Abstract
Plant diversity and composition in urban parks is determined by socio-economics, environmental, and ecological drivers. These drivers give rise to urban spaces with unique compositions of flora, consisting of planted and spontaneous species. The present study aimed to determine the contribution of native and exotic species in urban parks of a Latin American city, and to assess the related role of park area, age and socio-economic status. We also evaluated administration type (public or private) and the effect of environmental factors on plant richness. We hypothesized that the composition of park vegetation differs according to urban-rural gradient. To determine flora composition, two transects (100 m long by 1–3 m wide) per park were selected in 49 parks. In each of these, we identified all the vascular plants found (woody and herbs, planted and spontaneous), which were then classified as native or exotic. We conducted ANCOVAs in order to determine the effect of five independent variables and one factor, on native and exotic plant richness. Of 550 recorded species, 16.2% were native and 83.8% exotic. Number of plant species per park varied between 42 and 146. The parks are known urban habitats containing the highest planted and spontaneously occurring exotic diversity in the city of Santiago, contrasting with other types of habitats. Likewise, parks in Santiago are habitats with low levels of planted and spontaneously occurring native diversity, compared to the parks of Europe, America North and Asia. Ours results show that park area and age affected native plant richness, while exotic plant richness was determined only by park age. We rejected all other possible drivers. Finally, according to the low frequency of native species, we propose that Santiago’s parks could be gradually reoriented towards ex-situ conservation of native plants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urban parks can play an important role in biological conservation because they can provide a refuge habitat for native species, including those with compromised conservation status (Tam and Bonebrake 2016; Simmons et al. 2016). Additionally, plants in urban parks offer other important environmental services such as reducing solar radiation under the trees (Kotzen 2003; Oliveira et al. 2011) and urban air temperatures (Bowler et al. 2010), retaining atmospheric particulates (Nowak et al. 2014; Guerrero-Leiva et al. 2016), protecting and stabilizing the soil (Bae and Ryu 2015; Sarah et al. 2015), and protecting water sources (Bryant 2006). Furthermore, they provide basic social and humanitarian services, as well as sanitary, recreational and educational uses (Bedimo-Rung et al. 2005; Muratet et al. 2015). Thus, there is a great interest in recognizing the factors that determine plant diversity in urban parks (Nielsen et al. 2014; Lososová et al. 2011a; Fischer et al. 2016).
In this context, knowledge of native and exotic flora composition in parks could be a first approach to implementing ex-situ conservation of native plant species in urban habitats (Schmitt and Goetz 2010; Bonthoux et al. 2014). This information is also useful for monitoring the presence of exotic plant species, which may potentially be invasive (Pyšek et al. 2009). Urban public parks of Europe and North America have traditionally favored European and Asian plant species for ornamental and landscape design purposes (García 1989; Rosenzweig and Blackmar 1992; Säumel et al. 2010; Abendroth et al. 2012). This also happened in South America, up until the last years of the twentieth century (Collins 1997; Rossetti 2009). In public and private parks, native plant species have rarely been used, due to the influence of the European style (Philippi 1882; Ceballos 1997; Hoffmann 1998; Serra et al. 2002; Rossetti 2009; Alvarado et al. 2013). Thus, an important contrast is observed when comparing South American cities to those of Europe and North America, because the former contains a much higher representation of exotic plant species (Garcia 1997; Méndez 2005; Moro and Farias-Castro 2015; Figueroa et al. 2016).
Plant diversity and composition in urban parks is determined by socio-economics, environmental, and ecological drivers (Smith et al. 2006; Walker et al. 2009; Ramalho and Hobbs 2012). However, the concurrence of these factors has been scarcely and partially studied (Fischer et al. 2016). It is widely recognized that urban parks have flora composed by both planted (i.e. ornamental species) and spontaneously regenerating plants (i.e. mostly weedy species), which may be native or exotic (Mason 2000; McKinney 2008; Walker et al. 2009). Diverse studies have shown that the age, area of parks, along with administration type, and socio-economic and environmental factors (altitude and urban-rural gradient) affect plant diversity in North American and European parks (Gustafson and Gardner 1996; Li et al. 2006; Nielsen et al. 2014; Schwarz et al. 2015). However, it is currently unknown whether these factors jointly determine the richness of native and exotic plants including planted and spontaneously regenerating species of trees, shrubs and herbs. Thus, it is important to establish which factors determine the floristic diversity of urban parks, and then consider the relevance of each factor. Although several studies have focused on advancing this area of research (Nielsen et al. 2014), few analyze the complete composition of plants within parks, incorporating both planted and spontaneously growing species. In effect, information about the relative distribution of spontaneously regenerating native and exotic plants in urban parks can be found in Turner et al. (2005), Lososová et al. (2011a, b), and Fischer et al. (2016). However, studies that consider different floristic components provide a more complete model or scenario regarding the factors that determine plant species composition in urban areas (Li et al. 2006).
This research aimed to determine the representation of native and exotic species in urban parks in Santiago (Chile), in response to factors traditionally considered drivers of diversity in this type of urban habitat. Specifically, we analyzed area, altitude and age of the parks, as well as socio-economic conditions associated with adjacent neighborhoods, and administration type (public or private). By studying 49 parks located within metropolitan Santiago, we expected to record the quantitative importance of those factors on plant species richness and the effects of urbanization through an urban-rural gradient approach. A previous study (Fischer et al. 2016) analyzing only spontaneously regenerating herb diversity in 15 parks of Santiago showed that socio-economic factors and the age of the parks were important determinants of species richness, while urban-rural gradient was not. Therefore, we question whether these results extend to broader spectrum of plant life forms, such as trees, shrubs and perennial herbs. We tested this hypothesis for a wider floristic spectrum, including native and exotic species, as well as spontaneous and planted species, and also studied a greater sample of the parks in Santiago. In addition, we hypothesized that the composition of urban park vegetation differs along an urban-rural gradient, because urbanization may act as an environmental filter that excludes plant species (Vallet et al. 2010; Mouillot et al. 2013).
Methods
Study area
Santiago, the Chilean capital (33° S; 70° W; 728 km2; 500 m.a.s.l.) has a Mediterranean climate type (McPhee et al. 2014). Currently, the metropolitan area of Santiago has about 5.8 million people, with a population density of approximately 93.3 inhabitants per hectare (INE 2005). Additionally, in the late 20th and early 21th centuries, urban growth has spread to surrounding areas, mostly consisting of agricultural lands and smaller remnants of semi-natural vegetation (De Mattos 2003; Romero and Vásquez 2005; Romero et al. 2007; Figueroa et al. 2016).
We chose Santiago as a study site due to its rapid growth, large surface area, and because it is located in a region considered a global biodiversity hotspot (Myers et al. 2000).
The parks
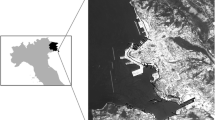
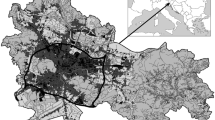
In this study, we defined parks as delineated green spaces embedded in urban areas, surrounded by houses, buildings, roadways, streets, peri-urban infrastructure and/or other types of anthropogenic components, and generally determined by public use (Nielsen et al. 2014). Parks were selected from 35 districts (henceforth called comunas), throughout Santiago, resulting in 49 sampling units (Fig. 1). Separated by walls and fences from the rest of the neighborhood, all these parks represent management units administered independently from the surrounding public infrastructure and activities. Park area varied between approximately a minimum of 6000 to a maximum of 654,000 m2; 50% have an area lesser than or equal to 25,000 m2, with a tree canopy covering at least 25%. Altitude varied between a minimum of 442 m.a.s.l. to a maximum of 853 m.a.s.l. Altogether, 51.3% of the parks are public and administered by municipal, educational or healthcare institutions, while the remaining 46.9% are private, belonging mostly to higher education or recreational institutions (Appendix Table 3).
Area of the 49 parks was determined using tools available on Google Earth Pro (Version 2016), which has recent 3D images of Santiago. With the same software, we determined for each park the distances to the Plaza de Armas or city centre, which is the urban core and corresponds to the historic site where Santiago was founded in the sixteenth century. This approach spatially assorted environmental variation along transects from inner city (urban core) to surrounding (peripheral rural matrix). We obtained the age of each parks by interviewing park administrators. Finally, socio-economic status was determined according to average income index of comuna inhabitants associated with the location of each park, based on the National Socioeconomic Characterization survey (1994 and 2003) (see PNUD 2003 for details).
The park flora
We recorded all vascular plants (trees, shrubs and herbs) present in each park between September 2014 and May 2015. At each park we recorded the plants in two transects (100 m × 1–3 m) randomly placed and separated by an interval of at least 100 m. In each transect, we collected and photographed all available woody plants, herbs, and succulents. For each recognized species we assigned the taxonomic category according to Zuloaga et al. (2009) and Figueroa et al. (2016), and the native and exotic origin according to Marticorena and Quezada (1985), Matthei (1995), and Zuloaga et al. (2009), and biogeographical origin according to Matthei (1995) and Castro et al. (2005). Species were classified as either native flora present in Chile prior to Spanish colonization or exotic flora that arrived afterwards. On the other hand, spontaneous species were defined as those established without direct human intervention, according to Matthei (1995) and Figueroa et al. (2016). Finally, the planted species were those established in Chile within the parks by humans, usually for ornamental purposes, according to Hoffmann (1998) and Alvarado et al. (2013).
Statistical analyses
In order to determine the effects of park area, park age, neighborhood socio-economic status, administration type (public and private), and environmental determinants on the richness of native and exotic plant species we used analyses of covariance (ANCOVA) by XLSTAT (Version 2015). Thus, we used the administration type variable as a factor (public or private) and five covariates (park age, park area, average income index, park altitude and distance to Plaza de Armas). These analyses were determined based on native and exotic richness as dependent variable, in two separate analyses of covariance. To comply with the requirements of homoscedasticity, we transformed the park area, park age, number of native and exotic species by log (x). We did not find the age of four parks, therefore ANCOVAs were done with 45 parks.
Results
Richness, frequency and taxonomic patterns
Our study registered a total of 550 plant species in the 49 parks studied. Of these, 89 (16.2%) species were native and 461 (83.8%) were exotic. The number of plant species per park varied between 42 and 146, and the proportion of native species per park fluctuated between 8% and 33%. Overall, native species had a frequency (100 × number of parks occupied/49) that ranged between 2% and 69%, while exotic species showed a frequency ranging between 2% and 92%. The difference between frequency of exotic and native species was statistically significant (P < 0.01, U = 17.208), showing that together exotic species occupied a greater proportion of parks compared to native species. Among the species with frequency greater than 50%, we recorded three native species (Cryptocary alba, Quillaja saponaria and Schinus molle) and 22 exotic species (Table 1), including Oxalis corniculata, Taraxacum officinale, Cynodon dactylon, Prunus cerasifera, Dichondra repens, Trifolium repens, Poa annua, Phoenix canariensis, Plantago major, and Euphorbia peplus (Table 1). Most of the exotic species found in Santiago parks were of Eurasian origin. On the other hand, 50% of the species of higher frequency were spontaneous plant species and annual herbs (e.g. Euphorbia peplus, Polygonum aviculare, Modiola caroliniana) (Table 1).
We recognized 131 plant families in the 49 parks. Exotic plants represented 115 families and native ones 41. The six most diverse families of native species (Poaceae, Fabaceae, Asteraceae, Salicaceae, Solanaceae and Myrtaceae) accounting to about 45% (40 species) of the native flora recorded in the parks, whereas the six most diverse families of exotic (Asteraceae, Fabaceae, Rosaceae, Poaceae, Pinaceae and Oleaceae) accounted to about 35% (163 species) of the exotic flora.
Factors associated with plant richness in parks
The ANCOVA (Table 2) proved that the number of native species can be explicated significantly by a model representing the linear combination of park age (F = 13.54; P = 0.001) and park area (F = 4.93; P = 0.032). Contrary to our expectation, distance to city centre was not significantly related with exotic species richness. Furthermore, ANCOVA proved that administration type, income, and park elevation were not related to native species richness (Table 2). Consistent with our results, the explicative model showed that bigger and older parks contain more native plant species than smaller and newer parks (Fig. 2a, c).
Relationship between (a) Log10 number of native species and Log10 park age, (b) Log10 number of exotic species and Log10 park area, (c) Log10 number of native species and Log10 park age, and (d) Log10 number of exotic species and Log10 park area in Santiago of Chile. The relationships shown in (a), (b) and (c) are statistically significant (P < 0.05)
On the other hand, ANCOVA (Table 2) proved that number of exotic species can be significantly explicated only by park age (F = 9.031; P = 0.005). In contrast to our hypothesis, distance to city centre was not related to exotic richness. Further, ANCOVA proved that administration type, income, park elevation, and park area were not related to richness of exotic plants species (Table 2). Consequently, the explicative model showed that the older parks contain more exotic species than newer parks (Fig. 2b).
Discussion
The present study showed that the park flora of Santiago was diverse, with 550 vascular plant species recorded across 49 urban parks. This species richness exceeds the diversity of 508 species previously recorded in 200 sampling sites (public square, residential open area, and abandoned successional sites) in public areas in Santiago (Figueroa et al. 2016). Furthermore, spontaneously occurring herbs recorded in this study (156 species) exceed the total of spontaneous herbs (43 species) recorded in the 15 parks studied by Fischer et al. (2016) and in the 41 sites allocated in sidewalks/pavement (95 species) in Santiago studied by Gärtner et al. (2015). Likewise, in our study, the average number of spontaneously occurring plant species (26.3 approx.) is greater than the average number of plant species in central European parks (< 20 species) according to Lososová et al. (2011a). Indeed, the urban parks of Santiago were the richest urban habitats, contrasting with the parks of central Europe (Lososová et al. 2011a). Thus, parks in Santiago constitute suitable habitats for spontaneously occurring plant species.
Lososová et al. (2011b) found that the low number of spontaneously occurring plant species in parks of central Europe is due to the low number of exotic rather than native species. We found that in Santiago there is a very high percentage of exotic plants (83.8%), even higher than parks in North America, Europe and Asia (Nielsen et al. 2014). In a synthesis of seven studies reviewed by Nielsen et al. (2014), it was shown that exotics accounted for between 6.2% and 66.3% (woody species) and between 17.2% and 66.0% (all vascular plants). Together with our results this suggests that parks in Santiago are exposed to a high propagule pressure of exotic plants. Including, this high exotic pressure was repeated in the Santiago public spaces (Figueroa et al. 2016), agricultural fields surrounding Santiago (Figueroa et al. 2013; Martín-Forés et al. 2016), and in semi-natural site of central Chile (Teillier et al. 2010; Figueroa et al. 2011) likely resulting in a high pressure of exotic plants on the urban parks in Santiago.
On the contrary, the scarce representation of planted and spontaneously regenerating native species in Santiago parks (< 20%) suggests that these sites may not be suitable for the establishment of spontaneously occurring native species, or they were not used for the cultivation of native species, or simply excluded as a result of management decisions. However, these reasons are not mutually exclusive and together explain the reduced representation of native species in the parks of Santiago. Also, our results differ from those regarding central European cities, where urban parks are important for the survival of native plant species (Lososová et al. 2011a, b).
The present study found that park area is a determinant of native species richness, but not of exotic species. We suggest that the relationship between native species richness and park area can be explained in a way that is similar to island area (Ames et al. 2012) and fragments of vegetation (Gustafson and Gardner 1996). Few studies have found a significant relationship between plant species richness and park area (Cornelis and Hermy 2004; Li et al. 2006; Nielsen et al. 2014; Fischer et al. 2016), although this subject has been well studied for animals (Nielsen et al. 2014). Bigger parks could offer more resources and surface area for the growth and establishment of native plants. In our study, the bigger parks contained a greater number of spontaneously established native species than the smaller parks (R2 = 0.11, P = 0.02), although the relationship was not significant for cultivated native plants (R2 < 0.01, P > 0.05). Fischer et al. (2016) studied herb layer for 15 parks in Santiago, Chile and found consistent results for wooded habitats, regarding the effect of area on richness on native species spontaneous establishment. Although these authors explain this diversity pattern as an effect of maintenance practices on spontaneous herbs in bigger parks, we also suggest the higher availability of suitable sites for the spontaneous establishment of native plants.
Furthermore, according to our hypothesis, the results also support the idea that richness of both native and exotic plants increases with park age. The time that passed from the initial management or disturbance might explain this relationship in the urban park. Similarly, for natural vegetation, succession increases the richness of plant species with time after disturbance. We propose that the older parks are more stable and contain more plant species than those at an earlier stage (newer parks). Nineteenth-century parks would likely have had long-life ornamental species planted from the outset, and later added plant species according to contemporary landscaping trends (Rossetti 2009). Simultaneously, as time passed, newly planted species came together with species of spontaneously regenerating plants, which could include native long-lived woody plants. For example, some parks in our study had wild, spontaneously regenerating Acacia caven, Cissus striata and Muehlenbeckia hastulata specimens.
It is important to note, contrary to what was expected considering the literature (e.g. Schwarz et al. 2015; Fischer et al. 2016), that the socio-economic factor did not affect the richness of native and exotic plant species in Santiago parks. There are at least two possible explanations for this outcome. Rather than the socio-economic status of the comunas, the richness of species in parks may also be affected by either: 1) the economic resources available to the park administration and/or, 2) the socio-economic status of inhabitants in the immediate park vicinity. Unfortunately, this study lacked the datasets necessary to test those hypotheses, since the information is not public.
Our results did not associate plant richness with altitude or urban-rural gradient, suggesting that the city of Santiago would have a distribution of spatially heterogeneous and dynamic plant species that would not correspond to gradients in linear environments (Ramalho and Hobbs 2012). In the case of spontaneously regenerating plants, it is difficult to establish explanations for these results. One possible explanation is that these trends would be removed by strong propagule pressures of exotic and generalist plants in Santiago (Magura et al. 2010; Nielsen et al. 2014). The propagule pressures for spontaneously regenerating Eurasian plants in regions of central Chile could be so strong that it cancels the effects of other drivers. In addition, this study supports the idea that urban grassland in a growing South America megacity represents a rapidly evolving urban vegetation of Eurasian type with a global distribution (Rapoport and López-Moreno 1987, Gaertner et al. 2009, Ignatieva 2011, Figueroa et al. 2016, Fischer et al. 2016). On the other hand, in the case of ornamental planted species, the absence of a linear gradient for species distribution could be related to cultural-historical causes, which would imply the use of a unique stock of ornamental species for the central Chilean region (Rossetti 2009; Alvarado et al. 2013).
Until recently, few studies had evaluated the drivers that would be determining the plants diversity in urban parks in Latin America. In addition, most works in parks have studied either spontaneously growing species or species established for ornamental purposes. In effect, the results of this study show that historical-cultural drivers in urban areas would reduce the importance of environmental drivers that affect flora distribution, which have been evaluated and recognized in the past (Nielsen et al. 2014). For example, in our study the distribution and richness of exotic plants, constituting more than 80% of the total flora, was explained only by park age and all remaining evaluated environmental drivers were finally rejected. Community-wide studies should explore these dynamic and highly heterogeneous urban scenarios more in-depth (Luck et al. 2009; Goddard et al. 2010). Finally, we found that the relationship between richness of native and exotic plant species was positive and significant in city parks (results not shown). In urban areas larger than 1 ha, this positive tendency is expected (Friedley et al. 2004) and common in successional urban areas, as well as in areas out of equilibrium (Lososová et al. 2011a). Accordingly, this positive relationship shows that Santiago’s parks may still provide resources and habitat for the establishment of more native and exotic plant species. However, it supports the idea that resources and maintenance practices are not used efficiently in order to increase the establishment of native and threatened flora.
In conclusion, city parks contain the highest planted and spontaneously occurring exotic diversity in the city of Santiago, contrasting with other habitats inside of this city, such as residential public areas, squares and abandoned successional sites. Likewise, parks in Santiago are habitats with low levels of planted and spontaneously occurring native diversity, compared to the parks of Europe, America North and Asia. The results of our study showed that the richness of native species is determined by park area and age, and for exotic plants is determined only by park age. Given the low frequency of native species, we propose that the vegetation composition of Santiago’s parks could be gradually reoriented to support the ex-situ conservation of native, endemic and threatened plant species in central Chile (Zerbe et al. 2003), a biogeographical region considered a global biodiversity hotspot (Myers et al. 2000).
References
Abendroth S, Kowarik I, Muller N, von der Lippe M (2012) The green colonial heritage: Woody plants in park of Bandung, Indonesia. Landsc Urban Plan 106:12–22
Alvarado A, Bandini A, Guajardo F (2013) Árboles urbanos de Chile. Guía de reconocimiento. 2d. edn. Corporación Nacional Forestal. Santiago de Chile
Ames S, Pischke K, Schoenfuss N, Snobl Z, Weiher E, Wellnitz T (2012) Biogeography patterns of lichens and tree on island of Boundary Waters Canoe Area Wilderness. Bios 83:145–154
Bae J, Ryu Y (2015) Land use and land cover changes explain spatial and temporal variations of the soil organic carbon stocks in a constructed urban park. Landsc Urban Plan 136:57–67
Bedimo-Rung AL, Mowen AJ, Cohen DA (2005) The significance of parks to physical activity and public health: A conceptual model. Am J Prev Med 28:159–168
Bonthoux S, Brun M, di Pietro F, Greulich S, Bouché-Pillon S (2014) How can wastelands promote biodiversity in cities? A review. Landsc Urban Plan 132:79–88
Bowler DE, Buyung-Ali L, Knight TM, Pullin AS (2010) Urban greening to cool towns and cities: A systematic review of the empirical evidence. Landsc Urban Plan 97:147–155
Bryant MM (2006) Urban landscape conservation and the role of ecological greenways at local and metropolitan scales. Landsc Urban Plan 76:23–44
Castro SA, Figueroa JA, Muñoz-Schick M, Jaksic FM (2005) Minimum residence time, biogeographical origin, and life cycle as determinants of the geographical extent of naturalized plants in continental Chile. Divers Distrib 11:183–191
Ceballos W (1997) Enverdecimiento urbano en Chile. In: Krishnamurthy L, Rente Nascimento J (eds) Área verdes urbanas en Latinoamérica y el Caribe. Banco Interamericano de Desarrollo, Ciudad de México, pp 231–251
Collins CC (1997) Oscar Prager, jardines en el paisaje. ARQ 37:62–68
Cornelis J, Hermy M (2004) Biodiversity relationship in urban and suburban parks in Flanders. Landsc Urban Plan 69:385–401
de Mattos C (2003) Globalización y transformación metropolitana en el caso de Santiago. In: Hidalgo R, Arenas F, Coll JL (eds) Los nuevos modos de gestión de la metropolización. Serie Geolibros. Instituto de Geografía. P. Universidad Católica de Chile, Santiago de Chile, pp 27–55
Figueroa JA, Teillier S, Castro SA (2011) Diversity patterns and composition of native and exotic floras in central Chile. Acta Oecol 37:103–109
Figueroa JA, Teillier S, Carvallo G, Castro SA (2013) Especies de plantas exóticas en los campos agrícolas y en los sitios perturbados en Chile Central. In: Reveco O (ed) Más allá de lo dicho: hallazgos desde la investigación. RIL Editores, Santiago de Chile, pp 369–384
Figueroa JA, Teillier S, Guerrero N, Ray C, Rivano S, Saavedra D, Castro SA (2016) Vascular flora in public space of Santiago, Chile. Gayana Bot 73:85–103
Fischer LK, Rodorff V, von der Lippe M, Kowarik I (2016) Drivers of biodiversity patterns in parks of a growing South American megacity. Urban Ecosyst 19:1231–1249
Friedley JD, Brown RL, Bruno JE (2004) Null models of exotic invasion and scale-dependent patterns of native and exotic species richness. Ecology 86:1848–1855
Gaertner M, Breeyen AD, Hui C, Richardson M (2009) Impacts of alien plants invasions on species richness in Mediterranean-type ecosystems: a meta-analysis. Prog Phys Geogr 33:319–338
García A (1989) El parquet urbano como espacio multifunctional: origen, evolución y principales funciones. Paralelo 37:105–111
Garcia E (1997) Composicion floristica y ecologica de las comunidades ruderales de las calles de la ciudad de La Paz. Ecología en Bolivia 29:1–75
Gärtner E, Rojas G, Castro SA (2015) Compositional patterns of ruderal herbs in Santiago, Chile. Gayana Bot 72:192–202
Goddard MA, Dougill AJ, Benton TG (2010) Scaling up from gardens: biodiversity conservation in urban environments. Trends Ecol Evol 25:90–98
GOOGLE EARTH PRO (Version 2016) (2016) Copyright Google. https://support.google.com/earth/. Accessed 1 Dec 2016
Guerrero-Leiva N, Castro SA, Rubio MA, Ortiz-Calderón C (2016) Retention of atmospheric particulates by three woody ornamental species in Santiago, Chile. Air Soil Poll 227:435
Gustafson EJ, Gardner RH (1996) The effect of landscape heterogeneity on the probability of patch colonization. Ecology 77:94–107
Hoffmann A (1998) El árbol urbano en Chile, 3rd edn. Fundación Claudio Gay, Santiago de Chile
Ignatieva ME (2011) Plant material for urban landscapes in the era of globalization: roots, challenges and innovative solutions. In: Richte M, Weiland U (eds) Applied urban ecology: a global framework. John Wiley & Sons, Chichester, pp 139–151
INE (2005) Chile: ciudades, pueblos, aldeas y caseríos. Instituto Nacional de Estadística. Dirección Nacional, Santiago de Chile
Kotzen B (2003) An investigation of shade under six different tree species of the Negev desert towards theirs potential use for enhancing micro-climatic condition in landscape architectural development. J Arid Environ 2:231–274
Li W, Ouyang Z, Meng X, Wang X (2006) Plant species composition in relation to green cover configuration and function of urban parks in Beijing, China. Ecol Res 21:221–237
Lososová Z, Horsák M, Chytrý M, Čejka T, Danihelka J, Fajmon K, Hájek O, Juřičková L, Kintrová K, Lániková D, Otýková Z, Řehořek V, Tichý L (2011a) Diversity of Central European urban biota: effect of human-made habitat types on plants and snails. J Biogeogr 38:1152–1163
Lososová Z, Chytrý M, Tichý L, Danihelka J, Fajmon K, Hájek O, Kintrová K, Kühn I, Láníková D, Otýpková Z, Řehořek V (2011b) Native and alien floras in urban habitats: a comparison across 32 cities of central Europe. Glob Ecol Biogeogr 21:545–555
Luck GW, Smallbone LT, O’Brien R (2009) Socio-economics and vegetation change in urban ecosystems: patterns in space and time. Ecosystems 12:604–620
Magura T, Lövei GL, Tóthmérész B (2010) Does urbanization decrease diversity in ground beetle (Caarabidae) assemblages? Glob Ecol Biogeogr 19:16–26
Marticorena C, Quezada M (1985) Catálogo de la flora vascular de Chile. Gayana Botánica 42:1–157
Martín-Forés I, Castro I, Acosta-Gallo B, del Pozo A, Sanchez-Jardon L, de Miguel JM, Ovalle C, Casado MA (2016) Alien plant species coexist over time with native ones in Chilean Mediterranean grasslands. J Plant Ecol. https://doi.org/10.1093/jpe/rtw043
Mason CF (2000) Thruhes now largely restricted to the built environment in Eastern England. Diver Distrib 6:189–194
Matthei O (1995) Manual de malezas que crecen en Chile. Alfabeta Impresores, Santiago de Chile
McKinney M (2008) Effects of urbanization on species richness: A review of plants and animals. Urban Ecosyst 11:161–176
McPhee J, Cortés G, Rojas M, García L, Descalzi A, Vargas L (2014) Downscaling Climate Changes for Santiago: What Effects can be Expected? In: Krellenberg K, Hansjürgens B (eds) Climate Adaptation Santiago. Springer-Verlag, Berlin Heidelberg, pp 19–41
Méndez E (2005) Flora y vegetación del centro urbano de Luján de Cuyo. Mendoza (Argentina). Revista de la Facultad de Ciencias Agrarias, Universidad Nacional de Cuyo 37:67–74
Moro MF, Farias-Castro AS (2015) A check list of plant species in the urban forestry of Fortaleza, Brazil: where are the native species in the country of megadiversity? Urban Ecosyst 18:47–71
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR (2013) A functional approach reveals community responses to disturbances. Trends Ecol Evol 28:167–177
Muratet A, Pellegrini P, Dufour AB, Arrif T (2015) Perception and knowledge of plant diversity among urban park users. Landsc. Urban Plan 137:95–106
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nielsen AB, van den Bosch M, Maruthaveeran S, van den Bosch CK (2014) Species richness in urban parks and its drivers: a review of empirical evidence. Urban Ecosyst 17:305–327
Nowak DJ, Hirabayashi S, Bodine A, Greenfield E (2014) Tree and forest effects on air quality and human health in the United States. Environ Pollut 193:119–129
Oliveira S, Andrade H, Vaz T (2011) The cooling effect of green space as a contribution to the mitigation of urban heat: A case study in Lisbon. Build Environ 46:2186–2194
Philippi RA (1882) Catálogo de las plantas cultivadas para el Jardín Botánico de Santiago hasta el 1 de mayo de 1881. Anales de la Universidad de Chile 59:519–581
PNUD (2003) Las trayectorias del desarrollo humano en las comunas de Chile (1994–2003). Temas de Desarrollo Humano Sustentable N° 11. PNUD Chile
Pyšek P, Jarošík V, Pergl J, Randall R, Chytrý M, Kühn I, Tichý L, Danihelka J, Chrtek J, Sádlo J (2009) The global invasion success of Central European plants is related to distribution characteristics in their native range and species traits. Diver Distrib 15:891–903
Ramalho CE, Hobbs RJ (2012) Time for a change: dynamic urban ecology. Trends Ecol Evol 27:179–188
Rapoport EH, López-Moreno I (1987) Aportes a la Ecología Urbana de la Ciudad de México. Limusa, Ciudad de México
Romero H, Vásquez A (2005) Evaluación ambiental del proceso de urbanización de las cuencas del piedemonte andino de Santiago de Chile. Eur Secur 94:97–118
Romero H, Molina M, Moscoso C, Sarricolea P, Smith P, Vásquez A (2007) Caracterización de los cambios de usos y coberturas de suelo causados por la expansión urbana de Santiago, análisis de sus factores explicativos e inferencias ambientales. In: de Mattos C, Hidalgo R (eds) Santiago de Chile: Movilidad espacial y Reconfiguración Metropolitana. Ediciones Pontificia Universidad Católica de Chile, Santiago de Chile, pp 251–270
Rosenzweig R, Blackmar E (1992) The park and the people. A history of Central Park. Cornell University Press, Ithaca
Rossetti F (2009) Arquitectura del paisaje en Chile. Ocho Libros Editores, Santiago de Chile
Sarah P, Zhevelev HM, Oz A (2015) Urban park soil and vegetation. Effects of natural and anthropogenic factors Pedosphere 25:392–404
Säumel I, Kowarik I, Butenschön S (2010) Green traces from past to future: the interplay of culture and ecological process in European historical parks. Acta Hortic 881:933–938
Schmitt JL, Goetz MNB (2010) Species richness of fern and lycophyte in an urban park in the Rio dos Sinos basin, Southern Brazil. Braz J Biol 70:1161–1167
Schwarz K, Fragkias M, Boone CG, Zhou W, McHale M, Morgan J, O’Neil-Dunne J, JP MF, Buckley GL, Childers D, Ogden L, Pincetl S, Pataki D, Whitmer A, Cadenasso ML (2015) Tree grow on money: urban tree canopy cover and environmental justice. PLoS One. https://doi.org/10.1371/journal.pone.0122051
Serra MT, Torres J, Grez I (2002) Breve historia de la introducción en Chile del álamo (Populus nigra L. var. italica (Moench.) Koehne) y el desarrollo de ejemplares siempreverdes. Chloris Chilensis 5 (2) http://www.chlorischile.cl. Accessed 1 Dec 2016
Simmons BL, Hallett RA, Falxa N, Auyeung DSN, Lu JWT (2016) Long-term outcomes of forest restoration in an urban park. Restor Ecol 24:109–118
Smith RM, Thompson K, Hodgson JG, Warren PH, Gaston KJ (2006) Urban domestic gardens (IX): Composition and richness of the vascular plan flora, and implications for native biodiversity. Biol Conserv 129:312–322
Tam KC, Bonebrake TC (2016) Butterfly diversity, habitat and vegetation usage in Hong Kong urban parks. Urban Ecosyst 19:721–733
Teillier S, Figueroa JA, Castro SA (2010) Especies exóticas de la vertiente occidental de la cordillera de la Costa, Provincia de Valparaíso, Chile central. Gayana Bot 67:27–43
Turner K, Lefler L, Freedman B (2005) Plant communities of selected urbanized areas of Halifax, Nova Scotia, Canada. Landsc Urban Plan 71:191–206
Vallet J, Daniel H, Beaujouan V, Rozé F, Pavoine S (2010) Using biological traits to assess how urbanization filters plant species of small woodlands. Appl Veg Sci 13:42–424
Walker JS, Grimm NB, Briggs JM, Gries C, Dugan L (2009) Effects of urbanization on plant species diversity in central Arizona. Front Ecol Environ 7:465–470
XLSTAT Pro (Version 2015) (2015) Copyright Addinsoft. http://www.xlstat.com. Accessed 1 Dec 2016
Zerbe S, Maurer U, Schmitz S, Sukopp H (2003) Biodiversity in Berlin and its potential for nature conservation. Landsc Urban Plan 62:139–148
Zuloaga F, Morrone O, Belgrano M (2009) Catálogo de las Plantas Vasculares del Cono Sur. Versión online. http://www.darwin.edu.ar/Proyectos/FloraArgentina/FA.asp. Accessed 1 Dec 2016
Acknowledgements
This study was funded by an intern project of the Universidad Central de Chile. We thank park managers who facilitated us to collect plants. SA Castro gratefully acknowledges the support of the Center for the Development of Nanoscience and Nanotechnology, CEDENNA FB0807 – Line 6, DICYT 021543CM, and USA 1498.04. In addition, we thank JF Hernández who revised the manuscript translation and J Macaya for help with species determination.
Author information
Authors and Affiliations
Corresponding authors
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Figueroa, J.A., Castro, S.A., Reyes, M. et al. Urban park area and age determine the richness of native and exotic plants in parks of a Latin American city: Santiago as a case study. Urban Ecosyst 21, 645–655 (2018). https://doi.org/10.1007/s11252-018-0743-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-018-0743-0