Abstract
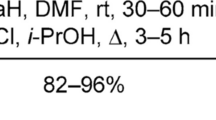
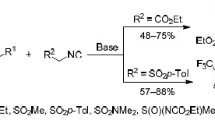
1-Alkynyl-1-chlorocyclopropanes undergo chlorine-lithium exchange on treatment with BunLi in THF at -40—0 °C. Thus generated organolithium species react with carbon dioxide (dry ice) and acetone to give selectively hitherto unknown 1-alkynylcyclopropanecarboxylic acids and the corresponding alcohols in up to 74% yields. Similar reactions involving methyl chloroformate result in the mixtures of cyclopropylacetylenic and vinylidenecyclopropanic esters, while the use of aliphatic aldehydes as electrophiles provides secondary allenic alcohols in up to 64% yields.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
V. D. Gvozdev, K. N. Shavrin, A. A. Ageshina, O. M. Nefedov, Russ. Chem. Bull., 2017. 67, 862.
G.-Q. Chen, W. Fang, Y. Wei, X.-Y. Tang, M. Shi, Chem. Commun., 2016. 52, 10799.
H.-H. Liao, R.-S. Liu, Chem. Commun., 2011. 47, 1339.
G.-Q. Chen, W. Fang, Y. Wei, X.-Y. Tang, M. Shi, Chem. Set., 2016. 7, 4318.
F. Yi, B. Huang, Q. Nie, M. Cai, Tetrahedron Lett, 2016. 57, 4405.
C. Zhang, M. Xu, J. Ren, Z. Wang, Eur. J. Org. Chem, 2016. 2467.
Y. Bai, W. Tao, J. Ren, Z. Wang, Angew. Chem., Int. Ed, 2012. 51, 4112.
S. Ye, Z.-X. Yu, Chem. Commun., 2011. 47, 794.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Russ. Chem. Bull., 2010. 59, 396.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Russ. Chem. Bull., 2010. 59, 1451.
A. Chen, R. Lin, Q. Liub, N. Jiao, Chem. Commun., 2009. 45, 6842.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Mendeleev Commun., 2008. 18, 300.
B. M. Trost, J. Xie, N. Maulide, J. Am. Chem. Soc, 2008. 130, 17258.
J. Barluenga, E. Tudela, R. Vicente, A. Ballesteros, M. Tomas, Angew. Chem., Int. Ed., 2011. 50, 2107.
J. P. Markham, S. T. Staben, F. D. Toste, J.Am. Chem. Soc, 2005. 127, 9708.
A. Zampella, M. V. Drauria, L. Minale, C. Debitus, C. Rous-sakis, J.Am. Chem. Soc, 1996. 118, 11085.
C. E. Tedford, J. G. Philips, R. Gregory, G. P. Pawlowski, L. Fadnis, M. A. Khan, S. M. Ali, M. K. Handley, S. L. Yates, J. Pharmacol. Exp. Titer., 1999. 289, 1160.
J. W. Corbett, S. S. Ko, J. D. Rodgers, L. A. Gearhart, N. A. Magnus, L. T. Bacheler, S. Diamond, S. Jeffrey, R. M. Klabe, B. C. Cordova, S. Garber, K. Logue, G. L. Trainor, P. S. Anderson, S. K. Erickson-Viitanen, J. Med. Chem., 2000. 43, 2019.
S. Yang, M. Shi, Ace. Chem. Res., 2018. 51, 1667.
K. N. Shavrin, I. V. Krylova, I. B. Shvedova, G. P. Okon-nishnikova, I. E. Dolgy, O. M. Nefedov, J. Chem. Soc.,Perkin Trans. 2, 1991. 1875; DOI: https://doi.org/10.1039/P29910001875.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Russ. Chem. Bull, 2002. 51, 1237.
S. Braese, S. Schoemenauer, G. McGaffm, A. Stolle, A. de Meijere, Chem. Eur. J., 1996. 2, 545.
P. Menningen, C. Harcken, B. Stecker, S. Koerbe, A. de Meijere, M. R. Lopes, J. Ollivier, J. Salauen, Synlett, 1999. 1534.
K. N. Shavrin, V. D. Gvozdev, D. V. Budanov, S. V. Yurov, O. M. Nefedov, Mendeleev Commun., 2006. 16, 73.
G. Bengtson, S. Keyaniyan, A. de Meijere, Chem. Ber., 1986. 119, 3607.
A. Stolle, J. Ollivier, P. P. Piras, J. Salauen, A. de Meijere, J.Am. Chem. Soc, 1992. 114, 4051.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Russ. Chem. Bull, 2008. 57, 2117.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Russ. Chem. Bull., 2009. 58, 2432.
K. N. Shavrin, V. D. Gvozdev, O. M. Nefedov, Mendeleev Commun., 2008. 18, 300.
T. Liese, A. de Meijere, Chem. Ber., 1986. 119, 2995.
A. de Meijere, S. I. Kozhushkov, Chem. Eur. J., 2002. 8, 3195.
H. J. Reich, J. E. Holladay, T. G. Walker, J. L. Thompson, J.Am. Chem. Soc, 1999. 121, 9769.
M. Miao, J. Cao, J. Zhang, X. Huang, L. Wu, Org. Lett, 2012. 14, 2718.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1384–1390, July, 2019.
Rights and permissions
About this article
Cite this article
Gvozdev, V.D., Shavrin, K.N. & Nefedov, O.M. Lithiation of 1-alkynyl-1-chlorocyclopropanes and subsequent reactions with electrophilic reagents: synthesis of functionalized alkynyl- and vinylidenecyclopropanes. Russ Chem Bull 68, 1384–1390 (2019). https://doi.org/10.1007/s11172-019-2566-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-019-2566-4