Res 46:4121–4129
Trang NTD, Konnerup D, Schierup HH, Chiem NH, Tuan LA, Brix H (2010) Kinetics of pollutant removal from domestic wastewater in a tropical horizontal subsurface flow constructed wetland system: effects of hydraulic loading rate. Ecol Eng 36(4):527–535
Urtiaga AM, Pérez G, Ibáñez R, Ortiz I (2013) Removal of pharmaceuticals from a WWTP secondary effluent by ultrafiltration/reverse osmosis followed by electrochemical oxidation of the RO concentrate. Desalination 331:26–34
Vymazal J (2001) In: Vymazal J (ed) Transformations on nutrients in natural and constructed wetlands. Leiden, Netherlands, Backhuys Publishers
Vymazal J (2005a) Constructed wetlands for wastewater treatment. Ecol Eng 25:475–477
Vymazal J (2005b) Horizontal sub-surface flow and hybrid constructed wetlands systems for wastewater treatment. Ecol Eng 25:478–490
Vymazal J (2009) Horizontal sub-surface flow constructed wetlands Ondřejov and Spálené Poříčí in the Czech Republic—15 years of operation. Desalination 246:226–237
Vymazal J (2013) The use of hybrid constructed wetlands for wastewater treatment with special attention to nitrogen removal: a review of a recent development. Water Res 47:4795–4811
Vymazal J (2014) Constructed wetlands for treatment of industrial wastewaters: a review. Ecol Eng 73:724–751
Wang Y, Sheng HF, He Y (2012) Comparison of the levels of bacterial diversity in freshwater, intertidal wetland, and marine sediments by using millions of illumina tags. Appl Environ Microbiol 78:8264–8271
Yang C, Li L, Shi J, Long C, Li A (2015) Advanced treatment of textile dyeing secondary effluent using magnetic anion exchange resin and its effect on organic fouling in subsequent RO membrane. J Hazard Mater 84:50–57
Zhai J, Xiao HW, Kujawa-Roeleveld K, He Q, Kerstens SM (2011) Experimental study of a novel hybrid constructed wetland for water reuse and its application in Southern China. Water Sci Technol 64:2177–2184
Zhang T, Xu D, He F, Zhang Y, Wu ZB (2012) Application of constructed wetland for water pollution control in China during 1990-2010. Ecol Eng 47:189–197
Zhao X, Hu HY, Yu T, Su C, Jiang H, Liu S (2014) Effect of different molecular weight organic components on the increase of microbial growth potential of secondary effluent by ozonation. J Environ Sci 26:2190–2197
Zhao YJ, Hui Z, Chao X, Nie E, Li HJ, He J, Zheng Z (2011) Efficiency of two-stage combinations of subsurface vertical down-flow and up-flow constructed wetland systems for treating variation in influent C/N ratios of domestic wastewater. Ecol Eng 37:1546–1554
Zupančič Justin M, Vrhovšek D, Stuhlbacher A, Griessler Bulc T (2009) Treatment of wastewater in hybrid constructed wetland from the production of vinegar and packaging detergents. Desalination 246:100–109
Acknowledgments
The authors wish to thank the Jiangsu Province Key Laboratory of Environmental Engineering (Grant No.ZX2014001) and the Advanced Treatment and Reclamation of Eutrophic Pollutants in Park Industrial Wastewater Technology and Demonstration (2012ZX07101-003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gerald Thouand
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 1911 kb)
Rights and permissions
About this article
Cite this article
Xu, M., Liu, W., Li, C. et al. Evaluation of the treatment performance and microbial communities of a combined constructed wetland used to treat industrial park wastewater. Environ Sci Pollut Res 23, 10990–11001 (2016). https://doi.org/10.1007/s11356-016-6181-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6181-8