Abstract
In the debate on the impact of urbanisation on biodiversity, two characteristics of urbanisation have been identified that mainly determine the extent of the impact, namely the level of urbanisation and the landscape context. More recently, it has been theorised that urban sprawl in an intensive agricultural landscape has a positive influence on pollinators by increasing habitat and resource availability. Using the eastern Po Plain (north-eastern Italy) as a model system, we investigated the relationship between attributes of landscape composition and configuration, pollinator richness and visits in 39 randomly selected permanent plots. Contrary to expectations, we found no relationship between urban sprawl and pollinator species richness. Conversely, descriptors of urban sprawl such as landscape heterogeneity and the proportion of urban green spaces had a positive influence on the number of pollinator visits. This suggests that urban sprawl, when occurring in an intensive agricultural land, has a positive effect on the abundance of local pollinator populations, while it may not promote pollinator richness due to limited immigration opportunities in the matrix of intensive agricultural land. Our results emphasise the importance of urban green spaces in supporting pollinator communities, but also the need to improve the heterogeneity and permeability of the landscape matrix for biodiversity to enhance pollinator conservation in human-modified landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation is a complex phenomenon involving a redistribution of people between agricultural and urban areas (van der Merwe and Bekker 2014), resulting in profound landscape transformations. The social, economic, and political dimensions of urbanisation lead to changes in the landscape in terms of both composition (i.e., different types of land cover and land use) and configuration (i.e., distribution, size and abundance of different land cover-use types) (e.g., Ruas et al. 2022).
When it comes to examining the impact of urbanisation on biodiversity, two factors are crucial: the level of urbanisation (e.g., urban densification, urban sprawl) and the landscape context (e.g., agricultural, natural) in which urbanisation takes place (Wenzel et al. 2020). Urban densification is the process of concentrating urban development in a limited area (Vergnes et al. 2014), which is characterised by high residential density, compact development with impervious surfaces covering more than 50% of the built-up area (Zhang et al. 2009), and a shortage of urban green spaces (Jim 2004). On the other side, urban sprawl is characterised by scattered settlements, often single-family houses with residential gardens (Artmann et al. 2019) and low residential density; compared to urban densification, urban sprawl holds a more widespread built-up area yet also more green spaces (Fuller and Gaston 2009; Brunner and Cozens 2013; EEA 2021). The dichotomy between dense and sprawling urbanisation has often been used to assess the impacts of different urban development schemes on biodiversity and to support sustainable urban planning (Haaland and van den Bosch 2015). Urban densification has been shown to have a significant impact on biodiversity in a limited area, in contrast to the comparatively low impact observed with urban sprawl, which typically occurs with low intensity over a large area (Sushinsky et al. 2013). Nevertheless, the impacts of urban densification to surrounding large areas of remaining natural and semi-natural habitats are limited, allowing urban sensitive species to find refuge and maintain their populations (Sushinsky et al. 2013; Soga et al. 2014; Concepción et al. 2016).
When the nature of the landscape context in which urbanisation takes place is considered, the response of organisms to different urban development schemes may become more complex. In fact, studies have shown that responses to urbanisation are often species-specific (e.g., Fenoglio et al. 2020), being also modulated by such species traits as resource requirements, mobility, and specialisation, that are expected to influence ecological processes such as metapopulation dynamics and ecological interactions (Öckinger et al. 2010; Concepción et al. 2015). When accounting for species traits it becomes thus clear that, besides the density of impervious surfaces, other features may modulate the impact of urbanisation, namely the landscape context in which urbanisation occurs, in terms of both landscape composition and configuration. Substantial empirical support has been given to the hypothesis that biodiversity decline is at least partly due to processes occurring at landscape scales, with such landscape attributes as the size, shape and spatial arrangement of land use-land cover patches, and their relative proportion being key factors in affecting biodiversity patterns and ecological processes (Betts et al. 2019; Martin et al. 2019). As such, the landscape context is expected to strongly influence species movements, especially when suitable habitat patches are surrounded by a matrix of unsuitable land (e.g., impervious surfaces or areas of intensive agriculture) that limit species dispersal (Revilla et al. 2004).
Species traits and the landscape context become even more crucial when considering species interactions (Mody et al. 2020), with effects increasing when strongly interacting organisms are involved, such as e.g., in the pollination process. Since the pollination process involves interactions between species belonging to two different trophic levels, any event of species loss, habitat loss or degradation that alters the distribution and abundance of a species poses potential risks to the associated partners. The complexity of plant-pollinator interactions and the large differences in terms of species traits may explain why pollinators show contrasting effects to urbanisation (Baldock 2020). While some studies considered urbanised areas as a potential refuge for pollinators and thus plant-pollinator interactions (Baldock et al. 2015; Hall et al. 2017; Theodorou et al. 2017), other saw urbanisation as a major cause of species loss and pollination contact reduction (Andrieu et al. 2009; Martins et al. 2013; Lagucki et al. 2017; Cardoso and Gonçalves 2018).
Based on an extensive review, recently Wenzel et al. (2020) hypothesised that urban sprawl in intensive agricultural landscapes may mitigate pollinator loss. More specifically, they theorised a hump-shaped relationship between pollinator richness and urbanisation. The core of Wenzel et al. (2020) hypothesis is that intermediate level of urbanisation, associated with urban sprawl (namely, with impervious surfaces covering less than 50% of the built-up area; Wenzel et al. 2020) in a resource-poor intensive agricultural land, would ensure greater landscape heterogeneity and increase habitat and resource availability for pollinators, thereby leading to positive diversity responses.
So far, studies have addressed the dichotomy of agricultural and urban landscapes (e.g., Kaluza et al. 2016) or agricultural-urban gradients where natural and semi-natural habitats were still relevant landscape components (e.g., Verboven et al. 2014; Theodorou et al. 2017). However, as emphasised by Wenzel et al. (2020), these approaches may fail to capture the complexity of the urbanisation process by only considering the urban vs. agricultural dichotomy, thereby ignoring both the urban development scheme and the nature of the agricultural areas (e.g., intensive vs. extensive).
In the present study, we empirically tested the hypothesis of Wenzel et al. (2020) in order to understand the response of pollinator communities and plant-pollinator interactions to the landscape context along an urbanisation gradient, associated with urban sprawl in an intensive agricultural land. In particular, we hypothesise that a positive role of urban sprawl for pollinator communities is mediated by the improvement of landscape heterogeneity and the availability of urban green spaces rather than urbanisation per se. Furthermore, we tested whether such a positive influence is exerted both on pollinator richness and on the number pollination visits. Since large, intensively used agricultural areas generally provide few resources for pollinators, we hypothesise that the positive effect of urban sprawl on pollinator communities will improve their local abundance rather than their richness, as the movement of pollinators across intensive agricultural land is limited, thus restricting immigration opportunities. The results may help to fill the knowledge gaps on the effects of urban sprawl in a highly modified landscape and provide insights into how to plan more pollinator-oriented landscapes.
Materials and methods
Study site and data collection
The study took place in the Po Plain floodplain, in northern Italy. Floodplains dominate large parts of European land and represent one of the most distinctive landscapes of cultural and economic relevance. Today, floodplains are among the most heavily modified landscapes in the world, mostly to increase the production of crops, livestock, and urban development (Tockner and Stanford 2002). Among the floodplains, the Po Plain is one of the most populated and urbanised areas in Europe and is representative of them. With an area of over 45,000 km2 and a population of over 25 million people (Lonati and Riva 2021), it is one of the largest lowland areas in Europe. The Po Plain has experienced centuries of intensive agricultural activities and urbanisation that have reduced the extent of natural and semi-natural habitats (Marchetti 2002) and created a landscape characterised by large intensive agricultural areas intermingled with impervious urban surfaces (Pristeri et al. 2020).
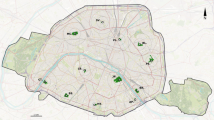
Data were collected in the eastern Po Plain and specifically in the Veneto Region (Fig. 1). A total of 15 sampling sites were selected in the study area, with an average minimum distance of 7,356.27 ± 8,450.29 m between them (mean ± SD). Sampling sites are representative of the Po Plain landscape, which is characterised by sprawl urbanisation over intensive agricultural land (Pristeri et al. 2020). At the selected sites, 39 permanent plots (ranging from 1 to 6 per site) were established using the stratified random method, with spontaneous herbaceous vegetation of urban green spaces (i.e., roadsides, ditches’ banks, overmanaged grasslands, field margins, uncultivated areas, recently planted hedges) serving as strata. The number of plots was proportional to the number and surface of the different types of urban green spaces. Plots were characterised by ruderal vegetation dominated by Cynodon dactylon (L.) Pers., Dactylis glomerata L. Lolium multiflorum Lam., Plantago lanceolata L., Poa pratensis L. subsp. pratensis, Potentilla reptans L., and Trifolium pratense L. Plots were 2 m × 2 m or 8 m × 0.5 m in size if the sampling areas had a narrow shape and longitudinal development (e.g., ditches’ banks).
Each plot was monitored three times in 2022, once between late April and early May, once in mid-July and once between late August and early September, to account for plants with different flowering times and pollinators with different yearly periods of activity (Fantinato 2019). During each survey, in each plot, the list of plant species in bloom was recorded and the number of floral displays of each blooming species was counted. Depending on the plant species, a floral display consisted of a single flower (e.g., Ranunculus bulbosus L.), a flower head (e.g., Bellis perennis L.), or a group of flowers that together formed a recognisable visual unit (e.g., Trifolium pratense L.; Hegland and Totland 2005).
To assess plant-pollinator interactions, floral visitors were recorded during each survey (on sunny and windless days; Fortel et al. 2014) by observing their interactions with floral displays for 16 min, 8 min between 8 a.m. and 1 p.m., and 8 min between 1 p.m. and 6 p.m., respectively, to capture visitors with different daily periods of activity (Fantinato et al. 2021). If a visitor had direct contact with the reproductive organs of flowers for more than 1 s, they were considered and recorded as a potential pollinator (Hegland and Totland 2005). Pollinators were identified either in the field or captured and subsequently identified in the laboratory at the species or morphospecies level. Following Villa-Galaviz et al. (2023), the number of visits has been used as a proxy for pollinator abundance.
The landscape context of each sampling site was derived from a high-resolution land cover-use map with a radius of 500 m, which has been shown to be consistent with the flight range of most pollinators (Bennett and Lovell 2019), using QGIS 2.18.18. The centre of the map for each site was located on the plot or on the centroid of the plots contained in the site if more than one plot occurred. Based on aerial photographs provided by the Veneto Region (size: 17,751 × 15,351 px; resolution: 0.2 m × 0.2 m), every appreciable landscape element on a scale of 1:500 was drawn and categorised according to CORINE Land Cover classification (Bossard et al. 2000). For each map, we calculated compositional attributes, i.e., the percentage cover of patches for each CORINE Land Cover category (Wei et al. 2017) and landscape heterogeneity based on Shannon diversity (Hˈ) of land cover-use types (Steckel et al. 2014), as well as configurational attributes, i.e., the number of patches of urban green spaces and semi-natural areas (Lami et al. 2021). We only counted the number of patches in the categories of urban green spaces and semi-natural areas, as these are the landscape elements crucial for biodiversity and their number can be used as a proxy of habitat fragmentation, which has a major impact on biodiversity conservation (Fischer and Lindenmayer 2007).
Data analysis
Prior to the analysis, landscape heterogeneity was calculated based on the Shannon diversity (Hˈ) of land cover-use types taking into account all mapped CORINE categories. Then, the land cover-use types for each map were grouped into five macro-categories (see Appendix 1, Table 3): semi-natural areas, water bodies, intensive agricultural land, urban green spaces, and impervious surfaces. All collected data were quality controlled and outliers were identified and removed (i.e., eight plots with a number of flowers 20 times higher than the average number of floral displays). To determine whether landscape composition (i.e., surface of semi-natural areas, water bodies, intensive agricultural land, urban green spaces, impervious surfaces, and landscape heterogeneity) and configuration (i.e., number of patches of semi-natural areas and urban green spaces) affected the richness of plant species in bloom and the number of floral displays we ran two separate generalised linear mixed models (GLMMs; function glmer; R version 4.1.1; package lme4). Firstly, we checked for possible collinearity between the independent variables using Spearman’s rank correlations and retained those variables that were not highly correlated with each other (i.e., Spearman’s |r| < 0.75). As the cover of impervious surfaces, intensive agricultural land and urban green spaces were collinear (see Appendix I), we chose to include in the models only the cover of urban green spaces being the distinctive characteristic of urban sprawl. Therefore, models with the richness of plant species in bloom and the number of floral displays as dependent variables included the percent cover and the number of patches of urban green spaces and semi-natural areas, the surface of water bodies and landscape heterogeneity. Since the variables had different units, they were scaled (z-scores; i.e., scaled to a mean of 0 and standard deviation of 1). Data collected during each 16 min survey were used as replicates. Two random factors were used in the models, i.e., sampling site to avoid pseudo-replication (Fantinato 2019) and plot identity (nested within site). The models were simplified by backward elimination of non-significant independent variables. Since data distribution of the dependent variables was not overdispersed (function dispersiontest; R version 4.1.1; package AER; Cameron and Trivedi 1990), the models were run using the Poisson error distribution and log link functions. Likelihood ratio tests (LRT; function drop1; R version 4.1.1; package stats; Chambers 1992) were used to determine the significance of the models.
We then tested the influence of landscape composition and configuration, the richness of plant species in bloom and the number of floral displays on pollinator species richness and visits. The distribution of pollinator species richness and visits showed an excess of zero counts. To deal with this distribution, we opted for a zero-inflated Poisson model (Zuur et al. 2009; Buffa et al. 2021). The Zero inflated Poisson model is the result of two stochastic models. The first model generates zero counts of pollinator species in a plot with probability π. The second model generates counts with a non-zero probability 1-π according to a Poisson distribution with mean λ. The predicted number of pollinator species is given by (1-π)⋅λ. The absence of species is promoted by high values of π while species richness is fostered by larger values of λ. In our modelling framework, the parameters π and λ are a function of some covariates and are estimated jointly. Since the richness of plant species in bloom and the number of floral displays were collinear (see Appendix I), only the richness of plant species in bloom was included in the model. We specified a zero-inflated model for pollinator species richness by including the richness of plant species in bloom, the percent cover and the number of patches of urban green spaces and semi-natural areas, the surface of water bodies and landscape heterogeneity as covariate variables. As the variables had different units, they were scaled (z-scores). The zero inflated Poisson models (zeroinfl function; package pscl; R version 4.1.1; Jackman 2017) were simplified by backward elimination of non-significant covariates. The same was done for the pollinator visits’ model.
Results
The dominant macro land cover categories in studied sites were represented by intensive agricultural land (with a mean cover across sites of 51.21 ± 14.80%; mean ± SD), followed by impervious surfaces (22.40 ± 11.05%), and urban green spaces (19.07 ± 8.11%). The percent cover of semi-natural areas and of water bodies was always very low (3.56 ± 4.20% and 3.18 ± 1.75%, respectively). Impervious surfaces ranged from 5.44 to 40.18%, covering almost the total spectrum of urban sprawl phenomenon. Urban green spaces ranged from 4.66 to 31.55%, and their average patch area, although not statistically significant, was negatively correlated to the number of patches (Pearson’s correlation coefficient; r = -0.48, p = 0.07).
Although each plot was covered by vegetation, plants in bloom were only present in 75.86% of the plots. A total of 91 plant species in bloom were identified (see Appendix II). The number of plant species in bloom per plot ranged from 0 to 12 and the number of floral displays per plot ranged from 0 to 788. The average number of plant species in bloom per plot was 3.06 ± 2.83 with an average number of floral displays of 97.97 ± 151.55. The most frequent animal-pollinated plant was Veronica persica Poir., which was observed in 22 plots.
A total of 176 species or morphospecies of pollinators were recorded (see Appendix II) and 797 pollinator visits were observed between pollinators and 66 of the 91 plant species in bloom (i.e., 72.53% of flowering plant species). Pollinators were recorded in 68.97% of the plots, with an average number of 3.60 ± 3.79 species and an average number of visits of 7.37 ± 10.06 per plot. Pollinator species richness per plot ranged from 0 to 14 species, while the number of visits per plot ranged from 0 to 55 visits. Pollinator species mostly involved in pollination contacts was Apis mellifera L. with a total of 169 visits.
Richness of plant species in bloom showed a significant positive relationship with the percent cover of urban green spaces (Table 1; Fig. 2). The same trend held true for the number of floral displays (Table 1; Fig. 2). This means that the richness of plant species in bloom and the number of floral displays increased with increasing area covered by urban green spaces.
According to the zero-inflated model, the probability of absence of pollinator species and of pollinator visits was strongly related to the richness of plant species in bloom. Specifically, both the probability of absence of pollinator species and pollinator visits decreased with increasing richness of plant species in bloom (Table 2; Fig. 3). In particular, the probability of the absence of pollinator species and pollinator visits dropped to 0% once at least four plant species in bloom were present in a plot (i.e., z-score = 0.33; Fig. 3). In addition to influencing the probability of absence of pollinator species and of pollinator visits, the richness of plant species in bloom also significantly influenced the expected richness of pollinators and expected number of pollinator visits (Table 2; Fig. 4), with expected richness of pollinator species and expected number of visits increasing with increasing richness of plant species in bloom. Regarding the relationships between landscape composition and configuration and pollinator species richness, no significant result was found. On the other hand, the expected number of pollinator visits was positively associated with the percent cover of urban green spaces and landscape heterogeneity and negatively associated with the number of patches of urban green spaces (Table 2; Fig. 4). This means that the number of visits increased with increasing area of urban green spaces and with increasing landscape heterogeneity but decreased with increasing number of patches of urban green spaces. No significant relationships were found between pollinator richness and visits, and the cover of semi-natural areas and their number of patches.
Relationship between the cover of urban green space [z-score] and the richness of plant species in bloom (a), the cover of urban green space [z-score] and the number of floral displays (b). Line represents the estimate of the generalised linear mixed-effect models. Black points are original data point
Association between the probability of absence of pollinator species and the richness of plant species in bloom [z-score] (a), and between pollinator visits and the richness of plant species in bloom [z-score] (b). For each covariate, the probability of absence was estimated as function of the selected covariate, setting the other covariates equal to their mean values
Association between the expected pollinator richness and the richness of plant species in bloom [z-score] (a), and association between the expected number of visits and the richness of plant species in bloom [z-score] (b), the cover of urban green spaces [z-score] (c), the landscape heterogeneity [z-score] (d), and the number of patches of urban green spaces [z-score] (e). For each covariate, the expected number of species or visits was estimated as function of the selected covariate, setting the other covariates equal to their mean values
Discussion
In our study, we did not find a positive response of pollinator species richness to increasing urbanisation in the context of urban sprawl. In particular, pollinator species richness did not show any recognisable patterns clearly associated with urban green spaces, which are used here as a proxy for the urban sprawl process. Instead, both the richness of plant species in bloom and the number of floral displays showed a significant positive correlation with the cover of urban green spaces. Urban green spaces are often only appreciated as features vital for human health and well-being (Lepczyk et al. 2017); however, urban green spaces have also been proven to significantly contribute to biodiversity conservation by harbouring self-sustaining native plant populations (Kowarik and von der Lippe 2018; Wintle et al. 2019), including rare and threatened species (Ives et al. 2016; Planchuelo et al. 2019).
Not surprisingly, our results showed that the presence of pollinators was positively associated with the richness of plant species in bloom, with the probability of absence decreasing with increasing plant richness. This result is in accordance with previous research that consistently indicated that more floral resources such as pollen and nectar attract pollinators and favour their populations (Tscharntke et al. 1998; Ebeling et al. 2008; Hudewenz et al. 2012; Langlois et al. 2020), both in urban (e.g., Mata et al. 2021) and agricultural (e.g., Jönsson et al. 2015) settings. Specifically, the zero-inflated Poisson model allowed us to define a threshold value at which the probability of absence of pollinator species (and thus of pollination visits) tended to zero, i.e. when at least four plant species in bloom were present in a plot.
The number of pollinator visits was not only positively related to the richness of plant species in bloom, but was also positively influenced by landscape attributes such as the cover of urban green spaces and landscape heterogeneity. Accordingly, the effects of urbanisation on pollinators lies in the presence of flowering communities that enhance landscape suitability for these organisms (Wenzel et al. 2020). Urban green spaces are usually very heterogeneously structured (Baldock 2020) and differ in the type, intensity, and timing of the management practises to which they are subject due to the different actors involved (Kowarik and von der Lippe 2018). The interplay of structural heterogeneity and differentiated management helps to create diverse land cover-use types and thus provide a wide variety of living conditions that increase the availability of niches suitable to pollinators (Kühn et al. 2004; Geslin et al. 2016; Parmentier 2023). Therefore, the effects of urban sprawl on pollinator visits, used here as a proxy for pollinator abundance (Villa-Galaviz et al. 2023), are mediated by the effects that urban green spaces have on local resources, as flowers, nesting and overwintering sites, which have a cascading effect on pollinator communities.
Although our approach differs substantially from that of Wenzel et al. (2020) by using green spaces as a proxy for urban sprawl instead of using impervious surface cover, we can conclude that urban green spaces surrounded by intensive agricultural land can have a positive effect on local pollinator abundance, while on the other hand they cannot promote pollinator species richness, possibly due to the limited immigration opportunities in the matrix of intensive agricultural land. This is especially true for central pollinators or non-migratory species (Jauker et al. 2009), as was the case for the species found in our study.
Noteworthy, the number of patches of urban green spaces showed a negative influence on pollinator abundance. Although not significant, the relationship between the number of patches and the mean patch size was negative. The decrease in pollinator visits with increasing number of patches might thus be associated with changes in patch size and increasing edge effects that reduce habitat suitability (Lázaro et al. 2020; Olynyk et al. 2021) and/or affect pollinator movements (González et al. 2016), thereby making pollinator communities more vulnerable to any type of external disturbance.
Despite research repeatedly evidenced the importance of natural and semi-natural areas (e.g., Garibaldi et al. 2011; Kennedy et al. 2013) in supporting pollinator communities, we did not find any relationship between either pollinator richness or pollinator visits and semi-natural areas. This result is likely to be linked to the negligible percent cover of natural and semi-natural areas across the study sites. Although unusual, this confirms the crucial role of urban green spaces associated with urban sprawl. In heavily modified and homogeneous landscapes, where human activities have drastically reduced the extent of natural and semi-natural habitats, urban green spaces may represent the sole green landscape elements. Thus, although fragmented and often of limited surface, urban green spaces may represent small biodiversity hotspots (Kabisch et al. 2022), becoming beneficial microhabitats for pollinators.
A final remark deserves the hump-shaped relationship between pollinator richness and urban sprawl, theorised by Wenzel et al. (2020). We argue that urban sprawl and urban densification are successive steps of the urbanisation gradient, substantially differing in the proportion of urban green spaces. In this regard, urban sprawl represents the rising part of the hump-shaped curve, having its peak at 50% of built-up areas, while the descending part of the curve corresponds to increasing (i.e., > 50%) impervious surface and loss of green areas, flowering communities, and landscape heterogeneity.
In conclusion, the positive effects of urban sprawl on pollinators should be taken with caution. Our results undoubtedly confirm that when taking place in an intensive agricultural landscape, urban sprawl may become beneficial to pollinators by increasing the proportion of such resources they depend on, as flowers, nesting and overwintering sites, thereby making intensive agricultural land more heterogeneous and permeable. However, it has to be considered that urban sprawl determines drastic and long-lasting changes to the landscape, including habitat transformation, degradation and fragmentation, and as such is considered as one of the most serious threats to sustainable land use.
Data availability
The data that support the findings of this study are available from the authors upon reasonable request.
References
Andrieu E, Dornier A, Rouifed S et al (2009) The town Crepis and the country Crepis: how does fragmentation affect a plant-pollinator interaction? Acta Oecol 35:1–7. https://doi.org/10.1016/j.actao.2008.07.002
Artmann M, Kohler M, Meinel G et al (2019) How smart growth and green infrastructure can mutually support each other — A conceptual framework for compact and green cities. Ecol Indic 96:10–22. https://doi.org/10.1016/j.ecolind.2017.07.001
Baldock KCR (2020) Opportunities and threats for pollinator conservation in global towns and cities. Curr Opin Insect Sci 38:63–71. https://doi.org/10.1016/j.cois.2020.01.006
Baldock KCR, Goddard MA, Hicks DM et al (2015) Where is the UK’s Pollinator Biodiversity? The importance of Urban Areas for Flower-Visiting insects. Proc R Soc B 282:20142849. https://doi.org/10.1201/9781315366098-17
Bennett AB, Lovell S (2019) Landscape and local site variables differentially influence pollinators and pollination services in urban agricultural sites. PLoS ONE 14:1–21. https://doi.org/10.1371/journal.pone.0212034
Betts MG, Hadley AS, Kormann U (2019) The landscape ecology of pollination. Landsc Ecol 34:961–966. https://doi.org/10.1007/s10980-019-00845-4
Bossard M, Feranec J, Otahel J (2000) CORINE land cover technical guide – Addendum 2000. Technical Report No. 40. European Environment Agency, Copenhagen
Brunner J, Cozens P (2013) Where have all the trees gone? Urban Consolidation and the demise of Urban Vegetation: a Case Study from Western Australia. Plan Pract Res 28:231–255. https://doi.org/10.1080/02697459.2012.733525
Buffa G, Gaetan C, Piccoli S et al (2021) Using fine-scale field data modelling for planning the management of invasions of Oenothera Stucchii in coastal dune systems. Ecol Indic 125:107564. https://doi.org/10.1016/j.ecolind.2021.107564
Cameron AC, Trivedi PK (1990) Regression-based tests for overdispersion in the Poisson model. J Econom 46:347–364. https://doi.org/10.1016/0304-4076(90)90014-K
Cardoso MC, Gonçalves RB (2018) Reduction by half: the impact on bees of 34 years of urbanization. Urban Ecosyst 21:943–949. https://doi.org/10.1007/s11252-018-0773-7
Chambers JM (1992) Linear models. In: Chambers JM, Hastie TJ (eds) Statistical models in S. Wadsworth and Brooks/Cole, California, pp 220–221
Concepción ED, Moretti M, Altermatt F et al (2015) Impacts of urbanisation on biodiversity: the role of species mobility, degree of specialisation and spatial scale. Oikos 124:1571–1582. https://doi.org/10.1111/oik.02166
Concepción ED, Obrist MK, Moretti M et al (2016) Impacts of urban sprawl on species richness of plants, butterflies, gastropods and birds: not only built-up area matters. Urban Ecosyst 19:225–242. https://doi.org/10.1007/s11252-015-0474-4
Ebeling A, Klein AM, Schumacher J et al (2008) How does plant richness affect pollinator richness and temporal stability of flower visits? Oikos 117:1808–1815. https://doi.org/10.1111/j.1600-0706.2008.16819.x
EEA (2021) Land take and land degradation in functional urban areas. Report 17. Luxembourg, European Environment Agency. https://www.eea.europa.eu/publications/land-take-and-land-degradation. Accessed 18 December 2023
Fantinato E (2019) The impact of (mass) tourism on coastal dune pollination networks. Biol Conserv 236:70–78. https://doi.org/10.1016/j.biocon.2019.05.037
Fantinato E, Sonkoly J, Török P, Buffa G (2021) Patterns of pollination interactions at the community level are related to the type and quantity of floral resources. Funct Ecol 35:2461–2471. https://doi.org/10.1111/1365-2435.13915
Fenoglio MS, Rossetti MR, Videla M (2020) Negative effects of urbanization on terrestrial arthropod communities: a meta-analysis. Glob Ecol Biogeogr 29:1412–1429. https://doi.org/10.1111/geb.13107
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16:265–280. https://doi.org/10.1111/j.1466-8238.2006.00287.x
Fortel L, Henry M, Guilbaud L et al (2014) Decreasing abundance, increasing diversity and changing structure of the wild Bee Community (Hymenoptera: Anthophila) along an urbanization gradient. PLoS ONE 9. https://doi.org/10.1371/journal.pone.0104679
Fuller RA, Gaston KJ (2009) The scaling of green space coverage in European cities. Biol Lett 5:352–355. https://doi.org/10.1098/rsbl.2009.0010
Garibaldi LA, Steffan-Dewenter I, Kremen C et al (2011) Stability of pollination services decreases with isolation from natural areas despite honey bee visits. Ecol Lett 14:1062–1072. https://doi.org/10.1111/j.1461-0248.2011.01669.x
Geslin B, Le Féon V, Folschweiller M et al (2016) The proportion of impervious surfaces at the landscape scale structures wild bee assemblages in a densely populated region. Ecol Evol 6:6599–6615. https://doi.org/10.1002/ece3.2374
González E, Salvo A, Defagó MT, Valladares G (2016) A moveable feast: insects moving at the forest-crop interface are affected by crop phenology and the amount of forest in the landscape. PLoS ONE 11:1–19. https://doi.org/10.1371/journal.pone.0158836
Haaland C, van den Bosch CK (2015) Challenges and strategies for urban green-space planning in cities undergoing densification: a review. Urban Urban Green 14:760–771. https://doi.org/10.1016/j.ufug.2015.07.009
Hall DM, Camilo GR, Tonietto RK et al (2017) The city as a refuge for insect pollinators. Conserv Biol 31:24–29. https://doi.org/10.1111/cobi.12840
Hegland SJ, Totland Ø (2005) Relationships between species’ floral traits and pollinator visitation in a temperate grassland. Oecologia 145:586–594. https://doi.org/10.1007/s00442-005-0165-6
Hudewenz A, Klein AM, Scherber C et al (2012) Herbivore and pollinator responses to grassland management intensity along experimental changes in plant species richness. Biol Conserv 150:42–52. https://doi.org/10.1016/j.biocon.2012.02.024
Ives CD, Lentini PE, Threlfall CG et al (2016) Cities are hotspots for threatened species. Glob Ecol Biogeogr 25:117–126. https://doi.org/10.1111/geb.12404
Jackman S (2017) pscl: Classes and methods for R developed in the political science computational laboratory. R package version, 1(2). United States Studies Centre, University of Sydney. Sydney. New South Wales, Australia. https://github.com/atahk/pscl/. Accessed 16 December 2023
Jauker F, Diekötter T, Schwarzbach F, Wolters V (2009) Pollinator dispersal in an agricultural matrix: opposing responses of wild bees and hoverflies to landscape structure and distance from main habitat. Landsc Ecol 24:547–555. https://doi.org/10.1007/s10980-009-9331-2
Jim CY (2004) Green-space preservation and allocation for sustainable greening of compact cities. Cities 21:311–320. https://doi.org/10.1016/j.cities.2004.04.004
Jönsson AM, Ekroos J, Dänhardt J et al (2015) Sown flower strips in southern Sweden increase abundances of wild bees and hoverflies in the wider landscape. Biol Conserv 184:51–58. https://doi.org/10.1016/j.biocon.2014.12.027
Kabisch N, Frantzeskaki N, Hansen R (2022) Principles for urban nature-based solutions. Ambio 51:1388–1401. https://doi.org/10.1007/s13280-021-01685-w
Kaluza BF, Wallace H, Heard TA et al (2016) Urban gardens promote bee foraging over natural habitats and plantations. Ecol Evol 6:1304–1316. https://doi.org/10.1002/ece3.1941
Kennedy CM, Lonsdorf E, Neel MC et al (2013) A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol Lett 16:584–599. https://doi.org/10.1111/ele.12082
Kowarik I, von der Lippe M (2018) Plant population success across urban ecosystems: a framework to inform biodiversity conservation in cities. J Appl Ecol 55:2354–2361. https://doi.org/10.1111/1365-2664.13144
Kühn I, Brandl R, Klotz S (2004) The flora of German cities is naturally species rich. Evol Ecol Res 6:749–764
Lagucki E, Burdine JD, McCluney KE (2017) Urbanization alters communities of flying arthropods in parks and gardens of a medium-sized city. PeerJ 5:e3620. https://doi.org/10.7717/peerj.3620
Lami F, Bartomeus I, Nardi D et al (2021) Species – habitat networks elucidate landscape effects on habitat specialisation of natural enemies and pollinators. Ecol Lett 24:288–297. https://doi.org/10.1111/ele.13642
Langlois A, Jacquemart AL, Piqueray J (2020) Contribution of extensive farming practices to the supply of floral resources for pollinators. Insects 11:1–19. https://doi.org/10.3390/insects11110818
Lázaro A, Fuster F, Alomar D, Totland Ø (2020) Disentangling direct and indirect effects of habitat fragmentation on wild plants’ pollinator visits and seed production. Ecol Appl 30:1–13. https://doi.org/10.1002/eap.2099
Lepczyk CA, Aronson MFJ, Evans KL et al (2017) Biodiversity in the City: fundamental questions for understanding the Ecology of Urban Green Spaces for Biodiversity Conservation. Bioscience 67:799–807. https://doi.org/10.1093/biosci/bix079
Lonati G, Riva F (2021) Regional scale impact of the covid-19 lockdown on air quality: gaseous pollutants in the Po valley, northern Italy. Atmosphere 12. https://doi.org/10.3390/atmos12020264
Marchetti M (2002) Environmental changes in the central Po Plain (northern Italy) due to fluvial modifications and anthropogenic activities. Geomorphology 44:361–373. https://doi.org/10.1016/S0169-555X(01)00183-0
Martin EA, Dainese M, Clough Y et al (2019) The interplay of landscape composition and configuration: new pathways to manage functional biodiversity and agroecosystem services across Europe. Ecol Lett 22:1083–1094. https://doi.org/10.1111/ele.13265
Martins AC, Gonçalves RB, Melo GAR (2013) Changes in wild bee fauna of a grassland in Brazil reveal negative effects associated with growing urbanization during the last 40 years. Zoologia 30:157–176. https://doi.org/10.1590/S1984-46702013000200006
Mata L, Andersen AN, Morán-Ordóñez A et al (2021) Indigenous plants promote insect biodiversity in urban greenspaces. Ecol Appl 31:1–17. https://doi.org/10.1002/eap.2309
Mody K, Lerch D, Müller AK et al (2020) Flower power in the city: replacing roadside shrubs by wildflower meadows increases insect numbers and reduces maintenance costs. PLoS ONE 15:1–29. https://doi.org/10.1371/journal.pone.0234327
Öckinger E, Schweiger O, Crist TO et al (2010) Life-history traits predict species responses to habitat area and isolation: a cross-continental synthesis. Ecol Lett 13:969–979. https://doi.org/10.1111/j.1461-0248.2010.01487.x
Olynyk M, Westwood AR, Koper N (2021) Effects of Natural Habitat loss and Edge effects on Wild bees and Pollination services in Remnant prairies. Environ Entomol 50:732–743. https://doi.org/10.1093/ee/nvaa186
Parmentier L (2023) Three-strip management: introducing a novel mowing method to generate architectural complexity in perennial flower margins to better support pollinators. J Pollinat Ecol 34:267–283. https://doi.org/10.26786/1920-7603(2023)747
Planchuelo G, von der Lippe M, Kowarik I (2019) Untangling the role of urban ecosystems as habitats for endangered plant species. Landsc Urban Plan 189:320–334. https://doi.org/10.1016/j.landurbplan.2019.05.007
Pristeri G, Peroni F, Pappalardo SE et al (2020) Mapping and assessing soil sealing in Padua Municipality through Biotope Area factor index. Sustainability 12:1–21. https://doi.org/10.3390/su12125167
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. https://www.R-project.org/. Accessed 16 December 2023
Revilla E, Wiegand T, Palomares F et al (2004) Effects of matrix heterogeneity on animal dispersal: from individual behavior to metapopulation-level parameters. Am Nat 164:E130–E153. https://doi.org/10.1086/424767
Ruas RB, Costa LMS, Bered F (2022) Urbanization driving changes in plant species and communities – A global view. Glob Ecol Conserv 38:e02243. https://doi.org/10.1016/j.gecco.2022.e02243
Soga M, Yamaura Y, Koike S, Gaston KJ (2014) Land sharing vs. land sparing: does the compact city reconcile urban development and biodiversity conservation? J Appl Ecol 51:1378–1386. https://doi.org/10.1111/1365-2664.12280
Steckel J, Westphal C, Peters MK et al (2014) Landscape composition and configuration differently affect trap-nesting bees, wasps and their antagonists. Biol Conserv 172:56–64. https://doi.org/10.1016/j.biocon.2014.02.015
Sushinsky JR, Rhodes JR, Possingham HP et al (2013) How should we grow cities to minimize their biodiversity impacts? Glob Chang Biol 19:401–410. https://doi.org/10.1111/gcb.12055
Theodorou P, Albig K, Radzevi R et al (2017) The structure of flower visitor networks in relation to pollination across an agricultural to urban gradient t e. 838–847. https://doi.org/10.1111/1365-2435.12803
Tockner K, Stanford JA (2002) Riverine flood plains: present state and future trends. Environ Conserv 29:308–330. https://doi.org/10.1017/S037689290200022X
Tscharntke T, Gathmann A, Steffan-Dewenter I (1998) Bioindication using trap-nesting bees and wasps and their natural enemies: community structure and interactions. J Appl Ecol 35:708–719. https://doi.org/10.1046/j.1365-2664.1998.355343.x
van der Merwe I, Bekker S (2014) Urbanization: an interdisciplinary approach to a complex phenomenon. Soc Work 39:18–25. https://doi.org/10.15270/39-1-376
Verboven HAF, Uyttenbroeck R, Brys R, Hermy M (2014) Different responses of bees and hoverflies to land use in an urban-rural gradient show the importance of the nature of the rural land use. Landsc Urban Plan 126:31–41. https://doi.org/10.1016/j.landurbplan.2014.02.017
Vergnes A, Pellissier V, Lemperiere G et al (2014) Urban densification causes the decline of ground-dwelling arthropods. Biodivers Conserv 23:1859–1877. https://doi.org/10.1007/s10531-014-0689-3
Villa-Galaviz E, Cirtwill AR, Gibson R et al (2023) What makes a good pollinator? Abundant and specialised insects with long flight periods transport the most strawberry pollen. Ecol Solut Evid 4:1–13. https://doi.org/10.1002/2688-8319.12253
Wei X, Xiao Z, Li Q et al (2017) Evaluating the effectiveness of landscape configuration metrics from landscape composition metrics. Landsc Ecol Eng 13:169–181. https://doi.org/10.1007/s11355-016-0314-6
Wenzel A, Grass I, Belavadi VV, Tscharntke T (2020) How urbanization is driving pollinator diversity and pollination – a systematic review. Biol Conserv 241:108321. https://doi.org/10.1016/j.biocon.2019.108321
Wintle BA, Kujala H, Whitehead A et al (2019) Global synthesis of conservation studies reveals the importance of small habitat patches for biodiversity. Proc Natl Acad Sci U S A 116:909–914. https://doi.org/10.1073/pnas.1813051115
Zhang Y, Odeh IOA, Han C (2009) Bi-temporal characterization of land surface temperature in relation to impervious surface area, NDVI and NDBI, using a sub-pixel image analysis. Int J Appl Earth Obs Geoinf 11:256–264. https://doi.org/10.1016/j.jag.2009.03.001
Zuur AF, Ieno EN, Walker NJ et al (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
This work was supported by EU in the framework of the European LIFE project LIFE19 IT/NAT/000848 PollinAction (https://lifepollinaction.eu/).
Funding
L.L, E.F., D.S. and G.B. were supported by the European LIFE project LIFE19 IT/NAT/000848 PollinAction (https://lifepollinaction.eu/). S.F. was supported by PON “Ricerca e Innovazione” 2014–2020.
Author information
Authors and Affiliations
Contributions
L.L.: Conceptualization, Methodology, Formal analysis, Investigation, Data curation, Writing - Original Draft, Writing - Review & Editing, Visualization. E.F.: Conceptualization, Methodology, Software, Investigation, Writing - Review & Editing, Supervision. D.S.: Methodology, Investigation, Writing - Review & Editing. S.F.: Investigation, Writing - Review & Editing. G.B.: Conceptualization, Methodology, Writing - Review & Editing, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lorenzato, L., Fantinato, E., Sommaggio, D. et al. Pollinator abundance, not the richness, benefits from urban green spaces in intensive agricultural land. Urban Ecosyst 27, 1949–1959 (2024). https://doi.org/10.1007/s11252-024-01565-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-024-01565-7