Abstract
It has been demonstrated that biochar has a great potential to reduce volatilisations of ammonia (NH3) from fertilised agricultural soils. While there have been several laboratory studies to demonstrate the effects of biochar on gaseous NH3 emissions, there is hardly any data on the influence of biochar amendments on NH3 volatilisations from the soils under a field environment. Modifying biochar or enriching it with nitrogen (N) may maximise its capacity to abate gaseous NH3 emissions from the soil. Three biochars, i.e. SAB, HPB, and KHB modified through post-pyrolysis treatment with sulphuric acid, hydrogen peroxide, and potassium hydroxide, respectively as well as two biochars enriched with either molten urea (URB) or ammonium nitrate (ANB), were used alongside the pristine biochar (PRB) for comparisons. The quantity of gaseous NH3 evolved from each the biochar amendments including their effects on the growth and yield of the Chinese cabbage plus selected soil chemical properties were evaluated through a field experiment. The control experiment consisted of urea applied alone. In comparison with the control, PRB, KHB, HPB, SAB, URB, and ANB amendments abated NH3 volatilisations from the soil by 44.18%, 45.91%, 63.23%, 65.62%, 72.66%, and 76.71%, respectively. Additionally, PRB, SAB, KHB, HPB, ANB, and URB amendments increased Chinese cabbage yields by 138.5%, 172.0%, 117.3%, 181.1%, 194.0%, and 181.1%, respectively in comparison with the control. The strong linear relationship (r2 = 0.97) between cumulative NH3 emissions and nitrogen use efficiencies indicates that biochar-induced reductions in emissions of gaseous NH3 concomitantly increased the use-efficiencies of the applied N.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Biochar, a product of pyrolysis of biomass under conditions of no or limited oxygen supply at temperatures above 250 °C, has attracted a lot of interest as a soil amendment or a material for general environmental management (Lehmann & Joseph, 2015). While biochar has been used as a soil amendment in several parts of the world for centuries or even millennia, the current surge in its interest resulted from the discovery of the Terra Preta (the dark earths of the Amazon rich in charred biomass) (Lee et al., 2019; Wiedner & Glaser, 2015). Biochar amendment to the soil has been proposed as one of the measures that could sustainably support food production systems to ensure uninterrupted supply of food to the ever growing global population in the face of climate change (Wiedner & Glaser, 2015). This conviction is premised on the fact that biochar can sequester carbon for extended periods of usually 1000–10,000 years and can thus potentially aid in mitigating climate change (Cowie et al., 2015; Wiedner & Glaser, 2015). Alongside this positive effect on net greenhouse gas balance, biochar has been reported to increase crop yields especially in the tropics (Jeffery et al., 2017a, b).
Over time, more beneficial functions of biochar have been explored and discerned including remediation and or stabilisation of contaminants including heavy metals and emerging pollutants, attenuation of emissions of greenhouse gases, etc. The efficiencies of biochar in fulfilling the aforementioned functions have been elucidated through meta-analyses of the data from the piles of published literature. For example, a meta-analysis by Jeffery et al. (2016) found that only biochar produced at temperatures higher than 600 °C can reduce methane emissions when applied to acidic flooded (paddy) soils and only when nitrogen is applied at low application rates of less than 120 kg/ha. Another meta-analysis by Van Zwieten et al. (2017) found that biochar abated emissions of nitrous oxide in both laboratory and field studies but very low application rates did not produce any noticeable effect on the reduction of gaseous emissions. Additionally, Chen et al. (2018) found that biochar reduced uptakes of cadmium, lead, copper, and zinc into the plant tissues by 38%, 39%, 25%, and 17%, respectively through a meta-analysis study.
Recently, biochar has been explored for the abatement of gaseous ammonia emissions from the soil (Mandal et al., 2016; Luyima et al., 2020, 2022) and stored or composting waste (Baitong et al., 2021; Janczak et al., 2017; Malińska et al., 2014). Ammonia is an important air pollutant because it forms particulate matter (PM) through a series of complex reactions in the atmosphere (Egyir et al., 2022; Luyima et al., 2022; Pinder & Adams, 2007) and hence, has direct impacts on both human health and environmental quality. It is important to note, however, that the previous studies have generated both negative (increment) and positive (decrement) results. For example, while studies by Mandal et al. (2016) and Luyima et al. (2020b) observed decreased volatilisations of NH3 from the biochar-amended soils, those by Subedi et al. (2015), Feng et al. (2017), Fan et al. (2017), and Sun et al. (2017) reported increased emissions. A meta-analysis review study by Sha et al. (2019) is possibly the best summary of the effects of biochar on gaseous NH3 emissions and it discovered that only highly acidic (pH ≤ 4) woody biochar could reduce gaseous NH3 emissions and only when applied to fine textured soils rich in organic matter/carbon (Soil organic carbon > 3%).
To improve or optimise the potential of biochar to attenuate gaseous NH3, a recent incubation study by Egyir et al. (2022) proposed the usage of both modified and nitrogen-enriched biochars and they found that the biochars reduced gaseous emissions by up to 73.23% and 82.88%, respectively. The modified and nitrogen-enriched biochars, therefore, outperformed the non-modified biochar which reduced the gaseous emissions by only 57.62% in comparison with the control (urea-only amendment). Given that incubation experiments take place under controlled laboratory conditions, it is difficult to generalise the findings of such studies to real field conditions (Fisher & Wood, 2007; Brüggemann and Bizer 2016). It is against that background that we proposed the evaluation of the gaseous NH3 emissions from the modified and nitrogen-enriched biochar-amended soils under the field conditions. Along with the gaseous emissions, the effects of the biochars on the agronomic performance of Chinese cabbage were assessed. This is important because although biochar has been applauded for increasing crop yields, such high yield increases are more of an exception than the rule and a wide range of crop yield responses, including negative responses in some cases, have been reported (Jeffery et al., 2011, 2017a, b). Benign and or negative yield responses may dissuade farmers from adopting the usage of modified and nitrogen-enriched biochars if they do not provide tangible financial benefits in form of marketable yields. The objective of this study, therefore, was to assess the effects of both modified and nitrogen-enriched biochars on gaseous NH3 evolutions, agronomic performance of the Chinese cabbage, and selected soil chemical properties.
2 Materials and methods
2.1 Biochar preparations and analysis
The details of the biochar modifications and nitrogen-enrichments are outlined in Egyir et al. (2022). Briefly, the modifications were achieved through post-pyrolysis treatment with hydrogen peroxide, potassium hydroxide, and sulphuric acid whereas both urea and ammonium nitrate were used to prepare the nitrogen-enriched biochar. Forty grams of the rice husk biochar freshly produced at 500 °C for 1 h in a muffle furnace (Lindberg/Blue M) was soaked in 1.5 L of 20% of each of the sulphuric acid, potassium hydroxide, and hydrogen peroxide solutions at room temperature for 8 h. Urea-enrichment involved impregnating non-modified biochar with molten urea whereas the ammonium nitrate-enrichment involved soaking the non-modified biochar in an ammonium nitrate solution. The nitrogen-enrichment in both cases was done in such a way that the 3% application rate of biochar supplied 150 kg of nitrogen per hectare of land (considering an incorporation depth of 20 cm). All the biochars were thoroughly dried in an oven before usage. KHB, HPB, and SAB denote biochar modified with potassium hydroxide, hydrogen peroxide, and sulphuric acid, respectively while URB and ANB denote biochar enriched with molten urea and ammonium nitrate, respectively. The non-modified biochar is denoted as PRB. The pH and EC values of the biochar were determined with a pH and EC meter (Orion Versa Star Pro; Thermo Scientific, Inc., USA) after mixing 5 g of biochar with 100 mL of distilled water and shaking for 30 min. The available phosphorus was determined following the vanadate-molybdate method after extraction with 2% formic acid. The contents of the cations in the biochar were quantified by ICP-OES (Thermo Scientific iCAP 7000 Series, Waltham, MA USA) after extraction from the biochar ashes as instructed by Enders et al. (2017). The carbon (C), hydrogen (H), nitrogen (N), and oxygen (O) contents of biochar were determined with the automated elemental analysers (Thermo Fisher Scientific EA 1112 and FLASH 2000, Waltham, MA USA (for O and CHN, respectively)). The resultant properties of the biochars are given in Table 1 whereas the methods used for the soil analysis and resultant properties of the soil of the experimental field used are given in Egyir et al. (2022)
2.2 Experimental set-up
The study was conducted through a field experiment that was set up at the Chungnam national university farm located at Eoeun-dong, Daejeon, South Korea (latitude, 127° 35´ E; longitude, 36° 36´ N). The soil at the farm is a sandy loam and belongs to the Inceptisol and Udepts order and suborder, respectively, according to the IUSS’s Working Group World Reference Base for Soil Resources (WRB). The study was set up in a completely randomized design with three replicates for each treatment. Each replicate was set up on a 2 m × 1 m furrow (plot) which equates to an area of 2 m2. There was a 1-m wide ridge between the adjacent plots to prevent the contents of one plot from contaminating the neighbouring plot. The experiment utilised Chunkwang variety of Chinese cabbage whose seeds were purchased from Nonghyup, Seoul, South Korea. The seeds were, germinated on a moist filter paper inserted inside a petri dish. Two-day old seedlings were transferred to a plastic seedling tray and grown under the controlled greenhouse conditions for 3 weeks. The growing media used to raise the seedlings in the seedling tray were exactly similar to the field treatments, i.e., seedlings raised on the modified biochar-amended soil media were transplanted to the modified biochar-amended plots. Each plot was planted with two rows of Chinese cabbage with spacings of 0.4 m and 0.5 m within and between the plant rows, respectively. Along with the six biochar treatments which included KHB, HPB, SAB, URB, ANB, and PRB, a control (urea-only amendment) was included. All biochars were applied to the soil at a rate of 3% (v/v) and nitrogen in form of urea was applied at a rate 150 kg per ha. The nitrogen was split into two applications for all other treatments except URB and ANB. The field experiment lasted for 9 weeks after transplanting 3 weeks old seedlings. All proper agronomic practices including irrigation, weed management, pests and diseases control, etc. were observed throughout the experimentation period.
2.3 Quantification of the ammonia emissions
The gaseous NH3 emissions were quantified by strictly following the venting method as outlined by Yang et al. (2018). Briefly, the sampling device was made of polyvinyl chloride rigid plastic bucket with a diameter of 15 cm and a height of 10 cm. Two sponges uniformly impregnated with 15 mL of glycerol solution made by mixing 50 mL of phosphoric acid with 40 mL of glycerol and diluted to 1000 mL were placed in the plastic bucket. The sponges had a thickness of 2 cm and a diameter of 16 cm. The lower sponge was 5 cm from the bottom of the bucket whereas the upper sponge was flat with the top of the bucket. The role of the upper sponge was to block the atmospheric ammonia from entering the sampling device while the lower sponge absorbed the gaseous NH3 emitted from the soil. The gaseous NH3 absorbed was extracted in excess 2 M KCl solution, filtered through a 0.45-µm filter paper using a vacuum filter and then analysed colourimetrically following the indophenol blue method. Sampling lasted for 60 days and was carried out daily in the first 7 weeks and every after 2 days from the 7th week until the end of the experiment.
2.4 Assessment of the Chinese cabbage yield parameters and soil chemical properties
After 8 weeks, the Chinese cabbage was harvested and its yield parameters which included fresh and dry leaf weight, fresh and dry root weight, leaf length, leaf diameter, root length, and chlorophyll content of the leaves were assessed. The weights of leaves and roots were determined using a portable electric weighing scale (Daihan Scientific, South Korea). The weighing of the roots followed thorough washing to remove all the soil and gentle rubbing with the serviette to remove water. The dry shoot and root weights were determined after drying of the materials for 24 h at 105 °C in a forced convection oven (ON-12Gw L080125, Jeio Tech, Daejeon, South Korea). Root and leaf lengths including leaf width were measured with a metre rule. The chlorophyll content was determined using a chlorophyll meter (SPAD-502, Konica Minolta, Tokyo, Japan). Along with the yield parameters, the nitrogen use efficiencies (NUE) of the Chinese cabbage grown on the different amendments were computed using the equation adopted from Anas et al. (2020).
The effects of both the modified and N-enriched biochars on the selected chemical properties of the soil including pH, EC, organic carbon, available phosphorus, available N, and cations were also evaluated. The soil organic carbon content was determined colourimetrically at 645 nm using a UV–Vis spectrophotometer (Genesys 50, Thermo Scientific Inc., Waltham, Massachusetts, USA) following a method espoused by Jones (2001) but the standard curve was prepared from absorbance values of the glucose solutions of varying concentrations. Available N was computed as a sum of ammonium and nitrate contents of the soil which were extracted with 2 M KCl solution and then determined colourimetrically. The ammonium content was determined following the indophenol blue method whereas the nitrate content was determined by paying strict adherence to the Chromotropic Acid Procedure as outlined in Jones (2001). The available phosphorus was extracted with the Olsen solution and determined colourimetrically following the vanadate molybdate method. The soil cations were extracted with 1 M neutral ammonium acetate solution and quantified with the ICP-OES (iCAP 7000 series, Thermo Scientific Inc., Waltham, Massachusetts, USA). The soil pH and EC were determined with a pH and EC meter from the soil slurry prepared by dissolving 1 g of dry soil in 5 ml of water.
2.5 Data analysis
The analysis of the data obtained from the experiments was executed through a one-way analysis of variance (Anova) using GenStat 12th edition. The significantly different mean values at p < 0.05 were subjected to a post hoc t-test at 5% level of significance to quantify the differences amongst the treatments. Variability of the data was expressed as the standard deviation. Additionally, linear relationships between the cumulative gaseous NH3 volatilisations and the pH of the biochar, and N use efficiency were evaluated using Microsoft Excel 16 data analysis ToolPak.
3 Results
3.1 Ammonia emissions
As seen in Figs. 1 and 2, the daily emissions of gaseous NH3 from the urea-only amendment by far exceeded those of the biochar amendments. Usually, the emissions are expected to either cease or greatly diminish after the first week of applying the N fertilisers to the soil but our observation contravenes that assumption since the emissions remained relatively high through the third week. Additionally, the emissions from the first instalment of urea peaked on the 13th day of the experiment which was late since the emissions from the second urea instalment peaked only after 4 days. While urea was equally split into two applications, the emissions ensuing from the first instalment were far higher than those that resulted from the second application. Cumulatively, the control (urea-only amendment) volatilised 3066.7 µg of gaseous NH3 for every gram of N fertiliser applied to the soil. The emissions from the control dwarfed those of the biochar amendments which cumulatively volatilised 1711.90 µg, 1054.28 µg, 1658.64 µg, and 1127.61 µg of NH3 per gram of N fertiliser applied in the PRB-, SAB-, KHB-, and HPB-amended soils, respectively. The cumulative emissions from the soils amended with N-enriched biochar were even lower, standing at just 714.17 µg and 838.42 µg of NH3 per gram of N applied.
3.2 Chinese cabbage yields and soil chemical properties
As shown in Table 2, biochar application resulted in higher Chinese cabbage marketable yields. In comparison with the urea-only amendment, PRB, SAB, KHB, HPB, ANB, and URB amendments increased Chinese cabbage yields by 138.5%, 172.0%, 117.3%, 181.1%, 194.0%, and 181.1%, respectively. Consequently, the weights of the dry shoots of the cabbage produced with the biochar amendments were heavier than the dry shoot weight of the control with both ANB and URB producing the heaviest dry shoots with no significant statistical differences between them. There were no significant statistical differences between SAB and HPB, and KHB and PRB amendments regarding the dry shoot weights. Additionally, the marketable yields of the Chinese cabbage grown with HPB and URB amendments were statistically similar and did not significantly differ from the marketable yields of both ANB and SAB statistically. In the same vein, there were no statistically significant differences between the yields obtained from the PRB and KHB amendments.
ANB and URB amendments produced the Chinese cabbage with the longest leaves of all the treatments with no significant statistical differences between them. These were followed by HPB and SAB amendments with no significant statistical differences between them. It is worthy a note that the lengths of the leaves produced with HPB, and SAB did not differ significantly from those of ANB and URB statistically. The shortest leaves came from the Chinese cabbage produced with the urea only amendment followed by those of the PRB and KHB amendments which did not exhibit any significant statistical differences between them. The widths of the leaves followed a similar trend as the lengths of the leaves but there were no significant statistical differences between the control and PRB, and between KHB and SAB amendments. Additionally, the average widths of the leaves of the cabbage produced with HPB amendment were not statistically different from those of the cabbage produced with ANB, URB, SAB, and KHB amendments.
The heaviest fresh roots came from the cabbage grown on SAB, HPB, ANB, and URB amendments with no significant statistical differences amongst them. The lightest fresh roots were obtained from the cabbage grown on the control (urea-only) amendment followed in an ascending order by the roots of the cabbage grown on the KHB and PRB amendments. The ANB and URB produced the Chinese cabbage with the heaviest dry roots with no significant statistical differences between them followed by the HPB amendment. The weight of the dry roots of the cabbage grown on the SAB amendment was statistically similar to the weights of PRB, KHB and HPB amendments. The control produced Chinese cabbage with the lightest dry roots followed by PRB and KHB with no significant statistical differences observed between them. The lengths of the roots of the Chinese cabbage produced from all the amendments did not statistically differ significantly from one another.
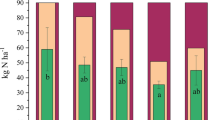
The chlorophyll content was highest in all biochar amended soils with no significant statistical differences amongst them except the KHB amendment whose Chinese cabbage contained lower chlorophyll content than the rest of biochars. The control produced the Chinese cabbage with the lowest content of chlorophyll. As shown in Fig. 3, the N-use efficiencies were generally higher in the biochar amendments than the control with the ANB amendment registering the highest N-use efficiency. The urea-only amendment resulted into an N-use efficiency of 33.83%. The lowest N-use efficiency amongst the biochar amendments came from the KHB amendment which registered an N-use efficiency of 73.53%. This was followed by the PRB amendment with an N-use efficiency of 80.69%. Even though the HPB amendment registered higher N-use efficiency of 94.12% than the SAB amendment whose efficiency stood at 92. 03%, there were no significant statistical differences between the amendments. N-enriched biochar amendments of URB and ANB resulted in the highest N-use efficiencies of all the biochar amendments registering efficiencies amounting to 95.09% and 99.48%, respectively.
As can be seen from Table 3 and soil data outlined in Egyir et al. (2022), SAB, HPB, and ANB amendments decreased soil pH whereas KHB, URB, and the control amendments increased it. The reduction from the pH value of 7.20 before experiment to the value of 6.60 after experiment registered in the SAB amendment was the largest reduction of all. There were no significant statistical differences amongst the pH values of the soil amended with KHB, URB, and the control after the experiment. The pH values of the soil amended with HPB and ANB did not significantly differ statistically from those of SAB and KHB, URB, and the control. All amendments increased soil EC with the highest increment recorded in the KHB amended soil whereas the control amendment led to the lowest increment. Generally, the modified biochar amendments increased soil EC higher than any other amendment. All biochar amendments increased the soil organic carbon (SOC) content of the soil whereas there were no noticeable differences in the SOC content of the soil in the control amendment.
Generally, the modified biochars resulted into the lowest increments in SOC contents of all the biochars. The PRB amendment produced the highest increment in the SOC content which increased from 0.71% before the experiment to 4.46% after the experiment. All the biochar amendments increased the available phosphorus content with no significant statistical differences amongst them whereas the control amendment led to the reduction in the content of available phosphorus. All amendments increased the concentration of available N. The available N content was highest in the N-enriched biochar amendments of ANB and URB with no significant statistical differences between them. The lowest concentration of available N was detected in the control and KHB amendments with no significant statistical differences between them followed by the PRB amendment. The PRB and the control amendments elicited the highest and lowest increases, respectively, in the concentrations of Ca, K and Mg. However, there were no noticeable differences in the concentrations of Na both before and after experiments.
4 Discussions
4.1 Ammonia emissions
In agreement with several formerly published studies, biochar amendments reduced gaseous NH3 emissions from the soil. PRB, KHB, HPB, SAB, URB, and ANB amendments abated NH3 volatilisations from the soil by 44.18%, 45.91%, 63.23%, 65.62%, 72.66%, and 76.71% respectively in comparison with the control amendment. These reductions were lower than those reported in the incubation experiments by Egyir et al. (2022) where the modified biochars abated NH3 volatilisations by 63.06–74.85% while URB and ANB amendments attenuated gaseous emissions by 79.93% and 82.88%, respectively. These observations underpin the limitations of the incubation studies in providing accurate real-field trends which calls for our caution to avoid making general conclusions with data from laboratory experiments (Fisher & Wood, 2007; Brüggemann and Bizer 2016). Additionally, the daily NH3 emissions contravened the observations made by several previous studies including Mandal et al. (2016), Luyima et al. (2020), etc., since the emissions did not greatly reduce after the first week of applying the N fertilisers to the soil as expected.
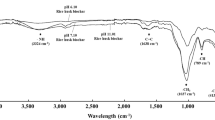
An earlier study by Egyir et al. (2022) indicated that modifying biochar with hydrogen peroxide and sulphuric acid increased the content of oxygen-containing surface groups especially the carboxylic and sulphoxide groups in HPB and SAB, respectively. The increase in the oxygen-containing surface groups enhanced the protonation of gaseous NH3 to ammonium and oxidation of ammonium to nitrates hence optimising the abatement of NH3 volatilisation. In the incubation study, KHB reduced gaseous NH3 emissions from the soil better than the PRB amendment but in this field study, there were no significant statistical differences between the two amendments. Egyir et al. (2022) opined that modification of the biochar with potassium hydroxide increased the sorption of nitrates into the biochar through the potassium exchange sites which resulted into higher reduction of NH3 volatilisation than the non-modified biochar. However, that mechanism seemed ineffective under the field conditions but it’s hard for us to come up with the reasons to explain this observation.
Similar to what was observed in the incubation studies, the N-enriched biochar amendments caused the largest reductions in the emissions of gaseous NH3. This observation concurred with that of Luyima et al. (2020) who observed that nutrient-enriched biochar abated gaseous NH3 emissions by as high as 85.93% and was more effective than the urea-only amendment in reducing the emissions. The instrumental analysis conducted by Egyir et al. (2022) indicated that N in the N-enriched biochars reacted with the oxygen-containing surface groups which slowed its release, concomitantly abating gaseous NH3 emissions. In a study reported by Nutt (2019), a group of scientists discovered the formation of covalent bonds between nitrogen and biochar carbon with an accompanying reduction in the amount of gaseous NH3 volatilised.
4.2 Chinese cabbage yields and soil chemical properties
Biochar has been reported to increase crop yields in tropical but not temperate soils (Jeffery et al., 2017a, b). Contrary to this observation, however, all biochar amendments increased the yields of the Chinese cabbage with the worst performing amendment producing more than twice the yield obtained from the urea-only amendment. The increase in crop yields observed upon biochar applications may be ascribed to a variety of reasons including; biochar’s role in the dynamics of plant nutrients (Jeffrey et al., 2017b; Luyima et al., 2021), its liming effect (Jeffery et al., 2011), and its effect on both soil hydrology and biotic interactions (Jeffrey et al., 2017b). Biochars supply important macro and micro plants nutrients (Jeffrey et al., 2017b; Luyima et al., 2021) while its liming effect reduces the concentrations of Fe and Al in the soil solution with a resultant increase in the availability of the previously bound P to plants (Cui et al., 2011). Biochar supports the growth of arbuscular mycorrhizal fungi (AMF) important for the increased acquisition of P by plants as Shen et al. (2016) observed.
Biochar plays a huge role in the sustainable utilisation of the applied N through reducing gaseous emissions and leaching (Taghizadeh-Toosi et al., 2012). It also increases N inputs through increased N fixation (Rondon et al., 2007), the net outcome of which is a higher N availability to the growing crop and higher N-use efficiency. Indeed, there was a strong negative linear relationship (r2 = 0.97) between the cumulative gaseous ammonia evolved from the different amendments and the N-use efficiency (see Fig. 4a) in the present study. This implies that biochar amendment increased use efficiencies of the applied N by abating gaseous NH3 emissions. The higher use-efficiencies of the applied N obtained from the biochar amended soils resulted from higher N uptake by the Chinese cabbage. The higher N uptake in turn resulted into higher Chinese cabbage yields as confirmed by the high positive linear relationship (r2 = 0.97) between the Chinese cabbage yields and the quantity of N taken up by the Chinese cabbage (see Fig. 4b).
The KHB amendment produced lower yields than even the non-modified biochar possibly because it contained higher amounts of chloride which competed with nitrate ions for uptake into the growing Chinese cabbage, thus affecting its yielding potential (Geilfus, 2018). Additionally, both KHB and the non-modified biochars were ineffective at abating N losses which limited the amount of nutrients available to the growing crops and hence lower yields than the rest of the biochar amendments. The non-modified biochar might have contained high amounts of labile carbon (C) at its surface which immobilised some of the applied N as Jeffrey et al. (2017b) opined, leading to lower yields than those of the modified biochars. The chlorophyll contents were higher in the Chinese cabbage grown on the modified and N-enriched biochars than on the control probably because they supplied adequate nutrients required in the formation of chlorophyll. N, magnesium (Mg), and iron (Fe) have huge direct impacts on the formation of chlorophyll as Ciompi et al. (1996) and Laing et al. (2000) demonstrated.
Therefore, since biochar amendments greatly reduced loss of N from the soil in form of NH3 and possibly through leaching, they supplied it to the growing Chinese cabbage in quantities sufficient for the normal chlorophyll formation process. Additionally, the biochar amendments contained substantial quantities of Mg which they supplied to the growing Chinese cabbage resultantly boosting the formation of chlorophyll. Given the sheer importance of chlorophyll in influencing customer choice and their recognised roles in health such as prevention of several diseases associated with oxidative stress, including cancer, cardiovascular diseases, and other chronic diseases (Sangeetha et al., 2010; Yang et al., 2021), it is commendable to conclude that the biochar amendments produced Chinese cabbage with the highest health value and marketability. The dry matter accumulation (dry shoot and root weights) was higher in the N-enriched biochars than the rest of the biochars which indicates that the N-enriched biochars supplied more N to the growing Chinese cabbage than the rest of the amendments. This is because studies by Mehrotra and Singh (1982), and Kumar and Sharma (1999) demonstrated that the dry matter accumulation in rice and wheat increased with increasing application rates of N which underpins the irreplaceable role of N in dry matter accumulation in crops.
The increases in soil pH and EC upon biochar application have been demonstrated by several studies including Chintala et al. (2013), Shen et al. (2016), Trupiano et al. (2017), Luyima et al. (2019), Lee et al. (2019), and others. The biochar-induced increases in soil pH have been ascribed to the liming power of biochar by Trupiano et al. (2017) whereas the increases in EC are attributed to the exchange of electrons between the soil solution and the added biochar (Joseph et al., 2010). In the present study, only PRB, KHB, and URB amendments caused considerable increases in soil pH, SAB caused considerable decreases in soil pH while the rest of the amendments generally had a benign effect on soil pH. The modification processes de-ashed the biochar which negated the liming potentials of the biochar but the alkalinity of potassium hydroxide might have compensated for the lost liming potential of the KHB biochar. The strong acidity of ammonium nitrate in solution might have masked the liming potential of biochar in ANB, hence the benign effect on soil pH.
The increases in the soil EC observed across all the amendments were a result of the compounding effect of the redox reactions and cation and anion exchange capacities of the biochar amendments as Joseph et al. (2010) demonstrated. The higher increments in soil EC observed with the modified biochar amendments concurred with the observations made by previous studies for example by Wang et al. (2021). On the other hand, the lower increments in soil organic carbon observed in soils amended with the modified biochar than those of the soils amended with pristine biochar may be due to the lack of readily releasable labile carbon contained in the PRB. In agreement with several previous studies by Shen et al. (2016), Lee et al. (2019), Luyima et al. (2019), and others, all biochar amendments increased the amount of soil available phosphorus with no statistically significant differences amongst the different amendments. This increase could have resulted from the biochar-induced increases in soil pH which stimulated increases in mobility and availability of phosphorus in the soil (Shen et al., 2016). However, given the fact that not all the biochar amendments increased soil pH, the increase in available phosphorus could be due to the direct release of phosphorus from the biochar itself as Deluca et al. (2009) opined. Another possible explanation for increases in available phosphorous is possibly the biochar-induced proliferation of arbscular mycorrhizal fungi which formed symbiotic relationships with the crops grown (Shen et al., 2016).
5 Conclusions
The N-enriched biochar amendments elicited the largest reductions in the amount of gaseous NH3 evolved and produced the highest Chinese cabbage yields. The biochar-induced reductions in the quantity of gaseous NH3 emitted had a direct positive impact on the use efficiencies of the applied N which in turn affected the Chinese cabbage yields. Generally, KHB was inferior over other modified biochars in both agronomic performance of the Chinese cabbage and the reduction of gaseous NH3 volatilisations.
Data availability
The data sets supporting the conclusions made are included in the article whereas a limited amount of the modified and nitrogen-enriched biochars can be provided to whoever may want to experiment with them upon request to the corresponding author.
References
Anas, M., Liao, F., Verma, K. K., Sarwar, M. A., Mahmood, A., Chen, Z.-L., Li, Q., Zeng, X.-P., Liu, Y., & Li, Y.-R. (2020). Fate of nitrogen in agriculture and environment: agronomic, eco-physiological and molecular approaches to improve nitrogen use efficiency. Biological Research, 53(1), 47. https://doi.org/10.1186/s40659-020-00312-4
Baitong, C., Koziel, J. A., Białowiec, A., Lee, M., Ma, H., O’Brien, S., Li, P., Meiirkhanuly, Z., & Brown, R. C. (2021). Mitigation of acute ammonia emissions with biochar during swine manure agitation before pump-out: proof-of-the-concept. Frontiers in Environmental Science, 9, 613–614. https://doi.org/10.3389/fenvs.2021.613614
Brüggemann, J., & Bizer, K. (2016). Laboratory experiments in innovation research: A methodological overview and a review of the current literature. Journal of Innovation and Entrepreneurship, 5, 24. https://doi.org/10.1186/s13731-016-0053-9
Chen, D., Liu, X., Bian, R., Cheng, K., Zhang, X., Zheng, J., Joseph, S., Crowley, D., Pan, G., & Li, L. (2018). Effects of biochar on availability and plant uptake of heavy metals – a meta-analysis. Journal of Environmental Management, 222, 76–85. https://doi.org/10.1016/j.jenvman.2018.05.004
Chintala, R., Mollinedo, J., Schumacher, T. E., Malo, D. D., & Julson, J. L. (2013). Effect of biochar on chemical properties of acidic soil. Archives of Agronomy and Soil Science, 60(3), 393–404.
Ciompi, S., Gentili, E., Guidi, L., & Soldatini, G. F. (1996). The effect of nitrogen deficiency on leaf gas exchange and chlorophyll fluorescence parameters in sunflower. Plant Science, 118, 177–184. https://doi.org/10.1016/0168-9452(96)04442-1
Cowie, A., Woolf, D., Gaunt, J., Brandão, M., Anaya de la Rosa, R., & Cowie, A. (2015). Chapter 27: Biochar, carbon accounting and climate change. In J. Lehmann, & J. Joseph (Eds.), Biochar for Environmental Management (2nd ed.). Routlenge
Cui, H. J., Wang, M. K., Fu, M. K., & Ci, E. (2011). Enhancing phosphorus availability in phosphorus-fertilized zones by reducing phosphate adsorbed on ferrihydrite using rice straw-derived biochar. Journal Soils and Sediments, 11, 1135–1141.
Deluca, T. H., MacKenzie, M. D., & Gundale, M. J. (2009). Biochar effects on soil nutrient transformations. In J. Lehmann & S. Joseph (Eds.), Biochar for Environmental Management: Science and Technology (pp. 251–270). Earth scan.
Enders, A., Sohi, S., Lehmann, J., & Singh, B. (2017). Chapter 9: Total elemental analysis of metals and nutrients in biochars. In B. Singh, M. Camps-Arbestain, & J. Lehmann (Eds.), Biochar: A Guide to Analytical Methods (1st ed., pp. 95–108). CRC Press.
Egyir, M., Luyima, D., Park, S.-J., Lee, K.-S., & Oh, T.-K. (2022). Volatilisations of ammonia from the soils amended with modified and nitrogen-enriched biochars. Science of the Total Environment, 835, 155453. https://doi.org/10.1016/j.scitotenv.2022.155453
Fan, C., Chen, H., Li, B., & Xiong, Z. (2017). Biochar reduces yield-scaled emissions of reactive nitrogen gases from vegetable soils across China. Biogeosciences, 14(11), 2851–2863.
Feng, Y., Sun, H., Xue, L., Liu, Y., Gao, Q., Lu, K., & Yang, L. (2017). Bio-char applied at an appropriate rate can avoid increasing NH3 volatilization dramatically in rice paddy soil. Chemosphere, 168, 1277–1284.
Fisher, C. G., & Wood, K. B. (2007). Introduction to and techniques of evidence-based medicine. Spine, 32(Supplement), S66–S72. https://doi.org/10.1097/brs.0b013e318145308d
Geilfus, C.-M. (2018). Review on the significance of chlorine for crop yield and quality. Plant Science, 270, 114–122.
Janczak, D., Malińska, K., Czekała, W., Cáceres, R., Lewicki, R., & Dach, A. (2017). Biochar to reduce ammonia emissions in gaseous and liquid phase during composting of poultry manure with wheat straw. Waste Management, 66, 36–45. https://doi.org/10.1016/j.wasman.2017.04.033
Jeffery, S., Abalos, D., Spokas, K. A., & Verheijen, F. G. A. (2017b). Chapter 12: Biochar effects on crop yield. In J. Lehmann, & J. Joseph (Eds.), Biochar for Environmental Management (2nd ed.). Routledge.
Jeffery, S., Verheijen, F. G. A., van der Velde, M., & Bastos, A. C. (2011). A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agriculture, Ecosystems and Environment, 144, 175–187.
Jeffery, S., Verheijen, F. G. A., Kammann, C., & Abalos, D. (2016). Biochar effects on methane emissions from soils: A meta-analysis. Soil Biology and Biochemistry, 101, 251–258. https://doi.org/10.1016/j.soilbio.2016.07.021
Jeffery, S., Abalos, D., Prodana, M., Bastos, A. C., van Groenigen, J. W., Hungate, B. A., & Verheijen, F. (2017a). Biochar boosts tropical but not temperate crop yields. Environmental Research Letters, 12(5), 053001. https://doi.org/10.1088/1748-9326/aa67bd
Jones, J. B., Jr. (2001). Laboratory guide for conducting soil tests and plant analysis; CRC Press: Boca Raton. FL.
Joseph, S. D., Camps-Arbestain, M., Lin, Y., Munroe, P., Chia, C. H., Hook, J., & Amonette, J. E. (2010). An investigation into the reactions of biochar in soil. Australian Journal of Soil Research, 48(7), 501.
Kumar, R., & Sharma, S. N. (1999). Effect of nitrogen on dry matter and nutrient accumulation pattern in wheat (Triticum aestivum) under different dates of sowing. Indian Journal Agronomy, 44(4), 738–744.
Laing, W., Greer, D., Sun, O., Lowe, A., & Payn, T. (2000). Physiological impacts of Mg deficiency in Pinus radiata: Growth and photosynthesis. New Phytologist, 146, 47–57. https://doi.org/10.1046/j.1469-8137.2000.00616.x
Lee, J. H., Luyima, D., Lee, J. Y., Kim, S. J., & Oh, T. K. (2019). Effects of two biochar-based organic amendments on soil chemical properties and productivity of selected vegetables. Journal Faculty Agricultute Kyushu University, 64(1), 39–46. https://doi.org/10.5109/2231632
Lehmann, J., & Joseph, S. (2015). Chapter 1: Biochar for environmental management: An introduction. In Biochar for Environmental Management 2nd ed., Lehmann, J and Joseph, J., Eds., Routlenge, London
Luyima, D., Lee, J.-H., An, J.-Y., Kwon, O.-S., Park, S.-Y., Lee, S.-J., Park, S.-Y., Shinogi, Y., Park, K.-W., & Oh, T.-K. (2019). impact of synchronizing the application of different biochar organic fertilisers with NPK on soil chemical properties and growth of leek (Allium ampeloprasum). Journal Faculty Agricultute Kyushu University, 64(1), 47–53. https://doi.org/10.5109/2231633
Luyima, D., Lee, J.-H., Sung, J.-K., & Oh, T.-K. (2020). Co-pyrolysed animal manure and bone meal-based urea hydrogen peroxide (UHP) fertilisers are an effective technique of combating ammonia emissions. Journal Mater Cycles Waste Managment, 22, 1887–1898. https://doi.org/10.1007/s10163-020-01074-7
Luyima, D., Egyir, M., Yun, Y.-U., Park, S.-J., & Oh, T.-K. (2021). Nutrient dynamics in sandy soil and leaf lettuce following the application of urea and urea-hydrogen peroxide impregnated co-pyrolyzed animal manure and bone meal. Agronomy, 11, 1664. https://doi.org/10.3390/agronomy11081664
Luyima, D., Egyir, M., Lee, J.-H., Yoo, J.-H., & Oh, T.-K. (2022). A review of the potentiality of biochar technology to abate emissions of particulate matter originating from agriculture. International Journal of Environmental Science and Technology, 9, 3411–3428. https://doi.org/10.1007/s13762-021-03267-5
Malińska, K., Zabochnicka-Świątek, M., & Dach, J. (2014). Effects of biochar amendment on ammonia emission during composting of sewage sludge. Ecological Engineering, 71, 474–478. https://doi.org/10.1016/j.ecoleng.2014.07.012
Mandal, S., Thangarajan, R., Bolan, N. S., Sarkar, B., Khan, N., Ok, Y. S., & Naidu, R. (2016). Biochar-induced concomitant decrease in ammonia volatilization and increase in nitrogen use efficiency by wheat. Chemosphere, 142, 120–127.
Mehrotra, O. M., & Singh, I. J. (1982). Effect of rate and time of nitrogen application on dry matter production, N-uptake and yield of rice. Indian Journal Agriculture Research, 16, 29–35.
Nutt, D. (2019). Biochar soaks up ammonia pollution, study shows. https://news.cornell.edu/stories/2019/03/biochar-soaks-ammonia-pollution-study-shows. Accessed 31 May 2022.
Pinder, R. W., & Adams, P. J. (2007). Ammonia emission controls as a cost-effective strategy for reducing atmospheric particulate matter in the Eastern United States. Environmental Science & Technology, 41(2), 380–386. https://doi.org/10.1021/es060379a
Rondon, M. A., Lehmann, J., Ramırez, J., & Hurtado, M. (2007). Biological nitrogen fixation by common beans (Phaseolus vulgaris L.) increases with bio-char additions. Biology and Fertility of Soils, 43, 699–708.
Sangeetha, R. K., & Baskaran, V. (2010). Carotenoid composition and retinol equivalent in plants of nutritional and medicinal importance: Efficacy of b carotene from Chenopodium album in retinol-deficient rats. Food Chemistry, 119, 1584–1590.
Sha, Z., Li, Q., Lv, T., Misselbrook, T., & Liu, X. (2019). Response of ammonia volatilization to biochar addition: A meta-analysis. Science of the Total Environment, 655, 1387–1396. https://doi.org/10.1016/j.scitotenv.2018.11.316
Shen, Q., Hedley, M., Camps Arbestain, M., & Kirschbaum, M. U. (2016). Can bio-char increase the bioavailability of phosphorus? Journal of Soil Science and Plant Nutrition. https://doi.org/10.4067/S0718-95162016005000022
Subedi, R., Kammann, C., Pelissetti, S., Taupe, N., Bertora, C., Monaco, S., & Grignani, C. (2015). Does soil amended with biochar and hydro-char reduce ammonia emissions following the application of pig slurry? European Journal of Soil Science, 66(6), 1044–1053.
Sun, X., Zhong, T., Zhang, L., Zhang, K., & Wu, W. (2019). Reducing ammonia volatilization from paddy field with rice straw derived biochar. Science of the Total Environment, 660, 512–518.
Taghizadeh-Toosi, A., Clough, T. J., Sherlock, R. R., & Condron, L. M. (2012). Biochar adsorbed ammonia is bioavailable. Plant and Soil, 350, 7–69.
Trupiano, D., Cocozza, C., Baronti, S., Amendola, C., Vaccari, F. P., Lustrato, G., & Scippa, G. S. (2017). The effects of biochar and its combination with compost on lettuce (Lactuca sativa L.) growth, soil properties, and soil microbial activity and abundance. International Journal of Agronomy, 2017, 1–12.
Van Zwieten, L., Kammann, C., Cayuela, M. L., Singh, B. P., Joseph, S., Kimber, S., Donne, S., Clough, T., & Spokas, K. A. (2017). Chapter 17: Biochar effects on nitrous oxide and methane emissions from soil. In J. Lehmann, & J. Joseph (Eds.), Biochar for Environmental Management (2nd ed.). Routlenge
Wang, Y., Zheng, K., Zhan, W., Huang, L., Liu, Y., Li, T., Yang, Z., Liao, Q., Chen, R., & Zhang, C. (2021). Highly effective stabilization of Cd and Cu in two different soils and improvement of soil properties by multiple-modified biochar. Ecotoxicology and Environmental Safety, 207, 111294.
Wiedner, K., & Glaser, B. (2015). Chapter 2: Traditional use of biochar. In J. Lehmann, & J. Joseph (Eds.), Biochar for Environmental Management (2nd ed.). Routlenge
Yang, J., Jiao, Y., Yang, W. Z., Gu, P., Bai, S. G., & Liu, L. J. (2018). Review of methods for determination of ammonia volatilization in farmland. IOP Conf. Ser. Earth Environ. Sci., 113, 012022.
Yang, J.-W., Luyima, D., Park, S.-J., Kim, S.-H., & Oh, T.-K. (2021). Mixing sodium-chloride-rich food waste compost with livestock manure composts enhanced the agronomic performance of leaf lettuce. Sustainability, 13, 13223. https://doi.org/10.3390/su132313223
Acknowledgements
This research was funded by the Cooperative Research Program for Agricultural Science & Technology Development of the Rural Development Administration, Republic of Korea, (Project No. PJ017028).
Author information
Authors and Affiliations
Contributions
Michael Egyir: conceptualization, methodology, investigation, writing—original draft. Deogratius Luyima: conceptualization, methodology, investigation, formal analysis. Seong-Heon Kim: conceptualization, resources, supervision. Taek-Keun Oh: conceptualization, supervision, project administration, funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Egyir, M., Luyima, D., Kim, SH. et al. Effects of Modified and Nitrogen-Enriched Biochars on Ammonia Emissions and Crop Yields Under a Field Environment. Water Air Soil Pollut 233, 439 (2022). https://doi.org/10.1007/s11270-022-05871-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05871-8