Abstract
Heavy metal (Cd, Cr, Cu, Fe, Mn, Ni, Pb and Zn) levels in red fox small intestine samples with or without Echinococcus multilocularis infection were studied. The red foxes were taken from the open countryside of northwest Bohemia (CR). Red foxes with E. multilocularis infection had lower levels of toxic metals (Cd, Pb); cadmium levels in infected foxes (0.0052 mg/kg) were twice as low as in uninfected foxes (0.0106 mg/kg). This was the same case for lead: 0.0288 mg/kg infected red foxes (inf.) and 0.0413 mg/kg uninfected (uninf.). Conversely, red foxes with E. multilocularis infection yielded higher concentrations in comparison to their uninfected counterparts: Cr (0.0087 mg/kg uninf. and 0.0116 mg/kg inf.), Cu (0.2677 mg/kg uninf. and 0.3205 mg/kg inf.), Fe (6.46 mg/kg uninf. and 10.89 mg/kg inf.), Mn (0.1966 mg/kg uninf. and 0.2029 mg/kg inf.), Ni (0.0415 mg/kg uninf. and 0.064 mg/kg inf.) and Zn (16.71 mg/kg uninf. and 20.25 mg/kg inf). This could support the hypothesis that tapeworms are able to absorb toxic heavy metals from the host body into their tissues, as well as to modify other element concentrations in the host body.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The red fox (Vulpes vulpes Linnaeus, 1758) is a representative of the canid family. It is widely distributed in the Northern Hemisphere, and it is the most abundant wild carnivore living in the territory of the Czech Republic. Annual captures in the Czech Republic currently range between 60,000 and 90,000 foxes, and populations continue to grow (Červený et al. 2004). The increase in the distribution and density of red fox (V. vulpes) in most European countries could be explained by a reduction in the mortality rate, which is due to an intensive campaign of vaccination against rabies (oral baits) as well as to the opportunist behaviour of the red fox (Hanosset et al. 2008; Červený et al. 2004). The red fox is a common host of the tapeworm Echinococcus multilocularis; this parasite is the causative agent of alveolar echinococcosis, a zoonotic parasitic disease that causes a severe hepatic disorder in humans (Letková et al. 2006). Several intestinal helminths are able to accumulate considerable concentrations of heavy metals (Sures et al. 2000, 2002, 2003). Because very few comparative studies on heavy metal concentrations in tissues of infected and uninfected hosts are available, it remains unclear whether conspicuous metal accumulation by parasitic worms affects the metal levels in the tissues of a definitive host. This information is also very important with respect to the use of the tissues of these animal tissues in monitoring of environmental pollution.
Hypothesis: The ability of a tapeworm to absorb toxic heavy metals from a host intestine can alter (decrease) those element concentrations in the host small intestine. Conversely, tapeworm presence can increase other elements (Cr, Cu, Fe, Mn, Ni and Zn) that are essential for the functioning of a number of immune system components.
Material and methods
Field research was conducted in northwest Bohemia near the city of Karlovy Vary. We examined a total of 48 red foxes collected in the winter of 2010–2011. The majority of animals were shot in the course of population control programmes. The carcasses were wrapped in plastic bags and stored at −20 °C until further examination. During the necropsy, the animals were sexed and their ages were evaluated. All of the animals used in this experiment were adults over 2 years old. The entire gut was removed and kept for at least 3 weeks at −78 °C in order to inactivate the infective material. The animals were dissected in the laboratory, and the presence of E. multilocularis was determined. Experimental material was obtained from a total of 12 red foxes (6 females and 6 males); 6 (3 females and 3 males) were infected with E. multilocularis alone (mean intensity of infection was about 1000 specimens per red fox) and 6 uninfected fox (3 females and 3 males). Samples of the small intestine were put into deep freeze until posterior processing for chemical analysis.
Determination of trace elements in animal tissues and parasites
The specific levels of cadmium (Cd), chromium (Cr), copper (Cu), iron (Fe), lead (Pb), manganese (Mn), nickel (Ni) and zinc (Zn) were determined in digested samples of the small intestines of red foxes by atomic absorption spectroscopy (AAS).
Frozen samples were dried by the lyophilisation using the LYOVAC GT 2 (LEYBOLD-HERAEUS, GmbH, Germany) and microwave digested in an acid solution using MWS-3+ (Berghof Products + Instruments, Germany) as described by Jankovska et al. (2011).
The concentrations of lead, cadmium, chromium and nickel in the digests were measured by ETAAS technique using Varian AA 280Z (Varian, Australia) with graphite tube atomizer GTA 120 and PSD 120 programmable sample dispenser. The concentrations of Mn, Fe and Zn in the same digests were measured by FAAS using Varian AA 110 (Varian, Australia). Standard solutions ASTASOL (Analytika, CR) of Cd, Pb, Cr, Ni, Fe, Mn and Zn were used in the preparation of calibration curves for the measurements.
The quality of analytical data was assessed by simultaneous analysis of certified reference material CRM 12-02-01 (Bovine Liver) (4 % of all the samples). Analytical data obtained for all determined elements were found in the confidence interval given by the producer of the CRM for this element. The background of the trace element laboratory was monitored by analysis of 17.5 % blanks prepared under the same conditions, but without samples, and experimental data were corrected by mean concentration of the elements in blanks and compared with detection limit (mean ±3 SD of blanks) which were 0.2 ng ml−1 for Pb, 0.08 ng ml−1 for Cd, 0.17 ng ml−1 for Cr, 0.21 ng ml−1 for Ni, 3.2 ng ml−1 for Cu, 2.7 ng ml−1 for Mn, 10.4 ng ml−1 for Fe and 7.5 ng ml−1 for Zn.
Statistical evaluation
Basic descriptive statistics were computed. The normality of the data was tested separately using a Shapiro-Wilk test. Considering the results of the normality test, a nonparametric Mann-Whitney U test was used for the evaluation of differences between males and females. Statistica ver. 12 (StatSoft, Inc 2013) was used for all statistical analyses.
Results and discussion
Cadmium is a widespread pollutant, largely increasing as a result of mining, smelting and burning activities. Unlike organic compounds, Cd is not biodegradable and has a very long biological half-life (Xuan et al. 2014). Red foxes with E. multilocularis infection had lower concentrations of toxic metals (Cd and Pb) in small intestine tissues than uninfected fox (Fig. 1); this could support the hypothesis that tapeworms absorb toxic heavy metals from the host body into their tissues.
Box plots illustrate the differences between concentrations of toxic metals (Pb and Cd) in the small intestinal tissues of foxes infected with tapeworms (“presence”) and those of foxes without tapeworms (“absence”). The graph presents the following values: black mark median, box 25–75 %, whiskers min-max
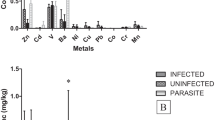
Conversely, red foxes with E. multilocularis infection yielded higher concentrations of Cr, Cu, Fe, Mn, Ni and Zn (Fig. 2).
Heavy metals are continuously being added to soils through various agricultural and industrial activities, which pose potential threat on food safety and health risks (Zhang et al. 2014). Chronic ingestion of environmental heavy metals is associated with the occurrence of various diseases. Intestinal absorption of heavy metals leads to accumulation in specific target organs, with severe detrimental effects on health. High concentrations of nonabsorbed heavy metals remain in the gut microenvironment, where they may have a direct impact on the gut ecosystem and its overall physiology. The gut microbiota has been described as a complex hidden organ, which plays a key role in the maintenance of health (Breton et al. 2013). Kazi et al. (2014) report that nutrition may play a role in reducing the adverse effects of Pb. According to our results, tapeworms in the small intestine may also play an important role in reducing toxic metals. In our experiment, red foxes with E. multilocularis infection had lower levels of toxic metals (Cd and Pb); cadmium levels in infected foxes were twice as low (0.0052 mg/kg) as in uninfected foxes (0.0106 mg/kg). This was the same case for lead: 0.0288 mg/kg infected red foxes (inf.) and 0.0413 mg/kg uninfected (uninf.; Fig. 1). Conversely, red foxes with E. multilocularis infection yielded higher concentrations in comparison to their uninfected counterparts: Cr (0.0087 mg/kg uninf. and 0.0116 mg/kg inf.), Cu (0.2677 mg/kg uninf. and 0.3205 mg/kg inf), Fe (6.46 mg/kg uninf. and 10.89 mg/kg inf.), Mn (0.1966 mg/kg uninf. and 0.2029 mg/kg inf.), Ni (0.0415 mg/kg uninf. and 0.064 mg/kg inf.) and Zn (16.71 mg/kg uninf. and 20.25 mg/kg inf.; Fig. 2). Our values were much lower than the maximum permissible values for the environment (soil) and food (muscle) in the Czech Republic and EU countries.
Therefore, these findings are essential when carrying out environmental pollution monitoring analysis, which must take into consideration important factors such as animal sex (Jankovská et al. 2014) and intestinal helminth presence (Sures et al. 1999; Sures et al. 2003; Sures 2001, 2004; Jankovska et al. 2010). In this study, there were no differences between male and female red fox heavy metal accumulation. Intestinal helminth presence is an important factor because one hypothesis (Sures et al. 1999) states that parasitic helminths without a digestive tract (Cestoda, Acanthocephala) and with a metabolic active body surface–tegument accumulate toxic metals (mainly Pb and Cd) to a higher degree than do the host tissues (Sures and Siddall 1999; Sures 2001; Sures et al. 2003; Jankovska et al. 2012).
We tried to analyse heavy metal concentrations in E. multilocularis tapeworms. However, the correct determination of elements in E. multilocularis was impossible due to the very small magnitude of the parasites (approximately 1 mm). If parasite mass is less than 1–2 mg dry weight, element concentrations are very difficult to detect using analytical methods. Nevertheless, we found the following heavy metal concentrations (mg/kg dry weight) in fox tapeworms from our previous analyses: Cr 0.140, Ni 0.827, Zn 354.0, Cd 0.427, Cu 36.03, Mn 87.54 and Pb 45.60 mg/kg dry weight. These values in fox tapeworms were 12.1, 12.9, 17.5, 82.1, 112.4, 431.4 and 1583.3 times higher than those in fox intestinal tissue for Cr, Ni, Zn, Cd, Cu, Mn and Pb, respectively. Because very few comparative studies on heavy metal concentrations in tissues of infected and uninfected hosts are available, it remains unclear whether conspicuous metal accumulation by parasitic worms affects the metal levels in the tissues of a definitive host.
Jankovska et al. (2010) tested the reliability of the cestode/red fox (V. vulpes) model as another bioindicator for heavy metal pollution detection under field (forest) conditions. These species were chosen since there are no other models that utilise carnivora and their parasites in environmental monitoring. The impact of the parasite burden on bioaccumulation of heavy metals in target organs of red foxes (the liver and kidneys) was studied (Jankovska et al. 2010); in that study, the accumulation of heavy metals in red fox parasites was accompanied by a significant lead content decrease in the host kidneys when compared to the host kidneys of noninfected animals. The infection of red fox (V. vulpes) by parasitic helminths was reflected in a significant increase of copper and manganese in the host livers in comparison with the nonparasitized group.
Trace elements are essential for health and immunity. Kilic et al. (2010) investigated the changes in the total content of the essential element (copper) in patients infected with Giardia intestinalis. The mean concentration of copper in serum was significantly higher in G. intestinalis-positive subjects than in their controls; this difference was evident in both females (p < 0.05) and males (p < 0.05). No correlation could be demonstrated between age and copper mean value in G. intestinalis-positive females and males (p > 0.05). When compared to control groups, copper levels clearly increased in patients infected with G. intestinalis. It is suggested that animals with parasite infection can have higher concentrations of essential elements because those elements positively influence the functioning of various immune system components (Yatoo et al. 2013). For example, zinc is an essential element for the maturation of T lymphocyte cells. The mammalian spleen is an important component of the immune system; it helps the body to resist parasites and induces immune responses by B and T lymphocytes. E. multilocularis growth appears to be controlled by the host T cell, and, at least in the early phase of the disease, T lymphocytes probably play the most important role in the immune response to the parasite (Hegglin 2003).
Our present results also confirm higher Cr, Cu, Fe, Mn, Ni and Zn concentrations and lower Cd and Pb concentrations in small intestine tissues of foxes with E. multilocularis infection. Unfortunately, the only statistically significant difference between element concentrations in foxes with tapeworms and those without tapeworms was with respect to iron (Fe). In all other cases, element concentration differences were statistically insignificant. Nevertheless, as seen in the box plots (Figs. 1 and 2), certain differences are evident. The reason these differences are insignificant could be due to the low number of animals killed for this analysis.
Trace elements act as cofactors of enzymes, which are necessary for an animal’s immunity (Yatoo et al. 2013). One possible reason for higher essential metal concentrations in infected red foxes is activation of the immune system due to tapeworm presence. These essential metals are contained in proteins, which take part in the immune system response. Primarily, zinc plays important role in anti-inflammatory activity which preserves the intestinal structure which is disrupted by the presence of parasites.
The mechanism whereby cestode-infected animals accumulate less toxicants than do their parasite-free counterparts is unknown; however, this may be (among other factors) partially due to their lower metabolic rate (Bergey et al. 2002). Our results can also suggest altered element metabolism due to pathophysiologic changes in intestinal parasite infection. Detailed biochemical and molecular studies on large samples are needed to clarify the relationship between intestinal parasitic diseases and element level changes in intestinal tissues.
Our present results also could support the hypothesis that tapeworms absorb toxic heavy metals from the host body into their tissues. The results of this study should always be taken into consideration when conducting environmental monitoring analysis.
References
Bergey L, Weis JS, Weis P (2002) Mercury uptake by the estuarine species Palaemonetes pugio and Fundulus heteroclitus compared with their parasites, Probopyrus pandalicola and Eustrongylides sp. Mar Pollut Bull 44(10):1046–1050
Breton J, Massart S, Vandamme P, De Brandt E, Pot B, Foligne B (2013) Ecotoxicology inside the gut: impact of heavy metals on the mouse microbiome. BMC Pharmacol Toxicol 14:62
Červený J, Kamler K, Kholová H, Koubek P, Martínková M (2004) Encyklopedie myslivosti (Hunting encyclopedy). Ottovo nakladatelství-Cesty, Praha, p 591 (in Czech)
Hanosset R, Saegerman C, Adant S, Massart L, Losson B (2008) Echinococcus multilocularis in Belgium: prevalence in red foxes (Vulpes vulpes) and in different species of potential intermediate hosts. Vet Parasitol 151:212–217
Hegglin D (2003) The fox tapeworm (Echinococcus multilocularis) and the red fox (Vulpes vulpes) in the urban habitat: ecological and epidemiological aspects and an evaluation of an intervention strategy. Mathematish-naturwissenschaftlichen Fakultat der Universitat Zurich, Dissertation, 229 pp
Jankovska I, Miholova D, Bejcek V, Vadlejch J, Sulc M, Szakova J, Langrova I (2010) Influence of parasitism on trace element contents in tissues of red fox (Vulpes vulpes) and its parasites Mesocestoides spp. (Cestoda) and Toxascaris leonina (Nematoda). Arch Environ Contam Toxicol 58:469–477
Jankovska I, Miholova D, Petrtyl M, Romocusky S, Kalous L, Vadlejch J, Cadkova Z, Langrova I (2011) Intestinal parasite Acanthocephalus lucii (Acanthocephala) from European perch (Perca fluviatilis) as a bioindicator for lead pollution in the stream “Jevansky potok” near Prague, Czech Republic. Bull Environ Contam Toxicol 86:342–346
Jankovska I, Miholova D, Lukesova D, Kalous L, Valek P, Romocusky S, Vadlejch J, Petrtyl M, Langrova I, Cadkova Z (2012) Concentrations of Zn, Mn, Cu and Cd in different tissues of perch (Perca fluviatilis) and in perch intestinal parasite (Acanthocephalus lucii) from the stream near Prague (Czech Republic). Environ Res 112:83–85
Jankovská I, Miholova D, Romočuský Š, Petrtyl M, Langrova I, Kalous L, Sloup V, Valek P, Vadlejch J, Lukešová D (2014) Importance of fish gender as a factor in environmental monitoring of mercury. Environ Sci Pollut Res Int 21:6239–6242
Kazi TG, Shah F, Shaikh HR, Afridi HI, Shah A, Naeemullah ASS (2014) Exposure of lead to mothers and their new born infants, residents of industrial and domestic areas of Pakistan. Environ Sci Pollut Res Int 21:3021–3030
Kilic E, Saraymen R, Miman O, Yazar S (2010) Evaluation of serum copper level during Giardia intestinalis infection. Afr J Microbiol Res 4:1013–1015
Letková V, Lazar P, Urlík J, Goldová M, Kocišová A, Košuthová L (2006) The red fox (Vulpes vulpes L.) as a source of zoonoses. Vet Arch 76:S73–S81
StatSoft, Inc. (2013). STATISTICA (data analysis software system), version 12. www.statsoft.com
Sures B (2001) The use of fish parasites as bioindicators of heavy metals in aquatic ecosystems: a review. Aquat Ecol 35:245–255
Sures B (2004) Environmental parasitology: relevancy of parasites in monitoring environmental pollution. Trends Parasitol 20:170–177
Sures B, Siddall R (1999) Pomphorhynchus laevis: the intestinal acanthocephalan as a lead sink for its fish host, chub (Leuciscus cephalus). Exp Parasitol 93:66–72
Sures B, Siddall R, Taraschewski (1999) Parasites as accumulation indicators of heavy metal pollution. Parasitol Today 15:16–22
Sures B, Jurges G, Taraschewski H (2000) Accumulation and distribution of lead in the archiacanthocephalan Moniliformis moniliformis from experimentally infected rats. Parasitology 121:427–433
Sures B, Grube K, Taraschewski H (2002) Experimental studies on the lead accumulation in the cestode Hymenolepis diminuta and its final host, Rattus norvegicus. Ecotoxicology 11:365–368
Sures B, Scheible T, Bashtar AR, Taraschewski H (2003) Lead concentrations in Hymenolepis diminuta adults and Taenia taeniaeformis larvae compared to their rat hosts (Rattus norvegicus) sampled from the city of Cairo, Egypt. Parasitology 127:483–487
Xuan R, Wu H, Li Y, Wang J, Wang L (2014) Sublethal Cd-induced cellular damage and metabolic changes in the freshwater crab Sinopotamon henanense. Environ Sci Pollut Res Int 21:1738–1745
Yatoo MI, Saxena A, Deepa PM, Habeab BP, Devi S, Jatav RS, Dimri U (2013) Role of trace elements in animals: a review. Veterinary World, EISSN: 2231–0916
Zhang W, Lin C, Liu K, Lei C, Lin K, Guo J, Liu L, Cui C, Yan Z (2014) Lead accumulations and toxic effects in earthworms (Eisenia fetida) in the presence of decabromodiphenyl ether. Environ Sci Pollut Res Int 21:3484–3490
Acknowledgments
This study was supported by Czech University of Life Sciences Prague internal grant agency (CIGA) project no. 20122028. The authors declare that the experiments comply with the current laws of the Czech Republic in which they were performed. We would also like to thank Mr. Brian Kavalir (Ontario, Canada) for his proofreading services.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Brožová, A., Jankovská, I., Miholová, D. et al. Heavy metal concentrations in the small intestine of red fox (Vulpes vulpes) with and without Echinococcus multilocularis infection. Environ Sci Pollut Res 22, 3175–3179 (2015). https://doi.org/10.1007/s11356-014-3733-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3733-7