Abstract
Microplastics (MPs) in sediments from the complex lagoon-channel of Bizerte were investigated, for the first time, to evaluate the occurrence and abundance of MPs in Tunisia. After density separation in saline solution, MPs were counted by a stereomicroscope. The number of MPs was at the range of 3–18 items/g sediment (3000–18,000 items/kg dry sediment) and the most contaminated site was of Menzel Abderrahmane (MA) followed by Carrier Bay (CB), Menzel Jemil (MJ) and Channel of Bizerte (C). The MPs gathered during the survey varied in size from 0.3 to 5 mm, and appear in a variety of shapes and colours. The dominant shape was fibre (88.88% in MA, 91.00% in CB, 82.35% in C and 21.05% in MJ). The rest of MPs are fragments whilst no micro beads were found. Colours are clear, white, blue, green, red and black. Cities discharges, fishing activity and industrial production sites are the most likely sources of MPs. This first work provides original data on the presence of MPs that determines their bioavailability to organisms as seafood, and then possibly transfers of to human. The high MP concentrations registered in the complex lagoon-channel of Bizerte suggest that this site is a hotspot for MP pollution and there is an urgency to understand their origins and effects on marine life. The results will provide useful background information for further investigations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Plastics, also called polymers, are produced by the conversion of natural products or by the synthesis from primary chemicals generally coming from natural gas and crude oil (Rios et al. 2007). Demand in this synthetic product is considerable since these materials started to be utilized. As a result, the global plastic production has increased dramatically from 1.5 million tonnes in the 1950s to approximately 311 million tonnes in 2014 (Plastics Europe 2015) and between 2 and 5% of this production has been estimated to end in the marine environment (Jambeck et al. 2015).
In the marine environment, the presence of plastic material has grown rapidly with the consequence that nowadays, 40–80% of debris are plastic (Barnes et al. 2009; Cole et al. 2011). This plastic requires several centuries, or even thousands of years, to degrade (Barnes et al. 2009). Debris items are usually subdivided into different size categories: mega-debris (>10 cm); macro-debris (2–10 cm); meso-debris (2 cm-5 mm) and micro-debris (<5 mm) (Barnes et al. 2009). The smallest form of plastic litter is called microplastic (MP). It has been discovered in fresh and saltwater ecosystems, sediments and wastewater effluent around the world (Chang 2015). Van Cauwenberghe et al. (2015) showed that MPs are omnipresent in the marine environment and sediments are hypothesised to be major sinks of these plastics. MPs enter the marine environment via multiple pathways. Their presence can result from either the degradation of macroplastics, in situ, due to mechanical, photolytic and/or chemical degradation processes (Mathalon and Hill 2014) or from the direct input by industrial processes and domestic uses (Costa et al. 2010). Browne et al. (2011) showed that the important source of MPs comes from synthetic clothing: a single synthetic garment can release up to 1900 fibres per washing cycle.
Environmental risks associated with MPs are their bioavailability for marine organisms (Wright et al. 2013). In fact, these compounds have been ingested by marine biota via direct and indirect ingestion (Gall and Thompson 2015; Gross 2015). Thompson et al. (2004), Ward and Shumway (2004), Murray and Cowie (2011) and Li et al. (2015) showed that MPs are known to be ingested by invertebrates such as polychaetes, crustaceans, echinoderms, bryozoans and bivalves. Browne et al. (2008) showed that the blue mussel Mytilus edulis can ingest and transport MP particles to the gut, where they then accumulate in the digestive cavity and tubules. Derraik (2002) and Eriksson and Burton (2003) reported that MPs can be ingested by vertebrates. Fossi et al. (2012, 2014) showed that contamination by MPs might affect the marine food chain from the smallest planktivores to the largest fish and marine mammal species. Watts et al. (2014) showed that MPs reach higher trophic levels including humans. Pedà et al. (2016) showed intestinal alterations in European sea bass Dicentrarchus labrax exposed to MPs. Cole et al. (2011) and Wright et al. (2013) showed that MPs affect many species of marine organisms, from suspension- and filter feeders to deposit feeders and their risks not only involve ingesting the plastic itself but also regard the contamination of organisms by chemical pollutants adsorbed on them. In fact, due to their hydrophobic nature, persistent organic pollutants such as polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs) and dichlorodiphenyltrichloroethane (DDTs) can be absorbed onto MP surfaces (Frias et al. 2010; Teuten et al. 2007) and can be released into the organisms’ tissues when ingested (Engler 2012). This allows MPs to act as reservoirs of toxic chemicals in the environment and as these toxins work their way up the food chain, we may begin to see concentration of toxins in humans.
Alomar et al. (2016) showed that MP concentrations do not increase with decreasing sediment grain size as it is the case for organic material and other contaminants. Browne et al. (2010) also did not observe any clear relationship between MP abundance and the proportion of clay in the analysed sediments.
In Tunisia, the presence of MPs as contaminants has not yet been investigated in any environmental compartment and currently, there is no regulation of MPs. Thus, the present study aims to examine, for the first time, the presence, abundance and composition of MPs in the sediments of the complex lagoon-channel of Bizerte (Northern Tunisia). The results will provide useful background information for further investigations.
2 Material and Methods
2.1 Sampling Site
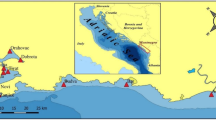
The lagoon of Bizerte covers roughly 150 km2 and represents an economically important body of water due to a variety of fishing and aquaculture activities. This lagoon is permanently connected to the Mediterranean Sea by a straight channel of 8.5 km long and communicates with the Ichkeul lake (110 km2) by the Tinja river. It is ranked second in terms of exports of seafood products. The Bizerte lagoon is bounded by the cities of Bizerte, Zarzouna, Menzel Jemil and Menzel Abderrahmane in the North side and Menzel Bourguiba and Tinja in the west and South west sides. Urban areas surrounding the complex lagoon-channel of Bizerte showed a very high demographic concentration and multiple industrial activities. Many industrial units related to various fields of activity such as petrochemicals, textiles, steel, plastics processing and ship repair and ship building facilities were created on the coasts of the lagoon (Yoshida et al. 2002; Ben Ameur et al. 2013; Lahbib et al. 2010, 2011; Abidli et al. 2013, 2015). Sediment samples were taken from four selected sites located in the complex lagoon-channel of Bizerte in January 2016. The locations of the sampling sites Menzel Abderrahmane (MA), Menzel Jemil (MJ), Carrier Bay (CB) and Channel (C) are shown in Fig. 1. The selection of these four sites was based on the demographic concentration and multiple industrial activities surrounding these locations and based also on the special value of these sites which represent most fish and shellfish productions.
Three 0.25 m × 0.25 m quadrats were used and wide pieces of natural debris (i.e. seaweed, stone, wood) were removed. Subsequently, the top layer of sediment (approximately 2–3 cm) was removed using a clean stainless steel spatula, from each sampling site and stored in closed containers, properly identified.
2.2 Laboratory Analysis
In the laboratory, sediment samples were dried at 50 °C during 48 h until complete dryness. Fifty grammes of dry sediment was introduced into 1-L beakers containing a high-density sodium chloride (NaCl; 140 g L−1) solution and stirred vigorously for 5 min, before collecting water to filter (Galgani et al. 2013). The supernatant water was then filtered onto Sartorius stedim biotech (particle retention 7–10 μm; diameter of 155 mm). The filters were analysed under a stereomicroscope to sort and measure the microscopic plastic particles using a calibrated eyepiece. Images of MPs were taken with a Casio (14 MP) camera attached to the stereomicroscope.
The most common MPs are fibres, fragments and pellets (Frias et al. 2016). In the present study, MP particles were classified into two types: fibre and fragment (Galgani et al. 2013) because pellets were not detected. In addition, the colour of MPs is of particular concern since it could easily be mistaken for food by marine organisms and seabirds (Thompson et al. 2004) and may give information about its origin following the examination of large-size plastics which may result, after their fragmentation, in the formation of MPs of the same colour. In the present study, colour of plastic items was recorded and divided into six groups: white, blue, clear, black, green and red. MPs per gramme of dry sediment were given. All material used was rinsed twice before use with distilled water and all working surfaces were cleaned with alcohol.
2.3 Statistical Analyses
A total of 12 sediment samples were taken from the four selected sites, including replicates (three replicates per site). From each replicate, one subsample (50 g of dry sediment) was taken for MP analysis. Analysis of variance (ANOVA) was performed using the software Statistica 8.0. After testing ANOVA assumptions, statistical significance was evaluated through one-way ANOVA. Whenever ANOVA detected significant differences, post hoc comparisons were made using the HSD Tukey test (THSD). Percentage was compared among samples using the χ 2 test in the software R. In all statistical analyses, significance level was considered for P < 0.05.
3 Results
3.1 Microplastic Abundances
MPs were found in all of the four sampling sites, with the types and amounts varying considerably between sites. The average MP concentration for the four sites was 7.96 ± 6.84 particles/g dry sediment (7960 ± 6840 particles/kg dry sediment). The highest MP concentration was observed at MA (18 ± 2.0 particles/g dry sediment) (18,000 ± 2000 particles/kg dry sediment). The lowest average MP concentration was found at the Channel of Bizerte at a concentration of 3.4 ± 0.2 particles/g dry sediment (3400 ± 200 particles/kg dry sediment) (Fig. 2).
3.2 Microplastic Shapes, Colours and Sizes
The MPs gathered during the survey varied in size and appeared in a variety of shapes and colours. As to the shapes, fibres and fragments were the only the shapes registered in this study, with no pellets found (Fig. 3). Fibres were found to be the most abundant in MA (88.88%), in CB (91.00%) and in C (82.35%). In MJ, the abundant shape was fragment (78.95%) (Fig. 4).
In each station, coloured particles were found. The majority of fibres are clear followed by white, blue, red, green and black (Figs. 3 and 5) whilst, fragments were dominated by blue, green, black and red (Figs. 3 and 6).
MP sizes varied from 0.3 to 5 mm and the average (±SE) size for collected fibres and fragments was 1.39 ± 0.27 and 0.51 ± 0.19 mm, respectively (Fig. 7). The difference in size between the two shapes was statistically significant (THSD: F = 3.18, P = 0.02). MPs smaller than 0.3 mm were not collected for this study. Among locations, fibre and fragment sizes did not vary significantly (THSD: F = 2.02, P = 0.14 for fibres, F = 1.27, P = 0.31 for fragments).
4 Discussion
Scientific literature has extensively explored the various anthropogenic impacts affecting the complex lagoon-channel of Bizerte, but the presence of MPs as contaminants has not yet been investigated. The results of this study reveal that MP particles are present in all examined sediments, indicating their extensive dispersion in the complex lagoon-channel of Bizerte. These results corroborates that MPs ubiquitously exist in the marine environment (Cole et al. 2011). The MP concentrations found in the complex lagoon-channel of Bizerte sediments were at the range of 3–18 items/g sediment (3000–18,000 items/kg sediment). Quantified MP concentrations in this study are comparable to those reported in the literature for other studies worldwide, although the wide array of existing techniques and quantification units limits the comparison of results (Qiu et al. 2015). However, it is evident that the abundance of MPs in the complex lagoon-channel of Bizerte is higher than that of most studied locations. In fact, in sediment obtained from the coastal shallow waters of Mallorca Island and Cabrera Island (Balearic Islands, western Mediterranean), the higher value of MP was 100.78 ± 55.49 MPs/kg of dry sediment. In the Lagoon of Venice, Italy, Vianello et al. (2013) showed concentrations ranging between 672 and 2175 items/kg dry sediment, which are less high than the concentrations reported in the present study. In the western Sardinian coast (Central-Western Mediterranean Sea), MP abundance value was 0.15 items/m3 (de Lucia et al. 2014). All sampled sites in the present study showed higher values than sediments sampled from the Belgium coast (97.2 items/kg dry sediment) and harbour area (166.7 items/kg dry sediment) (Claessens et al. 2011). Along the German Baltic coast, MP concentrations in beach sediments were ranging between 14 and 532 fibres/kg dry sediment (Stolte et al. 2015). In Portugal, Frias et al. (2016) found that MP abundance in coastal sediments from Southern Portuguese shelf waters was 0.01 ± 0.001 MP g−1 (10 ± 1 MP kg−1). In Slovenia, Laglbauer et al. (2014) revealed that the sediment of beach contains a concentration of 177.8 items/kg dry sediment. In the Canadian Lake Ontario nearshore, the abundance of MPs in sediment varied between 20 and 27,830 kg−1 (Ballent et al. 2016).
According to the type of MPs observed, identified MPs in this study were fibres and fragments, without plastic pellets. These results indicated that secondary MPs, deriving from the fragmentation of larger plastic debris through mechanical forces, by photolysis, thermo-oxidation, thermo-degradation and possibly via biodegradation processes (Laglbauer et al. 2014; Zhao et al. 2016), were the major sources of plastic debris in the studied locations. Our results corroborate the study of Frias et al. (2016) and Ballent et al. (2016) who showed that the majority of MPs in coastal sediments from Southern Portuguese shelf waters and from the Canadian Lake Ontario, respectively, were microfibers and fragments. In the present study, more than 82% of the MPs found in MA, CB and C were fibres. This type of MPs is indicative of sewage origin and synthetic garments from the textile industry that are an important source of MP contamination (Browne et al. 2011). Browne et al. (2011) showed that an increased microfibre load (>250%) was detected in sewage-sludge disposal sites compared to reference sites. In addition, Browne et al. (2011) concluded that up to 1900 fibres can be released into the environment from washing a single piece of clothing. Therefore, MP contamination in the populated areas of MA, CB and C could be attributed to sewage input and fragmentation of large plastic particles into smaller ones. In agreement with previous studies, fibres have been the most common shapes found in studies across the planet from the UK to Australia (Thompson et al. 2004; Browne et al. 2011). This is similar to the MP study conducted in Belgium (Claessens et al. 2011) that reported fibres as the most common type of prevailing MP particle. In Singapore’s coastal mangrove ecosystems, Nor and Obbard (2014) showed that fibres (72.0%) were the most common type of particle found. In the Canadian Lake Ontario nearshore, Ballent et al. (2016) revealed that the MP particles were primarily fibres and fragments. Contrarily, in MJ, more than 78% of the identified particles were fragments which are probably linked to fragmentation processes of bigger plastics and electrical plates which are thrown away by many industries that produce plastic and electronic plates. In addition, in this station, most shellfish production (Mediterranean mussel—Mytilus galloprovincialis and oyster—Crassostrea gigas) occurs and mussel/oyster farming used a lot of plastic for the cultivation of these bivalves. This plastic can be degraded by several factors as mechanical forces, photolysis, and possibly via biodegradation processes (Laglbauer et al. 2014; Zhao et al. 2016) leading to the formation of MPs in the shape of fragments.
MP concentrations in sediments found in this study show that higher levels were usually observed in areas of dense human population and high shipping and industrial activities as MA. In fact, the town of MA, with more than 16,000 inhabitants, is known for its fishing activity (harbour with a fishing fleet of 181 coastal boats) and with nearly 40% of the active population is working in the industry, especially the textile industry. Our results are in agreement with other results that showed that MPs appear to be more abundant in densely populated areas. In a study analysing sediments from 18 locations representing six continents, Browne et al. (2011) demonstrated a positive relationship between MP and human population density. Indeed, MPs are detected in large numbers in highly populated areas, such as at locations in the North Sea (Claessens et al. 2011; Liebezeit and Dubaish 2012; Thompson et al. 2004; Van Cauwenberghe et al. 2013) and the Mediterranean Sea (Kaberi et al. 2013; Vianello et al. 2013), as well as in Asia (Ismail et al. 2009; Ng and Obbard 2006; Nor and Obbard 2014; Reddy et al. 2006) and the highly populated coast of Brazil (Costa et al. 2010). Ballent et al. (2016) showed that MPs are concentrated in nearshore sediments in the vicinity of urban and industrial regions. On heavily polluted beaches, MPs can make up 3.3% of the sediment by weight, as opposed to 0.12% plastic by weight on control beaches (Carson et al. 2011). The link between MP pollution in sediments and human activities has also been demonstrated by Claessens et al. (2011), who detected particularly high concentrations of MP granules in the sediments of coastal harbours. Nevertheless, factors other than population density and industrial shipping activities may affect the accumulation of MPs in sediments. For instance, the level of MPs in sediment collected from the Channel of Bizerte was lower compared to other locations in this study, despite the very important human population (75,234 inhabitants), the important maritime traffic (~ 500 fishing boats and ~550 commercial boats), and industrial activities (petroleum refinery (Tunisian Refining Industries Company), lubricants processing unit (Sotulub), a cement plant (Cements of Bizerte) and the park of economic activities of the free zone of Bizerte which hosts several industrial and service companies). Some reasons can be cited to explain this fact, such as the strong upwelling currents in the Channel of Bizerte (during the sampling period). In fact, the Channel of Bizerte is a transition area between Bizerte Bay, which opens directly to the Mediterranean Sea, and Bizerte Lagoon, which is under the influence of terrestrial input. The low MP content in the sediment of this site might be related to the water current in the channel (personal observation) which can remove the surface sediment and reduce the MP accumulation which remains suspended in the water column.
The average MP size for collected fibres and fragments was 1.39 ± 0.27 and 0.51 ± 0.19 mm, respectively. The small size of MPs is of significance as it determines the potential impact of these contaminants on ecosystem biota. In fact, MPs of similar size to food have greater potential for ingestion by a wide range of organisms (Moore et al., 2001). Lusher et al. (2013) showed that the small size of MPs facilitates organisms’ intake compared to macroplastics. Van Cauwenberghe et al. (2015) revealed that particles accumulated in sediment are accessible to benthic suspension and deposit feeders and to other sediment-dwelling organisms (Wright et al. 2013). Thompson et al. (2004) showed that particles smaller than 20 μm have been seen to be actively ingested by small invertebrates. In addition, Rochman et al. (2013a) suggested that MPs ranging from 2 to 5 mm could take longer to pass from the stomachs of organisms and could be retained in the digestive system, potentially increasing the exposure time to adsorbed toxins. In the same context, Rochman et al. (2013b) found that polyethylene sorbs greater concentrations of PAHs and PCBs than other mass-produced polymers. The lagoon of Bizerte is ranked second in terms of exports of seafood products and based on our results concerning MP size, we can suggest that organisms living in this site are in danger. In the same context, Van Cauwenberghe et al. (2015) showed that many factors such as size of the plastic particles affect their potential bioavailability to a wide range of aquatic organisms and plastic particles may accumulate within organisms upon ingestion, resulting in direct effects caused by physical injury in the intestinal tract or even translocation to other tissues or organs, and indirect effect when MPs act as a vector for either chemicals or bacteria to marine organisms.
MPs reported in this study present different colours which is consistent with other studies on MPs (Nor and Obbard 2014; Zhao et al. 2014). In fact, the majority of the fibres identified were clear, white, red, blue, green and black. The green MP fibres recorded only in CB may result from the decomposition of ropes and fishing nets found in this site as well as this site is located near a military area and green fibres can be released from the washing of soldiers’ clothing. The majority of fragments were blue, green, black and red. The blue colour was recorded in all sites and probably is the result of the decomposition of blue bottles and plastic cans found on the beach of all sites. The green colour was recorded only in MA and MJ sites which are two sites under the influence of high urbanization and industrialization. Moreover, on the beach of these two sites, we found a lot of plastics of green colour (the majority are plastic electronic plates). Concerning the CB site, it is located in a military zone and it seems that this area is the main factor for MPs entering the site. The same result was registered by Stolte et al. (2015) who found that almost all samples of beach sediments (German Baltic coast) contained coloured fibres and 13 out of 22 sediment samples contained intensely coloured particles. Because of their resemblance of natural preys in oceanic waters, coloured MPs can be ingested preferably by mistaken for food by predator (Boerger et al. 2010; Browne et al. 2008; Lusher et al. 2013) and may result in internal damage of tissues in the digestive tract of organisms. These coloured MPs have been detected in fish, birds and turtles (Foekema et al. 2013; Hoarau et al. 2014).
5 Conclusions
The abundance of MPs found in the sediment of the complex lagoon-channel of Bizerte suggests that this contaminant may be entering the food chain through ingestion by marine organisms such as bivalves and fish and finally human following the consumption of these organisms. As MP abundances in the environment increase, further studies are planned to quantify the presence of MPs in all Tunisian coast, biota, notably fish and filter feeders such as bivalves, and assess their toxicological impact.
References
Abidli, S., Lahbib, Y., Rodríguez González, P., Ignacio García Alonso, J., & Trigui El Menif, N. (2013). Imposex and butyltin burden in Bolinus brandaris and sediment from the Tunisian coast. Hydrobiologia, 714, 13–24.
Abidli, S., Lahbib, Y., Rodríguez González, P., Ignacio García Alonso, J., & Trigui El Menif, N. (2015). Butyltin compounds in sediment and biota from the lagoon of Bizerte (northern Tunisia): potential risk for consumers? Human and Ecological Risk Assessment: An International Journal., 22, 337–349.
Alomar, C., Estarellas, F., & Deudero, S. (2016). Microplastics in the Mediterranean Sea: deposition in coastal shallow sediments, spatial variation and preferential grain size. Marine Environmental Research, 115, 1–10.
Ballent, A., Corcoran, P. L., Madden, O., Helm, P. A., & Longstaffe, F. J. (2016). Sources and sinks of microplastics in Canadian Lake Ontario nearshore, tributary and beach sediments. Marine Pollution Bulletin, 110, 383–395.
Barnes, D. K. A., Galgani, F., Thompson, R. C., & Barlaz, M. (2009). Accumulation and fragmentation of plastic debris in global environments. Philosophical Transactions of the Royal Society B, 364, 1985–1998.
Ben Ameur, W., Trabelsi, S., El Megdiche, Y., Hassine, S. B., Barhoumi, B., Hammami, B., Eljarrat, E., Barceló, D., & Driss, M. R. (2013). Concentration of polychlorinated biphenyls and organochlorine pesticides in mullet (Mugil cephalus) and sea bass (Dicentrarchus labrax) from Bizerte lagoon (northern Tunisia). Chemosphere, 90, 2372–2380.
Boerger, C. M., Lattin, G. L., Moore, S. L., & Moore, C. J. (2010). Plastic ingestion by planktivorous fishes in the North Pacific Central Gyre. Marine Pollution Bulletin, 60, 2275–2278.
Browne, M. A., Crump, P., Niven, S. J., Teuten, E., Tonkin, A., Galloway, T. S., & Thompson, R. C. (2011). Accumulation of microplastic on shorelines worldwide: sources and sinks. Environmental Science & Technology, 45, 9175–9179.
Browne, M. A., Dissanayake, A., Galloway, T. S., Lowe, D. M., & Thompson, R. C. (2008). Ingested microscopic plastic translocates to the circulatory system of the mussel, Mytilus edulis (L). Environmental Science & Technology, 42, 5026–5031.
Browne, M. A., Galloway, T. S., & Thompson, R. C. (2010). Spatial patterns of plastic debris along estuarine shorelines. Environmental Science & Technology, 44, 3404–3409.
Carson, H. S., Colbert, S. L., Kaylor, M. J., & McDermid, K. J. (2011). Small plastic debris changes water movement and heat transfer through beach sediments. Marine Pollution Bulletin, 62, 1708–1713.
Chang, M. (2015). Reducing microplastics from facial exfoliating cleansers in wastewater through treatment versus consumer product decisions. Marine Pollution Bulletin, 101, 330–333.
Claessens, M., De Meester, S., Van Landuyt, L., De Clerck, K., & Janssen, C. R. (2011). Occurrence and distribution of microplastics in marine sediments along the Belgian coast. Marine Pollution Bulletin, 62, 2199–2204.
Cole, M., Lindeque, P., Halsband, C., & Galloway, T. S. (2011). Microplastics as contaminants in the marine environment: a review. Marine Pollution Bulletin, 62, 2588–2597.
Costa, M. F., Ivar do Sul, J. A., Silva-Cavalcanti, J. S., Araúja, M. C. B., Spengler, A., & Tourinho, P. S. (2010). On the importance of size of plastic fragments and pellets on the strandline: a snapshot of a Brazilian beach. Environmental Monitoring and Assessment, 168, 299–304.
de Lucia, G. A., Caliani, I., Marra, S., Camedda, A., Coppa, S., Alcaro, L., Campani, T., Giannetti, M., Coppola, D., Cicero, A. M., Panti, C., Baini, M., Guerranti, C., Marsili, L., Massaro, G., Fossi, M. C., & Matiddi, M. (2014). Amount and distribution of neustonic micro-plastic off the western Sardinian coast (Central-Western Mediterranean Sea). Marine Environmental Research, 100, 10–16.
Derraik, J. G. B. (2002). The pollution of the marine environment by plastic debris: a review. Marine Pollution Bulletin, 44, 842–852.
Engler, R. E. (2012). The complex interaction between marine debris and toxic chemicals in the ocean. Environmental Science & Technology, 46, 12302–12315.
Eriksson, C., & Burton, H. (2003). Origins and biological accumulation of small plastic particles in fur seals from Macquarie Island. Ambio, 32, 380–384.
Foekema, E. M., De Gruijter, C., Mergia, M. T., Murk, A. J., van Franeker, J. A., & Koelmans, A. A. (2013). Plastic in North sea fish. Environmental Science & Technology, 47, 8818–8824.
Fossi, M. C., Coppola, D., Baini, M., Giannetti, M., Guerranti, C., Marsili, L., Panti, C., de Sabata, E., & Clò, S. (2014). Large filter feeding marine organisms as indicators of microplastic in the pelagic environment: the case studies of the Mediterranean basking shark (Cetorhinus maximus) and fin whale (Balaenoptera physalus). Marine Environmental Research, 100, 17–24.
Fossi, M. C., Panti, C., Guerranti, C., Coppola, D., Giannetti, M., Marsili, L., Panti, C., de Sabata, E., & Clo, S. (2012). Are baleen whales exposed to the threat of microplastics? A case study of the Mediterranean fin whale (Balaenoptera physalus). Marine Pollution Bulletin, 64, 2374–2379.
Frias, J. P. G. L., Gago, J., Otero, V., & Sobral, P. (2016). Microplastics in coastal sediments from Southern Portuguese shelf waters. Marine Environmental Research, 114, 24–30.
Frias, J. P., Sobral, P., & Ferreira, A. M. (2010). Organic pollutants in microplastics from two beaches of the Portuguese coast. Marine Pollution Bulletin, 60, 1988–1992.
Galgani, F., Hanke, G., Werner, S., & De Vrees, L. (2013). Marine litter within the European marine strategy framework directive. ICES Journal of Marine Science, 70, 1055–1064.
Gall, S. C., & Thompson, R. C. (2015). The impact of debris on marine life. Marine Pollution Bulletin, 92, 170–179.
Gross, M. (2015). Oceans of plastic waste. Current Biology, 25(3), R93–R96.
Hoarau, L., Ainley, L., Jean, C., & Ciccione, S. (2014). Ingestion and defecation of marine debris by loggerhead sea turtles, Caretta caretta, from by-catches in the South-West Indian Ocean. Marine Pollution Bulletin, 84, 90–96.
Ismail, A., Adilah, N. M. B., & Nurulhudha, M. J. (2009). Plastic pellets along Kuala Selangor-Sepang coastline. Malaysian Applied Biology, 38, 85–88.
Jambeck, J. R., Geyer, R., Wilcox, C., Siegler, T. R., Perryman, M., Andrady, A., Narayan, R., & Law, K. L. (2015). Plastic waste inputs from land into the ocean. Science, 347, 768–771.
Kaberi, H., Tsangaris, C., Zeri, C., Mousdisd, G., Papadopoulos, A., & Streftaris, N., 2013. Microplastics along the shoreline of a Greek island (Kea isl., Aegean Sea): types and densities in relation to beach orientation, characteristics and proximity to sources. In: 4th International Conference on Environmental Management, Engineering, Planning and Economics (CEMEPE) and SECOTOX Conference, Mykonos Island, Greece, pp. 197–202.
Laglbauer, B. J., Franco-Santos, M. R., Andreu-Cazenave, M., Brunelli, L., Papadatou, M., Palatinus, A., Grego, M., & Deprez, T. (2014). Macrodebris and microplastics from beaches in Slovenia. Marine Pollution Bulletin, 89, 356–366.
Lahbib, Y., Abidli, S., Chiffoleau, J. F., Averty, B., & Trigui El Menif, N. (2010). Imposex and butyltin concentrations in snails from the lagoon of Bizerta (Northern Tunisia). Marine Biology Research, 6, 600–607.
Lahbib, Y., Abidli, S., Rodríguez González, P., Ignacio García Alonso, J., & Trigui El-Menif, N. (2011). Potential of Nassarius nitidus for monitoring organotin pollution in the lagoon of Bizerta (northern Tunisia). Journal of Environmental Sciences, 23, 1551–1557.
Li, J., Yang, D., Li, L., Jabeen, K., & Shi, H. (2015). Microplastics in commercial bivalves from China. Environmental Pollution, 207, 190–195.
Liebezeit, G., & Dubaish, F. (2012). Microplastics in beaches of the East Frisian islands Spiekeroog and Kachelotplate. Bulletin of Environmental Contamination and Toxicology, 89, 213–217.
Lusher, A. L., McHugh, M., & Thompson, R. C. (2013). Occurrence of microplastics in the gastrointestinal tract of pelagic and demersal fish from the English Channel. Marine Pollution Bulletin, 67, 94–99.
Mathalon, A., & Hill, P. (2014). Microplastic fibers in the intertidal ecosystem surrounding Halifax Harbor, Nova Scotia. Marine Pollution Bulletin, 81, 69–79.
Moore, C. J., Moore, S. L., Leecaster, M. K., & Weisberg, S. B. (2001). A comparison of plastic and plankton in the North Pacific Central Gyre. Marine Pollution Bulletin, 42, 1297–1300.
Murray, F., & Cowie, P. R. (2011). Plastic contamination in the decapods crustacean Nephrops norvegicus (Linnaeus, 1758). Marine Pollution Bulletin, 62, 1207–1217.
Ng, K. L., & Obbard, J. P. (2006). Prevalence of microplastics in Singapore’s coastal marine environment. Marine Pollution Bulletin, 52, 761–767.
Nor, N. H. M., & Obbard, J. P. (2014). Microplastics in Singapore’s coastal mangrove ecosystems. Marine Pollution Bulletin., 79, 278–283.
Pedà, C., Caccamo, L., Fossi, M. C., Gai, F., Andaloro, F., Genovese, L., Perdichizzi, A., Romeo, T., & Maricchiolo, G. (2016). Intestinal alterations in European sea bass Dicentrarchus labrax (Linnaeus, 1758) exposed to microplastics: preliminary results. Environmental Pollution, 212, 251–256.
Plastics Europe (2015). Plastics- the facts 2015; an analysis of European latest plastics production, demand, and waste data (p. 30). Brussels: Plastics Europe.
Qiu, Q., Peng, J., Yu, X., Chen, F., Wang, J., & Dong, F. (2015). Occurrence of microplastics in the coastal marine environment: first observation on sediment of China. Marine Pollution Bulletin, 98, 274–280.
Reddy, M. S., Basha, S., Adimurthy, S., & Ramachandraiah, G. (2006). Description of the small plastics fragments in marine sediments along the Alang-Sosiya shipbreaking yard, India. Estuarine, Coastal and Shelf Science, 68, 656–660.
Rios, L. M., Moore, C., & Jones, P. R. (2007). Persistent organic pollutants carried by synthetic polymers in the ocean environment. Marine Pollution Bulletin, 54, 1230–1237.
Rochman, C. M., Hoh, E., Hentschel, B. T., & Kaye, S. (2013b). Long-term field measurements of sorption of organic contaminants to five types of plastic pellets: implications for plastic marine debris. Environmental Science & Technology, 47, 1646–1654.
Rochman, C. M., Hoh, E., Kurobe, T., & Teh, S. J. (2013a). Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Scientific Reports, 3, 3263.
Stolte, A., Forster, S., Gerdts, G., & Schubert, H. (2015). Microplastic concentrations in beach sediments along the German Baltic coast. Marine Pollution Bulletin, 99, 216–229.
Teuten, E. L., Rowland, S. J., Galloway, T. S., & Thompson, R. C. (2007). Potential for plastics to transport hydrophobic contaminants. Environmental Science & Technology, 41, 7759–7764.
Thompson, R. C., Olsen, Y., Mitchell, R. P., Davis, A., Rowland, S. J., John, A. W. G., McGonigle, D., & Russell, A. E. (2004). Lost at sea: where is all the plastic? Science, 304, 838–838.
Van Cauwenberghe, L., Claessens, M., Vandegehuchte, M. B., Mees, J., & Janssen, C. R. (2013). Assessment of marine debris on the Belgian Continental Shelf. Marine Pollution Bulletin, 73, 161–169.
Van Cauwenberghe, L., Devriese, L., Galgani, F., Robbens, J., & Janssen, C. R. (2015). Microplastics in sediments: a review of techniques, occurrence and effects. Marine Environmental Research, 111, 5–17.
Vianello, A., Boldrin, A., Guerriero, P., Moschino, V., Rella, R., Sturaro, A., & Da Ros, L. (2013). Microplastic particles in sediments of Lagoon of Venice, Italy: first observations on occurrence, spatial patterns and identification. Estuarine, Coastal and Shelf Science, 130, 54–61.
Ward, J. E., & Shumway, S. E. (2004). Separating the grain from the chaff: particle selection in suspension- and deposit-feeding bivalves. Journal of Experimental Marine Biology and Ecology, 300, 83–130.
Watts, A. J. R., Lewis, C., Goodhead, R. M., Beckett, S. J., Moger, J., Tyler, C. R., & Galloway, T. S. (2014). Uptake and retention of microplastics by the shore crab Carcinus maenas. Environmental Science & Technology, 48, 8823–8830.
Wright, S. L., Thompson, R. C., & Galloway, T. S. (2013). The physical impacts of microplastics on marine organisms: a review. Environmental Pollution, 178, 483–492.
Yoshida, M., Hamadi, K., & Ghrabi, A. (2002). Solid waste landfills and soil/sediment contamination around Bizerte lagoon: Possible pollution sources. In A. Ghrabi & M. Yoshida (Eds.), Study on environmental pollution of Bizerte Lagoon. Tunis: INRST-JICA Publishers.
Zhao, S., Zhu, L., & Li, D. (2016). Microplastic in three urban estuaries, China. Environmental Pollution, 206, 597–604.
Zhao, S., Zhu, L., Wang, T., & Li, D. (2014). Suspended microplastics in the surface water of the Yangtze Estuary System, China: first observations on occurrence, distribution. Marine Pollution Bulletin, 86, 562–568.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abidli, S., Toumi, H., Lahbib, Y. et al. The First Evaluation of Microplastics in Sediments from the Complex Lagoon-Channel of Bizerte (Northern Tunisia). Water Air Soil Pollut 228, 262 (2017). https://doi.org/10.1007/s11270-017-3439-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3439-9