Abstract
Vegetation gradients in Central Brazil encompass sharp transitions from savanna to forests, representing an iconic example of how interactions between plants and soils regulate biogeographical boundaries. Here we describe how canopy productivity regulates nutrient inputs to soils, affecting fertility and influencing ecosystem distribution. Based on soil and litter systematically collected during 12 months along a gallery forest-savanna transition, we determined associations between canopy cover (leaf area index—LAI) and the (re)cycling of essential macronutrients. This evaluation was aimed at aggregating information about biogeochemical controls of ecosystem distribution/productivity, to support conservation and management efforts in the region. We confirmed two hypotheses: (i) nutrient inputs via litterfall are significantly higher in forest than in adjacent savanna, and (ii) litter quality varies with canopy productivity and litter nutrient concentrations influence soil fertility reinforcing forest and savanna as alternate stable states. These observations delineate a productivity-efficiency tradeoff in which savannas communities are more efficient in the use of limiting nutrients, yet, less productive than forest communities. The relative importance of different nutrients, apparent on recovery rates in the litter regressed against LAI, revealed that the expansion of forest ecosystems is limited by P > Mg > K > N, with highest conservation observed for P. Differences in Ca input were also significant among ecosystems, but depended solely on the amount of deposited litter, with no differences in recovery rates observed between forests and savannas. A tradeoff-based framework could be used to predict ecotonal stability in the region, with transitions between savannas and forests marked by clear changes in species composition, productivity, litter deposition, and soil fertility.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In the cerrado region of Central Brazil, riparian forests and savannas, known as gallery forests and cerrado sensu stricto, respectively (Ribeiro and Walter 2008), occur side by side. Their transitions are characterized by abrupt changes in vegetation structure and many factors have been found to be associated with the long-term persistence of these sharp ecotones. For instance, groundwater availability (Eiten 1994; Rossatto et al. 2012, 2014), topography (Ruggiero et al. 2006), nutrient availability (Haridasan 1998; Silva et al. 2008, 2013), and fire frequency (Coutinho 2006; Hoffmann et al. 2012) are some of the main drivers of variations in vegetation physiognomies across the regional landscape. Since the strongest associations are found between vegetation structure and soil nutrient status (Furley 1992, 1999), it has been suggested that forest expansion or retraction result from vegetation-induced changes in soil fertility (Durigan and Ratter 2006; Furley 1999; Hopkins 1992; Ratter 1992; Silva and Anand 2011; Silva et al. 2008, 2010, 2013). These associations between soil and vegetation are also observed globally in other savanna ecosystems in Africa, Australia (Bond et al. 2004; Lehmann et al. 2011), and North America (Dahlgren et al. 2003). However, the mechanisms involved in this process remain poorly understood. Causal links between species performance, community composition, resource use, and nutrient (re)cycling at the ecosystem level remain to be established. Here we investigate how canopy production translates into nutrient depositional gradients along a sharp forest-savanna transition. Forest encroachment into ancient savanna and grassland ecosystems has been observed throughout South America (Franco et al. 2014; Silva 2014; Silva and Anand 2013), and represents a new challenge for developing sound management practices and conservation policies in the region.
Gallery forests cover approximately 5 % of the cerrado region, which encompasses approximately 2,000,000 km2 (Ribeiro and Walter 2008). These forests follow streambeds and rivers as corridors throughout the landscape and typically have greater soil water and nutrient availability than the neighboring savanna (Haridasan 1998). Gallery forests woody vegetation is mostly evergreen (Ribeiro and Walter 2008) while some savanna tree species lose leaves seasonally (Franco et al. 2005). Gallery forests also have higher productivity than savannas, as measured, for example, by total vegetation cover per land area (leaf area index—LAI; Silva et al. 2008, 2013), suppressing virtually all flammable grass cover that dominates open savannas (Hoffmann et al. 2005, 2012; Silva et al. 2013). Savanna and forest tree species represent different functional types, as tradeoffs between nutrient requirements and adaptations to resist fires reinforce savanna and forest as alternate stable states and explain the long-term persistence of vegetation mosaics in this region (Silva et al. 2013). Woody savanna species are adapted to low fertility conditions that predominate in the region (Haridasan 2008), whereas only a small group of forest species colonizes open areas leading to forest expansion (Geiger et al. 2011).
Current features of gallery forests and savannas such as soil organic matter content, litter production, and nutrients concentrations in soil and litter are important to understand how vegetation and soils interact to regulate boundary dynamics. Low nutrient concentrations are generally observed in litter produced under low soil nutrient availability (Vitousek 2004) (i.e., savannas on highly weathered oxisols), whereas the opposite is expected for more fertile sites, where individual plants and entire ecosystems tend to be less efficient, depositing more nutrients as litter per unit of biomass produced (Parron 2004; Parron et al. 2010; Silva and Anand 2011). Litter production and nutrient inputs have been previously evaluated in savanna (Kozovits et al. 2007; Nardoto et al. 2006; Peres et al. 1983; Schiavini 1983; Silva 2004; Silva et al. 2007; Villalobos-Vega et al. 2011) and forest communities (Parron 2004; Parron et al. 2010). Combined, these studies suggest the existence of a tradeoff between productivity and efficiency at the ecosystem level, which remains to be demonstrated.
Here, we investigate litter production and soil fertility as a way of characterizing nutrient dynamics across a typical gallery forest and savanna transition in Central Brazil, using a new approach to interpret litterfall and nutrient deposition as a proxy for nutrient use efficiency at the ecosystem level. Specifically, we tested two hypotheses: (i) variation in canopy productivity across the transition leads to marked differences in total nutrient input to savannas and forests, greater in the latter; and (ii) differences in nutrient use efficiency affect litter quality and influence soil fertility across the transition reinforcing the co-existence forest and savanna as alternate stable states. We analyzed litter and soil macronutrients across a natural (fire-protected) gradient spanning forest-savanna transitions, discussing the role of nutrients in influencing species composition and ecosystem productivity, which will be important to support conservation and management efforts in the region and elsewhere.
Methods
Site description
This study was performed at the Ecological Reserve of the Brazilian Institute of Geography and Statistics (IBGE) located at 35 km south from Brasilia in the Federal District of Brazil (15°57′05″–15°57′15″ S and 47°52′38″–47°52′54″ W), at an elevation of 1100 m a.s.l. and nearly flat terrains (5° slope), along forest-savanna transitions (Fig. 1a). The average annual rainfall recorded at IBGE meteorological station is 1461 mm (1980–2008), with a well-defined dry season from May to September and a mean annual temperature of 22.5 °C. At the time this study was performed, from October 2007 to September 2008, total rainfall was 1635 mm. Designated as a conservation area in 1975, the reserve harbors an area of 1350 hectares protecting different vegetation physiognomies, such as grasslands (open fields known as campo limpo or campo sujo), predominant savanna (cerrado sensu stricto), woodland savanna or dense wooded savannas (cerradão), and gallery forests associated with streams and springs (Furley 2007; Ribeiro and Walter 2008; Walter et al. 2008). Soils at the reserve are well characterized and deep well-drained oxisols predominate. The chosen area for this study was the gallery forest-savanna boundary of the Roncador stream (Fig. 1b), which was disturbed by fire only once in the past 35 years operation, in 1994, when non-prescribed burning occurred. The vegetation has recovered to its original state and there is now a clear (few meters) transition in vegetation structure and composition across this boundary, with a decline of 53 % of leaf area index (LAI) from gallery forest to savanna (Geiger et al. 2011). In total, 129 tree species from 51 botanical families occur along this transition (Geiger et al. 2011; Supplementary Material), including species of the family Fabaceae (Faboideae), typical N2-fixing species, among the most abundant in both forest and savanna (Felfili et al. 2004). This type of abrupt forest-savanna transition is commonly observed throughout the region, even in fire-protected areas, representing an integral component of the landscape.
Sampling design
Four transects of 60 m length separated by a distance of 40 m from each other were delineated according to the vegetation gradient at the Roncador site, so that they were positioned perpendicularly to the forest edge and centered at the border. The central point of each transect is defined as 0 (zero), where an unambiguous interruption of the forest canopy and the decreasing tree density coincide with the appearing of shrubs and grasses. From this central point, the four transects extended 30 m into forest and 30 m into savanna (Fig. 1c).
Litter traps of 0.25 m2 and 2.0 mm nylon mesh were arranged every 5 m along each transect (Fig. 1c). All traps were placed 15 cm above the ground. Since our goal was to evaluate changes in overstory litterfall and composition, litter from ground layer grasses and small eudicot species bellow 15 cm height were not included. In terms of mass and nutrient stock, the contribution of the herbaceous layer in both forest and savanna represents only a small fraction of total litter (Abdala et al. 1998), here better represented by the combination of contributions from the tree and shrub components. Over an entire year, paper bags were used to collect the litter intercepted and accumulated by each litter trap every 15 days. These samples were combined to obtain monthly samples. After sorting into two fractions: leaves and miscellaneous (leaf fragments, reproductive parts, and twigs with diameter ≤1 cm), the samples were washed using distilled water, oven-dried in paper bags at 70 °C for 72 h, and then weighed. Branches and wood debris greater than 1 cm diameter and occasional animal tissues were discarded.
Soil samples of 100 g were collected using a drill, along each transect immediately adjacent to each litter trap (Fig. 1c). Soils were sampled once at the end of the dry season (September 2008), from 0 to 10 cm and from 10 to 20 cm depths, totaling 52 samples per depth. Soil samples were sieved through a 2.0 mm mesh to separate fine roots, placed in plastic bags, and dried at room temperature.
Also adjacent to litter traps, LAI was obtained every 5 m along the four transects (Fig. 1c) using the CI-110 Plant Canopy Imager (CID Inc., Camas, WA, USA). This instrument captures LAI indirectly, based on digital hemispherical images (150° field of view) of the vegetation overhead to estimate canopy LAI as a measure of total vegetation cover per land area (vegetation density quantification rather than green leaf area per se). At each point of transects, images were taken at 30 cm above ground level in order to measure trees and shrubs LAI, excluding herbaceous layer. Images were taken in May 2008 under dawn conditions at the end of rainy season, when all species even deciduous ones had full foliage. For trees, our values included stem area, as well as leaf area.
Sample analysis
Samples obtained every fifteen days in the litter traps positioned along the four transects were combined to obtain monthly samples, totaling 52 samples per month (26 of leaves and 26 of miscellaneous). Chemical analyses of litter correspond to the leaf fraction (leaf litterfall), which was powdered and homogenized after being dried. Total N concentration in leaves was determined by the micro-Kjeldahl digestion and distillation method (Bremner and Mulvaney 1982), while P, K, Ca, and Mg concentrations were determined by digestion with nitric, sulfuric, and perchloric tri-acid solution (10:1:2). The cations were determined with atomic absorption spectrophotometry and available P by colorimetry (Allen 1989).
Soil organic carbon was determined using the Walkley and Black method (Walkley and Black 1934) and total N was determined by micro-Kjeldahl digestion and distillation method (Bremner and Mulvaney 1982). Exchangeable calcium and magnesium were extracted by a 1 M KCl solution and available P and K were determined using the method of Mehlich (Mehlich 1953). The cations concentrations in soils were determined with atomic absorption spectrophotometry and available P by colorimetry (Allen 1989). Soil physics data were not included in our analysis, since differences of soil texture (i.e., clay) along forest and savanna transitions at the IBGE Reserve are not crucial to determine the distribution of both vegetation types (Hoffmann et al. 2009, 2012).
Statistical analysis
Total nutrient input was calculated by multiplying leaf litterfall mass by the concentration of macronutrients at every sampling point along the transition. We performed repeated measures analyses of variance, using a mixed effect model to test the significance of fixed (distance from the border) and random (date) effects on leaves and miscellaneous, leaf litterfall nutrients, and soil nutrient concentrations. We tested fixed and random effects, using interaction terms to examine the significance of variations in litter deposition and nutrient input along the gradient at different sampling dates. Data were log transformed prior to analysis to meet assumptions of normality (Shapiro-Wilks test) (Shapiro and Wilk 1965) and homogeneity of variance, detected as departures from homoscedasticity (Bartlett’s test), both at <0.05 significance levels. Regression analyses were performed and angular coefficients of least square regressions used to interpret changes in canopy productivity (total annual leaf litterfall mass) and recovery of each measured macronutrient (dependent variables), separately regressed against LAI values across the transition. As such, litterfall mass and nutrient deposition could be interpreted as a proxy for plants and ecosystems efficiency and recycling patterns. All statistical analyses were performed using the SAS-created JMP statistical software (SAS Institute 2012).
Results
Leaf area index of trees and shrubs increased from savanna toward forest, with values ranging from 1.4 to 3.9, where the lowest values were found at 20 m from border into the savanna and the highest values measured at 30 m from border into the forest. Measurements of LAI characterized the differences in canopy productivity and vegetation structure across the transition, represented as the ratio of upper leaf surface per unit of land surface area (Fig. 2a).
Leaf area index (LAI) for trees and shrubs (a) and litterfall mass (b) produced along the Roncador gallery forest (white box) and savanna (gray box) transition in Central Brazil. Error bars represent standard errors of the average. Different letters represent statistical differences between means according to repeated measures analysis of variance. Positive correlation of litterfall regressed against LAI (R 2 = 0.88, p < 0.01), with an increase of 255.54 kg ha−1 year−1 (slope value) of leaf litter for each unit increased in LAI
Consistent with changes in vegetation structure, total annual litterfall was significantly higher in the forest physiognomy (6.3 t ha−1 year−1) than in the savanna (3.5 t ha−1 year−1) (Fig. 2b). The leaf fraction accounted for approximately 75 % of total litterfall, whereas the remaining fraction was classified as miscellaneous. The outputs of a repeated measures analysis showed highly significant (p < 0.01) effects of both date and distance from the border, in which the forest environment received in average 4.7 t ha−1 of leaves and 1.6 t ha−1 of miscellaneous litter throughout the year. No significant interactions occurred between leaf litter input along the gradient and sampling date (p = 0.59), but significant interactions (p = 0.01) occurred between miscellaneous litter input and time. In both cases, most of the litter input occurred between the months of August and September, which corresponds to the dry season. However, during the annual production of the miscellaneous component, it was observed an increase in the amount of twigs, wood debris, flowers, fruits, and seeds fragments due to the impact of the first rainfalls after the prolonged dry season (data not shown).
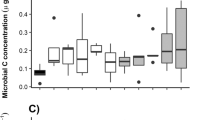
The total input of macronutrients from leaf litter along the forest-savanna transects followed the same trend as observed for the mass of leaves and miscellaneous produced across the transition, with all nutrients analyzed showing higher inputs in litter produced by the forest ecosystem (Fig. 3b). Only Ca concentration in leaf litterfall between savanna and forest was significantly different (p < 0.05), with higher values found in the forested habitat (Fig. 3a).
Nutrient concentration (a) and total nutrient input (b) in leaf litterfall along the Roncador gallery forest (white box) and savanna (gray box) transition in Central Brazil. Significant differences in leaf litterfall between ecosystems (p < 0.05) were only observed for Ca, but total nutrient inputs differed significantly in all cases. Error bars represent standard errors of the average
Using least square regressions using the total annual leaf litterfall against LAI, we found a strong positive relationship (R 2 = 0.88, p < 0.01), with an increase of approximately 255.54 kg of leaf litter per ha−1 year−1 for each unit increased in LAI measured during the rainy season (Fig. 2a). We used this relationship as a baseline for comparisons with least square regressions performed using litter nutrient inputs, by comparing the angular coefficients of each of these linear regressions as a proxy for nutrient use efficiency; i.e., biomass produced per unit of each nutrient recovered. In this sense, the angular coefficient of each regression (Fig. 4) was interpreted as an indicator of the degree of conservative use of each nutrient. The regression line closest to that of litterfall mass corresponds to the least conservative scenario. In this scenario, litter mass explains most of the variation in nutrient input, while the most conservative scenario (i.e., where nutrients are recycled within plants and variations in litter deposition do not affect input) would be represented by low angular coefficients (i.e., flat lines; Fig. 4).
Changes in total litter nutrient input along the Roncador gallery forest (white box) to the savanna (gray box) forest-savanna transition in Central Brazil. Least square regressions represent relationships between distance from the border and total nutrient input via leaf litterfall. Leaf area index (LAI) values reflect differences in canopy productivity between the two ecosystems
The results of these regressions revealed highly significant relationships between nutrient inputs and LAI (R 2 > 0.82) with slope values of 0.12 (P), 0.51 (Mg), 1.99 (K), 2.67 (N), and 4.35 (Ca), indicating the following sequence P > Mg > K > N as representative of high to low conservation, corresponding to differences in efficiency caused by distinct limitation, where P is the most limiting nutrient. Importantly, levels of deposited Ca cannot be compared directly with other nutrients through this method, as it is a structural component of the plant tissue that is not recycled (i.e., reassimilated) prior to litter deposition. This is evidenced by the large angular coefficient of Ca deposition, which closely matches total litterfall production along the gradient, even though Ca is considered a major limiting nutrient for biomass production and forest expansion in the region (Silva et al. 2013).
Soil organic carbon and total N varied significantly across the gradient with an abrupt change at the border and consistently higher values observed in the forest than in the adjacent savanna (Fig. 5). Reinforcing the notion of a P-limited transition, available P levels showed no significant changes across the gradient, ranging from 0.77 to 1.35 mg kg−1 (0 to 10 cm depth) and from 0.77 to 1.15 mg kg−1 (10 to 20 cm depth). Similarly, levels of K were not significantly different across the gradient, ranging from 0.18 to 0.30 cmol (+) kg−1 (0 to 10 cm depth) and from 0.16 to 0.23 cmol (+) kg−1 (10 to 20 cm depth). Exchangeable Ca and Mg concentrations at both soil depths showed divergent patterns. Levels of Ca were found to be depleted at the border, but reached maximum values 30 m deep into the savanna for both soil layers (5.86 and 2.75 cmol (+) kg−1); and at 25 m (7.88 cmol (+) kg−1, 0 to 10 cm depth) and at 20 m (4.44 cmol (+) kg−1, 10 to 20 cm depth) into the forest. Levels of Mg peaked at the border in both soil layers (1.88 and 1.23 cmol (+) kg−1) (Fig. 5), but no relationship was found between soil and leaf litterfall nutrient concentrations along the forest and savanna transition (Supplementary Material).
Soil organic carbon, total N, available P, K, and exchangeable Ca and Mg in the surface layers along the Roncador gallery forest (white box) and savanna (gray box) transition in Central Brazil. Error bars represent standard error of the average. For the two soil layers differences between ecosystems are as follows: organic carbon and N (p < 0.01); P (p = 0.45 and 0.83); K (p = 0.74 and 0.99); Ca (p = 0.61 and 0.70); and Mg (p = 0.53 and 0.54) according to repeated measures analyses of variance
Discussion
Litter deposition across forest-savanna boundaries
Our results confirmed the hypothesis that an increase in litter production from savanna to forest determines differences in total nutrient input. Inputs of N, P, K, Ca, and Mg significantly increase from open to closed canopies, mostly due to the effect of leaf litter mass, but also determining changes in litter quality across the forest-savanna transition. This corroborates the second hypothesis that differences in nutrient use efficiency and litter quality promote higher fertility in forest soils. A more in-depth analysis reveals that nutrient inputs vary along the gradient, with the influx of different macronutrients reflecting the degree in which each element limits productivity. These results show that patterns previously identified at the individual and species levels also hold at the ecosystem level, delineating a productivity-efficiency tradeoff, in which savannas communities are more efficient in the use of limiting nutrients, yet, less productive than forest communities. Under similar edaphic conditions, forest species grow faster and have higher leaf nutrient concentration than their congeneric savanna counterparts (Hoffmann et al. 2005; Rossatto et al. 2009). This is attributed to differences in stem to crown biomass allocation, which characterize forest and savanna trees as separate functional groups (Silva et al. 2013) and emphasize the role of species traits in generating gradients of resource accumulation through litter deposition.
Previous studies attempted to quantify the amount of litterfall produced in forest ecosystems of Central Brazil. At the same reserve, Parron (2004) registered an average of 6.7 t ha−1 year−1 for three gallery forest communities. These values are similar to ours (6.3 t ha−1 year−1), but litterfall in other forests under constraining moisture regime, range from 11.1 t ha−1 to 8.8 t ha−1 (Pagano and Durigan 2000), whereas in woodland savanna (cerradão) of the same region, annual litterfall has been reported to be 7.8 t ha−1 (Peres et al. 1983) and 5.6 t ha−1 (Cianciaruso et al. 2006). In regional deciduous forests litterfall ranges from values close to ours (6.1 t ha−1 year−1; Werneck et al. 2001) to almost twice as large (12.2 t ha−1 year−1; Hora et al. 2008). Notably, in all these cases litterfall produced by the canopies of forest species surpassed values that we observed in open savanna (3.5 t ha−1 year−1). This pronounced difference in total litterfall happens within a few meters from the forest border and makes the total amount of nutrients derived from litter to the soil surface much lower than values observed in forests. Other surveys in savannas areas ranged from 1.5 to 2.4 t ha−1 year−1 (Kozovits et al. 2007; Nardoto et al. 2006; Peres et al. 1983; Schiavini 1983), indicating an even lower contribution from litter inputs.
Differences in environmental conditions may also account for oscillations in litter deposition throughout the year (Peres et al. 1983). Seasonal water shortage, for instance, provokes greater shed of vegetative material in savannas (Dias and Oliveira-Filho 1997), while continuous water supplies in gallery forests buffers the effects of seasonality. Beyond such effects, phenology of the dominant species across the vegetation transition could be a potential cause for differences in seasonal litter production and nutrient input (Marimon-Júnior 2007).
Across the transition, the most important differences in soils were organic carbon and N contents. This contrasts with patterns of litter nutrient composition and indicates that nutrient fluxes via litterfall are not simply a function of soil fertility. Instead, they reflect the large differences in nutrient accumulation in forest and savanna vegetation, evidenced in the much greater aboveground biomass and nutrient stocks of forests (Silva et al. 2013). When litter quality is low, such as observed in the savannas, decomposition is expected to take longer, causing a lag in nutrient release and constraining both soil fertility and plant productivity (Dahlgren et al. 2003; Vitousek 2004). Here, corroborating this expectation, total nutrient inputs were found to be positively associated with canopy productivity, but there are important differences concerning each nutrient. The following sequence represents the order of limiting nutrients according to their degree of conservation (P > Mg > K > N), where P was the element used most conservatively across the vegetation gradient. Despite close associations between N and P levels in plant across biomes (Elser et al. 2007), our results show large N fluxes via leaf litterfall in contrast with low P fluxes as LAI increases, indicating a high P use efficiency.
As mentioned above, Ca cannot be compared directly with other nutrients based on its relationship with LAI, due to its structural role in plant cell wall (Lambers et al. 1998), which does not permit reassimilation prior to litter deposition (Vergutz et al. 2012). However, Ca is considered a major limiting nutrient for biomass production and forest expansion in the region (Silva et al. 2013). Due to its low mobility within plants (Kazakou et al. 2007), Ca input from litter is dependent solely on the total amount of deposited residues (König et al. 2002), which would be enhanced in forest soils as a result of greater canopy productivity and, consequently, mass of litter deposited (Fig. 4).
The role of litter nutrient inputs in driving forest expansion
Based on both individual level measurements of congeneric savanna and forest tree and estimates of soil and biomass nutrient stocks, Silva et al. (2013) concluded that Ca and P are the most limiting nutrients to build high-biomass forests in forest-savanna transitions. In fact, these nutrients are the most limiting ones to support a transition to any forested state and appear to be higher in the aboveground biomass than in soils of savannas and gallery forests. This is supported by our findings that around the border there is a decline in soil Ca availability (Fig. 5), which suggests a substantial constraint to forest expansion. The amount of Ca required to build low (woodland savanna) and high (gallery) biomass forests vary from 46 to 645 kg ha−1, whereas P requirements range from 3 to 57 kg ha−1 to allow the canopy cover typical of forest ecosystems found in the region (Silva et al. 2013). The slight variation of P concentrations in litter across the gradient (Fig. 3a), in spite of large variations in litter deposition, corroborates other studies using data collected from hundreds of surveys analyzing the response of diverse terrestrial ecosystems to P enrichment, considered particularly limiting in tropical ecosystems (Elser et al. 2007).
Soil stocks of N, K, and Mg are sufficient to allow forest cover at the study site, but the addition of P and Ca from litter would be required to allow the establishment and development of forest communities (Silva et al. 2013), favoring a transition to stable forested states (Hoffmann et al. 2009). The observed variation in litter nutrient content in relation to canopy production according to their respective degree of limitation has two important implications: (i) more litter would mean more nutrient inputs to the system except in the case of P which is recycled most efficiently; and (ii) soil nutrient gradients would be reinforced by vegetation with the most important nutrient determining forest distribution as a result of soil-plant feedbacks being Ca. Since Ca, as well as P, K, and Mg are originated from the parent material (Brady and Weil 2007), preexisting differences between organic nutrients (i.e., N) in the highly weathered soils typical of the studied region would also be important in determining the potential for forest expansion.
Implications for conservation and management
The continuous input of nutrient derived from litter is the main nutrients source maintaining gallery forest’s structure and species diversity. The fire-protected area of the Roncador vegetation boundary currently has forest species comprising 14 % of mature species and more than 33 % of juveniles (seedlings and saplings) in the adjacent savanna community, which supports the notion that floristic shifts precede structural changes (Geiger et al. 2011). Because of greater nutrient investments in leaf canopy and stem biomass, forest species differ from savanna species when both are growing under nutrient-poor soils. Intrinsic species differences lead to differences at the ecosystem level (Silva et al. 2013), determining new suppression and resistance thresholds (Hoffmann et al. 2012), which increase the possibility of forest encroachment (Hoffmann et al. 2003). Causal links between efficiency-productivity tradeoffs at the species level and community dynamics can influence ecosystem responses to environmental change at large spatiotemporal scales, as, for example, when refugia populations expand or contract in response to climate fluctuations (Silva and Anand 2011, 2013). Similarly, we provide evidence that a tradeoff-based approach is useful to explain ecosystem dynamics and ecotonal stability, with transitions between savanna and gallery forest marked by measurable changes in litter and soil composition. Long-term assessments and fertilization experiments would be useful in validating the generality of these patterns and to guide conservation and management efforts based on nutritional controls of species performance, as it propagates to communities and ecosystems.
References
Abdala GC, Caldas LS, Haridasan M, Eiten G (1998) Above and belowground organic matter and root: shoot ratio in a cerrado in Central Brazil. Braz J Ecol 2:11–23
Allen SE (1989) Chemical analysis of ecological materials. Blackwell Scientific Publications, Oxford
Bond WJ, Woodward FI, Midgley GF (2004) The global distribution of ecosystems in a world without fire. New Phytol 165:525–538
Brady NC, Weil RR (2007) The nature and properties of soils, 14th edn. Prentice Hall, New Jersey
Bremner JM, Mulvaney CS (1982) Nitrogen total. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis: chemical and microbiological properties, 2nd edn. American Society of Agronomy, Madison, pp 595–624
Cianciaruso MV, Pires JSR, Delitti WBC, Silva EFLP (2006) Produção de serapilheira e decomposição do material foliar em um cerradão na Estação Ecológica de Jataí, município de Luiz Antônio, SP, Brasil. Acta Bot Bras 20:49–59
Coutinho L (2006) O conceito de Bioma. Acta Bot Bras 20:13–23
Dahlgren RA, Horwath WR, Tate KW, Camping TJ (2003) Blue oak enhance soil quality in California oak woodlands. Calif Agric 57:42–47
Dias HCT, Oliveira-Filho AT (1997) Variação temporal e espacial da produção de serapilheira em uma área de Floresta Estacional Semidecídua Montana em Lavras, MG. Rev Arvore 21:11–26
Durigan G, Ratter JA (2006) Successional changes in cerrado and cerrado/forest ecotonal vegetation in western São Paulo state, Brazil, 1962–2000. Edinb J Bot 63:119–130
Eiten G (1994) Vegetação do Cerrado. In: Pinto MN (ed) Cerrado: caracterização, ocupação e perspectivas. UnB/SEMATEC, Brasília, pp 17–73
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harlope WS, Hillebrand H et al (2007) Global analysis of nitrogen and phosphorous limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1–8
Felfili JM, Silva-Júnior MC, Sevilha AC, Fagg CW, Walter BMT, Nogueira PE, Rezende AV (2004) Diversity, floristic and structural patterns of cerrado vegetation in Central Brazil. Plant Ecol 175:37–46
Franco AC, Bustamante M, Caldas LS, Goldstein G, Meinzer FC, Kozovits AR, Rundel P, Coradin VTR (2005) Leaf functional traits of Neotropical savanna trees in relation to seasonal water deficit. Trees Struct Funct 19:326–335
Franco AC, Rossatto DR, Silva LCR, Ferreira CS (2014) Cerrado vegetation and global change: the role of functional types, resource availability and disturbance in regulating plant community responses to rising CO2 levels and climate warming. Theor Exp Plant Physiol 26:19–38
Furley PA (1992) Edaphic changes at the forest-savanna boundary with particular reference to the neotropics. In: Furley PA, Proctor J, Ratter JA (eds) Nature and dynamics of forest-savanna boundaries. Chapman and Hall, London, pp 91–117
Furley PA (1999) The nature and diversity of neotropical savanna vegetation. Glob Ecol Biogeogr 8:223–241
Furley PA (2007) Tropical savannas and associated forests: vegetation and plant ecology. Prog Phy Geogr 31:203–211
Geiger EL, Gotsch SG, Vale GD, Haridasan M, Franco AC, Hoffmann WA (2011) Distinct roles of savanna and forest tree species in regeneration following fire suppression in a Brazilian savanna. J Veg Sci 22:312–321
Haridasan M (1998) Solos de mata de galeria e nutrição mineral de espécies arbóreas em condições naturais. In: Ribeiro JF (ed) Cerrado: caracterização e recuperação de matas de galeria. EMBRAPA Cerrados, Planaltina, pp 19–28
Haridasan M (2008) Nutritional adaptations of native plants of the cerrado biome in acid soils. Braz J Plant Physiol 20:183–195
Hoffmann WA, Orthen B, Nascimento PKV (2003) Comparative fire ecology of tropical savanna and forest trees. Funct Ecol 17:720–726
Hoffmann WA, Silva-Júnior ER, Machado GC, Bucci SJ, Scholz FG, Goldstein G, Meinzer FC (2005) Seasonal leaf dynamics across a tree density gradient in a Brazilian savanna. Oecologia 145:307–316
Hoffmann WA, Adasme R, Haridasan M, Carvalho M, Geiger EL, Pereira MAB, Gotsch SG, Franco AC (2009) Tree topkill, not mortality, governs the dynamics of alternate stable states at savanna-forest boundaries under frequent fire in Central Brazil. Ecology 90:1326–1337
Hoffmann WA, Geiger LG, Gotsch SG, Rossatto R, Silva LCR, Lau OL, Haridasan M, Franco AC (2012) Ecological thresholds at the savanna-forest boundary: how plant traits, resources and fire govern the distribution of tropical biomes. Ecol Lett 15:759–768
Hopkins B (1992) Ecological processes at the forest-savanna boundary. In: Furley PA, Proctor J, Ratter JA (eds) Nature and dynamics of forest-savanna boundaries. Chapman and Hall, London, pp 21–33
Hora RC, Primavesi O, Soares JJ (2008) Contribuição das folhas de lianas na produção de serapilheira em um fragmento de floresta estacional semidecidual em São Carlos, SP. Rev Bras Bot 31:277–285
Kazakou E, Garnier E, Navas ML, Roumet C, Colin C, Laurent G (2007) Components of nutrient residence time and the leaf economics spectrum in species from Mediterranean old-fields differing in successional status. Funct Ecol 21:235–245
König FG, Brun EJ, Schumacher MV, Longhi SJ (2002) Devolução de nutrientes via serapilheira em um fragmento de Floresta Estacional Decidual no município de Santa Maria, RS. Bras Florest 74:45–52
Kozovits AR, Bustamante MMC, Garofalo CR, Bucci S, Franco AC, Goldstein G et al (2007) Nutrient resorption and patterns of litter production and decomposition in a Neotropical Savanna. Funct Ecol 21:1034–1044
Lambers H, Chapin FS, Pons TL (1998) Plant physiological ecology, 2nd edn. Springer, New York
Lehmann CER, Archibald SA, Hoffmann WA, Bond WJ (2011) Deciphering the distribution of the savanna biome. New Phytol 191:197–209
Marimon-Júnior BH (2007) Relação entre diversidade arbórea e aspectos do ciclo biogeoquímico de uma floresta mondominante de Brosimum rubescens Taub. e uma floresta mista no leste mato-grossense. Thesis, University of Brasilia
Mehlich A (1953) Determination of P, Ca, Mg, K, Na and NH4. North Carolina Department of Agriculture, Raleigh
Nardoto GB, Bustamante MMC, Pinto AS, Klink CA (2006) Nutrient use efficiency at ecosystem and species level in savanna areas of Central Brazil and impacts of fire. J Trop Ecol 22:191–201
Pagano SN, Durigan G (2000) Aspectos da ciclagem de nutrientes em Matas Ciliares do oeste do Estado de São Paulo, Brasil. In: Rodrigues RR, Leitão-Filho HF (eds) Matas Ciliares: conservação e recuperação. Editora da USP/FAPESP, São Paulo, pp 109–123
Parron LM (2004) Aspectos da ciclagem de nutrientes em função do gradiente topográfico em uma mata de galeria no Distrito Federal. Thesis, University of Brasilia
Parron LM, Bustamante MMC, Markewitz D (2010) Fluxes of nitrogen and phosphorus in a gallery forest in the Cerrado of central Brazil. Biogeochemistry 105:89–104
Peres JRR, Suhet AR, Vargas MAT, Drozdowicz A (1983) Litter production in areas of Brazilian ‘Cerrados’. Pesq Agropecu Bras 18:1037–1043
Ratter JA (1992) Transitions between cerrado and forest vegetation in Brazil. In: Furley PA, Proctor J, Ratter JA (eds) Nature and dynamics of forest-savanna boundaries. Chapman and Hall, London, pp 417–430
Ribeiro JF, Walter BMT (2008) As principais fitofisionomias do Bioma Cerrado. In: Sano SM, Almeida SP, Ribeiro JF (eds) Cerrado: ambiente e flora. EMBRAPA Cerrados, Planaltina, pp 151–212
Rossatto DR, Hoffmann WA, Franco AC (2009) Differences in growth patterns between co-occurring forest and savanna trees affect the forest savanna boundary. Funct Ecol 23:689–698
Rossatto DR, Silva LCR, Villalobos-Vega R, Sternberg LSL, Franco AC (2012) Depth of water uptake in woody plants relates to groundwater level and vegetation structure along a topographic gradient in a neotropical savanna. Environ Exp Bot 77:259–266
Rossatto DR, Silva LCR, Sternberg LSL, Franco AC (2014) Do woody and herbaceous species compete for soil water across topographic gradients? Evidence for niche partitioning in a Neotropical savanna. S Afr J Bot 91:14–18
Ruggiero PGC, Pivello VR, Sparovek G, Teramoto E, Pires-Neto AG (2006) Relação entre solo, vegetação e topografia em área de cerrado (Parque Estadual de Vassununga, SP): como se expressa em mapeamentos? Acta Bot Bras 20:383–394
SAS Institute (2012) JMP version 10. SAS Institute, Cary, North Carolina, USA
Schiavini I (1983) Alguns aspectos da ciclagem de nutrientes em uma área de cerrado (Brasília, DF): chuva, produção e decomposição de liter. Dissertation, University of Brasilia
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591–611. doi:10.1093/biomet/52.3-4.591
Silva MRSS (2004) Produção de serapilheira, biomassa e diversidade de comunidades bacterianas do solo em áreas de Cerrado sob diferentes usos e manejos. Dissertation, Universidade de Brasília
Silva LCR (2014) The importance of climate-driven forest-savanna biome shifts in anthropological and ecological research. Proc Natl Acad Sci 111:3831–3832
Silva LCR, Anand M (2011) Mechanisms of Araucaria (Atlantic) forest expansion into Southern Brazilian grasslands. Ecosystems 14:1354–1371
Silva LCR, Anand M (2013) Historical links and new fronteirs in the study of forest-atmosphere interactions. Community Ecol 14:208–218
Silva CJ, Sanches L, Bleich ME, Lobo FA, Nogueira JS (2007) Produção de serapilheira no Cerrado e Floresta de Transição Amazônia-Cerrado do Centro-Oeste Brasileiro. Acta Amaz 37:543–548
Silva LCR, Sternberg LSL, Haridasan M, Hoffmann WA, Miralles-Wilhelm F, Franco AC (2008) Expansion of gallery forests into central Brazilian savannas. Glob Change Biol 14:1–11
Silva LCR, Haridasan M, Sternberg LSL, Franco AC, Hoffmann WA (2010) Not all forests are expanding over central Brazilian savannas. Plant Soil 333:431–442
Silva LCR, Hoffmann WA, Rossatto DR, Haridasan M, Franco AC, Horwath WR (2013) Can savannas become forests? A coupled analysis of nutrient stocks and fire thresholds in central Brazil. Plant Soil 373:829–842
Vergutz L, Manzoni S, Porporato A, Novais RF, Jackson RB (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82:205–220
Villalobos-Vega R, Goldstein G, Haridasan M, Franco AC, Miralles-Wilhelm F, Scholz FG, Bucci SJ (2011) Leaf litter manipulations alter soil physicochemical properties and tree growth in a Neotropical savanna. Plant Soil 346:385–397
Vitousek PM (2004) Nutrient cycling and limitation: Hawai’i as a model system. Princeton University Press, Oxford
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid tritation method. Soil Sci 37:29–38
Walter BMT, Carvalho AM, Ribeiro JF (2008) O conceito de savana e seu componente cerrado. In: Sano SM, Almeida SP, Ribeiro JF (eds) Cerrado: ambiente e flora. EMBRAPA Cerrados, Planaltina, pp 19–45
Werneck MS, Pedralli G, Gieseke LF (2001) Produção de serapilheira em três trechos de uma floresta semidecídua com diferentes graus de perturbação na Estação Ecológica do Tripuí, Ouro Preto, MG. Rev Bras Bot 24:195–198
Acknowledgments
We thank the staff of IBGE Ecological Reserve for infrastructure and logistical support. We also thank William Horwath for valuable discussion, William Hoffmann, Augusto Franco, Erika Geiger, Sybil Gotsh, and many other colleagues who helped with data collection, and Mara Rúbia Suzana Chaves who helped with laboratory analysis. This research was supported by CAPES Brazil.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erik P Hamerlynck.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paiva, A.O., Silva, L.C.R. & Haridasan, M. Productivity-efficiency tradeoffs in tropical gallery forest-savanna transitions: linking plant and soil processes through litter input and composition. Plant Ecol 216, 775–787 (2015). https://doi.org/10.1007/s11258-015-0466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0466-8