Abstract
The seeds of most tropical plants are dispersed by animals, many of which also act as seed predators. Shifts in animal community composition, such as those driven by the clearing of native vegetation, are therefore likely to drive changes in plant recruitment. We used manipulative experiments excluding ants, small rodents, and birds to quantify the relative impacts of these granivores on animal-dispersed pioneer trees (ADPT) in fragments of savanna vegetation and adjacent soy plantations in Brazil’s Cerrado. We found that ants were the main consumers of ADPT seeds, that the rates of seed removal varied with seed size, and that removal rates were higher in savanna fragments than in soy plantations. However, we also found significant interactions between habitat type, seed species, and the type of seed predator being excluded. Our results underscore how challenging it can be to predict the influence of human disturbances on the interactions between plant and animal communities. Because ants, rodents, and birds are Cerrado’s the main seed dispersers and granivores, seedling recruitment in Cerrado landscape mosaics will depend on how these distinct but related processes are each influenced by species-specific patterns of seed size and seed abundance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat loss and fragmentation are the main causes of declines in biodiversity, especially in species-rich tropical countries (Vié et al. 2009). The structural integrity of habitat remnants and the regeneration of cleared habitats may depend in part on plant–animal relationships such as seed dispersal and seed predation that mediate seedling recruitment (Bello et al. 2015). These relationships can themselves be altered in fragments (García and Chacoff 2007; Gallegos et al. 2014) or in the matrix that surrounds them (Prevedello and Vieira 2010; Craig et al. 2011). Although studies evaluating how plant–animal interactions influencing recruitment are modified in either fragments or surrounding matrix are common (Martinson and Fagan 2014), those comparing these interactions in both fragments and the matrix in which they are embedded are surprisingly rare (Herrera et al. 2011).
Most seed dispersal in tropical ecosystems is by animals, with both vertebrates and invertebrates acting as primary and secondary dispersers (Vander Wall and Longland 2004). While some of these animals disperse seeds incidentally as a byproduct of frugivory, others are also consuming the seeds themselves and hence also act as seed predators. Both the dispersal and predation of seeds are strongly influenced by their size (Cramer et al. 2007) and the local composition of the disperser/granivore community (García and Chacoff 2007). Because animals of different sizes are differentially affected by fragmentation (Terborgh 1992), the dispersal and predation of plants with different seed sizes could be affected as well. For instance, large-seeded plant species depend on a limited number of larger, vertebrate species for primary dispersal—species which are often locally extinct due to hunting or larger home-range requirements (Melo et al. 2006; Dirzo et al. 2007). While invertebrate dispersers/granivores may be more resistant to the effects of fragment size or isolation, their abundance or diversity in highly modified matrix habitat can be dramatically reduced (Brandão et al. 2011). Consequently, the regeneration of plant communities in human-modified landscapes will ultimately depend on both the disperser/granivore found in a site and the local diversity of functionally relevant plant traits (e.g., seed size) represented there (Christianini and Oliveira 2010; Ferreira et al. 2011).
Brazil’s Cerrado comprises a mosaic of vegetation physiognomies ranging from savannas to forests (Oliveira-Filho and Ratter 2002). The Cerrado biome has high species richness and endemism of both plants and vertebrates (Myers et al. 2000). It originally covered ~2 million km2, but the expansion of agriculture and other human activities has resulted in a 50% loss of this biome with most of the remaining habitat highly fragmented (Françoso et al. 2015). Cerrado landscapes dominated by a matrix of crops are the most detrimental to the conservation of Cerrado biodiversity (Carvalho et al. 2009). Seed germination and recruitment of forest species can be dramatically reduced in the matrix habitat of these landscapes (Carvalho et al. 2009). Furthermore, plant communities in disturbed Cerrado fragments that are embedded in crop-dominated landscapes have a lower proportion of species dispersed by animals than those in nearby protected areas (Vasconcelos et al. 2014). While this has been attributed to landscape structure and habitat isolation reducing the efficacy of animals as seed dispersers, it could also result from increased seed predation.
Ants, rodents, and birds are both the main dispersers and post-dispersal predators of many Cerrado plant species (Christianini and Oliveira 2010; Ferreira et al. 2011). As in other biomes, their relative importance as dispersers or granivores will depend on such factors as their feeding behavior and the traits of the available seeds (Rey et al. 2002; Ordóñez and Retana 2004; Briones-Salas et al. 2006). For example, rodents and birds remove larger seeds than do ants (Suazo et al. 2013), but ants frequently deposit seeds in refuse piles where some will subsequently germinate (Gallegos et al. 2014). It can also depend on how foraging behavior is influenced by habitat characteristics. For instance in contrast to birds, whose foraging appears to be primarily influenced by fruits and seed characteristics (Christianini and Galetti 2007), ants and rodents often concentrate their foraging in densely vegetated areas to reduce the risk of predation (Christianini and Galetti 2007). Notwithstanding this understanding, studies comparing predation by the Cerrado’s different granivore taxa across a range of habitat types remain rare.
Because “pioneer” or “early-successional” tree species can rapidly become established in the matrix surrounding fragments (Laurance et al. 1998), they are often critical to the regeneration of degraded sites (see Viani et al. 2015). While the dispersal of animal-dispersed pioneer trees (ADPT) is often well understood, almost nothing is known about the subsequent predation of their seeds, including whether it is higher or lower than in natural vegetation (García and Chacoff 2007). In this study, we experimentally evaluated the relative amount of ADPT seed removal by ants, small rodents, and birds in a crop-dominated Cerrado landscape. We addressed the following questions: (1) What is the relative proportion of ADPT seeds removed by birds, rodents, and ants? (2) How do removal rates vary with seed size? (3) Do these patterns vary among Cerrado fragments and the agricultural matrix? (4) To what extent is post-dispersal removal influenced by landscape characteristics such as fragments size, connectivity, and isolation?
Materials and methods
Study system
The study was conducted in central Brazil near the city of Uberlândia in Minas Gerais State (Fig. 1). The region is characterized by two well-defined seasons: a dry winter (May–September) and a rainy summer (October–April). The mean annual temperature and precipitation are approx. 22 °C and 1600 mm, respectively. Vegetation is influenced by climate and geomorphology at the regional level and by variations in soil fertility, topography, and incidence of fire at the local level (Oliveira-Filho and Ratter 2002).
The post-dispersal removal experiment was conducted in a crop-dominated landscape (169,135 ha) consisting of around 60 remnants of different Cerrado physiognomies (mean size = 30 ha, SD = 39.2 ha) and one protected area (Panga Ecological Station, 404 ha). In total 99.18% of the landscape is crop monoculture—primarily soy plantations, with some corn or sorghum in a rotational scheme. We chose 10 pairs of Cerrado fragment/nearby monoculture for our experiments. Sites were at least 17 km from each other, and none of the monocultures were being harvested during the experiment.
We used seeds of five pioneer shrub/tree species in our experiments: Siparuna guianensis Aublet (Siparunaceae), Xylopia aromatica Lam. (Annonaceae), Solanum lycocarpum A. St.-Hil. (Solanaceae), Matayba guianensis Aublet (Sapindaceae), and Tapirira guianensis Aublet (Anacardiaceae). We chose them based on seed size, seed availability, and their abundance in the region (Table 1). Although seeds of all species could be considered relatively small compared to those of other Cerrado trees (see Bello et al. 2015), we found that the average of diameter and weight between five species are not uniform using a test of homogeneity of variance (F 1,8 = 6.6, p = 0.03).
Post-dispersal removal experiment
We used a series of selective exclosure treatments to identify and quantify the taxa responsible for the removal of the different seed species (i.e., ants, rodents, birds) in different habitats (monoculture, Cerrado fragment). In Treatment 1, seeds were covered with an inverted transparent plastic container (250 ml) fixed to the ground in which three 1 cm2 openings allowed only ants to access the seeds. In Treatment 2, seeds were covered with a 500 ml container with three 4.5 × 4.5 cm openings through which rodents (and hence also ants) could gain access, but not granivorous birds. Treatment 3 was a control treatment in which seeds were accessible to all seed predators; seeds were placed on a 5 cm2 piece of transparent mesh flush with the soil surface. At each site of Cerrado fragment and monoculture, we established four parallel 40-m-long transects along each of which we positioned five sampling stations, one every 10 m. At each station, we placed 30 pulp-free seeds of each of the five ADPT species: 10 accessible to all three granivore taxa (Treatment 3), 10 accessible only to ants and rodents (Treatment 2), and 10 accessible only to ants (Treatment 1). The different treatments at each station were arranged 3 m apart from each other in a triangle. Seeds from all five species were offered simultaneously and remained exposed to consumers for two days, after which they were collected and classified as (1) in place and uneaten or (2) removed or eaten. All experimental trails were conducted between March and June 2015.
Landscape configuration metrics at patch scale
For each of the 10 Cerrado/monoculture pairs, we calculated three landscape metrics—isolation, area, and adjacent perimeter of the nearest Cerrado remnant—using Google Earth Pro Landsat Images (2016) (https://www.google.com.mx/intl/es-419/earth/download/gep/agree.html). As metric of isolation, we modified Gustafson and Parker’s (1994) proximity index (PI) by using the nearest-neighbor distance to each monoculture and Cerrado fragment divided by its area. Because the index is dimensionless its absolute value has little interpretive value. Instead it is used as a relative index: the values are large when larger and/or closer patches surround the focal patch and decrease as surrounding patches become smaller and/or sparser. As a metric of connectivity we used perimeter (in meters) of adjacent Cerrado remnants to our sample sites, since adjacent remnants may act as an ecological corridor facilitating animal movement through our study sites (Tewksbury et al. 2002). Area was only calculated for the Cerrado fragments.
Data analysis
A generalized linear mixed-effect model (GLMM) with a negative binomial error distribution was used to evaluate the effect of the following independent variables on the rate of seed removal: predator exclosure treatment, seed species, and habitat type. Because we found an interaction between the three independent variables, we determined the effects of habitat type and experimental treatment on the predation rates of each species using a separate GLMM for each species. To remove the effect of spatial pseudo-replication generated by non-independent replicates, we treated individual transects as a random effect.
To test for effects of landscape configuration at patch scale on rate of seed removal by species and treatment, we used generalized linear models (GLM) with a binomial distribution and a logit transformation. First, we examined effects on each species of habitat type (i.e., monoculture vs. Cerrado fragment), the isolation of sample sites (i.e., the proximity index) and connectivity (i.e., the adjacent perimeter of Cerrado remnants). In a separate analysis, we similarly tested for the effect of predator exclosure treatment. Second, we examined the effects of Cerrado fragment area on the rate of seed removal by species and then by predator exclosure treatment. Models were built using forward and backward stepwise selection of explanatory variables based on their significance. We used partial regression to calculate the independent effects of multiple explanatory variables in each model (Legendre and Legendre 1998). The best model was identified with Akaike’s Information Criterion (AIC). All analyses were carried out in the R programming language (R Core Development Team 2016) using the glmer functions from lme4 package (Bates et al. 2015), glm from stats and stepAIC from MASS.
Results
Across treatments and sites, the greatest removal was of Siparuna guianensis and Xylopia aromatica (4.82 seeds ± 3.58 SD and 4.77 seeds ± 3.85 SD out of 10, respectively), followed by Solanum lycocarpum (2.61 ± 3.36 SD), Tapirira guianensis (2.08 ± 3.28 SD), and Matayba guianensis (1.39 ± 2.32 SD). Across sites and species the most seeds were removed from the treatment in which seeds were exposed to all predators (Treatment 3: 3.41 ± 3.67 SD out of 10), followed by exposure to ants and small mammals (Treatment 2: 3.41 ± 3.07 SD) and the treatment in which only ants had access to seeds (Treatment 1: 2.58 ± 3.37 SD). Finally, seed removal rates were 10% higher in the Cerrado than in the plantations (3.62 ± 3.88 SD vs. 2.65 ± 3.22 SD; all sites and treatments pooled).
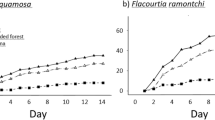
Our first model showed that predator exclosure treatment, tree species, and habitat type all have significant effects on seed removal rates. All interactions were also significant, except the one between treatment and habitat (Supplementary Material 1). In the separate models for each species, there was an effect of predator exclosure treatment on seed removal rates for all five species (Table 2; Fig. 2). Overall, the percent of seed removal in Treatment 1 (ants only) was 45.1%, in Treatment 2 (ants and rodents) 17.6%, and in Treatment 3 (ants, rodents and birds) 2.2%, indicating that ants were the primary seed removers. Rodents (but not birds) were important removers of Siparuna guianensis, Solanum lycocarpum, and Xylopia aromatica seeds, whereas birds (but not rodents) were important consumers of Matayba guianensis and Tapirira guianensis seeds (Fig. 2; Supplementary Material 2). The smallest seeds—Siparuna guianensis and Xylopia aromatica—had significantly higher removal rates than those of other larger-seeded species (Fig. 2).
Mean proportion (±SE) of seeds for each animal-dispersed pioneer trees (ADPT) species in three seed predator treatments inside two habitat types in a Cerrado landscape, Brazil. ADPT species: a Siparuna guianensis, b Xylopia aromatica, c Solanum lycocarpum, d Matayba guianensis, and e Tapirira guianensis
The overall seed removal rate differed among habitat types: 15.9 ± 3.2% in monocultures versus 22 ± 3.8% in Cerrado Fragments. For four of the five tree species, there was a significant effect of habitat type on removal rates (Table 2; Fig. 2), with removal higher in the fragments than monocultures. For Solanum lycocarpum and Siparuna guianensis, there was also an interaction between habitat and predator exclosure treatment. In the monocultures, rodents were comparatively more important removers of the seeds of these species than ants (Table 2; Fig. 2).
All analyses of landscape configuration indicated a difference in removal rates between Cerrado fragments and matrix of monoculture for each ADPT species (Tables 3, 4) and for predator exclosure treatment (Tables 5, 6). However, landscape metrics had idiosyncratic effects on the rates of removal of each species’ seeds. Seed removal rate of Siparuna guianensis increased in patches that were more isolated (z = 1.43, p < 0.001) and smaller (z = 2.074, p < 0.001), but decreased with greater connectivity (z = −5.52, p < 0.001). For Xylopia aromatica seed removal rate decreased inside monocultures (z = −6.49, p < 0.001) and in smaller Cerrado fragments (z = −6.49, p < 0.001). Solanum lycocarpum seed removal decreased inside monocultures (z = −4.12, p < 0.001), with greater isolation (z = −1.2, p < 0.001) and in smaller Cerrado fragments (z = −9.35, p < 0.001), but it increased with greater connectivity (z = 8.85, p < 0.001). Seed removal of Matayba guianensis decreased in smaller fragments (z = −8.23, p < 0.001), while that of Tapirira guianensis decreased inside monocultures (z = −7.88, p < 0.001), with greater fragment isolation (z = −7.82, p < 0.001) and in smaller fragments (z = −6.41, p < 0.001). In contrast, habitat type and fragment size were the most important landscape metrics influencing seed removal rates across predator exclosure treatments (Tables 5, 6). In all cases, seed removal rates decreased inside monocultures (Treatment 1, z = −0.31, p < 0.001; Treatment 2, z = −6.26, p < 0.001; Treatment 3, z = −6.46, p < 0.001) and in smaller Cerrado fragments (Treatment 1, z = −6.94, p < 0.001; Treatment 2, z = −5.77, p < 0.001; Treatment 3, z = −7.86, p < 0.001).
Discussion
Studies of post-dispersal seed removal in the Neotropics have rarely evaluated the magnitude of seed removal by different types of granivores (Christianini and Galetti 2007; Pufal and Klein 2013), or compared removal in native vegetation with that in the agricultural matrix replacing this habitat. We did so assessing the removal rates of seeds from five pioneer tree species by three different animal taxa in both Cerrado remnants and the crop monocultures in which they were embedded. It is important to recognize that seed removal could reflect either secondary dispersal or seed predation (Vander Wall et al. 2005)—missing seeds could have been consumed at the stations, moved and consumed later, or cached or discarded in locations favoring germination and seedling establishment. Observations of ants–diaspore interactions in the Cerrado have emphasized the ants’ role as dispersers (Christianini and Galetti 2007; Christianini and Oliveira 2010), but field experiments have shown that the recruitment of woody plants is higher in locations where ants are excluded (Ferreira et al. 2011). In concert with previous work showing rodents are major granivores in tropical regions (Briones-Salas et al. 2006), including some locations in the Cerrado (Briani and Guimarães 2007), our results lead us to conclude that the removal we observed is most likely predation rather than dispersal.
Our findings are consistent with those of other studies finding ants are the main consumers of seeds of Cerrado trees (e.g., Ferreira et al. 2011; Gallegos et al. 2014) and further underscore their importance for plant population and community dynamics in this biome (Costa et al. 2008, 2017). One possible explanation for the limited role of mammalian or avian granivores is that their abundance in our study region is low (Bruna et al. 2010). We did not quantify the abundance of the different taxa in our study. However, in previous surveys in one of our study sites—Panga Ecological Station—we found that the abundance of small mammals was extremely low (Bruna et al. 2010). In addition, while some avian pre-dispersal seed predators can be locally abundant (Mercival et al. 2008), pulp-free seeds on the ground are most likely consumed by granivorous birds, such as tinamous and doves, that our observations and prior results (Ferreira et al. 2011) suggest are present in low abundance.
The dominance of ants as granivores in our sites makes seed size an especially important trait in determining seed survival and seedling establishment. Small-seeded species (e.g., Siparuna guianensis, Xylopia aromatic, Solanum lycocarpum) are most likely to lose seeds to granivores (see Ordóñez and Retana 2004; Suazo et al. 2013), likely due to the morphological inability of ants to carry heavier seeds (Pirk and Lopez de Casenave 2010). However, it is also consistent with the general conclusion that seed predation rates decline as seed size increases (Moles et al. 2003). Our result suggests that efforts at the restoration of degraded tropical sites by direct seeding should consider how seed size and local granivore diversity might interact to influence seedling establishment in addition to factors such as microsite conditions (Doust et al. 2006; Iacona et al. 2010, 2012).
Birds are important dispersers in many ecosystems (Perez and Bulla 2000; Kelt et al. 2004, Suazo et al. 2013), including the Cerrado (Christianini and Oliveira 2010). Although ants are the primary remover of seeds in our study, two plant species with relatively larger seeds—Tapirira guianensis and Matayba guianensis—were removed more in the control treatment, suggesting that birds were important consumers of these species. Whether this removal results in seeds being consumed or dispersed is unclear, but if some seeds are secondarily dispersed by birds they are likely to move far greater distances than those removed by ants. Even if this secondary dispersal does not have the impact on the population dynamics of Cerrado tree species that primary dispersal by birds putatively has (Christianini and Oliveira 2010), it could still influence the genetic structure of populations and their ability to colonize new sites (Côrtes and Uriarte 2013).
On average, seed removal was significantly lower in agricultural sites than Cerrado vegetation. Populations of ants and rodents may be lower in monocultures than in Cerrado fragments due to the intensive land-use history (Christianini and Galetti 2007; Frizzo and Vasconcelos 2013), use of pesticides (Motzke et al. 2013), or increased exposure of rodents to predators (Sunyer et al. 2013). Nevertheless, rodents may be important seed predators of some species in monocultures (sensu Fischer and Türke 2016). For instance, we found an interaction between predator exclosure treatment and habitat for Siparuna guianensis and Solanum lycocarpum, with rodents comparatively more important than ants as seed removers in the monocultures than in fragments. Although our results differ from previous work finding a trend towards greater seed predation in open (Gallegos et al. 2014) or disturbed habitats (Myster 2004), they nevertheless suggest that both the natural and managed establishment of larger-seeded species could be favored in our study landscape. This is particularly true in monocultures, because both ants and rodents appear to prefer smaller seeds.
The distance between fragments and the contrast between matrix and fragment structure have been put forward as important landscape features influencing the outcome of plant–animal interactions in remnants of natural habitat (Tewksbury et al. 2002; Donoso et al. 2004; Herrera et al. 2011). However, we detected no systematic effects of fragment connectivity or isolation on the results of our experiments. Instead we found that the effects of landscape structure and fragment properties on seed removal rates were species-, habitat-, and site-specific. This complicates the ability to develop more general restoration plans based on seeding that mitigate the effects of landscape structure on seed removal. They do suggest, however, that considering landscape structure may be critical in the success of management plans or restoration efforts focused on single-species seed additions.
Tropical regions contain most of the world biodiversity, but the destruction of tropical habitats for agriculture is one of the main causes of global biodiversity loss (Arroyo-Rodríguez et al. 2015). Secondary seed dispersal and granivory may play a critical role in the recovery of these ecosystems because of how they affect patterns of seedling establishment (Cole 2009; Gallegos et al. 2014). This makes understanding the interactions between plants and animals—many of which are both seed predators and secondary dispersers—of paramount importance for habitat restoration (Arroyo-Rodríguez et al. 2015). Our results add to the growing body of literature indicating ants are the main post-dispersal seed predators in the Cerrado, with the impacts of rodents and birds mediated in part by seed size, seed species, and level of habitat disturbance (Christianini and Galetti 2007; Christianini and Oliveira 2010; Ferreira et al. 2011). We also found that seed removal was lowest in areas covered by crops, which suggests the remaining fragments of habitat will be critical to the regeneration of Cerrado tree species when agricultural areas are abandoned. Finally, our results suggest that while direct seeding may result in recruitment for some species irrespective of location, for others it will be important to consider the local landscape context and the composition of the animal community.
References
Aguiar RWS, dos Santos SF, da Silva Morgado F, Ascencio SD, de Mendonça Lopes M, Viana KF (2015) Insecticidal and repellent activity of Siparuna guianensis Aubl. (Negramina) against Aedes aegypti and Culex quinquefasciatus. PLoS ONE 10:e0116765. doi:10.1371/journal.pone.0116765
Arroyo-Rodríguez V, Melo FPL, Martínez-Ramos Miguel, Bongers F, Chazdon RL, Meave JA, Norden N, Santos BA, Leal IR, Tabarelli M (2015) Multiple successional pathways in human-modified tropical landscapes: new insights from forest succession, forest fragmentation and landscape ecology research. Biol Rev. doi:10.1111/brv.12231
Bao F, Lima LB, Luz PB (2014) Caracterização morfológica do ramo, sementes e plântulas de Matayba guianensis Aubl. e produção de mudas em diferentes recipientes e substratos. Rev Árvore 38:63–71
Bates D, Maechler M, Bolker B (2015) Lme4: linear mixed-effect models using S4 classes. R package version 0.999375–39. http://CRAN.R-project.org/package=lme4. Accessed Oct 2015
Beckman NG (2013) The distribution of fruit and seed toxicity during development for eleven neotropical trees and vines in Central Panama. PLoS ONE 8:e66764. doi:10.1371/journal.pone.0066764
Bello C, Galetti M, Pizo MA, Magnago LF S, Rocha MF, Lima RAF, Jordano P (2015) Defaunation affects carbon storage in tropical forests. Science 1:1–11. https://doi.org/10.1126/sciadv.1501105
Brandão CRF, Silva RR, Feitosa RM (2011) Cerrado ground-dwelling ants (Hymenoptera: Formicidae) as indicators of edge effects. Zoologia 28:379–387
Briani DC, Guimarães PR (2007) Seed predation and fruit damage of Solanum lycocarpum (Solanaceae) by rodents in the cerrado of central Brazil. Acta Oecol 31:8–12
Briones-Salas M, Sánchez-Cordero V, Sánchez-Rojas G (2006) Multi-species fruit and seed removal in a tropical deciduous forest in Mexico. Can J Bot 84:433–442
Bruna EM, Guimarães JF, Lopes CT, Duarte P, Gomes ACL, Belentani SCS, Vasconcelos HL (2010) Mammalia, Estação Ecológica do Panga, a Cerrado protected area in Minas Gerais state, Brazil. Check List 6:668–675
Carvalho FMV, De Marco P, Ferreira LG (2009) The Cerrado into-pieces: habitat fragmentation as a function of landscape use in the savannas of central Brazil. Biol Conserv 142:1392–1403. https://doi.org/10.1016/j.biocon.2009.01.031
Castellani ED, Damião Filho CF, Aguiar IB (2001) Caracterização morfológica de frutos e sementes de espécies arbóreas do gênero Xylopia (Annonaceae). Rev Bras Sementes 30:102–113
Christianini AV, Galetti M (2007) Spatial variation in post-dispersal seed removal in an Atlantic forest: effects of habitat, location and guilds of seed predators. Acta Oecol 32:328–336
Christianini AV, Oliveira PS (2010) Birds and ants provide complementary seed dispersal in a neotropical savanna. J Ecol 98:573–582
Cole RJ (2009) Postdispersal seed fate of tropical montane trees in an agricultural landscape, Southern Costa Rica. Biotropica 41:319–327. doi:10.1111/j.1744-7429.2009.00490.x
Côrtes MC, Uriarte M (2013) Integrating frugivory and animal movement: a review of the evidence and implications for scaling seed dispersal. Biol Rev Camb Philos Soc 88:255–272. doi:10.1111/j.1469-185X.2012.00250.x
Costa AN, Vasconcelos HL, Vieira-Neto EHM, Bruna EM (2008) Do herbivores exert top-down effects in neotropical savannas? Estimates of biomass consumption by leaf-cutter ants. J Veg Sci 19:849–854
Costa AN, Vasconcelos HL, Bruna EM (2017) Biotic-drivers of plant early-recruitment success: selective herbivory by leaf-cutter ants as an ecological filter in neotropical savannas. J Ecol 105:132–141
Craig MT, Orrock JL, Brudvig LA (2011) Edge-mediated patterns of seed removal in experimentally connected and fragmented landscapes. Landsc Ecol 26:1373–1381
Cramer JM, Mesquita RCG, Williamson BG (2007) Forest fragmentation differentially affects seed dispersal of large and small-seeded tropical trees. Biol Conserv 137:415–423. https://doi.org/10.1016/j.biocon.2007.02.019
Cuevas-Reyes P, Gilberti L, González-Rodríguez A, Wilson Fernandes G (2013) Patterns of herbivory and fluctuating asymmetry in Solanum lycocarpum St. Hill (Solanaceae) along an urban gradient in Brazil. Ecol Indic 24:557–561
De Assis PA, Theodoro PNET, de Paula JE, Araújo AJ, Costa-Lotufo LV, Michel S, Grougnet R, Kritsanida M, Espindola LS (2014) Antifungal ether diglycosides from Matayba guianensis Aublet. Bioorg Med Chem Lett 24:1414–1416
De Oliveira AP, Machado CG, Sigrist MR (2013) Matayba guianensis (Sapindaceae): frugivoria por aves em remanescente de Cerrado do Centro-Oeste brasileiro. SITIENTIBUS Série Ciências Biológicas 13:1–7. https://doi.org/10.13102/scb319
Dirzo R, Mendoza E, Ortiz P (2007) Size-related differential seed predation in a heavily defaunated neotropical rain forest. Biotropica 39:355–362
Donoso DS, Grez AA, Simonetti JA (2004) Effects of forest fragmentation on the granivory of differently sized seeds. Biol Conserv 115:63–70
Doust SJ, Erskine PD, Lamb D (2006) Direct seeding to restore rainforest species: microsite effects on the early establishment and growth of rainforest tree seedlings on degraded land in the wet tropics of Australia. For Ecol Manag 234:333–343. https://doi.org/10.1016/j.foreco.2006.07.014
Ferreira AV, Bruna EM, Vasconcelos HL (2011) Seed predators limit plant recruitment in neotropical savannas. Oikos 120:1013–1022
Fischer C, Türke M (2016) Seed preferences by rodents in the agri-environment and implications for biological weed control. Ecol Evol 6:5796–5807. https://doi.org/10.1002/ece3.2329
Françoso R, Brandão R, Nogueirac CC, Salmonaa YB, Machadoa RB, Collia GL (2015) Habitat loss and the effectiveness of protected areas in the Cerrado biodiversity hotspot. Nat Conserv 13:35–40. doi:10.1016/j.ncon.2015.04.001
Frizzo TLM, Vasconcelos HL (2013) The potential role of scattered trees for ant conservation in an agriculturally dominated neotropical landscape. Biotropica 45:644–651
Gallegos SC, Hensen I, Schleuning M (2014) Secondary dispersal by ants promotes forest regeneration after deforestation. J Ecol 102:659–666. https://doi.org/10.1111/1365-2745.12226
García D, Chacoff NP (2007) Scale-dependent effects of habitat fragmentation on hawthorn pollination, frugivory and seed predation. Conserv Biol 21:400–411
Gonçalves VF, Silva AM, Baesse CQ, Melo C (2015) Frugivory and potential of birds as dispersers of Siparuna guianensis. Braz J Biol 75:300–304. doi:10.1590/1519-6984.11413
Google Earth Pro (2016) https://www.google.com/earth/download/gep/agree.html. Accessed Dec 2016
Gustafson EJ, Parker GR (1994) Using an index of habitat patch proximity for landscape design. Landsc Urban Plan 29:117–130
Herrera JM, García D, Morales JM (2011) Matrix effects on plant-frugivore and plant-predator interactions in forest fragments. Landsc Ecol 26:125–135. https://doi.org/10.1007/s10980-010-9541-7
Iacona GD, Kirkman LK, Bruna EM (2010) Effects of resource availability on patterns of seedling recruitment in a fire maintained savanna. Oecologia 163:171–180
Iacona GD, Kirkman LK, Bruna EM (2012) Experimental test for facilitation of seedling recruitment by the dominant bunchgrass in a fire-maintained savanna. PLoS ONE 7:e39108
Kelt DA, Meserve PL, Gutierrez JR (2004) Seed removal by small mammals, birds and ants in semi-arid Chile, and comparisons with other systems. J Biogeogr 31:931–942
Laurance WF, Ferreira LV, Rankin-de Mérona J, Laurance SG, Hutchings RG, Lovejoy TE (1998) Effects of forest fragmentation on recruitment patterns in Amazonian tree communities. Conserv Biol 12:460–464
Legendre P, Legendre L (1998) Numerical ecology, 2nd English edn. Elsevier Science BV, Amsterdam, The Netherlands
Lombardi JA, Junior M (1993) Seed dispersal of Solanum lycocarpum St. Hil. (Solanaceae) by the maned wolf, Chrysocyon brachyurus Illiger(Mammalia, Canidae). Ciencia e cultura 45:126–127
Lorenzi H (2009) Árvores brasileiras: manual de identificação e cultivo de plantas arbóreas nativas do Brasil 3 ed. Instituto Plantarum de Estudos da Flora
Martinson HM, Fagan WF (2014) Trophic disruption: A meta-analysis of how habitat fragmentation affects resource consumption in terrestrial arthropod systems. Ecol Lett 17:1178–1189. https://doi.org/10.1111/ele.12305
Melo FPL, Dirzo R, Tabarelli M (2006) Biased seed rain in forest edges: evidence from the Brazilian Atlantic forest. Biol Conserv 132:50–60
Mercival RF, Lunardi V, Guimarães PR Jr, Galetti M (2008) Factors affecting seed predation of Eriotheca gracipiles (Bombacaceae) by parakeets in a Cerrado fragment. Acta Oecol Int J Ecol 33:240–245
Miranda-Melo AA, Martins FR, Santos FAM (2007) Estrutura populacional de Xylopia aromatica (Lam.) Mart. e de Roupala montana Aubl. em fragmentos de cerrado no Estado de São Paulo. Rev Bras Bot 30:501–507
Moles AT, Warton DI, Westoby M (2003) Do small-seeded species have higher survival through seed predation than large-seeded species? Ecology 84:3148–3161
Motzke I, Tscharntke T, Sodhi NS, Klein AM, Wanger TC (2013) Ant seed predation, pesticide applications and farmers’ income from tropical multi-cropping gardens. Agric For Entomol 15:245–254. doi:10.1111/afe.12011
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Myster RW (2004) Regeneration filters in post-agricultural fields of Puerto Rico and Ecuador. Plant Ecol 172:199–209
Oliveira-Filho AT, Ratter JA (2002) Vegetation physiognomies and woody flora of the Cerrado biome. In: Oliveira PS, Marquis RJ (eds) The Cerrados of Brazil: ecology and natural history of a neotropical savanna. Columbia University Press, New York, pp 91–120
Ordóñez JL, Retana J (2004) Early reduction of post-fire recruitment of Pinus nigra by post-dispersal seed predation in different time-since-fire habitats. Ecography 27:449–458
Perez EM, Bulla L (2000) Dietary relationships among four granivorous doves in Venezuelan savannas. J Trop Ecol 16:865–882
Pinto LVA, da Silva EAA, Davide AC, Mendes V, Toorop PE, Hilhorst HWM (2007) Mechanism and control of Solanum lycocarpum seed germination. Ann Bot 100:1175–1187
Pirk GI, Lopez de Casenave J (2010) Influence of seed size on feeding preference and diet composition of three sympatric harvester ants in the central Monte Desert, Argentina. Ecol Restor 25:439–445
Prevedello JA, Vieira MV (2010) Plantation rows as dispersal routes: a test with didelphid marsupials in the Atlantic Forest, Brazil. Biol Conserv 143:131–135. https://doi.org/10.1016/j.biocon.2009.09.016
Pufal G, Klein AM (2013) Post-dispersal seed predation of three grassland species in a plant diversity experiment. J Plant Ecol 6:468–479
R Core Team (2016). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rey PJ, Garrido JL, Alcántara JM, Ramírez JM, Aguilera A, García L, Manzaneda AJ, Fernández R (2002) Spatial variation in ant and rodent post-dispersal seeds of vertebrate-dispersed predation. Funct Ecol 16:773–781
Santos SWM, Silva-Mann R, Ferreira RA, Arrigoni-Blank MF, Fitzgerald AB, Melo JCP (2009) Morphology of fruits, flowers and seeds of pau-pombo (Tapirira guianensis Aublet—Anacardiaceae) in São Cristóvão, SE, Brazil. Sci For 37:47–54
Silva LE, Reis RA, Moura EA, Amararal W, Sousa Jr, PT (2015) Plantas do Gênero Xylopia: Composição Química e Potencial Farmacológico. Revista Brasileira de Plantas Medicinais, 17:814–826. https://dx.doi.org/10.1590/1983-084X/14_076
Suazo AA, Craig DJ, Vanier CH, Abella SR (2013) Seed removal patterns in burned and unburned desert habitats: Implications for ecological restoration. J Arid Environ 88:165–174
Sunyer P, Muñoz A, Bonal R, Espelta JM (2013) The ecology of seed dispersal by small rodents: A role for predator and conspecific scents. Funct Ecol 27: 1313–1321. https://doi.org/10.1111/1365-2435.12143
Terborgh J (1992) Maintenance of diversity in tropical forests. Biotropica 24:283–292
Tewksbury JJ, Levey DJ, Haddad NM, Sargent S, Orrock JL, Weldon A, Danielson BJ, Brinkerhoff J, Damschen EI, Townsend P (2002) Corridors affect plants, animals, and their interactions in fragmented landscapes. Proc Nat Acad Sci USA 99:12923–12926
Valentini CMA, Rodríguez-Ortíz CE, Coelho MFB (2010) Siparuna guianensis Aublet (negramina): Uma revisão. Rev Bras Plantas Med 12:96–104
Vander Wall SB, Longland WS (2004) Diplochory: are two seed dispersers better than one? Trends Ecol Evol 19:155–161. https://doi.org/10.1016/j.tree.2003.12.004
Vander Wall SB, Kuhn KM, Beck MJ (2005) Seed removal, seed predation, and secondary dispersal. Ecology 86:801–806
Vasconcelos PB, Araujo GM, Bruna EM (2014) The role of roadsides in conserving Cerrado plant diversity. Biodivers Conserv 23:3035–3050
Viani RAG, Vidas NM, Pardib MM, Vásquez DCC, Gusson E, Brancalion PHS (2015) Animal-dispersed pioneer trees enhance the early regeneration in Atlantic Forest restoration plantations. Nat Conserv 13:41–46. doi:10.1016/j.ncon.2015.03.005
Vié J, Hilton-Taylor C, Stuart SN (2009) Wildlife in a changing world—an analysis of the 2008 IUCN red list of threatened species. IUCN, Gland, Switzerland, p 180
Acknowledgements
We thank the anonymous reviewers for comments on the manuscript and for assistance in the field. E. M. B. and M. R. O. were supported by the CAPES Special Visiting Researcher Fellowship Program (Grant AUXPE-PVE-0205/2013). H. L. V. was supported by Grants from the Brazilian Council of Research and Scientific Development (CNPq Grants 403733/2012-0 and 302588/2015-9).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Miguel Franco.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rocha-Ortega, M., Bartimachi, A., Neves, J. et al. Seed removal patterns of pioneer trees in an agricultural landscape. Plant Ecol 218, 737–748 (2017). https://doi.org/10.1007/s11258-017-0725-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-017-0725-y