Abstract
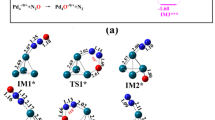
The reaction mechanisms of the decarbonylation and decarboxylation of propanoic acid on icosahedral and cuboctahedral clusters Pd55 were modeled in terms of the density functional theory using the PBE functional and the SBK pseudopotential. According to calculations, the hydrogen abstraction step of the decarboxylation reaction is the most sensitive to the cluster shape and proceeds more readily on the icosahedral cluster. The activation energy difference reaches a value of 4.4 kcal mol−1. In addition, the icosahedral cluster demonstrates a higher activity and selectivity toward decarboxylation compared to the cuboctahedral cluster.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Snåre, I. Kubicková, P. Mäki-Arvela, K. Eränen, D. Yu. Murzin, Ind. Eng. Chem. Res., 2006, 45, 5708; DOI: https://doi.org/10.1021/ie060334i.
A. S. Berenblyum, T. A. Podoplelova, R. S. Shamsiev, E. A. Katsman, V. Ya. Danyushevsky, V. R. Flid, Catalysis in Industry, 2012, 4, 209; DOI: https://doi.org/10.1134/S2070050412030026.
G. C. R. Silva, D. Qian, R. Pace, O. Heintz, G. Caboche, E. Santillan-Jimenez, M. Crocker, Catalysts, 2020, 10, 91; DOI: https://doi.org/10.3390/catal10010091.
J. Gopeesingh, R. Zhu, R. Schuarca, W. Yang, A. Heyden, J. Q. Bond, Ind. Eng. Chem. Res., 2021, 60, 16171; DOI: https://doi.org/10.1021/acs.iecr.1c03032.
K. A. Rogers, Y. Zheng, ChemSusChem, 2016, 9, 1750; DOI: https://doi.org/10.1002/cssc.201600144.
E. Santillan-Jimenez, M. Crocker, J. Chem. Technol. Biotechnol., 2012, 87, 1041; DOI: https://doi.org/10.1002/jctb.3775.
A. S. Berenblyum, V. Ya. Danyushevsky, P. S. Kuznetsov, E. A. Katsman, R. S. Shamsiev, Petroleum Chemistry (Engl. Transl.), 2016, 56, 663; DOI: https://doi.org/10.1134/S0965544116080028.
E. A. Katsman, V. Ya. Danyushevsky, P. S. Kuznetsov, R. S. Shamsiev, A. S. Berenblyum, Kinet. Catal. (Engl. Transl.), 2017, 58, 147; DOI: https://doi.org/10.1134/S0023158417020069.
J. P. Ford, J. G. Immer, H. H. Lamb, Top. Catal., 2012, 55, 175; DOI: https://doi.org/10.1007/s11244-012-9786-2.
K. Hengst, M. Arend, R. Pfitzenreuter, W. F. Hoelderich, Appl. Catal. B, 2015, 174–175, 383; DOI: https://doi.org/10.1016/j.apcatb.2015.03.009.
A. S. Berenblyum, T. A. Podoplelova, E. A. Katsman, R. S. Shamsiev, V. Ya. Danyushevsky, Kinet. Catal. (Engl. Transl.), 2012, 53, 595; DOI: https://doi.org/10.1134/S0023158412050023.
J. Fu, D. Mei, Catal. Today, 2021, 365, 181; DOI: https://doi.org/10.1016/j.cattod.2020.05.014.
R. S. Shamsiev, I. E. Sokolov, F. O. Danilov, V. R. Flid, Kinet. Catal. (Engl. Transl.), 2019, 60, 627; DOI: https://doi.org/10.1134/S0023158419050094.
J. Lu, S. Behtash, A. Heyden, J. Phys. Chem. C, 2012, 116, 14328; DOI: https://doi.org/10.1021/jp301926t.
J. Lu, S. Behtash, M. Faheem, A. Heyden, J. Catal., 2013, 305, 56; DOI: https://doi.org/10.1016/j.jcat.2013.04.026.
K. C. Chukwu, L. Árnadóttir, J. Phys. Chem. C, 2020, 124, 13082; DOI: https://doi.org/10.1021/acs.jpcc.0c00436.
R. S. Shamsiev, F. O. Danilov, V. R. Flid, Russ. Chem. Bull., 2022, 71, 220; DOI: https://doi.org/10.1007/s11172-022-3400-y.
S. K. Kundu, R. V. Solomon, W. Yang, E. Walker, O. Mamun, J. Q. Bond, A. Heyden, Catal. Sci. Technol., 2021, 11, 6163; DOI: https://doi.org/10.1039/D1CY01029H.
D. Uzio, G. Berhault, Catal. Rev. Sci. Eng., 2010, 52, 106; DOI: https://doi.org/10.1080/01614940903510496.
G. A. Somorjai, J. Y. Park, Angew. Chem. Int. Ed., 2008, 47, 9212; DOI: https://doi.org/10.1002/anie.200803181.
Y. Zhao, G. Fu, N. Zheng, Catal. Today, 2017, 279, 36; DOI: https://doi.org/10.1016/j.cattod.2016.05.017.
S. Sreedhala, V. Sudheeshkumar, C. P. Vinod, J. Catal., 2016, 337, 138; DOI: https://doi.org/10.1016/j.jcat.2016.01.017.
G. Collins, M. Schmidt, C. O’Dwyer, J. D. Holmes, G. P. McGlacken, Angew. Chem., Int. Ed., 2014, 53, 4142; DOI: https://doi.org/10.1002/anie.201400483.
A. Ruditskiy, S.-I. Choi, H.-C. Peng, Y. Xia, MRS Bull., 2014, 39, 727; DOI: https://doi.org/10.1557/mrs.2014.167.
Y. Wang, S. Xie, J. Liu, J. Park, C. Z. Huang, Y. Xia, Nano Lett., 2013, 13, 2276; DOI: https://doi.org/10.1021/nl400893p.
N. S. Kuzmina, S. V. Portnova, E. L. Krasnykh, Fine Chem. Technol., 2020, 15, No. 2, 47; DOI: https://doi.org/10.32362/2410-6593-2020-15-2-47-55.
D. N. Laikov, Chem. Phys. Lett., 1997, 281, 151; DOI: https://doi.org/10.1016/S0009-2614(97)01206-2.
D. N. Laikov, Yu. A. Ustynyuk, Russ. Chem. Bull., 2005, 54, 820; DOI: https://doi.org/10.1007/s11172-005-0329-x.
J. P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865; DOI: https://doi.org/10.1103/PhysRevLett.77.3865.
W. J. Stevens, H. Basch, M. Krauss, J. Chem. Phys., 1984, 81, 6026; DOI: https://doi.org/10.1063/1.447604.
F. L. Hirschfeld, Theoret. Chim. Acta, 1977, 44, 129; DOI: https://doi.org/10.1007/BF00549096.
P. Nava, M. Sierka, R. Ahlrichs, Phys. Chem. Chem. Phys., 2003, 5, 3372; DOI: https://doi.org/10.1039/B303347C.
R. S. Shamsiev, I. E. Sokolov, V. R. Flid, Russ. Chem. Bull., 2014, 63, 2585; DOI: https://doi.org/10.1007/s11172-014-0783-4.
R. S. Shamsiev, F. O. Danilov, Russ. Chem. Bull., 2017, 66, 395; DOI: https://doi.org/10.1007/s11172-017-1746-3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Calculations were carried out on computational facilities at the Joint Supercomputer Center of the Russian Academy of Sciences.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 9, pp. 1863–1869, September, 2022.
Rights and permissions
About this article
Cite this article
Shamsiev, R.S. Decarbonylation and decarboxylation of propanoic acid on Pd55 clusters: a quantum chemical modeling. Russ Chem Bull 71, 1863–1869 (2022). https://doi.org/10.1007/s11172-022-3603-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3603-2