Abstract
Introduction
Ursodeoxycholic acid (UDCA) is an intestinal bacterial metabolite with hepatoprotective effects. However, molecular mechanisms underlying its effects remain unclear.
Objectives
The aim of this study was to investigate the mechanisms underlying the therapeutic effects of UDCA by using global metabolomics analyses in healthy subjects.
Methods
Healthy Korean men were administered UDCA at dosage of 400, 800, or 1200 mg daily for 2 weeks. Serum samples were collected and used for liver function tests and to determine miR-122 expression levels. Urinary and plasma global metabolomics analyses were conducted using a liquid chromatography system coupled with quadrupole-time-of-flight mass spectrometry (LC/QTOFMS) and gas chromatography-TOFMS (GC/TOFMS). Unsupervised multivariate analysis (principal component analysis) was performed to identify discriminative markers before and after treatment.
Results
Alanine transaminase score and serum miR-122 levels decreased significantly after 2 weeks of treatment. Through LC- and GC-based metabolomic profiling, we identified 40 differential metabolites in plasma and urine samples.
Conclusions
Regulation of liver function scores and metabolic alternations highlight the potential hepatoprotective action of UDCA, which were primarily associated with amino acid, flavonoid, and fatty acid metabolism in healthy men.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Ursodeoxycholic acid (UDCA) is a steroid bile acid extensively used to treat cholesterol gallstones and primary biliary cirrhosis (PBC). UDCA is clinically predicted to improve biochemical parameters via its therapeutic effects, but its therapeutic efficacy remains still controversial (Beuers et al. 2015; Fiorucci et al. 2018). Numerous mechanistic studies have been conducted on UDCA; however, they have not yielded convincing evidence regarding UDCA-mediated hepatoprotection. For instance, the replacement of potentially toxic hydrophobic endogenous bile acids is theoretically considered the most popular therapeutic strategy (Roma et al. 2011; Sanchez-Garcia et al. 2018). Mechanisms underlying the inhibition of hepatocellular apoptosis have also been evaluated at the cellular level (Roma et al. 2011; Beuers et al. 2015; Kawata et al. 2010). Moreover, the antagonistic effects of farnesoid X receptor (FXR) regulate cholesterol synthesis in morbid obesity (Mueller et al. 2015). However, some researchers believed that cholic acid (CA) and chenodeoxycholic acid (CDCA) bind more strongly with the FXR than UDCA (Beuers et al. 2015). Given the state of the existing background knowledge, it is necessary to investigate further detailed therapeutic mechanisms of UDCA for its effective usage.
Global metabolomics—the profiling of metabolites in biofluids, cells, and tissues—has become a routine technique for diagnosing disease and monitoring progression. The identification of numerous biomarkers has furthered current understanding of metabolite systems-level effects, thereby providing novel insights into the underlying mechanisms for various physiological conditions and aberrant processes, including diseases (Johnson et al. 2016). Despite the widespread use of metabolomics in clinical research, limited information is available regarding the application of metabolomics to study the hepatoprotective effects of bile acids. Bile acid profiling is frequently used to investigate their pathophysiological effects on the progression of such diseases as Alzheimer’s disease (Pan et al. 2017), inflammatory bowel disease (Jahnel et al. 2014), and primary biliary cirrhosis (PBC) (Wunsch et al. 2014). Nevertheless, improvement of liver enzyme function, including aspartate transaminase (AST), alanine transaminase (ALT), and alkaline phosphatase (ALP), is a desired effect of UDCA against liver diseases (Beuers et al. 2015; Dufour et al. 2006; Kawata et al. 2010; Leuschner et al. 2010). However, studies of changes in endogenous metabolites and their metabolism upon UDCA administration are lacking in humans, thereby making it difficult to determine the therapeutic mechanisms of UDCA.
In the present study, we examined the dose-dependent therapeutic effect of UDCA in a dose-dependent manner and performed metabolomic profiling related to its therapeutic effects in healthy Korean male subjects. To conduct global metabolomics profiling, a liquid chromatography system coupled with quadrupole-time-of-flight mass spectrometry (LC/QTOFMS) and a gas chromatography-TOFMS (GC/TOFMS) system were used. For better differentiation of metabolites before and after treatment with UDCA, multivariate principal component analysis (PCA) was used. Using such new high-throughput technology, this study aimed to elucidate modulated endogenous metabolites potentially associated with UDCA-mediated hepatoprotection in healthy men.
2 Materials and methods
2.1 Clinical study and subjects
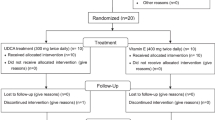
The participants in the present study were originally enrolled in a clinical trial to investigate the safety, tolerability, and pharmacokinetics of UDCA and tauro-UDCA in healthy male volunteers (clinicaltrials.gov identifier: NCT02622685). The samples analyzed herein were obtained from participants administered 400, 800, or 1200 mg UDCA per day for 2 weeks (URSA®, Daewoong Pharmaceutical Co., Ltd.) during this clinical trial. Each group was randomly assigned eight enrolled subjects. The study protocol was reviewed and approved by the Institutional Review Board of Seoul National University Hospital, Seoul, Korea (H-1503-134-660). This study was performed in accordance with the Declaration of Helsinki and Korean Good Clinical Practice. All subjects provided written informed consent prior to their participation in the study.
2.2 Sample collection
Blood samples were collected in EDTA-coated vials prior to initial UDCA administration and 24 h after the 14-day multiple UDCA administration treatment course for metabolomic analysis and to determine the miRNA, AST, and ALT levels. Urine samples were collected over two 24 h periods, the first starting 24 h prior to first administration and the second starting immediately after the final administration of the 14-day treatment course. Plasma was separated via centrifugation (4 °C, 3000 rpm, and 10 min) and stored at − 70 °C until analysis. For miRNA analysis, blood samples were collected in serum-separating tubes, centrifuged (25 °C, 2400 rpm, and 10 min), and stored at − 20 °C until analysis. Urine samples were stored at 4 °C after collection. At 24 h after collection, 10 mL sample aliquots was transferred to Falcon tubes for storage at − 70 °C until analysis.
2.3 LC/QTOFMS analysis
Briefly, frozen samples were diluted with either an acetonitrile:methanol:water (3:3:4) mixture (HPLC-grade, Millipore, Bedford, Massachusetts, USA) for plasma or 10% methanol for urine samples. Each sample (5 μL) was loaded onto an ACQUITY UPLC BEH C18 (1.7 μm, 2. 1 mm × 100 mm; Waters Corp., Milford, MA, USA) column and was analyzed using an Agilent 6530 QTOF mass spectrometer (Agilent Technologies). Detailed experimental procedure is described in our previous study (Kim et al. 2018). The level of Metabolomics Standard Initiative (MSI) and compound ID number are reported in supplementary materials.
2.4 GC/TOFMS analysis
Frozen samples were thawed at 4 °C and 50 µL of each sample was diluted with 1 mL of acetonitrile:isopropanol:water (3:3:2) mixture. Mixed samples were vortexed for 5 min and centrifuged at 18,341×g for 5 min at 4 °C. Thereafter, 400 µL of supernatant was evaporated to complete dryness at room temperature and reconstituted to 400 µL with 50% acetonitrile. Centrifugation was then repeated for 5 min at 18,341×g at 4 °C. The supernatant was then evaporated and the residue was reconstituted with 10 µL of methoxyamine dissolved in pyridine and maintained at 30 °C for 90 min under shaking. After cooling the samples to room temperature, 90 μL of mixture of MSTFA (N-Methyl-N-(trimethylsilyl) trifluoroacetamide) and FAME (fatty acid methyl esters) was added to each experimental sample and the pooled QC samples. The samples were incubated at 70 °C for 45 min under shaking, and then transferred to autosampler vials with glass inserts. Aliquots (1 µL) were then injected in splitless mode into an Agilent 7890A gas chromatograph coupled with a Pegasus HT time-of-flight mass spectrometer (Leco Corporation, St. Joseph, MI). Separation was achieved on a DB-5 ms capillary column (30 m × 250 µm i.d., 0.25 µm film thickness; (5%-phenyl)-methylpolysiloxane bonded and cross-linked; Agilent J&W Scientific, Folsom, CA, USA), with helium as the carrier gas at a constant flow rate of 1.5 mL/min. Oven temperature was initially set to 50 °C for 0.5 min and ramped to 330 °C at a constant rate of 20 °C/min up to 20 min. The intensity of each ion was normalized to the total sum of all detected metabolites using ChromaTof software, and then aligned according to retention time (v4.61 Leco Corporation, St. Joseph, MI). Candidates extracted from multivariate analysis (refer to Sect. 2.7) were selected for using similarity, reserve, and probability values (above 700, 800, and 3000, respectively). Thereafter, the data set was matched with existing metabolite in libraries. The levels of MSI and compound ID are listed in supplementary materials.
2.5 Multivariate analysis and pathway analysis
Multivariate analysis and pathway analysis were conducted using Metaboanalyst 4.0. Normalized data sets were entered and principal components analysis (PCA) was performed to examine differentiation in overall metabolite profiles between groups. Metabolic candidates were selected through the univariate analysis with the FDR-adjusted p value < 0.05. Pathway-associated metabolites were selected from an existing metabolite library and pathways with a Holm-corrected p value and an FDR-adjusted p value < 0.05 were considered potential pathways mediated by UDCA.
2.6 miRNA quantification
Total RNA was extracted to quantify the serum miRNA levels using a miRNeasy Mini kit (Qiagen, Hilden, Germany) in accordance with the manufacturer’s instructions. A detailed protocol was reported previously (Kim et al. 2018).
2.7 Statistical analysis
Statistical analysis of serum parameters was performed using GraphPad Prism version 7 (GraphPad Software Inc., La Jolla, CA, USA). To compare paired samples, non-parametric equivalent Wilcoxon signed-ranks tests were performed. P values < 0.05 were considered to indicate statistically significant findings.
3 Results
3.1 Biomarkers for liver function
To assess the hepatoprotective effect of drugs, we analyzed serum parameters in healthy subjects administered one of three different doses of UDCA (400, 800, and 1200 mg) for 2 weeks. As shown in Fig. 1a, ALT scores decreased following UDCA administration even in patients receiving low doses (400 mg). AST scores were also reduced in patients receiving 400 and 800 mg UDCA. However, no statistically significant change was observed for the 1200 mg group (Fig. 1b). We further evaluated circulating miR-122 levels, a potential biomarker of liver disease (Bandiera et al. 2015). All three dosages of UDCA resulted in well controlled serum miR-122 levels (Fig. 1c). These results indicate that UDCA administration resulted in diminished levels of the liver dysfunction biomarkers ALT, AST, and miR-122.
Changes in the ALT, AST, and miR-122 levels after UDCA treatment. The spaghetti plots represent the serum levels of a ALT and b AST before and after UDCA treatment (1200, 800, and 400 mg doses, respectively). The open-circles represent pre-dose levels and closed-circles represent post-dose levels. Changes in the miR-122 level upon UDCA treatment is represented with bar graph in c miR-122. The white squares represent the pre-dose levels and black squares represent the post-dose levels. The results are expressed as mean ± SEM. Asterisks represent statistical significance (*p value < 0.05 and **p value < 0.01) as determined via paired t test between pre-dose and post-dose values. FC indicates fold change
3.2 Metabolomic profiling via combined LC/QTOFMS and GC/TOFMS analyses
To assess systemic variations in small-molecule metabolites after drug treatment, we conducted a global metabolomics analysis using urine and plasma samples obtained before and after treatment with UDCA. Multivariate analysis was also performed to identify modulated metabolites in patients treated with 400 and 800 mg doses, but no models with acceptable predictability could be generated (data not shown). As such, we have instead provided the relative intensities of metabolites that were consistently modulated post-treatment in the 400 and/or 800 mg treatment groups (Fig. S1).
The results of the PCA score plots revealed different metabolomic signatures before and after treatment with 1200 mg UDCA (Fig. 2). Data for Fig. 2a, b were obtained from urine samples and those for Fig. 2c, d were from plasma samples. The PCA score plots in Fig. 2a, c were obtained by LC/QTOFMS and indicated different metabolome signatures upon UDCA treatment, with the overall accuracy of 22.9% and 25.5%, respectively. Figure 2b, d were obtained by GC/TOFMS, with the overall accuracy of 37.8% and 38.3%, respectively.
Score plot of PCA before and after UDCA treatment. Each score plot represents data obtained from a urine sample by LC/QTOFMS, b urine sample by GC/TOFMS, c plasma sample by LC/QTOFMS, and d plasma sample by GC/TOFMS. Pre-treatment samples are lined in blue and post-treatment samples are lined in red
All the metabolites fulfilled the criteria for univariate analysis, < 0.05 of FDR-adjusted P value. Based on the criteria, 28 metabolites via LC/QTOFMS and 12 metabolites via GC/TOFMS were finally selected as potentially modulated by UDCA administration (Table S2, S3). As shown in Figs. 3 and 4, the spaghetti plots represent the relative intensity values of identified metabolites in urine and plasma, respectively. Interestingly, we observed several flavonoid compounds such as fertaric acid, ferulic acid-sulfate, and vanillin, which were elevated in urine samples (Fig. 3 and Table S1, S2). Further, phenylalanine metabolites such as phenylalanyl-alanine and polyphenylalanine were also reduced after treatment. Upon urine sample analysis, the levels of certain amino acids such as l-threonine, alanine, fumaric acid, lysine, and histidine were reduced after UDCA treatment (Fig. 3). Similar patterns of metabolite alterations were observed in plasma samples, with a phenylalanine containing metabolite, glutamyl-phenylalanine, being decreased and the hydroxycinnamic acid containing metabolites p-hydroxyphenethyl trans-ferulate, cholesteryl ferulate, and cinnamyl alcohol presenting elevated levels (Fig. 4). Moreover, the levels of some fatty acids were decreased following UDCA administration, including lysophosphatidylcholine (lyso-PC), and phosphoethanolamine (PE). Linoleic acid, however, was the only fatty acid induced by UDCA at the plasma level (Fig. 4). Using GC/TOFMS analysis, glutamine and d-fructose were determined as metabolomics markers. Threonine levels were lower in plasma samples than in urine samples. Together, our data indicate that UDCA alters amino acid and fatty acid levels and increases levels of phenol-containing compounds in biological fluids.
Relative intensity of identified metabolites in urine. Spaghetti plots represent relative intensity of metabolites identified in the urine sample upon UDCA treatment. Representative urinary metabolites are alanine, l-threonine, l-lysine, l-histidine, l-fructose, fumaric acid, succinic acid, homovanillic acid sulfate, phenylalanyl-alanine, arginyl-asparagine, fertaric acid, and ferulic acid 4-sulfate. The FDR-adjusted p value < 0.05 were used for statistical evaluation
Relative intensity of identified plasma metabolites. The spaghetti plots represent relative intensity of the identified plasma metabolites upon UDCA treatment, including d-fructose, glutamine, l-threonine, glutamy-phenylalanine, linoleic acid, cinnamyl alcohol, bilirubin, choesteryl ferulate, p-hydroxyphenethyl trans-ferulate, lysophosphatidylcholine, and phosphoethanolamine. The FDR-adjusted p value < 0.05 were used for statistical evaluation
3.3 Pathway analysis of metabolite profiling data
To measure the centrality of a metabolite in a metabolic network, we performed pathway topological analysis using Metaboanalyst 4.0. All metabolites having Holm corrected and FDR adjusted p values < 0.05 were assessed for pathway analysis. As shown in Fig. 5 and Table S3, the candidate pathways with an FDR-adjusted p-value < 0.05 were (1) alanine, aspartate, and glutamate metabolism and (2) aminoacyl-tRNA biosynthesis. In addition, phenylalanine metabolism and tyrosine and histidine metabolism in the tricarboxylic acid (TCA) cycle also displayed p values < 0.05 but did not reach statistical significance upon FDR adjustment. Detailed statistical analysis is reported in Table S3. Further, we proposed a network analysis with visualization, as shown in Fig. 6. Web-based data from KEGG and HMDB were used for mapping. In conclusion, four major pathways were altered upon drug treatment: (1) amino acid metabolism, (2) flavonoid metabolism, (3) fatty acid metabolism and (4) the TCA cycle.
Analysis of metabolic pathways influenced by UDCA. A summary of pathway analysis representing the effect of identified metabolites on indicated pathways. The Y-axis represents the effect of the pathway and the X-axis presents a log scale of p values. Metabolic pathways displaying statistical significance (FDR-adjusted p value < 0.001) were alanine, aspartate, and glutamate metabolism and aminoacyl-tRNA biosynthesis
A schematic representation of proposed pharmacological mechanisms of UDCA. Metabolomic profiling of urine and plasma samples revealed that UDCA regulates levels of amino acids such as phenylalanine, glutamine, arginine, alanine, histidine, lysine, and threonine, as well as metabolites including fumarate and succinate. Levels of phenolic compounds such as p-coumaric acid, caffeic acid, ferulic acid, and vanillin acid were also elevated. In addition, the effect on fatty acid metabolism (phosphoethanolamine, lysophosphatidylcholine, monoacylglycerides, and diacylglycerides) is also shown. Metabolites elevated after UDCA treatment are indicated in red, while those decreased after UDCA treatment are indicated in blue
4 Discussion
In this study, we used global metabolomic approaches to assess hepatoprotection in healthy male subjects. Subjects were treated with high (1200 mg), intermediate (800 mg), or low (400 mg) doses of UDCA for 2 weeks, thereby reducing levels of the liver dysfunction biomarkers ALT, AST, and miR-122, even at low doses. Global metabolomics analyses using LC/QTOFMS and GC/TOFMS revealed metabolic signatures including amino acid, flavonoid, and fatty acid metabolism.
UDCA has long been used to treat PBC and non-alcoholic steatohepatitis (NASH), as it decreases liver biochemical levels, including alkaline phosphatase and gamma-glutamyltransferase (GGT), and delays the need for liver transplantation (Haedrich and Dufour 2011; Nevens et al. 2016). Changes in hydrophobic bile acid levels due to increased bile flow have been widely reported as a therapeutic effect of UDCA (Bachrach and Hofmann 1982; Roma et al. 2011). Accordingly, our previous study reported that 2 months of UDCA treatment significantly reduced the level of deoxycholic acid, one of the hydrophobic bile acids, in patients with liver dysfunction (Kim et al. 2018). Numerous studies have attempted to investigate the mechanisms underlying the beneficial effects of UDCA in liver dysfunction. Experimental evidence suggests three major mechanisms of action of UDCA: cholangiocyte protection against cytotoxicity of hydrophobic bile acids, stimulation of hepatobiliary secretion, and hepatocyte protection against bile acid-induced apoptosis. One or all of these mechanisms may occur in individual cholestatic disorders and/or different stages of cholestatic liver disease (Paumgartner and Beuers 2002).
In the present study, UDCA treatment effectively reduced the ALT/AST score even upon short-term administration, which was anticipated to a certain degree based on previous studies (Leuschner et al. 2010; Beuers et al. 2015; Parikh et al. 2016; Dufour et al. 2006). However, the scores were below the upper limits of normal ALT and AST values since this clinical trial was conducted in healthy men. However, miR-122 levels did not encounter this limitation. miR-122 is the most prevalent miRNA in the adult liver, accounting for approximately 70% of the total liver miRNA population (Bandiera et al. 2015). It regulates liver-enriched transcription factors, including hepatocyte nuclear factor-4 α and p53 (Mukherjee et al. 2010; Vincent and Sanyal 2014), thereby maintaining an appropriate balance between hepatocyte proliferation and differentiation. miR-122 is reportedly up-regulated in patients with hepatitis C and NAFLD (Bandiera et al. 2015). In our previous study, miR-122 was also used as an early biomarker of drug-induced liver injury (Lee et al. 2017). UDCA significantly decreased miR-122 levels, thereby conclusively indicating its hepatoprotective effect. Recently, we demonstrated the systemic downregulation of miR-122 levels by 8 weeks of UDCA (400 mg, twice daily) and vitamin E (400 IU, twice daily) in obese patients with liver dysfunction (Kim et al. 2018). However, it is interesting here that even short-term administration of a low UDCA dosage was sufficient to modulate miR-122 expression levels and liver function scores. This may indicate that lower doses of UDCA may be more effective for ameliorating or preventing liver dysfunction at pre-PBC stages with no adverse effects.
Global metabolomics analysis using LC/QTOFMS and GC/TOFMS enabled the evaluation of the mechanisms of action of UDCA. Among the new findings obtained herein, most of the identified metabolites categorized as amino acids were decreased upon UDCA treatment. In particular, urine and plasma levels of phenylalanine metabolites, including alanine and phenylalanine, were strongly regulated by UDCA. Amino acids are metabolized to provide energy or used to synthesize other bioactive molecules. In particular, aromatic amino acids are primarily metabolized in the liver (Rui 2014; Kamata et al. 1995). Increased plasma levels of these metabolites suggest an increase in the degradation of muscle proteins and decreased metabolism of these amino acids in the liver (Kamata et al. 1995). This implies that high concentrations of amino acids result in hepatotoxicity. Systemic phenylalanine and aminotransaminase levels are positively correlated with age (Jung et al. 2014). A recent in vivo study reported that FXR activation via the administration of obeticholic acid as part of high-protein diets enhanced hepatic amino acid catabolism via the regulation of amino acid-degrading enzymes (Massafra et al. 2017). Nevertheless, blunted FXR activity downregulated these enzymes and appeared to reduce phenylalanine, alanine, and glutamine levels. As reported previously, it is possible that the regulation of amino acids might be influenced by FXR activation. However, to ascertain whether FXR directly regulates amino acid catabolism or biosynthesis, further studies involving liver and/or intestinal-specific FXR deletion models are required.
Another possible contributor to amino acid levels is the transport of bile acid through the solute carrier (SLC) family of transport proteins during enterohepatic recirculation. The SLC gene superfamily encodes membrane-bound transporters which play a crucial role in passive transport, symport, and antiport with substrates including amino acids, inorganic cations, and anions. Solute carrier family 10 (SLC10) comprises two sodium-dependent bile acid transporters: the Na+/taurocholate cotransporting polypeptide (NTCP; SLC10A1) and the apical sodium-dependent bile acid transporter (ASBT; SLC10A2), located in the liver and small intestine, respectively (Balakrishnan and Polli 2006; Geyer et al. 2006). SLC36 enables the transport of proton-coupled amino acids and SLC43 represents the sodium-independent system-l-like amino acid (He et al. 2009). The most common feature shared by these transporters is that they rely on a sodium gradient to actively penetrate cells (Geyer et al. 2006). Therefore, sodium gradient-dependent bile flow may counteract the reabsorption of amino acids into the systemic circulation.
Elevated levels of phosphatidylcholine (PC), lyso-PC, phosphoethanolamine (PE), and sphingomyelins (SM) were previously reported as biomarkers of liver diseases including non-alcoholic fatty liver disease (NAFLD) (Ma et al. 2016; Chang et al. 2017; Li et al. 2005; Kalhan et al. 2011). In particular, PC accounts for ~ 95% of choline-containing compounds and is secreted into the plasma along with lipoproteins. These phospholipid classes are associated with inflammation and cellular apoptosis (Li et al. 2005; Kalhan et al. 2011). In the present study, plasma PC, lyso-PC, and PE levels decreased significantly after treatment with UDCA. This may be clinically acceptable because its therapeutic implications have been reported in patients with mutations in canalicular transporters including ATP8B1, ABCB11, and ABCB4 (Stapelbroek et al. 2010). Canalicular transporters are the major route for phospholipids and UDCA has been proven to propagate their flux, thereby decreasing systemic phospholipid levels (Stapelbroek et al. 2010). Furthermore, the elevation of linoleic acid (a long-chain ω-6 polyunsaturated fatty acid) may be associated with the regulation of these fatty acids and some cytokines by targeting sterol regulatory element-binding protein (SREBP)-1 isomers (Roche et al. 2002; Erdinest et al. 2012; Degirolamo et al. 2016).
Ferulic acid-, fertaric acid-, caffeic acid-, and vanillin-containing metabolites are hydroxycinnamic acids and are classified as a type of polyphenol. These phenolic compounds are ubiquitous in vegetables and fruits, thereby only capable of being consumed as/with food. The free forms of these phenolic acids are primarily distributed in the liver and conjugated primarily with glucuronide/sulfate and secreted into the bile for enterohepatic circulation (Zhao et al. 2004; Poquet et al. 2008; Lafay et al. 2006). Certain free compounds and their conjugates in the systemic circulation and tissues are then metabolized into other compounds, whereas most of them are ultimately eliminated through the kidneys (Zhao et al. 2004; Poquet et al. 2008; Zhao and Moghadasian 2008). Although the mechanisms underlying phenol metabolism are yet unclear, some studies have reported that dietary phenolic acids required bacterial enzymatic activity to be transformed or degraded. For example, Lactobacillus mucosae, Enterococcus faecium, and Bifidobacterium lactis possess enzymes for the esterification of hydroxycinnamates and isoflavones (Tapiero et al. 2002; Wiseman et al. 2004). Dehydroxylation of these compounds is associated with Clostridium scindens and Eggerthella lenta (Clavel et al. 2006, 2005). After transformation, the compounds passively permeate through the intestinal epithelium via transcellular diffusion into the blood or other tissues (Selma et al. 2009; Poquet et al. 2008; Zhao et al. 2004; Zhao and Moghadasian 2008; Olthof et al. 2001; Enright et al. 2018). Based on previous reports, we assumed two possible mechanisms explaining the high levels of phenolic compounds observed in this study. First, dietary phenols in bile might be reabsorbed in the same manner as UDCA—predominantly transported via passive diffusion in the small intestine. Alternatively, changes in the gut microbiota upon UDCA treatment may have influenced further metabolic reactions involving dietary phenols, thereby rendering them available for reabsorption or excretion via bile. Further investigations are, however, required to completely elucidate the interaction of dietary phenols and gut microbiota. Particularly, future studies should investigate whether microbiomes regulated by UDCA display enzymatic activity on dietary phenols for transformation.
We have reported microbiome metabolomic changes upon long-term treatment with UDCA and vitamin E in patients with liver dysfunction and higher body mass index values (Kim et al. 2018). Diminished phenylalanine levels were present in UDCA-treated individuals regardless of whether they had pre-existing conditions or were healthy. In addition, UDCA enabled the regulation of uremic toxins such as p-cresol sulfate and hippuric acid in patients. However, UDCA exerted anti-oxidative effects in healthy subjects. Therefore, we suggest with caution that short-term treatment with a non-toxic dose of UDCA may prevent liver dysfunction at a very early stage, presenting a possible new therapeutic strategy for healthy individuals or those with mild liver dysfunction.
Mechanistic studies on such therapeutic interventions in healthy subjects might be interpreted inconsistently relative to those involving patients with disease. However, this study reported changes in a putative early biomarker of liver injury (miR-122) upon UDCA treatment, therefore enabling the further evaluation of pharmacological mechanisms. However, our small sample size likely compromised the statistical power of our results, thereby serving as a study limitation. Future studies evaluating the interaction between gut microbiota and dietary phenolic acids are required to potentiate the therapeutic effects of UDCA administration on human health. Together, this study demonstrated changes in metabolite levels following UDCA treatment in healthy men, and our data indicate that a potentially novel therapeutic strategy using UDCA may be possible.
5 Conclusion
In summary, in this study, we used a metabolomics approach involving LC/QTOFMS and GC/TOFMS to evaluate potential therapeutic mechanisms of UDCA in healthy men. The findings suggest that UDCA effectively improved liver function and decreased circulating miR-122 levels. The possible mechanisms underlying this hepatoprotection may involve metabolic modifications in amino acid, flavonoid, and fatty acid metabolism.
Data Availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Bachrach, W. H., & Hofmann, A. F. (1982). Ursodeoxycholic acid in the treatment of cholesterol cholelithiasis. Part I. Digestive Diseases and Sciences, 27(8), 737–761.
Balakrishnan, A., & Polli, J. E. (2006). Apical sodium dependent bile acid transporter (ASBT, SLC10A2): A potential prodrug target. Molecular Pharmaceutics, 3(3), 223–230. https://doi.org/10.1021/mp060022d.
Bandiera, S., Pfeffer, S., Baumert, T. F., & Zeisel, M. B. (2015). miR-122–a key factor and therapeutic target in liver disease. Journal of Hepatology, 62(2), 448–457. https://doi.org/10.1016/j.jhep.2014.10.004.
Beuers, U., Trauner, M., Jansen, P., & Poupon, R. (2015). New paradigms in the treatment of hepatic cholestasis: From UDCA to FXR, PXR and beyond. Journal of Hepatology, 62(1 Suppl), 25–37. https://doi.org/10.1016/j.jhep.2015.02.023.
Chang, H., Meng, H. Y., Liu, S. M., Wang, Y., Yang, X. X., Lu, F., et al. (2017). Identification of key metabolic changes during liver fibrosis progression in rats using a urine and serum metabolomics approach. Scientific Reports, 7(1), 11433. https://doi.org/10.1038/s41598-017-11759-z.
Clavel, T., Henderson, G., Alpert, C. A., Philippe, C., Rigottier-Gois, L., Dore, J., et al. (2005). Intestinal bacterial communities that produce active estrogen-like compounds enterodiol and enterolactone in humans. Applied and Environmental Microbiology, 71(10), 6077–6085. https://doi.org/10.1128/AEM.71.10.6077-6085.2005.
Clavel, T., Henderson, G., Engst, W., Dore, J., & Blaut, M. (2006). Phylogeny of human intestinal bacteria that activate the dietary lignan secoisolariciresinol diglucoside. Fems Microbiology Ecology, 55(3), 471–478. https://doi.org/10.1111/j.1574-9641.2005.00057.x.
Degirolamo, C., Sabba, C., & Moschetta, A. (2016). Therapeutic potential of the endocrine fibroblast growth factors FGF19, FGF21 and FGF23. Nature Reviews Drug Discovery, 15(1), 51–69. https://doi.org/10.1038/nrd.2015.9.
Dufour, J. F., Oneta, C. M., Gonvers, J. J., Bihl, F., Cerny, A., Cereda, J. M., et al. (2006). Randomized placebo-controlled trial of ursodeoxycholic acid with vitamin e in nonalcoholic steatohepatitis. Clinical Gastroenterology and Hepatology, 4(12), 1537–1543. https://doi.org/10.1016/j.cgh.2006.09.025.
Enright, E. F., Griffin, B. T., Gahan, C. G. M., & Joyce, S. A. (2018). Microbiome-mediated bile acid modification: Role in intestinal drug absorption and metabolism. Pharmacological Research, 133, 170–186. https://doi.org/10.1016/j.phrs.2018.04.009.
Erdinest, N., Shmueli, O., Grossman, Y., Ovadia, H., & Solomon, A. (2012). Anti-inflammatory effects of alpha linolenic acid on human corneal epithelial cells. Investigative Ophthalmology & Visual Science, 53(8), 4396–4406. https://doi.org/10.1167/iovs.12-9724.
Fiorucci, S., Biagioli, M., & Distrutti, E. (2018). Future trends in the treatment of non-alcoholic steatohepatitis. Pharmacological Research, 134, 289–298. https://doi.org/10.1016/j.phrs.2018.07.014.
Geyer, J., Wilke, T., & Petzinger, E. (2006). The solute carrier family SLC10: More than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn-Schmiedebergs Archives of Pharmacology, 372(6), 413–431. https://doi.org/10.1007/s00210-006-0043-8.
Haedrich, M., & Dufour, J. F. (2011). UDCA for NASH: End of the story? Journal of Hepatology, 54(5), 856–858. https://doi.org/10.1016/j.jhep.2010.10.009.
He, L., Vasiliou, K., & Nebert, D. W. (2009). Analysis and update of the human solute carrier (SLC) gene superfamily. Human Genomics, 3(2), 195–206.
Jahnel, J., Fickert, P., Hauer, A. C., Hogenauer, C., Avian, A., & Trauner, M. (2014). Inflammatory bowel disease alters intestinal bile acid transporter expression. Drug Metabolism and Disposition, 42(9), 1423–1431. https://doi.org/10.1124/dmd.114.058065.
Johnson, C. H., Ivanisevic, J., & Siuzdak, G. (2016). Metabolomics: Beyond biomarkers and towards mechanisms. Nature Reviews Molecular Cell Biology, 17(7), 451–459. https://doi.org/10.1038/nrm.2016.25.
Jung, S., Kim, O. Y., Kim, M., Song, J., Lee, S. H., & Lee, J. H. (2014). Age-related increase in alanine aminotransferase correlates with elevated levels of plasma amino acids, decanoylcarnitine, Lp-PLA2 Activity, oxidative stress, and arterial stiffness. Journal of Proteome Research, 13(7), 3467–3475. https://doi.org/10.1021/pr500422z.
Kalhan, S. C., Guo, L., Edmison, J., Dasarathy, S., McCullough, A. J., Hanson, R. W., et al. (2011). Plasma metabolomic profile in nonalcoholic fatty liver disease. Metabolism, 60(3), 404–413. https://doi.org/10.1016/j.metabol.2010.03.006.
Kamata, S., Imura, K., Kawahara, H., Kubota, A., & Okada, A. (1995). Early postoperative change of plasma levels of amino acids in neonates with perforative peritonitis and its prognostic significance. Journal of Pediatric Surgery, 30(4), 559–562.
Kawata, K., Kobayashi, Y., Souda, K., Kawamura, K., Sumiyoshi, S., Takahashi, Y., et al. (2010). Enhanced hepatic Nrf2 activation after ursodeoxycholic acid treatment in patients with primary biliary cirrhosis. Antioxidants & Redox Signaling, 13(3), 259–268. https://doi.org/10.1089/ars.2009.2903.
Kim, D. J., Yoon, S., Ji, S. C., Yang, J., Kim, Y. K., Lee, S., et al. (2018). Ursodeoxycholic acid improves liver function via phenylalanine/tyrosine pathway and microbiome remodelling in patients with liver dysfunction. Scientific Reports, 8(1), 11874. https://doi.org/10.1038/s41598-018-30349-1.
Lafay, S., Morand, C., Manach, C., Besson, C., & Scalbert, A. (2006). Absorption and metabolism of caffeic acid and chlorogenic acid in the small intestine of rats. British Journal of Nutrition, 96(1), 39–46.
Lee, J., Ji, S. C., Kim, B., Yi, S., Shin, K. H., Cho, J. Y., et al. (2017). Exploration of biomarkers for amoxicillin/clavulanate-induced liver injury: Multi-omics approaches. Clinical and Translational Science, 10(3), 163–171. https://doi.org/10.1111/cts.12425.
Leuschner, U. F., Lindenthal, B., Herrmann, G., Arnold, J. C., Rossle, M., Cordes, H. J., et al. (2010). High-dose ursodeoxycholic acid therapy for nonalcoholic steatohepatitis: A double-blind, randomized, placebo-controlled trial. Hepatology, 52(2), 472–479. https://doi.org/10.1002/hep.23727.
Li, Z., Agellon, L. B., & Vance, D. E. (2005). Phosphatidylcholine homeostasis and liver failure. Journal of Biological Chemistry, 280(45), 37798–37802. https://doi.org/10.1074/jbc.M508575200.
Ma, D. W., Arendt, B. M., Hillyer, L. M., Fung, S. K., McGilvray, I., Guindi, M., et al. (2016). Plasma phospholipids and fatty acid composition differ between liver biopsy-proven nonalcoholic fatty liver disease and healthy subjects. Nutrition & Diabetes, 6(7), e220. https://doi.org/10.1038/nutd.2016.27.
Massafra, V., Milona, A., Vos, H. R., Ramos, R. J. J., Gerrits, J., Willemsen, E. C. L., et al. (2017). Farnesoid X receptor activation promotes hepatic amino acid catabolism and ammonium clearance in mice. Gastroenterology, 152(6), 1462–1476 e1410. https://doi.org/10.1053/j.gastro.2017.01.014.
Mueller, M., Thorell, A., Claudel, T., Jha, P., Koefeler, H., Lackner, C., et al. (2015). Ursodeoxycholic acid exerts farnesoid X receptor-antagonistic effects on bile acid and lipid metabolism in morbid obesity. Journal of Hepatology, 62(6), 1398–1404. https://doi.org/10.1016/j.jhep.2014.12.034.
Mukherjee, S., Vaidyanathan, K., Vasudevan, D. M., & Das, S. K. (2010). Role of plasma amino acids and gaba in alcoholic and non-alcoholic fatty liver disease-a pilot study. Indian Journal of Clinical Biochemistry, 25(1), 37–42. https://doi.org/10.1007/s12291-010-0007-0.
Nevens, F., Andreone, P., Mazzella, G., Strasser, S. I., Bowlus, C., Invernizzi, P., et al. (2016). A placebo-controlled trial of obeticholic acid in primary biliary cholangitis. The New England Journal of Medicine, 375(7), 631–643. https://doi.org/10.1056/NEJMoa1509840.
Olthof, M. R., Hollman, P. C., & Katan, M. B. (2001). Chlorogenic acid and caffeic acid are absorbed in humans. Journal of Nutrition, 131(1), 66–71. https://doi.org/10.1093/jn/131.1.66.
Pan, X., Elliott, C. T., McGuinness, B., Passmore, P., Kehoe, P. G., Holscher, C., et al. (2017). Metabolomic profiling of bile acids in clinical and experimental samples of alzheimer’s disease. Metabolites. https://doi.org/10.3390/metabo7020028.
Parikh, P., Ingle, M., Patel, J., Bhate, P., Pandey, V., & Sawant, P. (2016). An open-label randomized control study to compare the efficacy of vitamin e versus ursodeoxycholic acid in nondiabetic and noncirrhotic Indian NAFLD patients. Saudi Journal of Gastroenterologyrol, 22(3), 192–197. https://doi.org/10.4103/1319-3767.182451.
Paumgartner, G., & Beuers, U. (2002). Ursodeoxycholic acid in cholestatic liver disease: Mechanisms of action and therapeutic use revisited. Hepatology, 36(3), 525–531. https://doi.org/10.1053/jhep.2002.36088.
Poquet, L., Clifford, M. N., & Williamson, G. (2008). Transport and metabolism of ferulic acid through the colonic epithelium. Drug Metabolism and Disposition, 36(1), 190–197. https://doi.org/10.1124/dmd.107.017558.
Roche, H. M., Noone, E., Sewter, C., Mc Bennett, S., Savage, D., Gibney, M. J., et al. (2002). Isomer-dependent metabolic effects of conjugated linoleic acid: Insights from molecular markers sterol regulatory element-binding protein-1c and LXRalpha. Diabetes, 51(7), 2037–2044.
Roma, M. G., Toledo, F. D., Boaglio, A. C., Basiglio, C. L., Crocenzi, F. A., & Sanchez Pozzi, E. J. (2011). Ursodeoxycholic acid in cholestasis: Linking action mechanisms to therapeutic applications. Clinical Science (London), 121(12), 523–544. https://doi.org/10.1042/CS20110184.
Rui, L. (2014). Energy metabolism in the liver. Comprehensive Physiology, 4(1), 177–197. https://doi.org/10.1002/cphy.c130024.
Sanchez-Garcia, A., Sahebkar, A., Simental-Mendia, M., & Simental-Mendia, L. E. (2018). Effect of ursodeoxycholic acid on glycemic markers: A systematic review and meta-analysis of clinical trials. Pharmacological Research, 135, 144–149. https://doi.org/10.1016/j.phrs.2018.08.008.
Selma, M. V., Espin, J. C., & Tomas-Barberan, F. A. (2009). Interaction between phenolics and gut microbiota: Role in human health. Journal of Agricultural and Food Chemistry, 57(15), 6485–6501. https://doi.org/10.1021/jf902107d.
Stapelbroek, J. M., van Erpecum, K. J., Klomp, L. W., & Houwen, R. H. (2010). Liver disease associated with canalicular transport defects: Current and future therapies. Journal of Hepatology, 52(2), 258–271. https://doi.org/10.1016/j.jhep.2009.11.012.
Tapiero, H., Tew, K. D., Ba, G. N., & Mathe, G. (2002). Polyphenols: Do they play a role in the prevention of human pathologies? Biomedicine & Pharmacotherapy, 56(4), 200–207. https://doi.org/10.1016/S0753-3322(02)00178-6.
Vincent, R., & Sanyal, A. (2014). Recent advances in understanding of NASH: MicroRNAs as both biochemical markers and players. Current Pathobiology Reports, 2(3), 109–115. https://doi.org/10.1007/s40139-014-0049-8.
Wiseman, H., Casey, K., Bowey, E. A., Duffy, R., Davies, M., Rowland, I. R., et al. (2004). Influence of 10 wk of soy consumption on plasma concentrations and excretion of isoflavonoids and on gut microflora metabolism in healthy adults. American Journal of Clinical Nutrition, 80(3), 692–699.
Wunsch, E., Trottier, J., Milkiewicz, M., Raszeja-Wyszomirska, J., Hirschfield, G. M., Barbier, O., et al. (2014). Prospective evaluation of ursodeoxycholic acid withdrawal in patients with primary sclerosing cholangitis. Hepatology, 60(3), 931–940. https://doi.org/10.1002/hep.27074.
Zhao, Z. H., Egashira, Y., & Sanada, H. (2004). Ferulic acid is quickly absorbed from rat stomach as the free form and then conjugated mainly in liver. Journal of Nutrition, 134(11), 3083–3088.
Zhao, Z. H., & Moghadasian, M. H. (2008). Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review. Food Chemistry, 109(4), 691–702. https://doi.org/10.1016/j.foodchem.2008.02.039.
Funding
This study was supported in part by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (Grant No.: HI14C2770).
Author information
Authors and Affiliations
Contributions
DJK was responsible for experimental set up, measurements and analysis of data, interpretation of data, and manuscript writing. SCJ performed miR-122 measurements. HC, SHL, KSY, and IJJ helped with clinical study design, interpretation of data, and edited the manuscript. JYC contributed to the experimental design and manuscript editing. All authors participated sufficiently in the presented work and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, D.J., Chung, H., Ji, S.C. et al. Ursodeoxycholic acid exerts hepatoprotective effects by regulating amino acid, flavonoid, and fatty acid metabolic pathways. Metabolomics 15, 30 (2019). https://doi.org/10.1007/s11306-019-1494-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-019-1494-5