Abstract
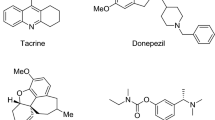
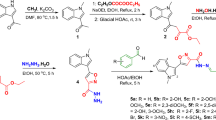
The synthesis of new compounds based on the domestic drug amiridine modified with derivatives of N-mono- and N,N-disubstituted thiourea is described. The compounds were investigated for the anticholinesterase and antioxidant activities and for the ability to inhibit the self-aggregation of β-amyloid (Aβ42). The structure—activity relationships were analyzed. The observed effects were consistent with the results of molecular docking of the compounds into cholinesterases and Aβ42. The compounds that effectively inhibit butyrylcholinesterase and demonstrate high antioxidant and antiaggregant activities were identified. The results show promise for further development of this class of compounds as potential multifunctional agents for the treatment of Alzheimer’s disease.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Cummings, G. Lee, A. Ritter, M. Sabbagh, K. Zhong, Alzheimer’s Dementia, 2019, 5, 272; DOI: https://doi.org/10.1016/j.trci.2019.05.008.
M. C. Carreiras, E. Mendes, M. J. Perry, A. P. Francisco, J. Marco-Contelles, Curr. Top. Med. Chem., 2013, 13, 1745; DOI: https://doi.org/10.2174/15680266113139990135.
Y. Huang, L. Mucke, Cell, 2012, 148, 1204; DOI: https://doi.org/10.1016/j.cell.2012.02.040.
L. Ismaili, B. Refouvelet, M. Benchekroun, S. Brogi, M. Brindisi, S. Gemma, G. Campiani, S. Filipic, D. Agbaba, G. Esteban, M. Unzeta, K. Nikolic, S. Butini, J. Marco-Contelles, Prog. Neurobiol., 2017, 151, 4; DOI: https://doi.org/10.1016/j.pneurobio.2015.12.003.
K. Spilovska, J. Korabecny, E. Nepovimova, R. Dolezal, E. Mezeiova, O. Soukup, K. Kuca, Curr. Top. Med. Chem., 2017, 17, 1006; DOI: https://doi.org/10.2174/1568026605666160927152728.
F. Mesiti, D. Chavarria, A. Gaspar, S. Alcaro, F. Borges, Eur. J. Med. Chem., 2019, 181, 111572; DOI: https://doi.org/10.1016/j.ejmech.2019.111572.
M. Girek, P. Szymański, Chem. Pap., 2019, 73, 269; DOI: https://doi.org/10.1007/s11696-018-0590-8.
R. T. Kareem, F. Abedinifar, E. A. Mahmood, A. G. Ebadi, F. Rajabi, E. Vessally, RSC Adv., 2021, 11, 30781; DOI: https://doi.org/10.1039/d1ra03718h.
E. E. Bukatina, I. V. Grigoréva, E. I. Sokolćhik, Neurosci. Behav. Physiol., 1993, 23, 83; DOI: https://doi.org/10.1007/BF01182643.
V. Kluša, J. Rumaks, Ñ. Karajeva, Proc. Latv. Acad. Sci., Sect. B, 2008, 62, 85; DOI: https://doi.org/10.2478/v10046-008-0024-z.
I. V. Damulin, D. A. Stepkina, A. B. Lokshina, Zhurn. Nevrologii i Psikhiatrii im. S. S. Korsakova [S. S. Korsakov J. Neurol. and Psych.], 2011, 111, No. 2, 40 (in Russian).
S. A. Zhivolupov, L. S. Onishchenko, N. A. Rashidov, I. N. Samartsev, E. V. Yakovlev, Zhurn. Nevrologii i Psikhiatrii im. S. S. Korsakova [S. S. Korsakov J. Neurol. and Psych.], 2018, 118, No. 2, 58–64; DOI: https://doi.org/10.17116/jnevro20181182158-64 (in Russian).
M. M. Oros, Int. J. Neurol., 2018, 23; DOI: https://doi.org/10.22141/2224-0713.6.100.2018.146454.
I. V. Litvinenko, S. A. Zhivolupov, I. N. Samartsev, A. Y. Kravchuk, M. N. Vorobyova, E. V. Yakovlev, Y. S. Butakova, Neurosci. Behav. Physiol., 2020, 50, 1112; DOI: https://doi.org/10.1007/s11055-020-01012-y.
J. Kojima, K. Onodera, M. Ozeki, K. Nakayama, CNS Drug Rev., 1998, 4, 247; DOI: https://doi.org/10.1111/j.1527-3458.1998.tb00067.x.
P. N. Shevtsov, E. F. Shevtsova, G. Sh. Burbaeva, S. O. Bachurin, Bull. Exp. Biol. Med., 2014, 156, 768; DOI: https://doi.org/10.1007/s10517-014-2445-9.
G. F. Makhaeva, S. V. Lushchekina, N. V. Kovaleva, T.Yu. Astakhova, N. P. Boltneva, E. V. Rudakova, O. G. Serebryakova, A. N. Proshin, I. V. Serkov, T. P. Trofimova, V. A. Tafeenko, E. V. Radchenko, V. A. Palyulin, V. P. Fisenko, J. Korábečný, O. Soukup, R. J. Richardson, Bioorg. Chem., 2021, 112, 104974; DOI: https://doi.org/10.1016/j.bioorg.2021.104974.
S. Yoshida, N. Suzuki, Eur. J. Pharmacol., 1993, 250, 117; DOI: https://doi.org/10.1016/0014-2999(93)90628-u.
A. M. Zhidkova, A. S. Berlyand, A. Z. Knizhnik, E. F. Lavretskaya, T. N. Robakidze, S. A. Sukhanova, T. P. Mufazalova, Pharm. Chem. J., 1989, 23, 709; DOI: https://doi.org/10.1007/BF00764431.
A. M. Zhidkova, M. S. Goizman, A. S. Berlyand, A. Z. Knizhnik, L. S. Khabarova, Khim.-Farm. Zhurn. [Pharm. Chem. J.], 1989, 23, 1401 (in Russian).
G. F. Makhaeva, N. V. Kovaleva, N. P. Boltneva, E. V. Rudakova, S. V. Lushchekina, T. Y. Astakhova, I. V. Serkov, A. N. Proshin, E. V. Radchenko, V. A. Palyulin, J. Korabecny, O. Soukup, S. O. Bachurin, R. J. Richardson, Molecules, 2022, 27, 1060; DOI: https://doi.org/10.3390/molecules27031060.
S. Bachurin, S. Tkachenko, I. Baskin, N. Lermontova, T. Mukhina, L. Petrova, A. Ustinov, A. Proshin, V. Grigoriev, N. Lukoyanov, V. Palyulin, N. Zefirov, Ann. N. Y. Acad. Sci., 2001, 939, 219; DOI: https://doi.org/10.1111/j.1749-6632.2001.tb03629.x.
G. L. Perlovich, A. N. Proshin, T. V. Volkova, S. V. Kurkov, V. V. Grigoriev, L. N. Petrova, S. O. Bachurin, J. Med. Chem., 2009, 52, 1845; DOI: https://doi.org/10.1021/jm8012882.
H. Pavan Kumar, H. K. Kumara, R. Suhas, D. Channe Gowda, Arch. Pharm., 2021, 354, e2000468; DOI: https://doi.org/10.1002/ardp.202000468.
B. Ozgeris, J. Antibiot., 2021, 74, 233; DOI: https://doi.org/10.1038/s41429-020-00399-7.
D. Q. Huong, M. V. Bay, P. C. Nam, J. Mol. Liq., 2021, 340, 117149; DOI: https://doi.org/10.1016/j.molliq.2021.117149.
M. Bajda, S. Filipek, Bioorg. Med. Chem. Lett., 2017, 27, 212; DOI: https://doi.org/10.1016/j.bmcl.2016.11.072.
T. Mohamed, P. P. N. Rao, Eur. J. Med. Chem., 2017, 126, 823; DOI: https://doi.org/10.1016/j.ejmech.2016.12.005.
G. F. Makhaeva, E. V. Rudakova, N. V. Kovaleva, S. V. Lushchekina, N. P. Boltneva, A. N. Proshin, E. V. Shchegolkov, Ya. V. Burgart, V. I. Saloutin, Russ. Chem. Bull., 2019, 68, 967; DOI: https://doi.org/10.1007/s11172-019-2507-2.
P. Taylor, S. Lappi, Biochemistry, 1975, 14, 1989; DOI: https://doi.org/10.1021/bi00680a029.
M. Bartolini, C. Bertucci, V. Cavrini, V. Andrisano, Biochem. Pharmacol., 2003, 65, 407; DOI: https://doi.org/10.1016/s0006-2952(02)01514-9.
P. Munoz-Ruiz, L. Rubio, E. Garcia-Palomero, I. Dorronsoro, M. Del Monte-Millan, R. Valenzuela, P. Usan, C. De Austria, M. Bartolini, V. Andrisano, A. Bidon-Chanal, M. Orozco, F. J. Luque, M. Medina, A. Martinez, J. Med. Chem., 2005, 48, 7223; DOI: https://doi.org/10.1021/jm0503289.
H. Safarizadeh, Z. Garkani-Nejad, J. Mol. Graph. Model., 2019, 87, 129; DOI: https://doi.org/10.1016/j.jmgm.2018.11.019.
S. Jokar, M. Erfani, O. Bavi, S. Khazaei, M. Sharifzadeh, M. Hajiramezanali, D. Beiki, A. Shamloo, Bioorg. Chem., 2020, 102, 104050; DOI: https://doi.org/10.1016/j.bioorg.2020.104050.
J. Wang, P. Cai, X.-L. Yang, F. Li, J.-J. Wu, L.-Y. Kong, X.-B. Wang, Eur. J. Med. Chem., 2017, 139, 68; DOI: https://doi.org/10.1016/j.ejmech.2017.07.077.
M. P. Williamson, Y. Suzuki, N. T. Bourne, T. Asakura, Biochem. J., 2006, 397, 483; DOI: https://doi.org/10.1042/BJ20060293.
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Rice-Evans, Free Radical Biol. Med., 1999, 26, 1231; DOI: https://doi.org/10.1016/s0891-5849(98)00315-3.
I. F. F. Benzie, J. J. Strain, Methods Enzymol., 1999, 299, 15; DOI: https://doi.org/10.1016/s0076-6879(99)99005-5.
S. Meir, J. Kanner, B. Akiri, S. Philosoph-Hadas, J. Agric. Food Chem., 1995, 43, 1813; DOI: https://doi.org/10.1021/jf00055a012.
G. F. Makhaeva, E. V. Radchenko, V. A. Palyulin, E. V. Rudakova, A.Yu. Aksinenko, V. B. Sokolov, N. S. Zefirov, R. J. Richardson, Chem.-Biol. Interact., 2013, 203, 231; DOI: https://doi.org/10.1016/j.cbi.2012.10.012.
G. F. Makhaeva, E. V. Rudakova, O. G. Serebryakova, A.Yu. Aksinenko, S. V. Lushchekina, S. O. Bachurin, R. J. Richardson, Chem.-Biol. Interact., 2016, 259, 332; DOI: https://doi.org/10.1016/j.cbi.2016.05.002.
G. L. Ellman, K. D. Courtney, V. Andres, Jr., R. M. Feather-Stone, Biochem. Pharmacol., 1961, 7, 88; DOI: https://doi.org/10.1016/0006-2952(61)90145-9.
G. F. Makhaeva, N. V. Kovaleva, N. P. Boltneva, S. V. Lushchekina, T.Yu. Astakhova, E. V. Rudakova, A. N. Proshin, I. V. Serkov, E. V. Radchenko, V. A. Palyulin, S. O. Bachurin, R. J. Richardson, Molecules, 2020, 25, 3915; DOI: https://doi.org/10.3390/molecules25173915.
G. F. Makhaeva, N. V. Kovaleva, N. P. Boltneva, S. V. Lushchekina, E. V. Rudakova, T. S. Stupina, A. A. Terentiev, I. V. Serkov, A. N. Proshin, E. V. Radchenko, V. A. Palyulin, S. O. Bachurin, R. J. Richardson, Bioorg. Chem., 2020, 94, 103387; DOI: https://doi.org/10.1016/j.bioorg.2019.103387.
M. Biancalana, S. Koide, Biochim. Biophys. Acta, Proteins Proteomis, 2010, 1804, 1405; DOI: https://doi.org/10.1016/j.bbapap.2010.04.001.
I. F. Benzie, J. J. Strain, Anal. Biochem., 1996, 239, 70; DOI: https://doi.org/10.1006/abio.1996.0292.
X. Pan, H. Wang, C. Li, J. Z. H. Zhang, C. Ji, J. Chem. Inf. Model., 2021, 61, 3159; DOI: https://doi.org/10.1021/acs.jcim.1c00075.
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, S. T. Elbert, M. S. Gordon, J. H. Jensen, S. Koseki, N. Matsunaga, K. A. Nguyen, S. Su, T. L. Windus, M. Dupuis, J. A. Montgomery, J. Comput. Chem., 1993, 14, 1347; DOI: https://doi.org/10.1002/jcc.540141112.
P.-O. Löwdin, in Advances in Quantum Chemistry, Ed. P.-O. Löwdin, Academic Press, New York, London, 1970, p. 185.
J. Cheung, M. J. Rudolph, F. Burshteyn, M. S. Cassidy, E. N. Gary, J. Love, M. C. Franklin, J. J. Height, J. Med. Chem., 2012, 55, 10282; DOI: https://doi.org/10.1021/jm300871x.
S. V. Lushchekina, G. F. Makhaeva, D. A. Novichkova, I. V. Zueva, N. V. Kovaleva, R. J. Richardson, Supercomput. Front. Innov., 2018, 5, 89; DOI: https://doi.org/10.14529/jsfi1804.
I. Zueva, S. Lushchekina, P. Shulnikova, O. Lenina, K. Petrov, E. Molochkina, P. Masson, Chem. Biol. Interact., 2021, 348, 109646; DOI: https://doi.org/10.1016/j.cbi.2021.109646.
Y. Nicolet, O. Lockridge, P. Masson, J. C. Fontecilla-Camps, F. Nachon, J. Biol. Chem., 2003, 278, 41141; DOI: https://doi.org/10.1074/jbc.M210241200.
P. Masson, S. Lushchekina, L. M. Schopfer, O. Lockridge, Biochem. J., 2013, 454, 387; DOI: https://doi.org/10.1042/bj20130389.
O. Crescenzi, S. Tomaselli, R. Guerrini, S. Salvadori, A. M. D’Ursi, P. A. Temussi, D. Picone, Eur. J. Biochem., 2002, 269, 5642; DOI: https://doi.org/10.1046/j.1432-1033.2002.03271.x.
H. Li, A. D. Robertson, J. H. Jensen, Proteins, 2005, 61, 704; DOI: https://doi.org/10.1002/prot.20660.
G. M. Morris, D. S. Goodsell, R. S. Halliday, R. Huey, W. E. Hart, R. K. Belew, A. J. Olson, J. Comput. Chem., 1998, 19, 1639; DOI: https://doi.org/10.1002/(sici)1096-987x(19981115)19:14<1639::aid-jcc10>3.0.co;2-b.
G. M. Morris, R. Huey, W. Lindstrom, M. F. Sanner, R. K. Belew, D. S. Goodsell, A. J. Olson, J. Comput. Chem., 2009, 30, 2785; DOI: https://doi.org/10.1002/jcc.21256.
Author information
Authors and Affiliations
Corresponding author
Additional information
The study was carried out within the state assignment of the Institute of Physiologically Active Compounds, Russian Academy of Sciences (No. FFSN-2021-0005) and was partially supported by the Russian Foundation for Basic Research (Project No. 19-53-26016a). Molecular modeling was carried out using the shared research facilities of the HPC computing resources at Lomonosov Moscow State University.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Based on the materials of the V Russian Conference on Medicinal Chemistry with international participation “MedChem-Russia 2021” (May 16–19, 2022, Volgograd, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2404–2415, November, 2022.
Rights and permissions
About this article
Cite this article
Makhaeva, G.F., Proshin, A.N., Kovaleva, N.V. et al. Synthesis and study of the biological activity of thiourea-containing amiridine derivatives as potential multi-target drugs for the treatment of Alzheimer’s disease. Russ Chem Bull 71, 2404–2415 (2022). https://doi.org/10.1007/s11172-022-3668-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3668-y