Abstract
Improving reproductive performance of repeat breeder buffalo cows due to clinical endometritis is crucial in overcoming infertility problems in buffalo cows. The aim of the present study was to use PGF2α and/or gentamicin 10% for treatment of endometritis and to determine biochemical parameters in serum that could be used to diagnose endometritis in buffalo cows. A total of 64 anestrous buffalo cows were assigned into one of five treatment groups: group one (n = 9) buffalo cows were physiological normal cows and served as a control group; group 2 buffalo cows had endometritis, but were not treated (n = 10); group 3 buffalo cows had endometritis and were treated with intrauterine delivery of 100 ml of a 10% gentamicin sulfate solution in three times within 1 week (n = 15); group 4 buffalo cows received two I.M. doses of PGF2α (2 ml Estrumate IM) at the time of corpus luteum dominance (n = 15) (treatment 4 does not make sense as you stated that the cows were in anestrus) on the ovary; group 5 buffalo cows received two IM doses of PGF2α at an 11 day interval and the 10% gentamicin solution as described for group 3. Serum samples were collected from control, pre-treated, and post-treated buffalo cows with endometritis to evaluate the diagnostic biochemical parameters. The days to first estrus (DFE), number of services per conception (S/C), days open (DO), and pregnancy rate (Preg) were the measures for determining reproductive performance for the buffalo cows. The buffalo cows treated by gentamicin and PGF2 exhibited their first estrus earlier than cows in the other four groups of cows. The number of days open for control cows was greater (P < 0.05) than for the other groups and the control cows, as were the number of services per conception (P < 0.05) and pregnancy rate (30%: P < 0.05). The receiver operating characteristic (ROC) curve was used to identify biochemical parameters in serum to predict endometritis. Creatine kinase (CK), aspartate aminotransferase (AST), and concentrations of total bilirubin and immunoglobulins in serum were greater (P < 0.05), while serum albumin values were lower (P < 0.05) in serum of buffalo cows with endometritis. From the ROC analyses, CK was the most predictable biomarker for endometritis with an area under the curve of 0.889, sensitivity of 80%, and specificity of 100% (P < 0.001). In conclusion, the use of gentamicin and PGF2 for treatment of endometritis improves the reproductive performance of buffalo cows, and concentrations of CK serve as an aid for diagnosing endometritis.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endometritis is a common reproductive disorder of buffalo cows and cattle. This problem affects milk yield and decreases reproductive efficiency. In cows, endometritis is defined as inflammation of the uterine endometrium for 21 days or more postpartum (Sheldon et al. 2009, Raliou et al. 2019). Endometritis may be either clinical or cytological endometritis according to the diagnostic criteria. Clinical endometritis is characterized by vaginal purulent or mucopurulent discharge and no systemic changes (LeBlanc et al. 2002, Sheldon et al. 2006, Bonsale et al. 2018, Arango-Sabogal et al. 2019). The diagnosis of cytological endometritis is based on the presence of a higher percentage of polymorphonuclear leukocytes (> 6%) on uterine cytology when compared with clinically healthy cows (Kaufmann et al. 2009, Machado et al., 2012a, b, Bogado Pascottini et al. 2017).
Clinical endometritis is highly prevalent in dairy cows with high milk yields and characterized by decreasing reproductive performance due to lower pregnancy rates, extended post-partum intervals to pregnancy, increased culling rates, and economic losses (Hay et al. 2019) (Helfrich et al. 2020) (Armengol and Fraile 2015) (Bartlett et al. 1995). The incidence of clinical endometritis ranges from 5.0 to 30% and affects about 20% of lactating dairy cows (Moraes et al. 2017) (Machado et al., 2012a, b). The causes of endometritis are multifactorial and more frequent during the peri-parturient period, (2 weeks pre-partum to 3 weeks post-partum). The incidence of uterine bacterial contamination post-partum increases and persists for 2 to 3 weeks. This is an inevitable and naturally occurring as the cervix dilates and the vestibule and vagina relaxes, resulting in the impairment of natural barriers and bacterial defenses after parturition (Pérez-Báez et al. 2019) (Cui et al. 2019) (Miranda-CasoLuengo et al. 2019) (Sheldon et al. 2019).
A number of risk factors play an important role in the prevalence of uterine diseases. These risk factors may be associated with damage to the uterus, metabolic stress, and/or deficits in hygiene (LeBlanc 2020) (Kelly et al. 2020) (Hay et al. 2019) (Giuliodori et al. 2017). The risk factors that are associated with uterine infection are those that likely lead to some trauma of the endometrium due to delivery of stillborn, twin, male and beef-sire calves, dystocia, cesarean section, and retention of the placenta (Gohar et al. 2018).
Other factors include endocrine disorders, deficiencies in selenium, vitamin E, vitamin A, and β-carotene, failure of the calf to suckle, hypocalcemia, and poor hygiene that predispose cows to uterine diseases during the early post-partum period (Borsberry and Dobson 1989, Spears 2000, Venjakob et al. 2018).
To overcome this problem, clinical endometritis in buffaloes was managed by the hormonal treatment either PGF2α (Lefebvre and Stock 2012) or PGF2α and oxytetracycline (Azawi et al. 2008, Gohar et al. 2018, Nehru et al. 2019), intrauterine infusion of ceftiofur (Nehru et al. 2019), intrauterine administration of mannose or bacteriophage (Machado et al., 2012a, b), intrauterine infusion of honey (M, 2019), and intrauterine infusion of oxytetracycline (Gohar et al. 2018).
In cows, the degree of endometritis is correlated with increases in creatine kinase (CK) and aspartate aminotransferase (AST) activities in serum (Sattler and Fürll 2004). There is also a positive relationship between uterine diseases and concentrations of albumin and blood urea nitrogen (BUN) (Walsh et al. 2007); (Raboisson et al. 2014, Ghanem et al. 2016). On other hand, Gahlot et al. 2018 (Gahlot et al. 2018) found that the concentrations of urea and bilirubin were altered in the uterine fluid of buffaloes with subclinical endometritis. The use of gentamicin for treatment of endometritis in buffaloes is not well documented. Therefore, the aim of the current study was to investigate changes in biochemical profiles in serum from buffalo cows with endometritis and evaluate the effects of treatment of cows with endometritis with PGF2α and/or intrauterine infusion of gentamicin on postpartum reproductive performance.
Material and methods
Animals and data collection
Animal experiments described in this article were conducted in accordance with the Guiding Principles for the Care and Use of Research Animals Veterinary Medicine Faculty, Mansoura University, Egypt (R. 35). Sixty-four buffalo cows, 3–7 years old and experiencing 1–5 parities were used in this study. The buffalo cows were in the village system of the Dakahlia Governorate from January 2016 until December 2018. The cows were fed a balanced ration consisting of ad libitum Berseem in winter and Darawa in summer and about 20 kg wheat straw and 5–8 kg concentrate mixture and milked two times daily. All animals were vaccinated and dewormed against the common infectious and parasitic diseases. Buffalo cows were admitted to our veterinary teaching clinic, Veterinary Medicine Faculty, Mansoura University, Egypt suffering from repeat breeder syndrome.
Estimation of serum biochemical parameters
The activity of CK and aspartate aminotransferase (AST) was measured by using Cobas Test Reagent, USA kits and Vitro Scientist, Germany kits, respectively. Total proteins and albumin were assayed using Stanbio Laboratory, USA kits. Concentrations of immunoglobulins were calculated by subtracting serum albumin from total protein. Serum urea and bilirubin kits were obtained from Diamond Company, Egypt. All assays were performed using a spectrophotometer (BM Co. Germany, 5010) according to the manufacturer’s instructions.
Diagnosis of endometritis and animal grouping
The experimental animals experienced normal parturition, but had a history of 3–4 inseminations not resulting in pregnancy indicative of repeat breeder syndrome after natural service. The cows also exhibited watery and/or mucopurulent vaginal discharges. Ultrasound imaging was used for further diagnosis of endometritis. The detection of intrauterine fluid using the SonoScape A5 ultrasound machine® (Shenzhen, Guangdong, China) is considered reliable for diagnosing endometritis (Kaya et al. 2016, Kelly et al. 2020).
The examination of buffaloes that came to our veterinary clinic was done when cows were in estrus. At that time, a 10 ml blood sample was obtained from the jugular vein. Blood samples were centrifuged at 3000 rpm for 20 min and the serum was stored at − 20 °C until analyzed for biochemical markers. Failure to conceive was diagnosed if the cow was not pregnant based on rectal examination and ultrasound imaging or if a subsequent breeding was required 17 to 24 days post-breeding. The buffalo cows were allocated into five groups and received one of the following treatments:
-
Group 1(control) cows with a normal parturition and physiologically normal uterus (n = 9);
-
Group 2 (endometritis control) cows with endometritis and did not receive any treatment (n = 10); group 3 cows received an intrauterine infusion of 10% gentamicin sulfate® 10% (Apigent, Amoun Pharmaceutical Co. El-Obour City, Cairo, Egypt) solution in 100 ml physiological saline every 3 days from the onset of estrus according to Kumar et al. (2019) (n = 15).
-
Group 4 cows received two intramuscular injections of 2 ml Estrumate® (500 mcg cloprostenol: Schering Plough, USA) 11 days apart in the presence of a corpus luteum (n = 15); and group 5 cows received intrauterine infusions of a 10% gentamicin sulfate solution as in group 3 and 2 injections of Estrumate at an 11 day interval (n = 15).
Buffalo cows were bred at the second estrus after evaluation of treatment outcomes based on rectal examination of the ovaries and uteri, the appearance of clear, translucent vaginal discharge, or no mucus 8–16 h after onset of estrus and normal reproductive organs based on ultrasound imaging. The dependent variables for reproductive performance were interval between treatment and first estrus, calving to conception (DO, the number of days between calving and conception), the number of services per conception (SC, defined as the number of services that a cow required to conceive) and pregnancy rate (Elmetwally et al. 2016, Elmetwally 2018). Pregnancy was diagnosed by rectal examination at 2 months post-breeding according to Gohar et al. 2018 (Gohar et al. 2018).
Statistical analysis
Normality of quantitative parameters (days to first estrus, days open, number of services per conception) was assessed using normal probability plots and the KolmogorovSmirnov test generated with the UNIVARIATE procedure of SAS according to Elmetwally et al. (Elmetwally et al. 2018, 2019). All data are reported as means ± SEM. Receiver operating characteristic (ROC) analysis was conducted using MedCalc software (version 16.4.3, Ostend, Belgium). Statistical analyses were done using SAS® (version 9.2, SAS Institute, Cary, NC, USA). For all analyses, P ≤ 0.05 was defined as significant.
Results
Effects of treatment of cows with endometritis with the combination of gentamicin and PGF2α treatments compared with control cows on biochemical changes in serum
Results for biochemical parameters in serum are summarized in Table 1. CK activity was greater (P < 0.05) in cows with endometritis prior to treatment (92.80 ± 2.13 U/l: P < 0.05) as compared with values post-treatment (60.80 ± 3.76 U/l) and values for control cows (54.20 ± 3.12 U/l). AST activity in serum was greater (P < 0.05) for cows with endometritis prior to treatment (108.01 ± 2.21 U/l) compared with values post-treatment (90.20 ± 2.67 U/l) and when compared with values for control cows (65.20 ± 2.22 U/l). Concentrations of albumin in cows with endometritis were greater prior to treatment and decreased (P ≤ 0.05) to (2.71 ± 0.17 g/dl) compared with post-treatment values (3.28 ± 0.05 g/dl) and values for control cows (3.31 ± 0.06 g/dl). Concentrations of immunoglobulins in serum were greater (P ≤ 0.05) in cows with endometritis prior to treatment (4.09 ± 0.22 g/dl) in contrast to values post-treatment (3.36 ± 0.15 g/dl) and in control cows (3.21 ± 0.07 g/dl). However, total protein and concentrations of urea in serum of cows with endometritis were not affected by treatment and were not different from values for control cows (P > 0.05). Prior to treatment for endometritis, cows had greater P < 0.05) concentrations of bilirubin (0.74 ± 0.02 mg/dl) compared with post-treatment values (0.62 ± 0.03 mg/dl) and values for control cows (0.61 ± 0.01 mg/dl).
ROC curve analysis
The area under curve (AUC) was greater for cows with endometritis for CK (Fig. 2A: AUC = 0.889, P < 0.001), urea (Fig. 2B: AUC = 0.811, P = 0.005), and total bilirubin (Fig. 2D: AUC = 0.783, P = 0.032), followed by albumin (Fig. 3A: AUC = 0.778, P = 0.018), total protein (Fig. 3B: AUC = 0.722, P = 0.075), globulin (Fig. 3C: AUC = 0.767, P = 0.026), and AST (Fig. 2C: AUC = 0.644, P = 0.341). The sensitivity (Se) %, specificity (Sp) %, and 95% confidence intervals (CI) were greater for CK (Se: 80%, Sp: 100%, CI: 0.791–0.985), total bilirubin (Se: 70%, Sp: 100%, CI: 0.538–0.936), albumin (Se: 60%, Sp: 100%, CI: 0.532–0.933), globulin (Se: 80%, Sp: 100%, CI: 00.520–0.927), and AST (Se: 60%, Sp: 100%, CI: 0.396–0.846), while urea (Se: 90%, Sp: 77.78%, CI: 0.568–0.951) and total protein (Se: 70%, Sp: 77.76%, CI: 0.473–0.899) had lower specificities % and 95% confidence intervals.
Overall, the AUC of 0.889 for CK (Se: 80%, Sp: 100%, CI: 0.791–0.985) indicates that it could serve as a valuable biomarker for differentiating endometritis-affected from healthy buffalo cows (P < 0.001). As well, the concentrations of total bilirubin, albumin, and globulin in serum, had high sensitivity and specificity percentages that may be used to support values for CK in the diagnosis of uterine tissue diseases including endometritis.
Effects of gentamicin and PGF2α treatments on postpartum reproductive performance in buffalo cows
-
a.
Days to the first estrous
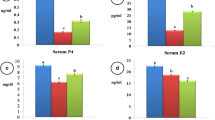
The buffalo cows treated with gentamicin and PGF2 exhibited fewer days to the first estrus (38.4 ± 1.35, P < 0.05) than for other treatment groups and control cows (gentamicin: 49.86 ± 1.19; PGF2: 51 ± 2.9, control: 51.55 ± 1.44 and endometritis control: 84.33 ± 1.65: Fig. 1A).
-
b.
Days open
Effect of different treatments on postpartum reproductive performance in buffalo cows with endometritis. A Days to the first estrus; B days open; C number of services per conception; D pregnancy rate. Significant effects are indicated by different superscript letters (P < 0.05). All quantitative data are presented as means and SEM
The number of days open for endometritis control buffaloes was greater (153.44 ± 2.98: P < 0.05) than for the other treatment groups (control: 77.22 ± 7.43; gentamicin: 126 ± 2.42; PGF2: 127.33 ± 2.9 and gentamicin + PGF2: 120.33 ± 2.69: Fig. 1B)
-
c.
Services per conception
The buffalo cows in the control endometritis group required a greater number of services per conception (4.3 ± 0.31: P < 0.05) than control cows and cows in the other treatment groups (control: 2.22 ± 0.25; gentamicin: 2.06 ± 0.18; PGF2: 2.26 ± 0.23 and gentamicin + PGF2: 1.93 ± 0.20: Fig. 1C).
-
d.
Pregnancy rate
The pregnancy rates were greater (P < 0.05) for buffalo cows following treatment with gentamicin only (73%) and gentamicin + PGF2 (80%) compared with control cows (66%) and cows receiving PGF2 alone (66%) (Fig. 1D). The lowest (P < 0.05) pregnancy rate was for buffalo cows with endometritis that were not treated (30%) (Figs. 2, 3).
Discussion
Endometritis is one of the most common reproductive disorders in buffalo cows and Bos taurus cows. Endometritis had a significant impact to decrease milk yield and reproductive efficiency including the interval between treatment and conception, days open, number of service per conception, pregnancy rate, culling rate, and economic losses (Rhoads et al. 2004, Sheldon et al. 2006).
Results of the current study showed that CK and AST are useful biomarkers for detection of endometritis; however, CK is much more sensitive than AST due to differences in the activities of these enzymes in uterine tissue (Sattler and Fürll 2004). Results of the present study are the first to reveal significant increases in CK and AST activities in serum of buffalo cows with endometritis that likely reflect the inflammatory status of the uterine endometrium (Azawi et al. 2008).
Albumin is considered a negative acute-phase protein with subnormal concentrations indicating impaired liver function due to increases in synthesis of acute-phase proteins (Bertoni et al. 2008). Results of the present study indicated significant changes in total protein in serum of cows with endometritis. Interestingly, concentrations of albumin decreased while concentrations of globulins increased in the serum of the buffalo cows with endometritis prior to treatment compared with values post-treatment and values for control cows. Similarly, lower concentrations of albumin and higher globulin levels were reported for serum from pasture-grazed cows with endometritis (Green et al. 2009, Burke et al. 2010, Raliou et al. 2019). The reduction in albumin may be a risk factor for fat infiltration of the liver as reported in cases of uterine diseases (Zerbe et al. 2000). The higher level of serum globulin may be attributed to the elevation in the serum acute-phase proteins such as amyloid A and haptoglobin in cows with subclinical uterine inflammation (Ahmadi et al. 2018). Moreover, greater concentrations of globulins in serum reflect increased activity of immunocompetent cells in the uterus, primarily in the circumferential blood (Brodzki et al. 2018).
There are negative impacts of increased levels of bilirubin and urea in serum on the uterine environment and the prevalence of endometritis (Benedet et al. 2019, Gruber and Mansfeld 2019). In the present study, the concentrations of urea in serum changed significantly and bilirubin was elevated prior to treatment of buffalo cows prior to treatment as compared with values post-treatment and for control cows. A previous study reported insignificant difference in urea serum from cows with and without endometritis (Burke et al. 2010). Concentrations of total bilirubin concentration were reportedly greater in buffalo cows with subclinical endometritis (Gahlot et al. 2018, Bogado Pascottini et al. 2020, LeBlanc 2020) and this may be related to alterations in liver function due to systemic toxins resulting from postpartum diseases that may result in hepatic failure (LeBlanc 2020).
In the current study, the metabolic parameters including CK, AST, total bilirubin, and globulin increased significantly while albumin decreased in buffalo cows with endometritis. The activity of CK may also increase in serum, skeletal muscle, and cardiac muscle in various diseases including diseases of the uterus (Sattler and Fürll 2004). Furthermore, CK, AST, and bilirubin increase in response to liver damage and Cholestasis (McSherry et al. 1984, Cebra et al. 1997). The decline in concentrations of albumin and increases in concentrations globulins was reported for cows suffering from metritis, mastitis, and other diseases (Overton et al. 2017).
Environmental variables and nutrition may also influence the biochemical parameters in serum of dairy cows. (Hanuš et al. 2018) revealed that values for calcium, AST, ALT, triglycerides, and cholesterol in serum of dairy cows differ among seasons especially during the first and third lactations (Hanuš et al. 2018).
In the current study, the Roc curve analysis was used for the first time to identify biochemical markers that are more specific and sensitive to endometritis in buffalo cows. The ROC analysis confirmed that CK (AUC: 0.889, Se: 80%, Sp: 100%, CI: 0.791–0.985) had greater sensitivity and specificity for predicting endometritis. Then, total bilirubin (AUC = 0.783, Se: 70%, Sp: 100%, CI: 0.538–0.936), albumin (AUC = 0.778, Se: 60%, Sp: 100%, CI: 0.532–0.933), and globulin (AUC = 0.767, Se: 80%, Sp: 100%, CI: 00.520–0.927) were less sensitive and specific. Although AST was significantly elevated in endometritis, the roc curve analysis showed it to be less sensitive (AUC = 0.644, Se: 60%, Sp: 100%, CI: 0.396–0.846). These variations may be correlated with variations in activities of these enzymes in the uterine tissue (Sattler and Fürll 2004).
In the current study, the buffalo cows treated with a combination of gentamicin and PGF2 had the fewest days to first estrus and higher pregnancy rates (80%) compared with cows treated with two injections of PGF2α (66%)or not treated (33%). Previous studies reported similar pregnancy rates (65.38%) after using PGF2α in treatment of clinical endometritis. Previous reports (Jeremejeva et al. 2012, López-Gatius et al. 2015, Voelz et al. 2018) indicated that the systemic administration of PGF2α significantly improved the pregnancy rate of cows with clinical endometritis, perhaps due to luteolysis of an active corpus luteum in cyclic cows that decreased concentrations of progesterone in serum. Furthermore, the estrogen in serum and increases in contractions of the myometrium are favorable for elimination of uterine infection (LeBlanc et al. 2002) (Sheldon et al. 2006) (Walsh et al. 2007) (Bogado Pascottini et al. 2020) (LeBlanc 2020). In addition, PGF2α causes relaxation of the cervix and expulsion of uterine contents (Hirsbrunner et al. 2003, Pascottini and LeBlanc 2020). Exogenous PGF2α may also enhance immune functions or increase uterine motility that help the uterus to resolve infections in animals that lack active corpus lutea (Hirsbrunner et al. 2003). Regression of CL allows the development of dominant follicle on the ovary and induces ovulation within 72–96 h after PGF2α administration so that the uterus becomes more resistant to infections under the influence of estrogen (Wulster-Radcliffe et al. 2003). Hence, the use of PGF2α may alter the endocrine state of cows to increase microbial resistance inside the uterus and enhance body defense mechanism including greater phagocytic activity and inflammatory responses in the endometrium (Kaufmann et al. 2010). In contrast, some studies found that PGF2α treatment is not effective in the treatment of endometritis (Choudhary et al. 2016, Mandhwani et al. 2017). The difference findings in different studies may be attributed to the hormonal imbalance at the time of inducing estrus as PGF2α controls the estrous cycle via shortening the luteal phase (Wenzinger and Bleul 2012, Nowicki et al. 2017).
Results of the present study clearly indicate that gentamicin is an effective treatment for endometritis in buffalo cows. The reproductive performance of cows was improved after intrauterine infusion of 10% gentamicin and intramuscular injections of PGF2α and was similar to that for buffalo cows without endometritis. Previous studies found similar effects following intrauterine infusion of 10% gentamicin in repeat breeder cows (LeBlanc et al. 2002, Tison et al. 2017). The current result for days open after intrauterine infusion of gentamicin was similar to that reported previously of 121 days (Daniels et al. 1976, Ensley and Hennessey 1979). The effects of gentamicin are attributed to the bactericidal effects through inhibition of synthesis of bacterial proteins (Mingeot-Leclercq et al. 1999, Khair et al. 2018). Gentamicin is one of the most effective intrauterine antibiotics for treatments of chronic or subclinical uterine infections in cattle (Ocal et al. 2004).
The results from the present study of reproductive performance in buffalo cows treated with gentamicin and PGF2α are similar to those reported previously for treatment of clinical endometritis (Jeremejeva 2015). In addition, similar results were obtained following the intrauterine infusion of oxytetracycline and a PGF2α injection for treatment of clinical endometritis (Sheldon and Noakes 1998, Ahmadi et al. 2018). Intrauterine infusion of cephapirin or systemic administration of PGF2α has improved pregnancy rates of cows with clinical endometritis (Kaufmann et al. 2010, Tison et al. 2017, Brodzki et al. 2018). The improvement may be attributed to luteolysis of the corpus luteum after PGF2α injection in cyclic cows resulting in lower progesterone and higher estrogen in serum and increases in myometrial contractions (Copelin et al. 1988, Stevenson and Phatak 2010). These events are favorable for clearance of uterine infections (Nehru et al. 2019). Regression of the corpus luteum allows growth of dominant follicles which results in estrus and ovulation 72–96 h after PGF2α administration. Under the influence of estrogen, the uterus becomes more resistant to infections (Wulster-Radcliffe et al. 2003). In addition, there are positive effects of PGF2α to enhance of phagocytosis, immune function, chemotaxis, cell-mediated cytotoxicity, and lymphocyte function of the uterus due to leukotriene B4 (Lewis 2003, Lefebvre and Stock 2012).
Altogether, the biochemical changes (CK, AST, total bilirubin, and albumin concentrations) in serum serve as an aid for diagnosing endometritis in the buffalo with more specificity for CK. Furthermore, Gentamicin and/or PGF2α tend to improve reproductive performance and are effective for treatment of clinical endometritis in buffalo cows. Hence, producers should pay close attention to prevention and control programs for endometritis in postpartum buffalo cows from both milk production and reproductive efficiency points of view, but also to decrease culling of high producing cows.
References
Ahmadi, M.R., A. Mogheiseh, and S. Nazifi. 2018. Changes in biomarkers serum amyloid A and haptoglobin following treatment of endometritis in dairy cows. Comparative clinical pathology, 27(6), 1659–65.
Arango-Sabogal, J.C., J. Dubuc, C. Krug, J. Denis-Robichaud, and S. Dufour. 2019. Accuracy of leukocyte esterase test, endometrial cytology and vaginal discharge score for diagnosing postpartum reproductive tract health status in dairy cows at the moment of sampling, using a latent class model fit within a Bayesian framework. Preventive Veterinary Medicine, 162, 1–10.
Armengol, R. and L. Fraile. 2015. Comparison of two treatment strategies for cows with metritis in high-risk lactating dairy cows. Theriogenology, 83(8), 1344–51.
Azawi, O.I., S.N. Omran, and J.J. Hadad. 2008. A study of endometritis causing repeat breeding of cycling Iraqi buffalo cows. Reproduction in domestic animals = Zuchthygiene, 43(6), 735–43.
Bartlett, P.C., M. Kopcha, P.H. Coe, N.K. Ames, P.L. Ruegg, and R.J. Erskine. 1995. Economic comparison of the pyloro-omentopexy vs the roll-and-toggle procedure for treatment of left displacement of the abomasum in dairy cattle. Journal of the American Veterinary Medical Association, 206(8), 1156–62.
Benedet, A., C.L. Manuelian, A. Zidi, M. Penasa, and M. De Marchi. 2019. Invited review: β-hydroxybutyrate concentration in blood and milk and its associations with cow performance. Animal : an international journal of animal bioscience, 13(8), 1676–89.
Bertoni, G., E. Trevisi, X. Han, and M. Bionaz. 2008. Effects of inflammatory conditions on liver activity in puerperium period and consequences for performance in dairy cows. Journal of Dairy Science, 91(9), 3300–10.
Bogado Pascottini, O., M. Hostens, P. Sys, P. Vercauteren, and G. Opsomer. 2017. Cytological endometritis at artificial insemination in dairy cows: Prevalence and effect on pregnancy outcome. Journal of Dairy Science, 100(1), 588–97.
Bogado Pascottini, O., M. Probo, S.J. LeBlanc, G. Opsomer, and M. Hostens. 2020. Assessment of associations between transition diseases and reproductive performance of dairy cows using survival analysis and decision tree algorithms. Preventive Veterinary Medicine, 176, 104908.
Bonsale, R., R. Seyed Sharifi, E. Dirandeh, N. Hedayat, A. Mojtahedin, M. Ghorbanalinia, and A. Abolghasemi. 2018. Endocannabinoids as endometrial inflammatory markers in lactating Holstein cows. Reproduction in domestic animals = Zuchthygiene, 53(3), 769–75.
Borsberry, S. and H. Dobson. 1989. Periparturient diseases and their effect on reproductive performance in five dairy herds. The Veterinary Record, 124(9), 217–9.
Brodzki, P., U. Lisiecka, A. Brodzki, R. Pyz-Łukasik, and L. Krakowski. 2018. Phagocytic and oxidative burst activity of phagocytic cells in peripheral blood and uterine washings in cows with clinical endometritis before and after intrauterine use of cephapirin and methisoprinol. Animal science journal = Nihon chikusan Gakkaiho, 89(10), 1389–97.
Burke, C.R., S. Meier, S. McDougall, C. Compton, M. Mitchell, and J.R. Roche. 2010. Relationships between endometritis and metabolic state during the transition period in pasture-grazed dairy cows. Journal of Dairy Science, 93(11), 5363–73.
Cebra, C.K., F.B. Garry, D.M. Getzy, and M.J. Fettman. 1997. Hepatic lipidosis in anorectic, lactating holstein cattle: a retrospective study of serum biochemical abnormalities. Journal of Veterinary Internal Medicine, 11(4), 231–7.
Choudhary, K.K., K.M. Kavya, A. Jerome, and R.K. Sharma. 2016. Advances in reproductive biotechnologies. Veterinary world, 9(4), 388–95.
Copelin, J.P., M.F. Smith, H.A. Garverick, R.S. Youngquist, W.R. McVey, and E.K. Inskeep. 1988. Responsiveness of bovine corpora lutea to prostaglandin F2 alpha: comparison of corpora lutea anticipated to have short or normal lifespans. Journal of Animal Science, 66(5), 1236–45.
Cui, L., H. Wang, Y. Ding, J. Li, and J. Li. 2019. Changes in the blood routine, biochemical indexes and the pro-inflammatory cytokine expressions of peripheral leukocytes in postpartum dairy cows with metritis. BMC veterinary research, 15(1), 157.
Daniels, W.H., D.A. Morrow, B.W. Pickett, and L. Ball. 1976. Effects of intrauterine infusion of gentamicin sulfate on bovine fertility. Theriogenology, 6(1), 61–8.
Elmetwally, M.A. 2018. Uterine involution and ovarian activity in postpartum holstein dairy cows. A review. Journal of Veterinary Healthcare, 1(4), 29–40.
Elmetwally, M. A., Lenis, Y., Tang, W., Wu, G., and Bazer, F. W. 2018. Effects of catecholamines on secretion of interferon tau and expression of genes for synthesis of polyamines and apoptosis by ovine trophectoderm. Biology of reproduction, 99(3), 611–628. https://doi.org/10.1093/biolre/ioy085
Elmetwally, M. A., Halawa, A. A., Lenis, Y. Y., Tang, W., Wu, G., and Bazer, F. W. 2019. Effects of BPA on expression of apoptotic genes and migration of ovine trophectoderm (oTr1) cells during the peri-implantation period of pregnancy. Reproductive toxicology (Elmsford, N.Y.), 83, 73–79. https://doi.org/10.1016/j.reprotox.2018.12.001
Elmetwally, M., A. Montaser, N. Elsadany, W. Bedir, M. Hussein, and S. Zaabel. 2016. Effects of parity on postpartum fertility parameters in holstein dairy cows. IOSR Journal of Nursing and Health Science, 09(08), 91–9.
Ensley, L.E. and P.W. Hennessey. 1979. Effects of a single intrauterine infusion of gentamicin or Utonex suspension on conception in normal and infected cows. Veterinary Medicine, Small Animal Clinician, 74(6), 864–70.
Gahlot, S.C., S. Kumar, A. Kumaresan, S. Vairamuthu, K.K. Saraf, L. Sreela, R.K. Baithalu, S.S. Lathwal, and T.K. Mohanty. 2018. Biochemical analysis of uterine fluid for identification of indicators for subclinical endometritis in the water buffalo (Bubalus bubalis). Reproduction in domestic animals = Zuchthygiene, 53(1), 48–53.
Ghanem, M.E., E. Tezuka, K. Sasaki, M. Takahashi, N. Yamagishi, Y. Izaike, and T. Osawa. 2016. Correlation of blood metabolite concentrations and body condition scores with persistent postpartum uterine bacterial infection in dairy cows. The Journal of reproduction and development, 62(5), 457–63.
Giuliodori, M.J., M. Magnasco, R.P. Magnasco, I.M. Lacau-Mengido, and R.L. de la Sota. 2017. Purulent vaginal discharge in grazing dairy cows: Risk factors, reproductive performance, and prostaglandin F2α treatment. Journal of Dairy Science, 100(5), 3805–15.
Gohar, M.A., M.A. Elmetwally, A. Montaser, and S.M. Zaabel. 2018. Effect of Oxytetracycline Treatment on Postpartum Reproductive Performance in Dairy Buffalo-Cows with Retained Placenta in Egypt. Journal of Veterinary Healthcare, 1(3), 45–53.
Green, M.P., A.M. Ledgard, M.C. Berg, A.J. Peterson, and P.J. Back. 2009. Prevalence and identification of systemic markers of sub-clinical endometritis in postpartum dairy cows. Proceedings of the New Zealand Society of Animal Production.
Gruber, S. and R. Mansfeld. 2019. Herd health monitoring in dairy farms - discover metabolic diseases. An overview. Tierarztliche Praxis. Ausgabe G, Grosstiere/Nutztiere, 47(4), 246–55.
Hanuš, O., E. Samková, L. Křížová, L. Hasoňová, and R. Kala. 2018. Role of Fatty Acids in Milk Fat and the Influence of Selected Factors on Their Variability-A Review. Molecules (Basel, Switzerland), 23(7).
Hay, M.J., A.J. Gunn, A. Abuelo, and V.J. Brookes. 2019. The effect of abnormal reproductive tract discharge on the calving to conception interval of dairy cows. Frontiers in Veterinary Science, 6, 374.
Helfrich, A.L., H.-D. Reichenbach, M.M. Meyerholz, H.-A. Schoon, G.J. Arnold, T. Fröhlich, F. Weber, and H. Zerbe. 2020. Novel sampling procedure to characterize bovine subclinical endometritis by uterine secretions and tissue. Theriogenology, 141, 186–96.
Hirsbrunner, G., B. Knutti, U. Küpfer, H. Burkhardt, and A. Steiner. 2003. Effect of prostaglandin E2, DL-cloprostenol, and prostaglandin E2 in combination with D-cloprostenol on uterine motility during diestrus in experimental cows. Animal Reproduction Science, 79(1–2), 17–32.
Jeremejeva, J. 2015. Prostaglandin F2α and Parenteral Antibiotics as a Treatment of Postpartum Metritis and Endometritis, and Possible Relation of Acute Phase Proteins With Subsequent Fertility in Dairy Cows.
Jeremejeva, J., T. Orro, A. Waldmann, and K. Kask. 2012. Treatment of dairy cows with PGF2α or NSAID, in combination with antibiotics, in cases of postpartum uterine inflammation. Acta veterinaria Scandinavica, 54, 45.
Kaufmann, T.B., M. Drillich, B.A. Tenhagen, D. Forderung, and W. Heuwieser. 2009. Prevalence of bovine subclinical endometritis 4h after insemination and its effects on first service conception rate. Theriogenology, 71(2), 385–91.
Kaufmann, T.B., S. Westermann, M. Drillich, J. Plöntzke, and W. Heuwieser. 2010. Systemic antibiotic treatment of clinical endometritis in dairy cows with ceftiofur or two doses of cloprostenol in a 14-d interval. Animal Reproduction Science, 121(1–2), 55–62.
Kaya, S., O. Merhan, C. Kacar, A. Colak, and K. Bozukluhan. 2016. Determination of ceruloplasmin, some other acute phase proteins, and biochemical parameters in cows with endometritis. Veterinary world, 9(10), 1056–62.
Kelly, E., C.G. McAloon, L. O’Grady, M. Duane, J.R. Somers, and M.E. Beltman. 2020. Cow-level risk factors for reproductive tract disease diagnosed by 2 methods in pasture-grazed dairy cattle in Ireland. Journal of Dairy Science, 103(1), 737–49.
Khair, A., M. Asaduzzaman, Z. Sultana, A.K. Talukder, Z.C. Das, M.G.S. Alam, and M. Shamsuddin. 2018. Economic benefit in repeat breeder cows using intrauterine infusion of penicillin and estrus synchronization followed by timed artificial insemination. Journal of Advanced Veterinary and Animal Research, 5(4), 454–8.
Kumar, D., S. ish, and G.N. Purohit. 2019. A discussion on risk factors, therapeutic approach of endometritis and metritis in cattle. International journal of current microbiology and applied sciences, 8(05), 403–21.
LeBlanc, S.J. 2020. Review: Relationships between metabolism and neutrophil function in dairy cows in the peripartum period. Animal : an international journal of animal bioscience, 14(S1), s44–54.
LeBlanc, S.J., T.F. Duffield, K.E. Leslie, K.G. Bateman, G.P. Keefe, J.S. Walton, and W.H. Johnson. 2002. The effect of treatment of clinical endometritis on reproductive performance in dairy cows. Journal of Dairy Science, 85(9), 2237–49.
Lefebvre, R.C. and A.E. Stock. 2012. Therapeutic efficiency of antibiotics and prostaglandin F2α in postpartum dairy cows with clinical endometritis: an evidence-based evaluation. The Veterinary clinics of North America. Food animal practice, 28(1), 79–96, ix.
Lewis, G.S. 2003. Steroidal regulation of uterine resistance to bacterial infection in livestock. Reproductive Biology and Endocrinology, 1, 117.
López-Gatius, F., I. López-Helguera, F. De Rensis, and I. Garcia-Ispierto. 2015. Effects of different five-day progesterone-based synchronization protocols on the estrous response and follicular/luteal dynamics in dairy cows. The Journal of reproduction and development, 61(5), 465–71.
Machado, V.S., M.L.S. Bicalho, R.V. Pereira, L.S. Caixeta, J.H.J. Bittar, G. Oikonomou, R.O. Gilbert, and R.C. Bicalho. 2012a. The effect of intrauterine administration of mannose or bacteriophage on uterine health and fertility of dairy cows with special focus on Escherichia coli and Arcanobacterium pyogenes. Journal of Dairy Science, 95(6), 3100–9.
Machado, V.S., W.A. Knauer, M.L.S. Bicalho, G. Oikonomou, R.O. Gilbert, and R.C. Bicalho. 2012b. A novel diagnostic technique to determine uterine health of Holstein cows at 35 days postpartum. Journal of Dairy Science, 95(3), 1349–57.
Mandhwani, R., A. Bhardwaz, S. Kumar, M. Shivhare, and R. Aich. 2017. Insights into bovine endometritis with special reference to phytotherapy. Veterinary world, 10(12), 1529–32.
McSherry, B.J., J.H. Lumsden, V.E. Valli, and J.D. Baird. 1984. Hyperbilirubinemia in sick cattle. Canadian journal of comparative medicine. Revue canadienne de medecine comparee, 48(3), 237–40.
Mingeot-Leclercq, M.P., Y. Glupczynski, and P.M. Tulkens. 1999. Aminoglycosides: activity and resistance. Antimicrobial Agents and Chemotherapy, 43(4), 727–37.
Miranda-CasoLuengo, R., J. Lu, E.J. Williams, A.A. Miranda-CasoLuengo, S.D. Carrington, A.C.O. Evans, and W.G. Meijer. 2019. Delayed differentiation of vaginal and uterine microbiomes in dairy cows developing postpartum endometritis. Plos One, 14(1), e0200974.
M, A.H., S. Sh, S. Am, and A.K. Ha. 2019. Intrauterine honey infusion in Holstein Frisian cows with purulent endometritis. International Journal of Complementary & Alternative Medicine, 12(1).
Moraes, J.G.N., P.R.B. Silva, L.G.D. Mendonça, A.A. Scanavez, J.C.C. Silva, and R.C. Chebel. 2017. Effects of intrauterine infusion of Escherichia coli lipopolysaccharide on uterine health, resolution of purulent vaginal discharge, and reproductive performance of lactating dairy cows. Journal of Dairy Science, 100(6), 4772–83.
Nehru, D.A., G.S. Dhaliwal, M.H. Jan, R.S. Cheema, and S. Kumar. 2019. Clinical efficacy of intrauterine cephapirin benzathine administration on clearance of uterine bacteria and subclinical endometritis in postpartum buffaloes. Reproduction in domestic animals = Zuchthygiene, 54(2), 317–24.
Nowicki, A., W. Barański, A. Baryczka, and T. Janowski. 2017. OvSynch Protocol and its Modifications in the Reproduction Management of Dairy Cattle Herds - an Update. Journal of Veterinary Research, 61(3), 329–36.
Ocal, H., M. Yuksel, and A. Ayar. 2004. Effects of gentamicin sulfate on the contractility of myometrium isolated from non-pregnant cows. Animal Reproduction Science, 84(3–4), 269–77.
Overton, T.R., J.A.A. McArt, and D.V. Nydam. 2017. A 100-Year Review: Metabolic health indicators and management of dairy cattle. Journal of Dairy Science, 100(12), 10398–417.
Pascottini, O.B. and S.J. LeBlanc. 2020. Modulation of immune function in the bovine uterus peripartum. Theriogenology.
Pérez-Báez, J., C.A. Risco, R.C. Chebel, G.C. Gomes, L.F. Greco, S. Tao, I.M. Thompson, B.C. do Amaral, M.G. Zenobi, N. Martinez, C.R. Staples, G.E. Dahl, J.A. Hernández, J.E.P. Santos, and K.N. Galvão. 2019. Association of dry matter intake and energy balance prepartum and postpartum with health disorders postpartum: Part I. Calving disorders and metritis. Journal of Dairy Science, 102(10), 9138–50.
Raboisson, D., M. Mounié, and E. Maigné. 2014. Diseases, reproductive performance, and changes in milk production associated with subclinical ketosis in dairy cows: a meta-analysis and review. Journal of Dairy Science, 97(12), 7547–63.
Raliou, M., D. Dembélé, A. Düvel, P. Bolifraud, J. Aubert, T. Mary-Huard, D. Rocha, F. Piumi, S. Mockly, M. Heppelmann, I. Dieuzy-Labaye, P. Zieger, D. G E Smith, H.-J. Schuberth, I.M. Sheldon, and O. Sandra. 2019. Subclinical endometritis in dairy cattle is associated with distinct mRNA expression patterns in blood and endometrium. Plos One, 14(8), e0220244.
Rhoads, M.L., R.O. Gilbert, M.C. Lucy, and W.R. Butler. 2004. Effects of urea infusion on the uterine luminal environment of dairy cows. Journal of Dairy Science, 87(9), 2896–901.
Sattler, T. and M. Fürll. 2004. Creatine kinase and aspartate aminotransferase in cows as indicators for endometritis. Journal of veterinary medicine. A, Physiology, pathology, clinical medicine, 51(3), 132–7.
Sheldon, I.M., J.G. Cronin, and J.J. Bromfield. 2019. Tolerance and innate immunity shape the development of postpartum uterine disease and the impact of endometritis in dairy cattle. Annual review of animal biosciences, 7, 361–84.
Sheldon, I.M., J. Cronin, L. Goetze, G. Donofrio, and H.-J. Schuberth. 2009. Defining postpartum uterine disease and the mechanisms of infection and immunity in the female reproductive tract in cattle. Biology of Reproduction, 81(6), 1025–32.
Sheldon, I.M., G.S. Lewis, S. LeBlanc, and R.O. Gilbert. 2006. Defining postpartum uterine disease in cattle. Theriogenology, 65(8), 1516–30.
Sheldon, I.M. and D.E. Noakes. 1998. Comparison of three treatments for bovine endometritis. The Veterinary Record, 142(21), 575–9.
Spears, J.W. 2000. Micronutrients and immune function in cattle. The Proceedings of the Nutrition Society, 59(4), 587–94.
Stevenson, J.S. and A.P. Phatak. 2010. Rates of luteolysis and pregnancy in dairy cows after treatment with cloprostenol or dinoprost. Theriogenology, 73(8), 1127–38.
Tison, N., E. Bouchard, L. DesCôteaux, and R.C. Lefebvre. 2017. Effectiveness of intrauterine treatment with cephapirin in dairy cows with purulent vaginal discharge. Theriogenology, 89, 305–17.
Venjakob, P.L., L. Pieper, W. Heuwieser, and S. Borchardt. 2018. Association of postpartum hypocalcemia with early-lactation milk yield, reproductive performance, and culling in dairy cows. Journal of Dairy Science, 101(10), 9396–405.
Voelz, B.E., L. Rocha, F. Scortegagna, J.S. Stevenson, and L.G.D. Mendonça. 2018. Response of lactating dairy cows with or without purulent vaginal discharge to gonadotropin-releasing hormone and prostaglandin F2α. Journal of Animal Science, 96(1), 56–65.
Walsh, R.B., J.S. Walton, D.F. Kelton, S.J. LeBlanc, K.E. Leslie, and T.F. Duffield. 2007. The effect of subclinical ketosis in early lactation on reproductive performance of postpartum dairy cows. Journal of Dairy Science, 90(6), 2788–96.
Wenzinger, B. and U. Bleul. 2012. Effect of a prostaglandin F2α analogue on the cyclic corpus luteum during its refractory period in cows. BMC veterinary research, 8, 220.
Wulster-Radcliffe, M.C., R.C. Seals, and G.S. Lewis. 2003. Progesterone increases susceptibility of gilts to uterine infections after intrauterine inoculation with infectious bacteria. Journal of Animal Science, 81(5), 1242–52.
Zerbe, H., N. Schneider, W. Leibold, T. Wensing, T.A. Kruip, and H.J. Schuberth. 2000. Altered functional and immunophenotypical properties of neutrophilic granulocytes in postpartum cows associated with fatty liver. Theriogenology, 54(5), 771–86.
Author information
Authors and Affiliations
Contributions
Mohammed Elmetwally: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing–original draft, writing–review, and editing;
Gehad E Elshopakey: conceptualization, analyses for blood, data analyses, writing, review, and editing;
Wael Eldomany: conceptualization, writing–review, and editing;
Ashraf El-Desouky: conceptualization, writing–review, and editing.
Fuller W Bazer: data curation, writing, review, and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elmetwally, M.A., Elshopakey, G.E., El-Desouky, A.M. et al. Serum biochemical profile in buffalo endometritis and impact of treatment with PGF2α and intrauterine gentamicin infusion on postpartum reproductive performance. Trop Anim Health Prod 52, 3697–3706 (2020). https://doi.org/10.1007/s11250-020-02406-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-020-02406-3