Abstract
Variation in the occurrence and the intensity of pre-dispersal seed predation can help understand ecological and evolutionary dynamics of plant populations. Pre-dispersal seed predation can vary in intensity, resulting in costs that differ in magnitude among populations. We examined variation in the cost of pre-dispersal seed predation across nine Yucca schidigera populations, by quantifying yucca moth infestation, damage on fertile seeds and seed mass in 650 fruits sampled from 73 Yucca plants. We recorded Tegeticula mojavella larvae on 100% of the Y. schidigera plants. Across populations, mean infestation rates ranged from 2.9 to 6.11 larvae per fruit. Despite slight variation in the level of fruit infestation, predation on mature seeds differed among the nine populations, ranging from 10 to 26%. Mean seed mass and germination rate differed among populations, but the effect of larvae infestation on these traits was not significant. Altogether, our results demonstrate that infestation and pre-dispersal seed predation by yucca moths differ significantly across populations of Y. schidigera. Variable ratios of seed predation result in different costs across plant populations, despite experiencing similar infestation ratios. It seems that, regardless of its spatial distribution, the interaction between Y. schidigera and T. mojavella is dependent on local ecological factors that regulate oviposition and seed predation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Variation in the occurrence and the intensity of insect–plant interactions is common in natural ecosystems. Understanding the nature of factors that influence the spatial structure of such interactions is central to the study of ecology and evolution simply because insects have been an important driver of the genetic and phenotypic diversity in plant populations and species (Farrell et al. 1992; Weber and Agrawal 2014). Interactions like pollination and indirect defense have beneficial effects for plants, whereas antagonistic interactions such as herbivory and pre-dispersal seed predation are accompanied by negative impacts on plant fitness (Kolb et al. 2007; Katz 2016). These antagonistic and mutualistic interactions have shown ample variation in their intensity across populations (Thompson and Cunningham 2002; Toju and Sota 2006; Castillo et al. 2014), generating geographic mosaics of fitness costs and benefits for the species involved (Thompson 2005). For example, the detrimental cost of pre-dispersal seed predation on plant populations has been found to be temporally and spatially heterogeneous (Leimu et al. 2002; Borchert and DeFalco 2016; Dimitri et al. 2018). In part, local conditions such as the abundance of plants, pollinators, and seed predators likely influence the intensity of species interactions.
Many plant species are involved in interactions with pollinators that act as pre-dispersal seed predators (Kato and Kawakita 2017). However, pollination and pre-dispersal seed predation exert opposite effects on plant fitness. While pollination positively influences plant fitness by increasing the number of fertilized seeds, pre-dispersal seed predation reduces seed production, and can even alter germination and seedling survival during early stages of growth (Koptur 1998; Han et al. 2018). In this sense, seed predation reduces plant fitness by affecting population growth (Katz 2016). The outcome of the simultaneous effects of pollination and seed predation on plant fitness can be difficult to quantify and the complexity of the effects are not equally predictable across populations. Documenting variation in the strength of a highly host-specific interaction by measuring seed predation and analyzing the influence on fitness of both participants in several populations has been suggested as an advantageous approach to understand how fitness costs can shape insect–plant interactions (Bronstein 2001).
In particular, studies of pre-dispersal seed predation have found that the intensity of infestation and seed predation are accompanied with different costs across populations (Leimu and Lehtilä 2006). Variation in the magnitude of these costs of pre-dispersal seed predation has been found to depend on the efficacy of pollination service. For example, seed production can be limited if pollinator abundance is low and/or the amount and type of pollen reduce the probability of ovule fertilization (Ashman et al. 2004; Baer and Maron 2018). In a scenario where pollen limitation results in plants with reduced seed production, the negative impacts of seed predation would increase in relation to plants with larger seed production. In terms of fitness, the cost for a plant that produces a low number of seeds would be higher in relation to a plant that can produce a larger number of seeds (Bello-Bedoy et al. 2011). Moreover, this condition would occur even when plants show similar infestation rates by seed predators (Bello-Bedoy et al. 2011). Thus, a variable seed production could result in the cost of pre-dispersal seed predation of a different magnitude.
As for all other Yucca species, Y. schidigera is engaged in an obligate mutualism with two species of yucca moths of the genus Tegeticula (Prodoxidae). In this system, adult female moths pollinate yucca flowers by actively pushing pollen into the stigma and then ovipositing into the ovary (Riley 1892). During pollination, the female moth lays its eggs, and latter hatching larvae feed on a fraction of the developing seeds. To prevent overexploitation by the pollinating moth, plants selectively abscise flowers with a relatively large number of eggs (Wilson and Addicott 1998; Marr and Pellmyr 2003; Shapiro and Addicott 2004) and flowers that receive low amounts of pollen (Pollination quality; Pellmyr and Huth 1994). The number of fertile seeds is expected to increase with the number of pollinator visits, which would confer fitness benefits for the plant. However, with each visit the moth will oviposit in the flower’s ovary, increasing the potential number of seed-consuming larvae, and reducing the probability of flower retention (Pellmyr and Huth 1994; Csotonyi and Addicott 2001). In addition, hatching larvae feed on a fraction of the seeds that develop within the fruit, thereby, reducing the benefits of pollination.
Despite the strong co-dependence between Yucca and its pollinator, studies quantifying the cost of mutualisms throughout pre-dispersal seed predation in Yucca have found differences in the intensity of fruit infestation and pre-dispersal seed predation when they compare between and within species (Keeley et al. 1984; Addicott 1986; Pellmyr and Huth 1994; Ziv and Bronstein 1996). Only a few studies have examined infestation and pre-dispersal seed predation in multiple plant populations of the same Yucca species (Dodd and Linhart 1994; Harrower and Gilbert 2018), limiting our understanding of the ecological and evolutionary dynamic processes driven by the cost seed consumption. Increasing the number of populations across heterogeneous landscape allows evaluation of the degree of variation in the cost of pre-dispersal seed predation for different Yucca populations and can help predict whether these differences are relevant for demographic or evolutionary processes in the populations.
In this study, we examined the variation in the intensity of fruit infestation and pre-dispersal seed predation in multiple populations of Y. schidigera and Tegeticula mojavella to examine the magnitude of the cost for the plant. Seed predation and its cost was quantified by recording the number of unfertile and fertile damaged seeds. Likewise, we examined variation of T. mojavella infestation of Y. schidigera fruits by counting the number of moth larvae within each yucca fruit. Finally, we discuss the evolutionary consequences of variation in seed predation for the maintenance of mutualism.
To examine the variation in the cost of pre-dispersal seed predation on fertile seed production, we asked the following specific questions:
-
(i)
Does production of fertile and infertile seeds vary between populations?
-
(ii)
Do fruit infestation rates and pre-dispersal seed predation vary among plant populations?
-
(iii)
Are seed mass and germination rates correlated with infestation by T. mojavella larvae?
Materials and methods
Study species
Yucca schidigera Roezel (Asparagaceae), is a long-lived caulescent, woody monocot that is native to the Mojave desert, fringes of the Sonoran desert in Arizona, California and Baja California, and chaparral throughout the coastal foothills and mountains of southern California and Baja California. Y. schidigera populations occur at altitudes of 100 m to ~ 2000 m, from coastal bluffs to interior deserts, and within a latitudinal range that extends from 30° to 36°N (Turner et al. 2005; Fig. 1).

(modified from Turner et al. 2005)
Geographic variation of pre-dispersal seed predation in nine Y. schidigera populations in Baja California. Pie charts depict population averages of the proportion of seeds per fruit for: fertile seeds that escaped predation (black), damaged fertile seeds (gray) and the sum of infertile seeds (white). Inset map shows the distribution range of Y. schidigera
In Y. schidigera, flowering occurs between February and May. During this period, plants produce large paniculate inflorescences with as many as 500 flowers. Fruits are indehiscent capsules that complete maturation three to four weeks after being fertilized (Baldwin et al. 2012). This plant is pollinated by two moth species, Tegeticula mojavella and T. californica (Lepidoptera: Prodoxidae; Pellmyr 1999). Reports of T. californica are mostly restricted to low altitudes sites within a 40 km coastal stretch in southern California (Althoff et al. 2006; Pellmyr et al. 2008). By means of molecular genotyping (850 bp, mtDNA, n = 50), we detected exclusively T. mojavella moths in our sample. Yucca plants are self-compatible, and although pollen transfer within a same inflorescence has been registered to be frequent (55% of total observations, N = 270; Marr et al. 2000), self-fertilized flowers are more likely to be aborted than flowers that were outcrossed (Huth and Pellmyr 2000).
Sample collection
We studied nine populations of Y. schidigera distributed in northwestern Baja California (Fig. 1). These locations have a semiarid, Mediterranean climate, and are located at altitudes of 850–1950 m (Table 1), with an annual regional precipitation mean of 234.7 mm ± 78.2 SD (data from the WorldClim2 dataset; Fick and Hijmans 2017). Precipitation annual averages during the winters of 2013, 2014 and 2015 were lower (58.5, 88.3 and 32.9 mm, respectively) than the 30 year average for this region (rain data taken from the CONAGUA weather station in Ojos Negros, Baja California).
In March 2015, we marked 15 flowering plants at each site during the flowering season. We registered latitude and longitude for each population using a GPS (Garmin eTrex 10). We registered plant density at each site by counting the number of Y. schidigera individuals within two adjacent 30 m × 50 m quadrats and then estimating the mean. Densities ranged from 28 to 129.5 ind./ha across all nine populations (Table 1). In late August, when fruits were fully ripe, we visited each site and counted the number of fruits produced by the marked plants. From each plant, we collected a sample of 10 fruits, whenever possible. To avoid losing any larvae during transportation, we placed the fruits in individual plastic cups that we covered with nylon mesh. The plastic cups were labeled to maintain population and plant identity. A total of 650 fruits belonging to 73 plants were collected from the nine populations (Table 1). In the laboratory, we dissected the fruits and recorded the number of Tegeticula larvae within each fruit. By examining the exocarp, we also recorded the number of perforations bored on fruits by the larvae when escaping mature fruits; each exit hole was scored as a surrogate of a larva that developed within the fruit.
Seed number
We recorded the number of fertile and infertile seeds per fruit. Fertile seeds have a black seed coat, whereas infertile seeds are white and lack endosperm. We quantified the number of intact and damaged seeds per fruit. Pre-dispersal seed predation was analyzed using fertile seeds only, and to have an indicator of the cost imposed on the plant by the yucca moth larvae, we calculated the proportion of damage on fertile seeds (d) as follows:
Seed mass and germination rate
We recorded seed mass and germination rate in a subset of 55 fruits belonging to eight populations (3–6 plants per population), with 20 seeds per fruit (Nseeds = 940). Each seed was weighed to the nearest 0.1 mg using an analytical balance (Mettler-Toledo, Columbus, OH, USA). To assess germination rate, we sowed all seeds ~ 3 cm deep in plastic trays filled with BM2 soil (Berger, Quebec, Canada) maintaining the identity of each seed (e.g., fruit, plant and population number). Trays were kept inside a greenhouse at environmental conditions and the plantings were watered daily. We conducted observations every 24 h and scored germination when the radicle was visible at the soil surface.
Statistical analyses
To assess differences in yucca moth infestation, seed production, and pre-dispersal seed predation across populations, we carried out an independent nested ANOVA for each variable. These models included the fixed effects of the site and the plant nested within its population to control individual variation in each variable. To examine the effect of larvae on seed production, we included the number of larvae per fruit as a covariate of the population. To assess the association of any plant density relationship on pre-dispersal seed predation, we carried out Pearson correlations of plant density with larvae number and damaged seed number. To test whether infestation by T. mojavella larvae affects seed mass and germination, we conducted independent ANCOVA models, including the fixed effect of the site, the plant nested within its population, and each plant’s mean number of larvae per fruit as a covariate. We fitted mean seed mass per fruit and the proportion of seeds that germinated after a 60-day trail as separate response variables. Seed damage ratio and germination rate were log transformed before conducting further statistical analysis (Warton and Hui 2011). In all cases, a post-hoc HSD Tukey test was used to compare means among populations, and we used an alpha level of 0.05 for statistical significance. Finally, we conducted independent linear regression of mean infestation and mean seed predation as response variable on latitude and altitude. Statistical analyses were performed with JMP 10 (SAS Institute, Cary, NC, USA).
Results
Variation in seed production
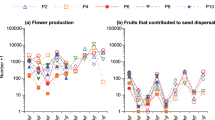
Population means for total seed production per fruit ranged from 53.10 ± 2.55 SE to 72.48 ± 3.19 SE across populations. The total number of seeds per fruit differed among populations (F8 = 8.60; P ≤ 0.0001, N = 650; Table 2) and within populations (F64 = 11.19, P < 0.0001). Plant density showed a negative correlation with seed production (r = − 0.7397, P = 0.02). Likewise, the number of fertile seeds and infertile seeds showed significant differences among plant populations (fertile: F8 = 21.7, P < 0.0001; infertile: F8 = 51.95, P < 0.0001; Table 2) and among individuals within populations (fertile: F64 = 11.53, P < 0.0001; infertile: F64 = 9.81, P < 0.0001). The mean number of fertile seeds in non-infested fruits was 34.98 ± 2.48 SE (N = 73), and ranged from 15.25 ± 3.5 SE to 59.66 ± 12.73 SE across populations. In turn, the mean number of fertile seeds across infested fruits was 46.86 ± 1.05 SE (N = 577), and ranged from 33.95 ± 2.47 SE to 60.55 ± 3.13 SE across populations.
Fruit infestation
We found T. mojavella larvae in 100% of plants and in 85.5% of the total fruit sample (N = 650). The number of larvae found within the fruits ranged from 0 to 26 and the grand mean was 4.06 ± 0.14 SE. Across populations, mean number of larvae per fruit ranged from 2.9 ± 0.541 SE to 6.11 ± 0.61 SE (Fig. 2) and larvae infestation differed between populations after controlling individual variation within populations (F8,64 = 5.81, P < 0.0001). Plant density did not correlate with the level of infestation (r = − 0.36, P = 0.33).
Pre-dispersal seed predation
The nested ANCOVA detected variation across populations in the number of fertile seeds damaged by T. mojavella larvae. Mean number of fertile damaged seeds per fruit differed between populations (F8 = 6.23, P ≤ 0.0001, N = 650; Table 3) and within populations (F8,64 = 2.51, P < 0.0001; Table 3). Plant density did not correlate with seed predation (r = − 0.12, P = 0.73). Moreover, the interaction between population and the number of larvae was statistically significant (F8 = 8; P < 0.024; Table 3), indicating that the effect of seed damage differed between populations, even at similar levels of fruit infestation. The proportion of damaged fertile seeds (d) ranged from 10 to 26% across populations (Table 2; Fig. 1), indicating that the cost of seed predation is variable across populations.
Seed mass and germination
Seed mass ranged from 22.3 to 276.1 mg and averaged 110.95 mg ± 1.08 SE across all populations. Overall germination rate was 32.68% and population germination rates ranged from 0.71% for site D, up to 90% for site I. Seeds began germination after 14 days, and out of the 353 seeds that germinated, 80% did so during the first 28 days. Mean seed mass and germination rate of fertile undamaged seeds were significantly different between populations (seed mass: F7 = 6.18, P = 0.01; germination rate: F7 = 10.28, P < 0.0001). However, mean number of larvae per fruit did not affect mean seed mass or germination rate (mean seed mass: F1 = 2.73, P = 0.13; germination rate: F1 = 0.12, P = 0.72).
Discussion
In this study we quantified seed production by Yucca schidigera and seed predation by its obligate pollinator, Tegeticula mojavella, across their distribution in Baja California. Our data found significant variation in the proportions of fertile and infertile seeds produced in fruits across populations suggesting that plants experienced variation in pollination environments at each site. Also, we found that the number of moth larvae developing in the fruits and the proportion of seeds consumed by yucca moth larvae differed among populations (Figs. 1, 2). Damage on fertile seeds caused by the larvae varied between populations, even at similar infestation levels (i.e. significant population × larvae interaction). Although variation in infestation intensity was relatively small, the number of damaged seeds across Yucca populations resulted in differences in the cost of seed predation (Fig. 1). This heterogeneous fitness cost for the plant suggests that larvae might exert natural selection of differing magnitudes between populations. We did not find differences in seed mass and germination rate in seeds derived from fruits with different level of infestation by larvae of the moth. Altogether, the interplay between the beneficial effects of pollination and the antagonistic effects of seed predation can account for the mutualism costs at the population level.
Variation in seed production
We found large differences in the production of fertile and infertile seeds across populations. Variation in infertile seed production could be explained by differences in the abundance of pollinators and/or in the quality of pollen available for pollination (Ashman et al. 2004). Studies in Yucca filamentosa have shown that manually pollinated flowers with selfed and low amounts of pollen resulted in lower fruit retention in relation to flowers pollinated with abundant and outcrossed pollen (Huth and Pellmyr 2000). Thus environments with low pollinator efficacy can reduce an individual’s seed production. Furthermore, our sampled populations occur in different environmental conditions, and this environmental heterogeneity could reflect in differences in pollinator abundance across populations.
Variation in fruit infestation by the Tegeticula larvae
We found significant differences in the rate of yucca moth larvae infestation across Y. schidigera populations. The level of infestation found in this study was considerably greater in relation to reported values for other Y. schidigera populations. For instance, Keeley et al. (1984) reported a mean of 0.6 larvae per fruit. In contrast, we found 4.06 larvae per fruit. Such difference in the level of infestation can be explained by differences in the moth's population density, as well as differences in oviposition rates. As is the case in several insect–plant systems, host plant density can influence population-level differences in infestation intensity (Skoracka et al. 2017; Harrower and Gilbert 2018). However, plant densities in our populations were not correlated with infestation or to fertile seed damage, ruling out the role of plant density.
Another explanation for variation in the number of larvae is that regulatory mechanisms that operate to limit yucca moth densities vary among populations. For instance, flower abscission is a common mechanism employed by Yucca plants to prevent overexploitation. Plants avoid opportunistic behavior of pollinators by abscising flowers in which ovaries have been damaged by an excessive number of ovipositor insertions (Marr and Pellmyr 2003), and by selectively aborting developing fruits with heavy egg loads (Pellmyr and Huth 1994; Wilson and Addicott 1998). However, a question is whether plants from all populations equally sensitive to ovary damage and ovipostion? In our study, three populations had large larvae numbers in relation to the rest (Fig. 2). It is possible that differences in moth infestation result from different abilities across plants to tolerate ovipositor damage. These three populations are candidates to test hypotheses of tolerance to oviposition in field experimental manipulation.
Variation in pre-dispersal seed predation
Pre-dispersal seed predation in Y. schidigera differed within and between populations. Overall, seed predation on fertile seeds was higher in relation to values previously reported for this species (Keeley et al. 1984). Two factors can explain variable rates of seed predation: first, large seed predation rates observed in this study are associated with a high number of larvae per fruit; and second, larvae could consume different amounts of seeds in each population, as indicated by a significant population × larvae interaction. Thus, when comparing among fruits with similar numbers of larvae, Yucca populations experienced seed predation differently. Studies in other Yucca species have reported similar results, suggesting that variation in consumption rate by seed predators is not unusual (Dodd and Linhart 1994; Harrower and Gilbert 2018). Some populations would experience higher costs even with similar levels of infestation, partially explaining variability in the cost of pre-dispersal seed predation.
Evolutionary implication of variation in pre-dispersal seed predation
Our data demonstrated that individuals within populations and between populations suffered different costs of seed predation, suggesting that natural selection might operate on these fitness-related traits if they are genetically based. Studies have demonstrated that natural levels of damage by pre-dispersal seed predators can select for structural traits, such as plant size (Kolb et al. 2007) or features related to the fruit (Toju and Sota 2006). Yucca plants interact with yucca moths through flowers, the developing fruit, and seeds, so selection exerted by T. mojavella should favor the evolution of reproductive structures that make the plant a better host for the moth’s larvae. Studies in Yucca species have reported that different moth species have preferences to oviposit eggs in different parts of the ovary (Pellmyr and Leebens-Mack 2000), and this behavior has evolved more than once during the evolution of the interaction (i.e., in the evolutionary transition from seed predator to a complete mutualism, and to parasitism in some cases). Differences between individuals and populations in fruit infestation and seed predation intensities in our study suggest that selection could promote phenotypic changes in traits that maximize the beneficial effect of pollination, while efficiently regulating larvae density (i.e., plant tolerance) to minimize seed loss (i.e., plant fitness). Thus, a study of the selective role of damage caused by pollinators and seed predators in the phenotypic evolution Y. schidigera flowers, fruits, and seeds could reveal if selection varies across populations.
Our results demonstrate that the intensity of infestation and pre-dispersal seed predation by T. mojavella can differ between Y. schidigera populations. Even when most populations showed similar levels of infestation, the fruits collected from three populations bore more larvae than the rest of the sites. This suggests that overexploitation is controlled in most populations, but plants from other populations can be more tolerant to ovipositor damage and host more larvae per fruit. In addition, we found that individuals within populations and between populations suffered different costs of seed predation. It is possible that natural selection might operate on fitness-related traits. Further studies in several populations in the Y. schidigera—T. mojavella system should examine whether variability in the cost of seed predation exerts selective pressure in reproductive traits, generating a complex geographic mosaic of coevolution.
Data availability
Our dataset is available using the https://doi.org/10.6084/m9.figshare.7215014
References
Addicott JF (1986) Variation in the costs and benefits of mutualism: the interaction between yuccas and yucca moths. Oecologia 70:486–494
Althoff DM, Segraves KA, Leebens-Mack J, Pellmyr O (2006) Patterns of speciation in the Yucca moths: parallel species radiations within the Tegeticula yuccasella species complex. Syst Biol 55:398–410
Ashman T-L, Knight TM, Steets JA, Amarasekare P, Burd M, Campbell DR, Dudash MR, Johnston MO, Mazer S, Mitchell RJ, Morgan MT, Wilson WG (2004) Pollen limitation of plant reproduction: ecological and evolutionary causes and consequences. Ecology 85:2071–2081
Baer KC, Maron JL (2018) Pre-dispersal seed predation and pollen limitation constrain population growth across the geographic distribution of Astragalus utahensis. J Ecol 106:1646–1659
Baldwin BG, Goldman DH, Keil DJ et al (eds) (2012) The Jepson manual: vascular plants of California. The University of California Press, Berkeley
Bello-Bedoy R, Cruz LL, Núñez-Farfán J (2011) Inbreeding alters a plant-predispersal seed predator interaction. Evol Ecol 25:815–829
Borchert MI, DeFalco LA (2016) Yucca brevifolia fruit production, predispersal seed predation, and fruit removal by rodents during two years of contrasting reproduction. Am J Bot 103:830–836. https://doi.org/10.3732/ajb.1500516
Bronstein JL (2001) The costs of mutualism. Am Zool 41:825–839
Bronstein JL, Ziv Y (1997) Costs of two non mutualistic species in a yucca/yucca moth mutualism. Oecologia 112:379–385
Castillo G, Cruz LL, Tapia-López R et al (2014) Selection mosaic exerted by specialist and generalist herbivores on chemical and physical defense of Datura stramonium. PLoS ONE 9:e102478
Csotonyi JT, Addicott JF (2001) Competition between mutualists: the role of differential flower abscission in yuccas. Oikos 94:557–565
Dimitri LA, Longland WS, Tonkel KC, Rector BG, Kirchoff VS (2018) Impacts of granivorous and frugivorous arthropods on pre-dispersal seed production of western juniper (Juniperus occidentalis). Arthropod Plant Interact 12:465–476
Dodd RJ, Linhart YB (1994) Reproductive consequences of interactions between Yucca glauca (Agavaceae) and Tegeticula yuccasella (Lepidoptera) in Colorado. Am J Bot 81:815–825
Farrell BD, Mitter C, Futuyma DJ (1992) Diversification at the insect-plant interface. Bioscience 42:34–42
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315
González-Abraham C, Garcillán P, Ezcurra E (2010) Ecorregiones de la península de Baja California: Una síntesis. Boletín la Soc Botánica México 82:69–82
Han YJ, Baskin JM, Tan DY et al (2018) Effects of predispersal insect seed predation on the early life history stages of a rare cold sand-desert legume. Sci Rep 8:3240–3250
Harrower J, Gilbert GS (2018) Context-dependent mutualisms in the Joshua tree–yucca moth system shift along a climate gradient. Ecosphere 9:e02439
Huth CJ, Pellmyr O (2000) Pollen-mediated selective abortion in Yuccas and its consequences for the plant-pollinator mutualism. Ecology 81:1100–1107
Kato M, Kawakita A (2017) Obligate pollination mutualism, ecological research monographs. Springer, Japan
Katz DSW (2016) The effects of invertebrate herbivores on plant population growth: a meta-regression analysis. Oecologia 182:43–53
Keeley JE, Keeley SC, Swift CC, Lee J (1984) Seed predation due to the Yucca-Moth symbiosis. Am Midl Nat 112:187–191
Kolb A, Ehrlén J, Eriksson O (2007) Ecological and evolutionary consequences of spatial and temporal variation in pre-dispersal seed predation. Perspect Plant Ecol Evol Syst 9:79–100
Koptur S (1998) Effect of seed damage on germination in the common vetch (Vicia sativa L.). Am Midl Nat 140:393–396
Leimu R, Lehtilä K (2006) Effects of two types of herbivores on the population dynamics of a perennial herb. Basic Appl Ecol 7:224–235
Leimu R, Syrjänen K, Ehrlén J, Lehtilä K (2002) Pre-dispersal seed predation in Primula veris: among-population variation in damage intensity and selection on flower number. Oecologia 133:510–516
Marr DL, Pellmyr O (2003) Effect of pollinator-inflicted ovule damage on floral abscission in the yucca-yucca moth mutualism: the role of mechanical and chemical factors. Oecologia 136:236–243
Marr DL, Leebens-Mack J, Elms L, Pellmyr O (2000) Pollen dispersal in Yucca filamentosa (Agavaceae): the paradox of self-pollination behavior by Tegeticula yuccasella (Prodoxidae). Am J Bot 87:670–677
Molau U, Eriksen B, Knudsen JT (1989) Predispersal seed predation in Bartsia alpina. Oecologia 81:181–185
Pellmyr O (1999) Systematic revision of the yucca moths in the Tegeticula yuccasella complex (Lepidoptera: Prodoxidae) north of Mexico. Syst Entomol 24:243–271
Pellmyr O, Huth CJ (1994) Evolutionary stability of mutualism between yuccas and yucca moths. Nature 372:257–260
Pellmyr O, Leebens-Mack J (2000) Reversal of mutualism as a mechanism for adaptive radiation in yucca moths. Am Nat 156:S62–S76
Pellmyr O, Balcázar-Lara M, Segraves KA et al (2008) Phylogeny of the pollinating yucca moths, with revision of Mexican species (Tegeticula and Parategeticula; Lepidoptera, Prodoxidae). Zool J Linn Soc 152:297–314
Riley CV (1892) The yucca moth and yucca pollination. Third Annual Report of the Missouri Botanical Garden.
Shapiro J, Addicott JF (2004) Re-evaluating the role of selective abscission in moth/yucca mutualisms. Oikos 105(3):449–460
Skoracka A, Lewandowski M, Rector BG, Szydlo W, Kuczyński L (2017) Spatial and host-related variation in prevalence and population density of wheat curl mite (Aceria tosichella) cryptic genotypes in agricultural landscapes. PLoS ONE 12:1–17
Thompson JN (2005) The geographic mosaic of coevolution. The University of Chicago Press
Thompson JN, Cunningham BM (2002) Geographic structure and dynamics of coevolutionary selection. Nature 417:735–738
Toju H, Sota T (2006) Imbalance of predator and prey armament: geographic clines in phenotypic interface and natural selection. Am Nat 167:105–117
Turner RM, Bowers JE, Burgess TL (2005) Sonoran Desert plants: an ecological atlas. University of Arizona Press
Warton DI, Hui FKC (2011) The arcsine is asinine: the analysis of proportions in ecology. Ecology 92:2049–2055
Weber MG, Agrawal AA (2014) Defense mutualisms enhance plant diversification. Proc Natl Acad Sci 111:16442–16447
Wilson RD, Addicott JF (1998) Regulation of mutualism between yuccas and yucca moths: is oviposition behavior responsive to selective abscission of flowers? Oikos 81:109–118
Ziv Y, Bronstein JL (1996) Infertile seeds of Yucca schottii: a beneficial role for the plant in the yucca-yucca moth mutualism? Evol Ecol 10:63–76
Acknowledgements
This manuscript constitutes a partial requirement for the doctoral dissertation of L. De la Rosa-Conroy at the Life Sciences Graduate Program in CICESE. We thank Sula Vanderplank, PhD for helpful comments on an earlier version of this manuscript, and E. López and M. Salazar for assisting with sample collection. We also thank Juan Cons and the students at Genética Ecológica Lab (UABC) and Genética de la Conservación Lab (CICESE) for participating in fieldwork and fruit dissections during 2015–2018.
Funding
Dr. Maria Clara Arteaga received funding from CONACYT (Project No. CB-2014-01-238843) and CICESE (Project No. 681-114); Dr. Rafael Bello-Bedoy received funding from CONACYT (Project No. INFRA2015_226239); and Leonardo De la Rosa-Conroy received a doctoral scholarship from CONACYT (Grant No. 19713).
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the preparation of this manuscript. MCA directed the project, MCA and RBB conceived the study, LDC and RBB contributed with sample collection and data analysis, LDC and RBB wrote the first draft of the manuscript, and MCA, SBH, and LEF contributed with the overall preparation of the manuscript and with interpretation of the results.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Philip Ladd.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De la Rosa-Conroy, L., Arteaga, M.C., Bullock, S.H. et al. Population variation in the intensity of fruit infestation and pre-dispersal seed predation in Yucca schidigera (Asparagaceae) by its obligate pollinator. Plant Ecol 220, 711–720 (2019). https://doi.org/10.1007/s11258-019-00946-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-019-00946-9