Abstract
The ornamental fish industry has continued to flourish since eighteenth century with increased fascination by enthusiasts in the striking body colours and patterns displayed in the fishes, a beneficial outcome of rigorous selective programmes. The expression of these pigmented colours is the result of the differentiation and orientation of specialised chromatophores located within the dermal layer. The different types of chromatophores found in many ornamental fish species, are the basis of the unique colour hues and patterns. This review discusses the current approaches for enhancing the body pigmentation and pattern of ornamental fishes. Two factors are considered to be the main drivers of body colour regulation: feed additives (pigments) and rearing environment setup, i.e. tank colour and light. Potential candidate pigment genes to manipulate the ornamental fish body pigmentation and pattern have been elucidated through mapping of putative regulatory pathways, buoyed by the rapid development of next generation sequencing technologies. The effects of feed additives, tank background colour and light on various ornamental fish species, and regulatory pathways of involved genes offer valuable insights for enhanced variety production prior to genetic engineering and are herein discussed. It is hoped that the systematic analysis of the current knowledge in this review would be a boon for the ornamental fish community to step up efforts to boost the ornamental fish breeding industry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ornamental fish petting is one of most popular hobbies of the global community, accelerating the ornamental fish industry since eighteenth century. With an annual worldwide growth rate of 14%, worth US $15 billion, it is an important economic activity in many developing nations (Yanar et al. 2019; Yue 2019). The value of an ornamental fish is determined by its morphological traits and features, in particular its body colours, pattern and shape. For instance, a discus fish (Symphysodon spp.) (Fig. 1) with unique disc-shaped body and assorted body colours and patterns would cost around USD100–400 locally, while discus varieties with pale and monotonous colour, such as snow white bred (Fig. 1a), receive less attention (personal communication, Aquacity Tropical Shd. Bhd., one of the pioneer discus breeding companies in South East Asia region).
The varieties of adult discus fish (Symphysodon sp.) with various body pigmented patterns. a Snow white discus with body entirely covered in white. b The spotted green classic discus with melanic vertical bar stripes, orange dotted spots and dorsal wavy stripes with white background. c Pigeon blood discus with dorsalvertically wavy orange stripes on top of the white background. d Golden tangerine discus with entire trunk covered in golden background colour, head appeared white with transparent fins. e Alenquer red discus with melanic vertical bar stripes, blue-white stripes above the lateral line on top of the orange background colour. f Blue snakeskin discus with entire body covered in spatial melanic markings with blue iridescent background. g Checkerboard discus with body covered in red wavy stripes, melanic vertical bars on top of the white background colour. h Wild brown discus with melanic vertical bar stripes and brown background colour, and fins with red wavy stripes. Pictures retrieved from Discus Catalogue (2004) supplied by Aquacity Tropical Sdn. Bhd. Photo credit: Patrik Tan
Much efforts have been geared towards enhancing body colour and patterns. In a typical scenario, the original stocks are wild captured and selectively crossbred to produce more attractive domesticated varieties for the ornamental fish trade industry (Ng 2016). Unlike maricultural fish, the selective breeding of ornamental fish emphasizes on beautifying the morphological appearance. Unfortunately, selective breeding is a time and cost-consuming process, which requires the interbreeding over many generations, with the inevitable production of undesirable progenies during the process, before attaining the targeted pure bred (Walster 2008). Several methods have been developed and widely applied for enhancing the morphological appearance of ornamental fishes. These include nutritional supplements such as carotenoid-based diet, varying stocking density and other environmental parameters which could effectively affect the colour hue and brightness (Klann et al. 2021). However, the positive effects may not be permanent, disappearing upon termination of carotenoid supplementation or environmental stimulus fading. Therefore, a multi-pronged strategy is necessary to manipulate the ornamental fish body colours and patterns, in order to expedite the production of new varieties.
Although advances in breeding techniques have positively impacted the ornamental fish industry (Pouil et al. 2020), the majority of marine species remain unsustainably sourced due to the complicated breeding processes involved (King 2019). One strategic approach is to elucidate the molecular mechanisms controlling the various morphological variants which could potentially be engineered for the desired outcomes. Numerous studies (Cerdá-Reverter et al. 2011; Cal et al. 2017; Kimura 2021; Luo et al. 2021; Kwon et al. 2022; Liu et al. 2022) on fish pigmentation have permitted a better understanding of the regulatory mechanisms of fish colour and pattern formations. To date, there are at least five types of pigment cells in fish, namely melanophores (black and brown colour), xanthophores (orange and yellow colour), erythophores (orange and red colour), iridophores (iridescence) and leucophores (white colour) (Luo et al. 2021). These pigment cells may share the same neural progenitor cell, which rely on a complicated regulatory network of multiple gene pathways and signalling control of the cell processes of proliferation, specification, migration and orientation (Luo et al. 2021).
Herein, we present a review of the scientific efforts on the effects of selective breeding, feed additives and tank environment, e.g. light and tank colours, on various ornamental fish species. We also discuss candidate genes for potential genetic engineering manipulation of ornamental fish body colours and patterns. This review aims to provide a crucial reference to the scientific community to accelerate future ventures with regional ornamental fish farmers for production of novel ornamental fish varieties.
Breeding of ornamental fish
Fish breeding had long been practised since thousands of years ago in China prior to the initiation of systematic studies and breeding programmes in the 1970s (Gjedrem and Baranski 2009; Zhou and Gui 2018). In the early days of the modern breeding programme, the more systematic documentation of parental lineages over generations gradually led to the development of pedigree breeding models (Gjedrem and Baranski 2009). In freshwater ornamental fish, most breeding approaches for genetic improvement were focused on members of family Cyprinidae particularly the common carp (Cyprinus carpio) and goldfish (Carassius auratus) (Gui and Zhou 2010; Chen et al. 2020a, b). Genetic trait improvement in common carp through artificial selection involving hybridization, crossbreeding and selective breeding accelerated the production of commercial breeds (Hulata 1995; Vandeputte 2003; Zhou and Gui 2018).
During the early phases of the artificial selection programme, farmers had focused on the breeding of closely related individuals, presumably due to their similarity in expressing the targeted traits. This led to inbreeding, with consequent reduction in productivity even over a span of a few generations. With greater understanding of genetics, crossbreeding techniques was introduced involving mating between distant variaties (Gjedrem and Baranski 2010). This led to the propagation of numerous new breeds of carps, particularly in China (Wu and Zhong 1964; Li and Wang 2001; Zhou and Gui 2018). Numerous novel ornamental red carps have since been produced, frequently with the red common carp contributing as one of the parents including Xingguo red common carp, Purse red common carp and glass red common carp (Li and Wang 2001). Furthermore, other ornamental species such as Swordtail fish variety with red eyes and body have also been propagated through family selection.
However, there have been concerns over the sustainability of the improvement resulting from the artificial selection approach. Genetic improvement through selective breeding is an accumulative and time-consuming process, where only minimal improvement is gained in each cycle; substantial genetic enhancement can only be achieved and maintained through repetitive breeding over many generations (Gjedrem and Baranski 2010). Moreover, the majority of selective breeding programmes were private efforts; unsystematic, with limited scientific basis and documentation, where accurate and detailed records of parental lineages were unavailable for reproducibility or further studies (Ng 2004; Komiyama et al. 2009). The welfare of the cultured fish was frequently of little concern to the aquaculturists, including the rampant practice of culling undesired progenies (Walster 2008; Kirkwood 2012). Furthermore, selection can only target populations with varying phenotypes that are governed by the variation within the genetic pool (Gjedrem and Baranski 2009). However, even with diverse parental lineages, effective population size tends to decrease over generations, thus lowering the likelihood for further improvement (Vandeputte 2003; Lind et al. 2012). One classic example is the case of the selective breeding of Symphysodon spp which are currently represented by more than 69 domesticated varieties. However, all are relatively homogeneous with little significant genetic variation among each other (Ng et al. 2021), Therefore, there is a need for proper breeding planning with genetic consideration, while trialling on novel alternatives for pigmentation improvement.
Feed additives
Colour variation in ornamental fishes is exhibited through the expression of dermal chromatophores, particularly xanthophore and erythrophore-based colours originating from the intake of dietary biological pigments. This pigment uptake, modification and deposition in the fish dermal tissue is deemed species-specific (Kop and Durmaz 2008), influenced by multiple factors such as the regulation of several genes (Ahi et al. 2020; Fang et al. 2022) and dietary pigment sources.
Red, yellow and orange enhancers
Carotenoids are the major group of pigments fed to ornamental fish to generate the attractive yellow, red and orange colours on the skin (Table 1) (Das et al. 2016). Besides colouration, carotenoids are essential nutrients for the growth, reproduction and disease resistance in fish (Mente et al. 2011; Sathyaruban et al. 2021). More than 1,100 carotenoids have been identified, which are classified into two groups: carotenes (e.g. beta-carotene and lycopene) and xanthophylls (e.g. astaxanthin, lutein and canthaxanthin). These dietary carotenoids are synthetically produced or naturally extracted from animals (Sathyaruban et al. 2021), microorganisms (Venil et al. 2013), algae (Hu 2019) and plant sources (Singh et al. 2021). As there is no common carotenoid metabolic pathway in fish, its influence on the colouration of various ornamental fish species differs (Table 2). For instance, algae-based carotenoids effectively enhanced the pigmentation in koi carp (Gouveia et al. 2003; Sun et al. 2012) but not in cichlid (Kop and Durmaz 2008).
Synthetic astaxanthin is the most traded carotenoid, accounting for 25.70% of global carotenoids in the market in 2017 (FIORMARKETS global industry analysis, report id: 376,034). It is also the most widely-used carotenoid in ornamental fish feed such as in cichlid (Kop and Durmaz 2008), dwarf gourami (Baron et al. 2008), koi carp (Sun et al. 2012) and red devil cichlid (Pan and Chien 2009) being most efficient in enhancing body skin carotenoid deposition, thus colouration and intensity, compared to other synthetic and natural sources of carotenoids. However, not all fish are capable of effectively absorbing or utilising astaxanthin directly. Kaur and Shah (2017) classified aquatic organisms into three categories based on their metabolic actions on carotenoids: (i) converts lutein into astaxanthin (e.g. red carp), (ii) accumulates lutein and carotene in the tissue but incapable of converting these carotenoids into other forms (e.g. sea bream), (iii) converts beta-carotene into astaxanthin (e.g. prawn).
Considering the above, the selection of carotenoid supplement should be based on two criteria: (i) the type of carotenoid metabolism and ii) the targeted colour hue to be enhanced (Table 1). For instance, red carp, goldfish and fancy red carp can convert lutein (but not beta-carotene) to astaxanthin and be absorbed into the body (Teruhisa et al. 1972). Therefore, a supply of either astaxanthin or lutein is expected to enhance the redness in these fishes. By constant feeding of the appropriate carotenoids, the production and targeted colour hue and brightness in the ornamental fish is maintained, subsequently increasing its value.
The cost of synthetic carotenoids ranged between USD 250–2,000 kg−1, lower than the cost of natural-derived ones that are priced between USD 350–7,500 kg−1 (FIORMARKETS global industry analysis in 2017, report id: 376034). However, natural-derived carotenoids are becoming more popular due to the emerging concerns on the use of synthetic carotenoids in aquaculture, such as inferior quality (Capelli et al. 2013), high market price, limited choice and risk of aquarium pollution from the petrochemical solvents used during the commercial production (Das et al. 2016; Li et al. 2020). Thus, natural-derived carotenoids from photosynthetic organisms, including microalgae and plants are considered excellent substitutions to synthetic carotenoids in ornamental fish feeds (Hu 2019; Yusoff et al. 2020; Singh et al. 2021). For instance, Wagde et al. (2018) recommended that a supply of raw carrot fruits for as little as 1.33 mg kg−1 or 1.82 mg kg−1 of raw spinach could efficiently enhance the orange hue in swordtail. However, natural whole foods such as tomato and carrot containing high beta-carotene contents may not be suitable as the sole carotenoid source, as beta-carotene are deemed ineffective for enhancement of body pigmentation in many ornamental fish, such as cichlids (Kop and Durmaz 2008) and betta fish (Thongprajukaew et al. 2014).
Microalgae serve as an excellent pigment source, especially for algae-eating fishes (Rout et al. 2013), as they are capable of accumulating high amounts of various carotenoids (referred to as carotenogenesis), such as astaxanthin, beta-carotene, phytoene, lutein under extreme growth conditions, e.g. nitrogen deficiency (Henríquez et al. 2016; Pérez-Legaspi et al. 2020; Lu et al. 2021). In addition, numerous technological breakthroughs such as industrial bioreactor reformation and strain improvement of microalgae through genetic engineering have led to substantial cost reduction in natural carotenoid pigment production (Lin et al. 2019), a great boost to the ornamental fish industry. Astaxanthin-producing microalgae species, such as Haematococcus pluvialis, Chlorococcum sp., C. vulgaris are commercially cultured in many countries for human and animal consumptions (Pérez-Legaspi et al. 2020). These microalgal carotenoids possess very high bioactivities in numerous physiological functions, playing pivotal roles in the pharmaceutical and cosmetic industries (Patel et al. 2022). Thus, the feeding of microalgal carotenoids not only render comparable or better colouration effects than synthetic carotenoids (Gouveia et al. 2003; Pan and Chien 2009), but also offers health benefits.
Blue and violet enhancers
Phycobiliproteins are light-harvesting compounds that are essential for photosynthetic organisms such as cyanobacteria, red algae and crytophytes (Eriksen 2016). Four main types of phycobiliproteins occur in nature; phycoerythrin, phycocyanin, phycoerythrocyanin and allophycocyanin that are classified based on the absorption peak and chemical structures (Pagels et al. 2019).
Cyanobacteria and plants are recognised as the major sources of phycobiliproteins. Commercial fish feeds such as Artkua® blue color (https://artakua.com/en/portfolio-items/blue-color/) and Dainichi COLOR SUPREME (https://dainichi.com/CichlidFood-ColorSupreme.dmx) developed for blue aquarium fish colour enhancement, consist of phycobiliproteins-producing organisms, such as spirulina. A feeding trial conducted on angelfish (P. scalare) demonstrated overall colour enhancement (including blue) with the supplementation of spirulina flour to its commercial basal diet (Lili et al. 2021). Even though commercial production has yet to be reported, scientific efforts are currently focused on optimising the culture parameters and extraction methods, with particular emphasis on phycocyanin from spirulina (Silva et al. 2020; Tan et al. 2021).
Plant pigments, anthocyanins and betalains are commonly found in the Caryophyllales plants (except Caryophyllaceae and Molluginaceae) (Tanaka et al. 2008; Khoo et al. 2017; Rahimi et al. 2019). Anthocyanins are the causative pigments for plant blue, purple, violet, magenta, red and orange colouration (Jackman and Smith 1996). One interesting property of the anthocyanins is its dynamic colour changes based on the pH; it is red in acid condition, change to purple in pH 7 and becomes blue in alkaline condition, all due to its ionic structural modification (Tanaka et al. 2008; Khoo et al. 2017). Betalains occur as two types of pigments; betaxanthins (yellow-orange) and betacyanins (red-violet) (Jackman and Smith 1996). These two plant pigments have long been used as natural colourants in food (Tanaka et al. 2008). Previous studies have shown that 450 mg microencapsulated and 160 mg roselle-extracted anthocyanin could elevate the pigment cell density, growth and feed conversion rates in goldfish (Pérez-Escalante et al. 2012; Vanegas-Espinoza et al. 2019).
Other potential blue enhancers highlighted in the last two decades, include pyocyanin (DeBritto et al. 2020) and marennine (Gastineau et al. 2014). Pyocyanin is a bacterial biologically active phenazine pigment exclusively produced by 95% of the Pseudomonas aeruginosa strains (DeBritto et al. 2020; Gonçalves and Vasconcelos 2021). Due to its antibacterial and accessibility properties (Gonçalves and Vasconcelos 2021), the effects of pyocyanin supplement have been examined for growth, cytotoxicity response and carotenoid content in broilers (Ashour et al. 2021), rainbow trout (Priyaja et al. 2016) and cichlid (Ezhil and Narayanan 2013). Another blue enhancer is the marennine or marennine-like blue pigments which were discovered particularly in the diatom, genus Haslea and is known to enhance the greening of oysters (Gastineau et al. 2014). Despite the lack of data on the effects of bacterial blue pigment on ornamental fishes, it appears a good blue enhancer candidate for the ornamental fish industry.
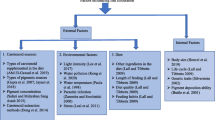
Rearing environment
The ornamental fish body colour is known to change dynamically in response to environmental stimuli such as changes in tank background colour and light (Table 3).
Tank colour
The effect of background (environment/tank) colour on cultured fish has been well studied since several decades ago and the evidence show strong correlation between fish colour pigments and tank colour (Eslamloo et al. 2015; Asra Nor Izaty and Norazmi-Lokman 2019; Díaz-Jiménez et al. 2021; McLean 2021). Rearing ornamental fish in colourful tank has been shown to induce temporary or permanent pigmentation, depending on the regulatory mechanisms associated with the skin chromatophores (Eslamloo et al. 2015; Asra Nor Izaty and Norazmi-Lokman 2019).
Most previous studies (Table 3) have demonstrated a decrease in hue brightness in fish reared in white coloured tank with varying growth rate. Skin response towards the tank colour is postulated to be mediated by the background matching mechanism, a neuroendocrine regulated process to control pigment transport, dispersal and aggregation in the chromatophores (McLean 2021). As colour change is energy-consuming (Rodgers et al. 2013), it is postulated that only tank colours that resemble the wild habitat of the species would favour growth. For instance, the natural habitat of yellow gourami fish is from the middle to the top layer of slow flowing rivers or lakes which is highly exposed to sunlight, thus a high affinity for white background. This species has been recorded with increased weight and paler body colour when reared in a white tank (Asra Nor Izaty and Norazmi-Lokman 2019). The body colour intensity of this species increased when reared in a green tank which matches the tank colour, but however, decreased in weight. In contrast, a different scenario was observed in the clownfish, Amphiprion ocellaris (Díaz-Jiménez et al. 2021). Clownfish lives in shallow seawater and is protected and sheltered by their sea anemone symbiont from direct sunlight; excessive light could hinder their reproduction (Fobert et al. 2019). The clownfish could still display a yellow body, although with almost invisible bands when grown in a white tank, but the intense red colour with defined black bands is only expressed when reared in the preferred black tank and this is coupled with a high survival rate. These evidences in yellow gourami (Asra Nor Izaty and Norazmi-Lokman 2019) and clownfish (Díaz-Jiménez et al. 2021) imply that background matching mechanism and fish global development are probably interdependently regulated.
Light
Light is indispensable for ornamental fish to function optimally, especially the influences of light intensity and photoperiod in a species that inhabit the top layer of an aquatic environment (Boeuf and Le Bail 1999). Light reflection provides vision and supports fish health (Stevens et al. 2017), where the optimum culture environment is attained when it is aligned to its natural habitat light spectrum. According to Marshall et al. (2019), in the presence of light, vision unlocks several crucial survival processes in fish, including mate choice, feeding, agonistic behaviours and camouflage. Studies have revealed that numerous ornamental fishes, such as goldfish (Baite et al. 2010) and angelfish (Rajeswari et al. 2017) exhibit poor growth and reduced body pigmentation when reared in poor or no lighting condition.
The level of light intensity influences the colour development and the effects may vary among species and is not permanent. For instance, the brightness of body and fins of clownfish increased when exposed to lower light intensity, while a more pronounced orange hue was reported at higher light intensity (Yasir and Qin 2009). In medaka fish, the skin xanthophore pigments aggregate (paler) under high light intensity exposure such as at 9,000 lx but the pigments disperse following a reduction in light intensity (Oshima et al. 1998). Similarly, the red body colour brightness of rainbow fish was also found to increase when exposed to low luminescence (Kelley et al. 2012).
The effects of different light sources on ornamental fish pigmentation reveal varying responses in different species. Artificial white light enhanced light brown and white bars to dark black with milky white bars in the clownfish (Uthayasiva et al. 2014) in contrast to the zebrafish, Danio rerio where melanosome aggregated temporarily and resulted in paler colour under the same lighting condition (Mueller and Neuhauss 2014). However, the melanosome aggregation in zebrafish was reversed when shifted to ultra-violet (UV) light, serving its protective function (increased pigmented layer) against UV light. Interestingly, body pattern could also be impacted by different lighting. The structural colour of lateral stripes on the neon tetra fish, Paracheirodon innesi changed from blue-green to indigo when moved away from light (Gur et al. 2015). Long wavelength light spectrum coupled with low light intensity was found to induce the spreading of pigmented area (Kelley et al. 2012).
Lighting greatly influences the fish circadian clock, affecting its development especially the melatonin rhythmic secretion which subsequently influences physiological activities, including skin pigmentation (Sánchez-Vázquez et al. 2019). The effects of photoperiod have been reported in several ornamental fishes, such as in the orange balloon molly, Poecilia latipinna (Zutshi and Singh 2021) and zebrafish (Shiraki et al. 2010). Both fish exhibited brighter skin colour, higher skin carotenoid content and delayed pigment aggregation under long photoperiod or continuous illumination and good growth development. However, most ornamental fish require a certain duration of dark period for their growth and breeding (Chen et al. 2020a, b).
Optimising rearing conditions: feed supplement, lighting and temperature conditions
In order to enhance pigmentation without compromising the health of the precious ornamental fish, the tank environment must emulate its wild habitat. This would require that the critical parameters such as feed supplement, lighting and temperature be jointly factored into the total environmental conditions and not to be considered in silos.
Sunlight accessibility and the background brightness (i.e. the dark leaf litters or the base soil colour) require full consideration. Rearing ornamental fish that favours darker environment is easier and straightforward, such as angelfish (Rajeswari et al. 2017) and yellow gourami fish (Asra Nor Izaty and Norazmi-Lokman 2019), as they grow well in the dark while pigmentation is enhanced. On the contrary, for fish that favour bright environment, pigment enhancement may be more challenging as they tend to turn paler under such conditions. Unfortunately, it is not ideal to rear these fishes under darker conditions to trigger the pigmentation enhancement, as this digresses from its natural habitat environment and hence require increased energy consumption (Rodgers et al. 2013), retarding its growth and interrupting social signalling (Kelley et al. 2016). Therefore, it would be better to opt for consistent carotenoid feeding to enhance the pigmentation.
Generally, the recommended minimum supplementation of the most common carotenoid, synthetic or natural astaxanthin, for pigmentation enhancement is 50 mg kg−1, a greater amount is likely to result in elevated colouration or deposition in the fish skin, as observed in cichlid (Kop and Durmaz 2008), dwarf gourami (Baron et al. 2008) and paradise fish (Lee and Lee 2008). Diler and Dilek (2002) suggested 35–100 ppm of carotenoid supplement in the breeding of several ornamental species at the juvenile stage, including goldfish and trout. However, beyond a certain limit, the improvement stops, but to date there has been no report of any lethal effect on the ornamental fish due to excess amounts. A certain duration is required for the colour intensity enhancement to occur after the onset of carotenoid feeding. In the orchid dottyback, Pseudochromis fridmani, at least 70 days of consistent astaxanthin feeding (vegetative H. pluvialis) is required to improve the magenta/raspberry colouration to achieve commercial standard (Jiang et al. 2019). A previous study also showed that inclusion of microbial carotenoid-rich food in X. helleri improved colouration and growth after 28-days (Dharmaraj and Dhevendaran 2011). The use of carotenogenic marine yeast, Rhodotorula paludigena VA 242 also enhanced the pigmentation and carotenoid content in Koi Carp in a 60-days feeding trial (Rekha et al. 2022).
As described earlier, blue colorants have shown positive results in preliminary blue colour enhancement studies. The current price of phycobiliprotein extract is estimated in the range of USD130 and USD 15,000 per gram, depending on the purity (Pagels et al. 2019). Thus, direct feeding with the blue pigment-rich organisms instead of purified pigments, such as spirulina (Marzorati et al. 2020) and Andean cyanobacteria (Galetović et al. 2020) may be more cost-efficient in improving blue colouration. In fact, spirulina is also an excellent source of other biological pigments (e.g. carotenoids), which may be fed to simultaneously enhance multi spectrum of colours, as observed on the angel fish in 10–40 days trials (Lili et al. 2021). Furthermore, the antibacterial and antioxidant properties of bacterial (pyocyanin and marennine) pigments confers resistance against common aquaculture pathogen such as Vibrio and Staphylococcus aureus (Ezhil and Narayanan 2013; Prasetiya et al. 2020). Thus, further research is needed to determine the optimum dosage that would offer health benefits without cytotoxicity induced in the ornamental fish.
Light settings including a minimum photoperiod of 12–14 h (Chen et al. 2020a, b) of white light under acceptable level of UV, coupled with light intensity similar to ambient sunlight and bright tank background, are recommended for ornamental fish species that naturally live in the top layer of aquatic environment. On the other hand, the opposite settings, such as darker tank base or background with limited light exposure and photoperiod is suitable for those naturally inhabiting near the underwater with limited sunlight exposure and leaf litters, for example the genus Betta (Ng 2016). As previously reported, constant exposure of light/darkness or red light increase the risks of growth abnormalities, while blue light induces better hatching rates and growth (Sánchez-Vázquez et al. 2019). Therefore, the light settings should be carefully designed to target both fish skin pigmentation and health.
Limited studies have been conducted on the effect of temperature on fish colouration although it is an important factor in fish generally, especially of exotic ornamental species. Careful setting and monitoring of temperature are essential to increase the survival rate of the ornamental fish. A study (Yanar et al. 2019) which investigated the thermal tolerance of 13 ornamental fish species showed that, goldfish, koi, doctorfish, sailfin molly and zebrafish could be reared outdoor in subtropical climate, whereas peppered cory, tiger barb and blue dolphin cichlid require shade in tropical and subtropical climates.
Advancing the ornamental fish industry through genetic engineering
Application of transgenic technology for genetic improvement of cultured fishes had been mooted in the early 1990s (Fletcher and Davies 1991), after the successful generation of transgenic goldfish (Zhu et al. 1985). Since the first report on the transgenic zebrafish (Glofish) (Wan et al. 2002), several transgenic lines of ornamental fish have been produced; medaka (Zeng et al. 2005; Cho et al. 2013, 2014) and tetra fish (Pan et al. 2008; Leggatt and Devlin 2020). As in Glofish, the fluorescent genes namely green fluorescent protein (gfp) (Wan et al. 2002; Zeng et al. 2005; Leggatt and Devlin 2020), yellow fluorescent protein (yfp), cyan fluorescent protein (cfp) (Cho et al. 2014) and red fluorescent protein (rfp) (Wan et al. 2002; Pan et al. 2008) have been successfully transferred to produce attractive luminous fish. The skeletal muscle gene promoters regulate the fluorescent genes to express various beautifully glowing colours under different light emission (Cho et al. 2014).
However, ethical concerns have been raised on the stability of the targeted traits, the sustainability of the transgenic fish as well as the environmental impacts of escapees, which could easily occur. While Snekser et al. (2006) reiterated that the social activities of Glofish was not affected, Howard et al. (2015) reported that the male Glofish was inferior in mate competition with wild type and the transgenic trait was completely wiped out after 15 generations. Furthermore, Glofish that were reared in natural headwater creeks showed early maturation and produced smaller eggs (Magalhães et al. 2022). Thus, the sustainability of the current transgenic technologies in ornamental fish industries remains in doubt.
Subsequently, technology advancement of next-generation sequencing has led to an explosion of new knowledge with wide applications, including for the improvement of the ornamental fish industry. Transcriptomic analysis has enabled the identification of genes responsible for the expression and regulation of skin colouration and pattern formation in numerous ornamental fish species and groups, such as zebrafish (Iwashita et al. 2006; Huang et al. 2021), Nile tilapia (Lu et al. 2022), Cyprinidae fish (Yu et al. 2021), goldfish (Gan et al. 2021) and red crucian carp (Zhang et al. 2017). In particular, extensive studies on the animal model zebrafish have generated massive output that have unravelled the regulation pathways of skin colouration. The detailed molecular mechanisms of fish pigmentation and the list of pigment genes are well-documented (Baxter et al. 2019; Luo et al. 2021). To date, several genetic tools have been developed to exploit gene editing of the targeted gene, such as zinc finger nucleases and TALENs (Barman et al. 2017). In particular. advanced tool such as CRISPR-Cas9 system (Huang et al. 2021; Yang et al. 2021; Yue et al. 2022), had facilitated the precise manipulation of skin pigmentation and body pattern in zebrafish and betta fish (Lu et al. 2022). Therefore, these pigment genes are excellent candidates in genetic engineering to manipulate fish colouration and patterns.
Quantitative trait locus (QTL) analysis is another potentially powerful tool in ornamental fish morphological enhancement. It has been explored in elucidating the causative pigment genes in ornamental fish species within populations (Irion and Nüsslein-Volhard 2019), such as in tilapia (Li et al. 2019; Liu et al. 2022; Xiong et al. 2022) and cichlid fishes (O’Quin et al. 2013; Albertson et al. 2014; Henning et al. 2014). Moreover, QTL mapping through genome-wide association studies (GWAS) has facilitated the simultaneous identification of multiple coloration and pattern traits that contribute to the diverse variation within a population (Zhang et al. 2022). For instance, the QTL mapping in GWAS of Betta fish identified the causative genes responsible for the various phenotypes produced by Royal Blue crossbred (Zhang et al. 2022). Certainly, this is another approach for moving forward in this area of study.
Manipulation of body colour through genetic engineering
Melanogenesis is the pathway that is responsible for melanin synthesis, which is modulated by several pathways such as MAPK, endothelin and WNT signalling pathways in the upstream. One of the crucial rate-limiting enzymes in this pathway is tyrosinase, encoded by tyr gene, which catalyses the oxidation of tyrosine to produce melanin and other pigments. This gene could be targeted to modify the black or brown colour in ornamental fish. For instance, tyr gene knockout via CRISPR-Cas9 in C. auratus successfully suppressed melanin production and downregulated the other melanin-producing genes such as mitf, dct and sox10, subsequently resulting in total albinism (Liu et al. 2019). The tyr was also mutated in cichlid, A. burtoni and inhibited melanophore differentiation (Li et al. 2021). Such approach would be useful to suppress the dominant top-embedded melanic black colour and permit the expression of the other attractive colourful pigments underneath the super-imposed layers of ornamental fish skin.
The microphthalmia-associated transcription factor (mitf), is the “master regulator” in the development and differentiation of melanophores (Luo et al. 2021). In ornamental red crucian carp, the decreased expressions of mitfa and other melanin synthesis-related genes are responsible for the body colour change from gray to red (Zhang et al. 2017). Moreover, the two variants of mitf, mitfa and mitfb, actively express and modulate the cutaneous and eye melanophores respectively (Luo et al. 2021), excellent candidate genes for the simultaneous manipulation of eye and skin black colour. Another potential gene for colour manipulation is the solute carrier family 24 member 5 (slc24a5), which is associated with the regulation of melanophore development and distribution in vertebrates. In particular, the slc24a5 mutation in ornamental Cyprinids lead to loss of melanophores in both larva and adult fish (Yu et al. 2021).
Aside from the black and brown colours, xanthophore-related genes revealed in transcriptomic studies also allow the manipulation of xanthophore-based colours (i.e. orange, red and yellow colour) in ornamental fish. In order to enhance the xanthophore-based colours, pathways leading to the accumulation of pteridines and carotenoids may be targeted. Transcriptomic studies conducted on red tilapia and Bengal loach, Botia dario, showed up-regulation in the genes related to lipid metabolism and carotenoid transport, i.e. scarb1, starb5, plin2, rdh12 and stard10 in the yellow and red pigmented regions (Tripathy et al. 2019; Fang et al. 2022). These genes may be considered to regulate carotenoid accumulation in the fish skin, subsequently increasing the carotenoid-based colour hue or intensity in the ornamental fish. On the other hand, genes involved in pteridine metabolism are also useful to engineer body colour change in ornamental fish. Both pts gene (yellow colour expression) and gchI (differentiation of xanthophore and melanophore) (Braasch et al. 2007) are crucial in pteridine metabolism and potential candidate genes for xanthophore-based colour enhancement.
Iridophore, a pigment cell that contains guanine or purine reflective plates, produces iridescence on the fish body. The iridophore-related genes are often linked to melanophore and xanthophores (Petratou et al. 2018). For instance, Higdon et al. (2013) identified 62 common enriched genes in melanophores and iridophores of zebrafish, implying that the development of melanophores and iridophores was regulated concurrently via cell–cell interaction. These genes are candidate genes that could potentially be altered to target the melanophores and iridophores simultaneously.
Several enzymes are involved in guanine metabolism during the formation of reflective iridescent plates within the iridophores (Higdon et al. 2013). The interactions among their multiple metabolic pathways recycles the phosphorobosyl pyrophosphate, a substrate for purine synthesis, completing the guanine cycle. One of these enzymes, leucocyte tyrosine kinase (ltk) acts as the core enzyme in modulation of iridophore establishment, proliferation and survival (Fadeev et al. 2016; Mo et al. 2017). During the modulation, the ligands of both leucocyte tyrosine kinase and anaplastic lymphoma kinase are speculated to induce iridophore differentiation (Mo et al. 2017), whereas transparent gene which encodes for the mitochondrial protein Mpv17 is responsible for iridophore survival (Krauss et al. 2013). Hence, the genes encoding these functional proteins could be targeted for iridophore density and survival.
The manipulation of iridophores may also be accomplished by targeting the transcriptional factors (TFs). Several TFs, sox10, tfec, pnp4a and mitfa have been identified as the key players in the complex gene regulatory network of iridophore (Petratou et al. 2018). In the neural crest, iridophore specification from the multiple neural crest progenitors involves a positive feedback loop between ltk and tfec which is sox10-dependent. The sox10 gene is also actively involved in driving the molecular mechanism in iridophore development and couples with tfec in regulating the expression of the key differentiation gene, pnp4a. As pnp4a mutation led to the total shutdown of guanine metabolism in the medaka iridophores (Kimura et al. 2017), the potential of pnp4a gene engineering in producing attractive see-through skin phenotype in ornamental fish could be exploited.
Manipulation of body pattern through genetic engineering
In addition to colours, the exquisite body patterns too are of high value (Fig. 1). Based on zebrafish and anemonefish models, these pigment patterns predominantly arise from the complex cellular interactions and molecular mechanisms underlying the melanophores, xanthophores and iridophores (Patterson and Parichy 2013; Klann et al. 2021). Therefore, establishing the desired pigment pattern through gene editing is deemed more complicated, since a common regulatory gene mutation may trigger undesirable changes in the arrangement of more than one pigment cell types. For instance, when the transparent gene in zebrafish was mutated, the melanic stripes turned into spots following the huge reduction in iridophores and melanophores (Krauss et al. 2013).
The melanic horizontal and vertical bar stripes are common pigment patterns observed in many ornamental fish, such as zebrafish, discus, red tilapia, clownfish etc. The stripes are the result of the unique organisation of different pigment cell types (Klann et al. 2021; McCluskey et al. 2021a), where melanophores form black stripes, xanthophores form light interstripes while iridophores exist in both regions (Krauss et al. 2013). A conserved zinc finger gene (bnc2) is postulated to be the “switch-gene” that determines the fate of stripe patterning in the zebrafish. It acts as the “master booster” in promoting development and establishment of the three chromatophores; a mutation in the bnc2 would lead to the loss of stripes and interstripes (Patterson and Parichy 2013). Furthermore, the properties of stripes could be controlled through asip and tspan genes. The asip1-mutant induces changes in the number of different chromatophores in the skin and scales, subsequently increasing the thickness of the melanic horizontal stripes. On the other hand, tspan is responsible in maintaining the stripe boundaries, where mutated tspan created intermingled stripes in the mutants (Inoue et al. 2014). White bar stripes are modulated through another different set of genes. The spatial white vertical bar stripes (iridophore-based) in clownfish displayed on top of the dark body background are regulated by several requisite genes including fhl2a, fhl2b, saiyan, gpnmb, and apod1a, through the maintenance and organisation of iridophores (Salis et al. 2019).
Manipulation of the number of horizontal and bar stripes, however, requires different approaches, as the mechanisms for their formation are different. Horizontal stripes are formed when melanophores proliferate along the horizontal myosepta, starting from metamorphosis into adult, whereas the bar number and position dynamically changes as the body length increases (Hendrick et al. 2019). The difference in the formation of the two patterns most probably depends on distinct regulatory pathways, therefore it is strongly recommended to target the genes associated with neural crest progenitor migration, during the metamorphosis stage, to reduce or increase the number of bars.
Fish display dorso-ventral countershading pattern, with dark and light colour, respectively dominant at the dorsal and ventral surfaces. The agouti-signalling protein, encoded by asip1, known to be the antagonist of melanisation (Cerdá-Reverter et al. 2005), acts as one of the components in maintaining the countershading pattern in zebrafish and goldfish (Cal et al. 2019). By disrupting the Asip1, the melanisation could spread and become established in the ventral region (Cal et al. 2019). Such axial developments (e.g. anterior–posterior, dorsal–ventral) are also under the influences of pattern developmental genes, as observed in the African cichlids (Ahi and Sefc 2017b, 2017a) and wild discus fish (Ng et al. 2023).
There is a scarcity of information on the mechanism of spot formation in ornamental fish. The manifestation of either stripes or spots on two closely-related zebrafish (D. quagga and D. kyathit) was investigated on early adult stage individuals, which coincides with the establishment and organisation period of the chromatophores (McCluskey et al. 2021b). Although the switching between stripes and spots could be achieved through a single gene change as reported in the widely studied D. rerio, complex mechanisms involving multiple genes and gene signallings were detected in these two zebrafish species (McCluskey et al. 2021b). As in red tilapia, wintering black spot was revealed to be regulated by multiple environmental factors under the influences of three potential causative genes, namely sena-4c, uvssa and htr2cl1 (Xiong et al. 2022).
Interestingly, growth rate may also be involved in fish pattern formation. Changes in growth rate and sequences of chromatophores could promote further pattern modulation and orientation on various pattern formation, including vertical stripes and maze-like patterns in the zebra fish (Owen et al. 2021). For instance, directional elongation of stripe patterning was driven by the spreading of the chromatophores during growth and subsequently affects the pigment orientation in zebrafish (Míguez and Muñuzuri 2006).
Nonetheless, it is definitely more challenging to manipulate body patterns in ornamental fish, compared to the body colour changes, due to the ambiguous and species-specific coordination among different chromatophores, which is deemed more complicated in some ornamental fish species. The advancement of next generation sequencing and efficient gene editing tool such as CRISPR could facilitate faster target gene discovery for pattern manipulation.
Challenges in pigment gene disruption
The modification of endogenous pigment genes can be hindered by pleiotropic effects of genes regulating the body colouration or patterns in some ornamental fish. For instance, the expression of melanic side-spotting patterns varies among members of family Poeciliidae due to the influences of multiple selective factors and polygenic control (Zerulla and Stoddard 2021). The concerted interaction among multiple genomic regions regulating pigmentation (hypermelanic phenotype), vision and sleep has been documented in the blind cave fish (Astyanax mexicanus) (Gross et al. 2016; O'Gorman et al. 2021). Under such pleiotropic effects of pigment genes, imprudent attempt of mutating a candidate pigment gene will rock the boat and likely lead to severe disruption on other survival traits. Although the identification and selection of causative genes for colouration and pattern modifications are highly challenging, it is a highly worthwhile effort in producing novel, attractive domesticated ornamental fish.
Conclusion
Manipulating the colour and patterning in ornamental fish could be achieved through multiple environmental stimuli and genetic factors. Focusing on high dietary supplement of natural carotenoids (versus synthetic) and non-carotenoid pigments has proven to be a more efficient approach to enhance the body colour hue and brightness while also improving the general health of the ornamental fish. Settings of optimum environmental variables, such as tank colour, light intensity, light sources or colour and photoperiod, are important considerations. With the flourishing of genetic engineering techniques, the manipulation of pigment genes would facilitate the production of mutated varieties in ornamental fish species with various body colours and patterns, circumventing time-consuming traditional selective breeding process. However, as many candidate genes are also crucially involved in other developmental and survival processes, careful consideration is warranted in many aspects (e.g. choice of genetic tools) to reduce the risk of severe disruption on the fish growth and survival. The effective collaboration among scientists and ornamental fish industry players is of prime importance to initiate knowledge-sharing, amelioration in fish breeding techniques and establish functional taxonomic classification on new ornamental fish varieties.
Data availability
All data generated or analysed during this study are included in this published article.
References
Ahi EP, Sefc KM (2017a) Anterior-posterior gene expression differences in three Lake Malawi cichlid fishes with variation in body stripe orientation. PeerJ 5:e4080
Ahi EP, Sefc KM (2017b) A gene expression study of dorso-ventrally restricted pigment pattern in adult fins of Neolamprologus meeli, an African cichlid species. PeerJ 5:e2843
Ahi EP, Lecaudey LA, Ziegelbecker A, Steiner O, Glabonjat R, Goessler W, Hois V, Wagner C, Lass A, Sefc KM (2020) Comparative transcriptomics reveals candidate carotenoid color genes in an East African cichlid fish. BMC Genom 21(1):1–15. https://doi.org/10.1186/s12864-020-6473-8
Albertson RC, Powder KE, Hu Y, Coyle KP, Roberts RB, Parsons KJ (2014) Genetic basis of continuous variation in the levels and modular inheritance of pigmentation in cichlid fishes. Mol Ecol 23(21):5135–5150. https://doi.org/10.1111/mec.12900
Ashour EA, Farsi RM, Alaidaroos BA, Abdel-Moneim A-ME, El-Saadony MT, Osman AO, Abou Sayed-Ahmed ET, Albaqami NM, Shafi ME, Taha AE (2021) Impacts of dietary supplementation of pyocyanin powder on growth performance, carcase traits, blood chemistry, meat quality and gut microbial activity of broilers. Ital J Anim Sci 20(1):1357–1372
Asra Nor Izaty A, Norazmi-Lokman NH (2019) Effects of tank colour on body colour intensity and growth of Yellow Gourami Trichopodus trichopterus. Universiti Malaysia Terengganu J Undergrad Res 1(4):9–18. https://doi.org/10.46754/umtjur.v1i4.87
Baite J, Verma AK, Prakash C, Chandrakant MH, Saharan N (2010) Effect of light intensity on growth, survival and skin colour of goldfish (Carassius auratus Linnaeus). J Aquacult Trop 25(1–4):47–59
Barman HK, Rasal KD, Chakrapani V, Ninawe AS, Vengayil DT, Asrafuzzaman S, Sundaray JK, Jayasankar P (2017) Gene editing tools: state-of-the-art and the road ahead for the model and non-model fishes. Transgenic Res 26:577–589
Baron M, Davies S, Alexander L, Snellgrove D, Sloman KA (2008) The effect of dietary pigments on the coloration and behaviour of flame-red dwarf gourami, Colisa Lalia. Anim Behav 75(3):1041–1051. https://doi.org/10.1016/j.anbehav.2007.08.014
Baxter LL, Watkins-Chow DE, Pavan WJ, Loftus SK (2019) A curated gene list for expanding the horizons of pigmentation biology. Pigment Cell Melanoma Res 32(3):348–358. https://doi.org/10.1111/pcmr.12743
Boeuf G, Le Bail PY (1999) Does light have an influence on fish growth? Aquac 177(1):129–152. https://doi.org/10.1016/S0044-8486(99)00074-5
Braasch I, Schartl M, Volff J-N (2007) Evolution of pigment synthesis pathways by gene and genome duplication in fish. BMC Evol Biol 7(1):74. https://doi.org/10.1186/1471-2148-7-74
Cal L, Suarez-Bregua P, Cerdá-Reverter JM, Braasch I, Rotllant J (2017) Fish pigmentation and the melanocortin system. Comp Biochem Physiol A Mol Integr Physiol 211:26–33. https://doi.org/10.1016/j.cbpa.2017.06.001
Cal L, Suarez-Bregua P, Comesaña P, Owen J, Braasch I, Kelsh R, Cerdá-Reverter JM, Rotllant J (2019) Countershading in zebrafish results from an Asip1 controlled dorsoventral gradient of pigment cell differentiation. Sci Rep 9(1):3449. https://doi.org/10.1038/s41598-019-40251-z
Capelli B, Bagchi D, Cysewski GR (2013) Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutraceutical supplement. Nutrafoods 12(4):145–152. https://doi.org/10.1007/s13749-013-0051-5
Cerdá-Reverter JM, Haitina T, Schiöth HB, Peter RE (2005) Gene structure of the goldfish agouti-signaling protein: a putative role in the dorsal-ventral pigment pattern of fish. Endocrinology 146(3):1597–1610. https://doi.org/10.1210/en.2004-1346%JEndocrinology
Cerdá-Reverter JM, Agulleiro MJ, Guillot R, Sánchez E, Ceinos R, Rotllant J (2011) Fish melanocortin system. Eu J Pharmacol 660(1):53–60
Chen JY, Zeng C, Jerry DR, Cobcroft JM (2020b) Recent advances of marine ornamental fish larviculture: broodstock reproduction, live prey and feeding regimes, and comparison between demersal and pelagic spawners. Rev Aquac 12(3):1518–1541. https://doi.org/10.1111/raq.12394
Chen D, Zhang Q, Tang W, Huang Z, Wang G, Wang Y, Shi J, Xu H, Lin L, Li Z, Chi W, Huang L, Xia J, Zhang X, Guo L, Wang Y, Ma P, Tang J, Zhou G, Liu M, Liu F, Hua X, Wang B, Shen Q, Jiang Q, Lin J, Chen X, Wang H, Dou M, Liu L, Pan H, Qi Y, Wu B, Fang J, Zhou Y, Cen W, He W, Zhang Q, Xue T, Lin G, Zhang W, Liu Z, Qu L, Wang A, Ye Q, Chen J, Zhang Y, Ming R, Van Montagu M, Tang H, Van de Peer Y, Chen Y, Zhang J (2020a) The evolutionary origin and domestication history of goldfish (Carassius auratus). Proc Natl Acad Sci USA 117(47): 29775–29785. https://doi.org/10.1073/pnas.2005545117
Cho YS, Lee SY, Kim DS, Nam YK (2013) Characterization of stable fluorescent transgenic marine medaka (Oryzias dancena) lines carrying red fluorescent protein gene driven by myosin light chain 2 promoter. Transgenic Res 22(4):849–859. https://doi.org/10.1007/s11248-012-9675-2
Cho YS, Lee SY, Kim DS, Nam YK (2014) A cyan fluorescent protein gene (cfp)-transgenic marine medaka Oryzias dancena with potential ornamental applications. Fish Aquat Sci 17(4):479–486. https://doi.org/10.5657/FAS.2014.0479
Costa DC, Mattioli CC, Silva WS, Takata R, Leme FOP, Oliveira AL, Luz RK (2016) The effect of environmental colour on the growth, metabolism, physiology and skin pigmentation of the carnivorous freshwater catfish Lophiosilurus alexandri. J Fish Biol 90(3):922–935. https://doi.org/10.1111/jfb.13208
Das A, Biswas SJJoA, Biology M (2016) Carotenoids and pigmentation in ornamental fish. J Aquac Mar Biol 4(4):00093. https://doi.org/10.15406/jamb.2016.04.00093
DeBritto S, Gajbar TD, Satapute P, Sundaram L, Lakshmikantha RY, Jogaiah S, Ito S-i (2020) Isolation and characterization of nutrient dependent pyocyanin from Pseudomonas aeruginosa and its dye and agrochemical properties. Sci Rep 10(1):1542. https://doi.org/10.1038/s41598-020-58335-6
Dharmaraj S, Dhevendaran K (2011) Application of microbial carotenoids as a source of colouration and growth of ornamental fish Xiphophorus helleri. World J Fish Mar Sci 3(2):137–144
Díaz-Jiménez L, Hernández-Vergara MP, Pérez-Rostro CI, Olvera-Novoa MÁ (2021) The effect of two carotenoid sources, background colour and light spectrum on the body pigmentation of the clownfish Amphiprion ocellaris. Aquac Res 52(7):3052–3061. https://doi.org/10.1111/are.15149
Diler I, Dilek K (2002) Significance of pigmentation and use in aquaculture. Turkish J Fish Aquat Sci 2:97–99
Ebeneezar S, Prabu DL, Chandrasekar S, Tejpal CS, Madhu K, Sayooj P, Vijayagopal P (2020) Evaluation of dietary oleoresins on the enhancement of skin coloration and growth in the marine ornamental clown fish, Amphiprion ocellaris (Cuvier, 1830). Aquac 529:735728. https://doi.org/10.1016/j.aquaculture.2020.735728
Eriksen NT (2016) Research trends in the dominating microalgal pigments, -carotene, astaxanthin, and phycocyanin used in feed, in foods, and in health applications. J Nutr Food Sci 6(3):507. https://doi.org/10.4172/2155-9600.1000507
Eslamloo K, Akhavan SR, Eslamifar A, Henry MA (2015) Effects of background colour on growth performance, skin pigmentation, physiological condition and innate immune responses of goldfish. Carassius Auratus Aquac Res 46(1):202–215. https://doi.org/10.1111/are.12177
Ezhil J, Narayanan M (2013) Pseudomonas aeruginosa as a potential probiont and pigment enhancer in the ornamental cichlid. Pseudotropheus Lombardoi Proc Zool Soc 66(2):154–158. https://doi.org/10.1007/s12595-012-0050-3
Fadeev A, Krauss J, Singh AP, Nüsslein-Volhard C (2016) Zebrafish leucocyte tyrosine kinase controls iridophore establishment, proliferation and survival. Pigment Cell Melanoma Res 29(3):284–296. https://doi.org/10.1111/pcmr.12454
Fang W, Huang J, Li S, Lu J (2022) Identification of pigment genes (melanin, carotenoid and pteridine) associated with skin color variant in red tilapia using transcriptome analysis. Aquac 547:737429. https://doi.org/10.1016/j.aquaculture.2021.737429
Fletcher GL, Davies PL (1991). Transgenic fish for aquaculture. genetic engineering: principles and methods. (ed) J. K. Setlow. Boston, MA, Springer US: 331–370
Fobert EK, Burke da Silva K, Swearer SE (2019) Artificial light at night causes reproductive failure in clownfish. Biol Lett 15(7):20190272. https://doi.org/10.1098/rsbl.2019.0272
Galetović A, Seura F, Gallardo V, Graves R, Cortés J, Valdivia C, Núñez J, Tapia C, Neira I, Sanzana S, Gómez-Silva B (2020) Use of phycobiliproteins from atacama cyanobacteria as food colorants in a dairy beverage prototype. Foods 9(2):244. https://doi.org/10.3390/foods9020244
Gan W, Chung-Davidson Y-W, Chen Z, Song S, Cui W, He W, Zhang Q, Li W, Li M, Ren J (2021) Global tissue transcriptomic analysis to improve genome annotation and unravel skin pigmentation in goldfish. Sci Rep 11(1):1815. https://doi.org/10.1038/s41598-020-80168-6
Gastineau R, Turcotte F, Pouvreau JB, Morançais M, Fleurence J, Windarto E, Prasetiya FS, Arsad S, Jaouen P, Babin M, Coiffard L, Couteau C, Bardeau JF, Jacquette B, Leignel V, Hardivillier Y, Marcotte I, Bourgougnon N, Tremblay R, Deschênes JS, Badawy H, Pasetto P, Davidovich N, Hansen G, Dittmer J, Mouget JL (2014) Marennine, promising blue pigments from a widespread Haslea diatom species complex. Mar Drugs 12(6):3161–3189. https://doi.org/10.3390/md12063161
Gjedrem T, Baranski M (2009) Selective breeding in aquaculture: an introduction. reviews: methods and technologies in fish biology and fisheries. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2773-3
Gjedrem T, Baranski M (2010) Selective breeding in aquaculture: an introduction (Vol. 10). Springer Science & Business Media
Gonçalves T, Vasconcelos U (2021) Colour me blue: the history and the biotechnological potential of pyocyanin. Molecules 26(4):927
Gouveia L, Rema P (2005) Effect of microalgal biomass concentration and temperature on ornamental goldfish (Carassius auratus) skin pigmentation. Aquac Nutr 11(1):19–23. https://doi.org/10.1111/j.1365-2095.2004.00319.x
Gouveia L, Rema P, Pereira O, Empis J (2003) Colouring ornamental fish (Cyprinus carpio and Carassius auratus) with microalgal biomass. Aquac Nutr 9(2):123–129. https://doi.org/10.1046/j.1365-2095.2003.00233.x
Gross JB, Powers AK, Davis EM, Kaplan SA (2016) A pleiotropic interaction between vision loss and hypermelanism in Astyanax mexicanus cave x surface hybrids. BMC Evol Biol 16(1):145. https://doi.org/10.1186/s12862-016-0716-y
Gui J, Zhou L (2010) Genetic basis and breeding application of clonal diversity and dual reproduction modes in polyploid Carassius auratus gibelio. Sci China Life Sci 53:409–415. https://doi.org/10.1007/s11427-010-0092-6
Gur D, Palmer BA, Leshem B, Oron D, Fratzl P, Weiner S, Addadi L (2015) The mechanism of color change in the neon tetra fish: a light-induced tunable photonic crystal array. Angew Chem Int Ed 54(42): 12426–12430. https://doi.org/10.1002/anie.201502268
Hendrick LA, Carter GA, Hilbrands EH, Heubel BP, Schilling TF, Le Pabic P (2019) Bar, stripe and spot development in sand-dwelling cichlids from Lake Malawi. EvoDevo 10(1):18. https://doi.org/10.1186/s13227-019-0132-7
Henning F, Lee HJ, Franchini P, Meyer A (2014) Genetic mapping of horizontal stripes in Lake Victoria cichlid fishes: benefits and pitfalls of using RAD markers for dense linkage mapping. Mol Ecol 23(21):5224–5240
Henríquez V, Escobar C, Galarza J, Gimpel J (2016) Carotenoids in Microalgae. In: Stange C (ed) Carotenoids in nature: biosynthesis, regulation and function. Springer International Publishing, Cham, pp 219–237
Higdon CW, Mitra RD, Johnson SL (2013) Gene expression analysis of zebrafish melanocytes, iridophores, and retinal pigmented epithelium reveals indicators of biological function and developmental origin. PLoS ONE 8(7):e67801. https://doi.org/10.1371/journal.pone.0067801
Howard RD, Rohrer K, Liu Y, Muir WM (2015) Mate competition and evolutionary outcomes in genetically modified zebrafish (Danio rerio). Evolution 69(5):1143–1157. https://doi.org/10.1111/evo.12662
Hu IC (2019) Chapter 14—production of potential coproducts from microalgae. In: Pandey A, Chang JS, Soccol CR, Lee DJ, Chisti Y (eds) Biofuels from Algae, 2nd edn. Elsevier, pp 345–358
Huang D, Lewis VM, Foster TN, Toomey MB, Corbo JC, Parichy DM (2021) Development and genetics of red coloration in the zebrafish relative Danio albolineatus. eLife 10:e70253. https://doi.org/10.7554/eLife.70253
Hulata G (1995) A review of genetic improvement of the common carp (Cyprinus carpio L.) and other cyprinids by crossbreeding, hybridization and selection. Aquac 129(1–4):143–155. https://doi.org/10.1016/0044-8486(94)00244-I
Inoue S, Kondo S, Parichy DM, Watanabe M (2014) Tetraspanin 3c requirement for pigment cell interactions and boundary formation in zebrafish adult pigment stripes. Pigment Cell Melanoma Res 27(2):190–200. https://doi.org/10.1111/pcmr.12192
Irion U, Nüsslein-Volhard C (2019) The identification of genes involved in the evolution of color patterns in fish. Curr Opin Genet Dev 57:31–38. https://doi.org/10.1016/j.gde.2019.07.002
Iwashita M, Watanabe M, Ishii M, Chen T, Johnson SL, Kurachi Y, Okada N, Kondo S (2006) Pigment pattern in jaguar/obelix zebrafish is caused by a kir7.1 mutation: implications for the regulation of melanosome movement. PLoS Genet 2(11):e197. https://doi.org/10.1371/journal.pgen.0020197
Jackman RL, Smith JL (1996). Anthocyanins and betalains. Natural Food Colorants. G. A. F. Hendry and J. D. Houghton. Boston, MA, Springer US: 244–309
Jiang J, Nuez-Ortin W, Angell A, Zeng C, de Nys R, Vucko MJ (2019) Enhancing the colouration of the marine ornamental fish Pseudochromis fridmani using natural and synthetic sources of astaxanthin. Algal Res 42:101596. https://doi.org/10.1016/j.algal.2019.101596
Kaur R, Shah TK (2017) Role of feed additives in pigmentation of ornamental fishes. Int J Fish Aquat Sci 5(2):684–686
Kelley JL, Phillips B, Cummins GH, Shand J (2012) Changes in the visual environment affect colour signal brightness and shoaling behaviour in a freshwater fish. Anim Behav 83(3):783–791. https://doi.org/10.1016/j.anbehav.2011.12.028
Kelley JL, Rodgers GM, Morrell LJ (2016) Conflict between background matching and social signalling in a colour-changing freshwater fish. R Soc Open Sci 3(6):160040. https://doi.org/10.1098/rsos.160040
Khoo HE, Azlan A, Tang ST, Lim SM (2017) Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr Res 61(1):1361779. https://doi.org/10.1080/16546628.2017.1361779
Kimura T, Takehana Y, Naruse K (2017) pnp4a is the causal gene of the medaka iridophore mutant guanineless. G3: Genes Genomes Genet 7(4):1357–1363. https://doi.org/10.1534/g3.117.040675
Kimura T (2021) Pigments in teleosts and their biosynthesis. In: Hashimoto H, Goda M, Futahashi R, Kelsh R and Akiyama T (eds) Pigments, pigment cells and pigment patterns. Springer Singapore, Singapore, 127–148
King TA (2019) Wild caught ornamental fish: a perspective from the UK ornamental aquatic industry on the sustainability of aquatic organisms and livelihoods. J Fish Biol 94(6):925–936. https://doi.org/10.1111/jfb.13900
Kirkwood JK (2012) Selective breeding: making the welfare consequences clear. Vet Rec 170(21):535–537. https://doi.org/10.1136/vr.e3344
Klann M, Mercader M, Carlu L, Hayashi K, Reimer JD, Laudet V (2021) Variation on a theme: pigmentation variants and mutants of anemonefish. EvoDevo 12(1):8. https://doi.org/10.1186/s13227-021-00178-x
Komiyama T, Kobayashi H, Tateno Y, Inoko H, Gojobori T, Ikeo K (2009) An evolutionary origin and selection process of goldfish. Gene 430(1–2):5–11. https://doi.org/10.1016/j.gene.2008.10.019
Kop A, Durmaz Y (2008) The effect of synthetic and natural pigments on the colour of the cichlids (Cichlasoma severum sp., Heckel 1840). Aquac Int 16(2):117–122. https://doi.org/10.1007/s10499-007-9130-1
Krauss J, Astrinides P, Frohnhöfer HG, Walderich B, Nüsslein-Volhard C (2013) transparent, a gene affecting stripe formation in Zebrafish, encodes the mitochondrial protein Mpv17 that is required for iridophore survival. Biol Open 2(7):703–710. https://doi.org/10.1242/bio.20135132
Kurnia A, Nur I, Muskita WH, Hamzah M, Iba W, Patadjai RS, Balubi AM, Kalidupa N (2019) Improving skin coloration of koi carp (Cyprinus carpio) fed with red dragon fruit peel meal. Aquac Aquar Conserv Legis 12(4):1045–1053
Kwon, YM, Vranken N, Hoge C, Lichak MR, Norovich AL, Francis KX, Camacho-Garcia J, Bista I, Wood J, Mccarthy S, Chow W, Tan HH, Howe K, Bandara S, Lintig JV, Rüber L, Durbin R, Svardal H, Bendesky A (2022). Genomic consequences of domestication of the Siamese fighting fish.. Sci Adv 8(10):eabm4950. https://doi.org/10.1126/sciadv.abm4950
Lee CR, Lee SM (2008) Effect of dietary supplementation of pigment sources on pigmentation of the round tailed paradise fish Macropodus chinensis and the pale chub Zacco platypus. J Aquac 21(4):213–217
Leggatt RA, Devlin RH (2020) Fluorescent protein transgenesis has varied effects on behaviour and cold tolerance in a tropical fish (Gymnocorymbus ternetzi): implications for risk assessment. Fish Physiol and Biochem 46(1):395–403. https://doi.org/10.1007/s10695-019-00725-3
Li CY, Steighner JR, Sweatt G, Thiele TR, Juntti SA (2021) Manipulation of the tyrosinase gene permits improved CRISPR/Cas editing and neural imaging in cichlid fish. Sci Rep 11(1):15138. https://doi.org/10.1038/s41598-021-94577-8
Li BJ, Zhu ZX, Gu XH, Lin HR, Xia JH (2019) QTL Mapping for Red Blotches in Malaysia Red Tilapia (Oreochromis spp.). Mar Biotechnol 21(3):384–395. https://doi.org/10.1007/s10126-019-09888-9
Li SF, Wang CH (2001) Genetic diversity and selective breeding of red common carps in China. Naga - The ICLARM Quarterly 24(3&4):56-63
Li X, Wang X, Duan C, Yi S, Gao Z, Xiao C, Agathos SN, Wang G, Li J (2020) Biotechnological production of astaxanthin from the microalga Haematococcus pluvialis. Biotechnol Adv 43:107602. https://doi.org/10.1016/j.biotechadv.2020.107602
Lili W, Rizal A, Herman RG, Ramadhan RM (2021) Effect of Spirulina flour on changes of color intensity in angelfish (Pterophyllum scalare) strain three color. Asian J Fish Aquat Res 11(2):52–58
Lin B, Cui Y, Yan M, Wang Y, Gao Z, Meng C, Qin S (2019) Construction of astaxanthin metabolic pathway in the green microalga Dunaliella viridis. Algal Res 44:101697. https://doi.org/10.1016/j.algal.2019.101697
Lind CE, Ponzoni RW, Nguyen NH, Khaw HL (2012) Selective breeding in fish and conservation of genetic resources for aquaculture. Reprod Domest Anim 47(s4):255–263. https://doi.org/10.1111/j.1439-0531.2012.02084.x
Liu Q, Qi Y, Liang Q, Song J, Liu J, Li W, Shu Y, Tao M, Zhang C, Qin Q, Wang J, Liu S (2019) Targeted disruption of tyrosinase causes melanin reduction in Carassius auratus cuvieri and its hybrid progeny. Sci China Life Sci 62(9):1194–1202. https://doi.org/10.1007/s11427-018-9404-7
Liu F, Sun F, Kuang GQ, Wang L, Yue GH (2022) Identification of Pmel17 for golden skin color using linkage mapping in Mozambique tilapia. Aquac 548(2):737703. https://doi.org/10.1016/j.aquaculture.2021.737703
Lu Q, Li H, Zou Y, Liu H, Yang L (2021) Astaxanthin as a microalgal metabolite for aquaculture: A review on the synthetic mechanisms, production techniques, and practical application. Algal Res 54:102178. https://doi.org/10.1016/j.algal.2020.102178
Lu B, Wang C, Liang G, Xu M, Kocher TD, Sun L, Wang D (2022) Generation of ornamental Nile tilapia with distinct gray and black body color pattern by csf1ra mutation. Aquac Rep 23:101077. https://doi.org/10.1016/j.aqrep.2022.101077
Luo M, Lu G, Yin H, Wang L, Atuganile M, Dong Z (2021) Fish pigmentation and coloration: molecular mechanisms and aquaculture perspectives. Rev Aquac 13(4):2395–2412
Magalhães ALB, Brito MFG, Silva LGM (2022) The fluorescent introduction has begun in the southern hemisphere: presence and life-history strategies of the transgenic zebrafish Danio rerio (Cypriniformes: Danionidae) in Brazil. Stud Neotrop Fauna and Environ 1–13. https://doi.org/10.1080/01650521.2021.2024054
Maiti MK, Bora D, Nandeesha T, Sahoo S, Adarsh B, Kumar S, Studies A (2017) Effect of dietary natural carotenoid sources on colour enhancement of Koi carp, Cyprinus carpio L. Int J Fish Aquat Stud 5(4):340–345
Margareta A, Nikhlani A (2021) The effectiveness of supplementation of spirulina sp and astaxanthin within feed to color quality of comet fish (Carassius auratus). Jurnal Ilmu Kelautan 7(1):1–6
Marshall NJ, Cortesi F, de Busserolles F, Siebeck UE, Cheney KL (2019) Colours and colour vision in reef fishes: past, present and future research directions. J Fish Biol 95(1):5–38. https://doi.org/10.1111/jfb.13849
Marzorati S, Schievano A, Idà A, Verotta L (2020) Carotenoids, chlorophylls and phycocyanin from Spirulina: supercritical CO2 and water extraction methods for added value products cascade. Green Chem 22(1):187–196. https://doi.org/10.1039/C9GC03292D
McCluskey BM, Liang Y, Lewis VM, Patterson LB, Parichy DM (2021a) Pigment pattern morphospace of Danio fishes: evolutionary diversification and mutational effects. Biol Open. https://doi.org/10.1242/bio.058814
McCluskey BM, Uji S, Mancusi JL, Postlethwait JH, Parichy DM (2021b) A complex genetic architecture in zebrafish relatives Danio quagga and D. kyathit underlies development of stripes and spots. PLoS Genet 17(4):e1009364. https://doi.org/10.1371/journal.pgen.1009364
McLean E (2021) Fish tank color: an overview. Aquac 530:735750. https://doi.org/10.1016/j.aquaculture.2020.735750
Mente E, Karalazos V, Karapanagiotidis IT, Pita C (2011) Nutrition in organic aquaculture: an inquiry and a discourse. Aquac Nutr 17(4):e798–e817. https://doi.org/10.1111/j.1365-2095.2010.00846.x
Míguez DG, Muñuzuri AP (2006) On the orientation of stripes in fish skin patterning. Biophys Chem 124(2):161–167. https://doi.org/10.1016/j.bpc.2006.06.014
Mo ES, Cheng Q, Reshetnyak AV, Schlessinger J, Nicoli S (2017) Alk and Ltk ligands are essential for iridophore development in zebrafish mediated by the receptor tyrosine kinase Ltk. Proc Natl Acad Sci USA 114(45):12027–12032. https://doi.org/10.1073/pnas.1710254114
Mohammadiazarm H, Maniat M, Ghorbanijezeh K, Ghotbeddin N (2021) Effects of spirulina powder (Spirulina platensis) as a dietary additive on Oscar fish, Astronotus ocellatus: Assessing growth performance, body composition, digestive enzyme activity, immune-biochemical parameters, blood indices and total pigmentation. Aquac Nutr 27(1):252–260. https://doi.org/10.1111/anu.13182
Monica J, Neelakantan V, Seenappa D (2019) Effect of dietary incorporation of anthocyanin pigments on the coloration and growth of orange sword tail fish (Xiphophorus helleri). Int J Fish Aquat Stud 7(5):144–149
Mueller KP, Neuhauss SCF (2014) Sunscreen for fish: co-option of uv light protection for camouflage. PLoS ONE 9(1):e87372. https://doi.org/10.1371/journal.pone.0087372
Nawang SUSM, Ching FF, Senoo S (2019) Comparison on growth performance, body coloration changes and stress response of juvenile river catfish, Pangasius hypophthalmus reared in different tank background colour. Aquac Res 50(9):2591–2599. https://doi.org/10.1111/are.14215
Ng TT, Lau CC, Tan MP, Wong LL, Sung YY, Tengku-Muhammad TS, Van de Peer Y, Sui LY, Danish-Daniel M (2023) Cutaneous transcriptomic profiling and candidate pigment genes in the wild discus (Symphysodon spp.). NZ J Zool. https://doi.org/10.1080/03014223.2023.2180763
Ng M (2004) Discus catalogue. AquaCare Worldwide
Ng C (2016) The ornamental freshwater fish trade in Malaysia. UTAR Agric Sci J 2(4)
O’Quin CT, Drilea AC, Conte MA, Kocher TD (2013) Mapping of pigmentation QTL on an anchored genome assembly of the cichlid fish. Metriaclima Zebra BMC Genom 14(1):287. https://doi.org/10.1186/1471-2164-14-287
O’Gorman M, Thakur S, Imrie G, Moran RL, Choy S, Sifuentes-Romero I, Bilandžija H, Renner KJ, Duboué E, Rohner N, McGaugh SE, Keene AC, Kowalko JE (2021) Pleiotropic function of the oca2 gene underlies the evolution of sleep loss and albinism in cavefish. Curr Biol 31(16):3694-3701.e3694. https://doi.org/10.1016/j.cub.2021.06.077
Oshima N, Nakata E, Kamagata MS (1998) Light-induced pigment aggregation in xanthophores of the medaka. Oryzias Latipes Pigment Cell Res 11(6):362–367. https://doi.org/10.1111/j.1600-0749.1998.tb00495.x
Owen JP, Yates CA and Kelsh RN (2021) Differential growth is a critical determinant of zebrafish pigment pattern formation. bioRxiv 2021.06.11.448058. https://doi.org/10.1101/2021.06.11.448058
Padhi N, Jena SK, Ail SKS, Ferosekhan S, Sahoo SN, Udit UK, Bairwa MK, Swain SK (2022) Does tank background colour influence the growth, survival, and carotenoid content in fishes? An illustration in filament barb, Dawkinsia filamentosa (Valenciennes, 1844). Aquac 560:738536. https://doi.org/10.1016/j.aquaculture.2022.738536
Pagels F, Guedes AC, Amaro HM, Kijjoa A, Vasconcelos V (2019) Phycobiliproteins from cyanobacteria: chemistry and biotechnological applications. Biotechnol Adv 37(3):422–443. https://doi.org/10.1016/j.biotechadv.2019.02.010
Pan CH, Chien YH (2009) Effects of dietary supplementation of alga Haematococcus pluvialis (Flotow), synthetic astaxanthin and β-carotene on survival, growth, and pigment distribution of red devil, Cichlasoma citrinellum (Günther). Aquac Res 40(8):871–879. https://doi.org/10.1111/j.1365-2109.2008.02153.x
Pan X, Zhan H, Gong Z (2008) Ornamental expression of red fluorescent protein in transgenic founders of white skirt tetra (Gymnocorymbus ternetzi). Mar Biotechnol (NY) 10(5):497–501. https://doi.org/10.1007/s10126-008-9094-9
Patel AK, Tambat VS, Chen CW, Chauhan AS, Kumar P, Vadrale AP, Huang CY, Dong CD, Singhania RR (2022) Recent advancements in astaxanthin production from microalgae: A review. Bioresour Technol 364:128030. https://doi.org/10.1016/j.biortech.2022.128030
Patterson LB, Parichy DM (2013) Interactions with iridophores and the tissue environment required for patterning melanophores and xanthophores during zebrafish adult pigment stripe formation. PLoS Genet 9(5):e1003561. https://doi.org/10.1371/journal.pgen.1003561
Pérez-Escalante V, Aguirre-Guzmán G, Vanegas-Espinoza PE, Villar-Martínez AAD (2012) Effect of Anthocyanin’s Extract from flour of Roselle calyx (Hibiscus sabdariffa) on growth and pigmentation of Goldfish (Carassius auratus). Wetchasan Sattawaphaet 42(1):107–111
Pérez-Legaspi IA, Valadez-Rocha V, Ortega-Clemente LA, Jiménez-García MI (2020) Microalgal pigment induction and transfer in aquaculture. Rev Aquac 12(3):1323–1343. https://doi.org/10.1111/raq.12384
Petratou K, Subkhankulova T, Lister JA, Rocco A, Schwetlick H, Kelsh RN (2018) A systems biology approach uncovers the core gene regulatory network governing iridophore fate choice from the neural crest. PLoS Genet 14(10):e1007402. https://doi.org/10.1371/journal.pgen.1007402
Pinto KS, Pires TH, Stefanelli-Silva G, Barros BS, Borghezan EA, Zuanon J (2020) Does soil color affect fish evolution? Differences in color change rate between lineages of the sailfin tetra. Neotrop Ichthyol 18(2):e190093. https://doi.org/10.1590/1982-0224-2019-0093
Pouil S, Tlusty MF, Rhyne AL, Metian M (2020) Aquaculture of marine ornamental fish: overview of the production trends and the role of academia in research progress. Rev Aquac 12(2):1217–1230. https://doi.org/10.1111/raq.12381
Prabhath GPWA, Shukla SP, Srivastava PP, Sawant PB, Chouksey MK, Nuwansi KKT (2019) Effects of dietary supplemented Spirulina (Arthrospira) platensis extracted pigments on the colouration of ornamental fish Koi Carp (Cyprinus Carpio Var. Koi). J Exp Zool 22(2):1287–1297
Prasetiya FS, Sunarto S, Bachtiar E, Agung MUK, Nathanael B, Pambudi AC, Lestari AD, Astuty S, Mouget J-L (2020) Effect of the blue pigment produced by the tropical diatom Haslea nusantara on marine organisms from different trophic levels and its bioactivity. Aquac Rep 17:100389. https://doi.org/10.1016/j.aqrep.2020.100389
Priyaja P, Jayesh P, Philip R, Bright Singh IS (2016) Pyocyanin induced in vitro oxidative damage and its toxicity level in human, fish and insect cell lines for its selective biological applications. Cytotechnology 68(1):143–155. https://doi.org/10.1007/s10616-014-9765-5
Rahimi P, Abedimanesh S, Mesbah-Namin SA, Ostadrahimi A (2019) Betalains, the nature-inspired pigments, in health and diseases. Crit Rev Food Sci Nutr 59(18):2949–2978. https://doi.org/10.1080/10408398.2018.1479830
Rajeswari MV (1833) Rajasree SRR and Balasubramanian T (2017) Effect of light levels on growth, survival and skin colour enhancement of marine angelfish, Apolemichthys xanthurus (Bennett. Turkish J Fish Aquat Sci 17(6):1083–1087
Rekha R, Nimsi KA, Manjusha K, Sirajudheen TK (2022) Marine yeast Rhodotorula paludigena VA 242 a pigment enhancing feed additive for the ornamental fish Koi Carp. Aquac Fish. https://doi.org/10.1016/j.aaf.2022.05.008
Rodgers GM, Kelley JL, Morrell AJ (2010) Colour change and assortment in the western rainbowfish. Anim Behav 79(5):1025–1030. https://doi.org/10.1016/j.anbehav.2010.01.017
Rodgers GM, Gladman NW, Corless HF, Morrell LJ (2013) Costs of colour change in fish: food intake and behavioural decisions. J Exp Biol 216(14):2760–2767. https://doi.org/10.1242/jeb.080879
Rout J, Sharma B, Swain S, Mishra S (2013) Algae in nutrition and colouration of ornamental fish: a review. Renew Sust Energ 14(2):578–597
Salis P, Lorin T, Lewis V, Rey C, Marcionetti A, Escande ML, Roux N, Besseau L, Salamin N, Sémon M, Parichy D, Volff JN, Laudet V (2019) Developmental and comparative transcriptomic identification of iridophore contribution to white barring in clownfish. Pigment Cell Melanoma Res 32(3):391–402. https://doi.org/10.1111/pcmr.12766
Sánchez-Vázquez FJ, López-Olmeda JF, Vera LM, Migaud H, López-Patiño MA, Míguez JM (2019) Environmental cycles, melatonin, and circadian control of stress response in fish. Front Endocrinol 10:279. https://doi.org/10.3389/fendo.2019.00279
Sathyaruban S, Uluwaduge DI, Yohi S, Kuganathan S (2021) Potential natural carotenoid sources for the colouration of ornamental fish: a review. Aquac Int 29(4):1507–1528. https://doi.org/10.1007/s10499-021-00689-3
Shiraki T, Kojima D, Fukada Y (2010) Light-induced body color change in developing zebrafish. Photochem Photobiol Sci 9(11):1498–1504. https://doi.org/10.1039/c0pp00199f
Silva SC, Ferreira ICFR, Dias MM, Barreiro MF (2020) Microalgae-derived pigments: a 10-year bibliometric review and industry and market trend analysis. Mol. https://doi.org/10.3390/molecules25153406
Singh MK, Borgohain P, Kaur K, Gogoi S (2021) Emergence of plants in fish pigmentation. Agric Res 10(4):535–542. https://doi.org/10.1007/s40003-021-00551-1
Snekser JL, McRobert SP, Murphy CE, Clotfelter ED (2006) Aggregation behavior in wildtype and transgenic zebrafish. Ethology 112(2):181–187. https://doi.org/10.1111/j.1439-0310.2006.01139.x
Song X, Wang L, Li X, Chen Z, Liang G, Leng X (2017) Dietary astaxanthin improved the body pigmentation and antioxidant function, but not the growth of discus fish (Symphysodon spp.). Aquac Res 48(4):1359–1367. https://doi.org/10.1111/are.13200
Stevens CH, Croft DP, Paull GC, Tyler CR (2017) Stress and welfare in ornamental fishes: What can be learned from aquaculture? J Fish Biol 91(2):409–428. https://doi.org/10.1111/jfb.13377
Sun X, Chang Y, Ye Y, Ma Z, Liang Y, Li T, Jiang N, Xing W, Luo L (2012) The effect of dietary pigments on the coloration of Japanese ornamental carp (koi, Cyprinus carpio L.). Aquac 342–343:62–68. https://doi.org/10.1016/j.aquaculture.2012.02.019
Tan HT, Yusoff FM, Khaw YS, Ahmad SA, Shaharuddin NA (2021) Uncovering research trends of phycobiliproteins using bibliometric approach. Plants. https://doi.org/10.3390/plants10112358
Tanaka Y, Sasaki N, Ohmiya A (2008) Biosynthesis of plant pigments: anthocyanins, betalains and carotenoids. Plant J 54(4):733–749. https://doi.org/10.1111/j.1365-313X.2008.03447.x
Teruhisa K, Toshiro M, Makoto S, Chichester CO (1972) The biosynthesis of astaxanthin.X. the carotenoids in the red carp, Cyprinus carpio linne, and the interconversion of β-[15,15′-3H2]carotene into their body astaxanthin. Int J Biochem Cell Biol 3(17):569–572. https://doi.org/10.1016/0020-711X(72)90013-4
Thongprajukaew K, Kovitvadhi S, Kovitvadhi U, Rungruangsak-Torrissen K (1910) Pigment deposition and in vitro screening of natural pigment sources for enhancing pigmentation in male Siamese fighting fish (Betta splendens Regan. Aquac Res 45(4):709–719. https://doi.org/10.1111/are.12009
Tripathy PS, Devi NC, Parhi J, Priyadarshi H, Patel AB, Pandey PK, Mandal SC (2019) Molecular mechanisms of natural carotenoid-based pigmentation of queen loach, Botia dario (Hamilton, 1822) under captive condition. Sci Rep 9(1):12585. https://doi.org/10.1038/s41598-019-48982-9
Unver AG, Hamzaçebi S (2020) Effect of natural pigment sources on colouration of red zebra cichlid (Maylandia estherae Konings, 1995). Aquac Res 51(10):4372–4380. https://doi.org/10.1111/are.14868
Uthayasiva M, Haq MB, Kumar TA (2014) Significance of light intensity to enhance the colour of marine ornamental fish Amphiprion clarkii (Bennett, 1830) in captivity. Int J Fauna Biol Stud 1(4):14–18
Vandeputte M (2003) Selective breeding of quantitative traits in the common carp (Cyprinus carpio): a review. Aquat Living Resour 16(5):399–407. https://doi.org/10.1016/S0990-7440(03)00056-1
Vanegas-Espinoza PE, Pérez-Escalante V, Aguirre-Guzman G, Hoyos-Leyva JD, Del Villar-Martínez AA (2019) Microencapsulation of anthocyanins from roselle (Hibiscus sabdariffa) and its application on a pigment supplied diet to fantail goldfish (Carassius auratus). Aquac Int 27(6):1801–1811. https://doi.org/10.1007/s10499-019-00430-1
Venil CK, Zakaria ZA, Ahmad WA (2013) Bacterial pigments and their applications. Process Biochem 48(7):1065–1079. https://doi.org/10.1016/j.procbio.2013.06.006
Wagde MS, Sharma SK, Sharma BK, Shivani AP, Keer NR (2018) Effect of natural β-carotene from-carrot (Daucus carota) and Spinach (Spinacia oleracea) on colouration of an ornamental fish-swordtail (Xiphophorus hellerii). J Entomol Zool Stud 6(6):699–705
Walster C (2008) Chapter 16—the welfare of ornamental fish. In: Branson EJ (ed) Fish welfare, Blackwell Publishing Ltd, United Kingdom, pp 271–289
Wan H, He J, Ju B, Yan T, Lam TJ, Gong Z (2002) Generation of two-color transgenic zebrafish using the green and red fluorescent protein reporter genes gfp and rfp. Mar Biotechnol 4(2):146–154. https://doi.org/10.1007/s10126-001-0085-3
Wu HW, Zhong L (1964) Progress and achievement on artificial proliferation of grass carp, black carp, silver carp and big head carp in China. Chin Sci Bull 9:900-907
Xiong YY, Ai CH, Zhu ZX, Xia JH (2022) Genetic architecture of wintering black spot trait in red tilapia as revealed by a genome-wide association study. Aquac 558:738358. https://doi.org/10.1016/j.aquaculture.2022.738358
Yanar M, Erdoğan E, Kumlu M (2019) Thermal tolerance of thirteen popular ornamental fish Species. Aquac 501:382–386. https://doi.org/10.1016/j.aquaculture.2018.11.041
Yang Z, Yu Y, Tay YX, Yue GH (2021) Genome editing and its applications in genetic improvement in aquaculture. Rev Aquac 14(1):178–191. https://doi.org/10.1111/raq.12591
Yasir I, Qin JG (2009) Effect of light intensity on color performance of false clownfish, Amphiprion ocellaris cuvier. J World Aquac Soc 40(3):337–350. https://doi.org/10.1111/j.1749-7345.2009.00254.x
Yu L, Chen H, Hu X, Chen X, Liu Z, Wang J, Wang C (2021) SLC24A5 plays fundamental roles in regulating melanophore development in Cyprinidae fish. Reprod Breed 1(3):167–173. https://doi.org/10.1016/j.repbre.2021.11.001
Yue GH, Wang L, Yang Z, Shen Y, Meng Z, Alfiko Y (2022) The ornamental fighting fish is the next model organism for genetic studies. Rev Aquac 14(4):1966–1977. https://doi.org/10.1111/raq.12681
Yue GH (2019) The ornamental fish industry in Singapore. J Fish China 43(1):116–127. https://doi.org/10.11964/jfc.20180911442
Yusoff FM, Banerjee S, Nagao N, Imaizumi Y, Shariff M, Toda T (2020) Use of microalgae pigments in aquaculture. In: Jacob-Lopes E, Queiroz MI and Zepka LQ (eds) Pigments from microalgae handbook. Springer International Publishing, Cham, pp 471–513
Zeng Z, Liu X, Seebah S, Gong Z (2005) Faithful expression of living color reporter genes in transgenic medaka under two tissue-specific zebrafish promoters. Dev Dyn 234(2):387–392. https://doi.org/10.1002/dvdy.20491
Zerulla TC, Stoddard PK (2021) The biology of polymorphic melanic side-spotting patterns in Poeciliid fishes. Front Ecol Environ 8:608289. https://doi.org/10.3389/fevo.2020.608289
Zhang Y, Liu J, Peng L, Ren L, Zhang H, Zou L, Liu W, Xiao Y (2017) Comparative transcriptome analysis of molecular mechanism underlying gray-to-red body color formation in red crucian carp (Carassius auratus, red var.). Fish Physiol Biochem 43(5):1387–1398. https://doi.org/10.1007/s10695-017-0379-7
Zhang W, Wang H, Brandt DYC, Hu B, Sheng J, Wang M, Luo H, Li Y, Guo S, Sheng B, Zeng Q, Peng K, Zhao D, Jian S, Wu D, Wang J, Zhao G, Ren J, Shi W, van Esch JHM, Klingunga S, Nielsen R, Hong Y (2022) The genetic architecture of phenotypic diversity in the Betta fish (Betta splendens). Sci Adv 8(38):eabm4955. https://doi.org/10.1126/sciadv.abm4955
Zhou L, Gui J-F (2018) Applications of genetic breeding biotechnologies in chinese aquaculture. Aquac China 463–496
Zhu Z, He L, Chen S (1985) Novel gene transfer into the fertilized eggs of gold fish (Carassius auratus L. 1758). J Appl Ichthyol 1(1): 31–34. https://doi.org/10.1111/j.1439-0426.1985.tb00408.x
Zutshi B, Singh A (2021) Artificial photoperiod influence on survivability, pigmentation and hematological parameters in live-bearer ornamental fish. Poecilia Sphenops Indian J Ecol 48(2):508–512
Acknowledgements
We would like to thank Aquacity Tropical Fish Sdn. Bhd for sharing commercial data collection.
Funding
We thank Universiti Malaysia Terengganu for providing funding support for this project (UMT/SRG/2019/55195).
Author information
Authors and Affiliations
Contributions
Conceptualization: CCL, MDD; Funding acquisition: YSY, MDD; Literature search and data analysis: CCL, MPT; Original draft preparation: CCL; Review and editing: SAMN, LLW, YVP, PS.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no conflicts of interest to declare for this project.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lau, C.C., Mohd Nor, S.A., Tan, M.P. et al. Pigmentation enhancement techniques during ornamental fish production. Rev Fish Biol Fisheries 33, 1027–1048 (2023). https://doi.org/10.1007/s11160-023-09777-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-023-09777-4