Abstract
Tracking the movements of aquatic animals is a primary means of understanding movement ecology and interactions with human activities such as fisheries. Despite the diverse spatiotemporal scales that various underwater tracking tools (e.g., acoustic, satellite, PIT, radio, archival telemetry) enable, there are still limitations associated with their application and ability to address diverse research questions. In many cases, supplementary methods are used to complement tracking approaches either to overcome such limitations or to optimize the data that can be collected in a study. In this review, we synthesize relevant literature between 2010 and 2019 to evaluate the different types of complementary methods used with one of the main approaches for tracking fishes—acoustic telemetry. We categorize broad and specific methods used, as well as the outcome or objectives targeted for each method. In addition to summarizing the use of complementary methods and their outcomes, we discuss how they supplement acoustic telemetry and other tracking approaches. Our review shows that using additional methods to support telemetry data helps expand the breadth of research questions that can be addressed regarding the complex and assorted factors influencing movement patterns. Doing so enables greater value in movement ecology research and adjacent fields such as population dynamics, physiology, trophic ecology, reproduction, and health and survival, to underpin management decisions. This review serves as a primer and guide for bolstering data collection and multidisciplinary research within the growing field of movement ecology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Movement ecology, the study of animal movements in relation to biotic and abiotic factors, is a rapidly growing field of scientific research that is directly applicable to the management of aquatic resources (Matley et al. 2022). Knowledge of aquatic animal movements and their interactions with humans contributes to many areas of resource management, including fisheries, marine protected areas, coastal development, tourism, and conservation (Hays et al. 2019; Lowerre-Barbieri et al. 2019a). As a result, interest in movement ecology has flourished, particularly in the last decade, with thousands of studies being conducted within private, academic, and government organizations. The data generated from these studies, in addition to expansions in environmental monitoring, have enabled comprehensive exploration of diverse ecological and human-related questions that have otherwise remained elusive due, in large part, to technological limitations (Crossin et al. 2017; Harcourt et al. 2019).
Despite these advances, questions remain associated with how best to apply tracking technologies in aquatic environments to benefit social (e.g., tourism, heritage, recreation), ecological (e.g., climate-related impacts, ecosystem-wide interactions), or economic (e.g., fisheries) priorities (Sequeira et al. 2021; Matley et al. 2022). Given the multitude of biological and environmental factors that influence animal space use and the resultant impacts on ecosystems, there is considerable potential to expand interpretation of movement patterns through the combination of distinct but associated methodological approaches. These complementary methods (also sometimes referred to as interdisciplinary or multidisciplinary), defined here based on Smith et al. (2016) as a mode of research that integrates sampling tools and techniques to advance understanding beyond the scope of movement ecology approaches alone, act to supplement common output of animal tracking research. The application of complementary methods can serve to bolster existing spatiotemporal findings as an alternative source of sampling or provide novel information altogether by applying related but independent sampling methods.
As movement ecology research is increasingly used to inform and direct management outcomes, there is an ever-growing need to optimize relevant data that can be retrieved during sampling. There have been several advancements in tracking technologies and analytical tools to interpret movement data over the last decade, but little attention has been directed towards the use of other methodologies in conjunction with traditional approaches such as acoustic, satellite, radio, and archival telemetry—see Harcourt et al. (2019) and Matley et al. (2022) for descriptions of each method. The purpose of this paper is to identify, summarize, and contextualize the different complementary methods used in conjunction with aquatic movement ecology research. We conducted a literature synthesis of peer-reviewed research articles that used acoustic telemetry (AT), one of the most widely used technologies for tracking underwater animals. By identifying complementary methods and the reasons for using them in movement ecology research, this review serves as a primer to guide different data collection options to help optimize application and pertinence of research questions in animal tracking studies.
Methods
A literature review of research articles published between 2010 and 2019 that employed acoustic telemetry to track the movements of aquatic animals was carried out to identify the use of complementary sampling methods. These articles were identified using a multicriteria syntax in Web of Science™ as well as journal repositories not listed within it—see Matley et al. 2022, Supplemental Information for more information. The abstract and methods sections of each article were read to identify whether a distinct sampling method was used in addition to AT. This complementary component did not require the same individuals to be sampled as those being tracked to be considered, but still needed to be applicable to the population or species being studied. Given their widespread application directly associated with movement ecology research, we did not incorporate environmental monitoring (e.g., temperature) or habitat delineation (e.g., habitat mapping) as distinct sampling (but see Ancillary complementary methods and outcomes section below). Similarly, the use of sensors built into AT transmitters (e.g., acceleration, depth) were not considered distinct as these typically provide spatially explicit information within AT research. Furthermore, the recapture of acoustically tagged fish was not considered distinct unless it was part of a broader and coordinated sampling effort. Finally, articles that focused on technical aspects of AT research (e.g., reviews, range testing, technological or analytical advancements) were not included in this synthesis.
For each article, we extracted the complementary method(s) used. These were broadly categorized as follows: traditional fisheries sampling, biological sampling, visual observations, hydroacoustics, experimental approaches, and other telemetry technologies (i.e., other than AT). Although the latter category provides a similar type of data as AT (i.e., movement), it was included because the spatial and temporal scales of other tracking techniques often differ from AT. We were also interested in determining the frequency with which they are used in combination with AT. Complementary methods were also categorized relative to associated outcomes or objectives that the method aimed to address. These outcome categories were identified as follows: population biology, reproduction, trophic ecology, health and survival, physiology, and space use. Again, the latter category is similar to the usual outcomes of AT but was included because other types of approaches also provide spatially relevant information distinct from AT. The complementary methods and outcome categories are outlined in Tables 1 and 2, respectively. The use of complementary method and method outcome categories (and specific subcategories) were quantified and presented as summary plots to explore their application with AT.
Complementary methods used in AT research
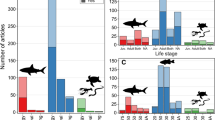
The literature search (and consequent verification and review) identified 1111 AT articles published between 2010 and 2019. Of these, 322 articles (29%) incorporated complementary methods with no apparent change in proportional use over the 10-year period reviewed (i.e., 22–35%; Fig. 1). In order of prevalence, the six broad complementary methods used with AT (Table 1) were: traditional fisheries sampling (n = 133 occurrences, 29% of total occurrences), biological sampling (n = 124, 27%), visual observations (n = 85, 18%), other telemetry technologies (n = 84, 18%), hydroacoustics (n = 22, 5%), and experimental approaches (n = 14, 3%; Fig. 2). The following sections briefly outline each of these methods relative to AT.
Traditional fisheries sampling
The traditional fisheries sampling group consisted of characterizing or quantifying conspecifics (e.g., individuals, eggs) or species of interest (e.g., prey) independent of animal tracking by capturing or collecting them from the water column, surface, or benthos (Table 1). Different metrics, such as the number of individuals captured or location of samples collected, were identified depending on the objectives of the study. Target species were usually captured non-lethally without the undue stress associated with internal tagging or external equipment (e.g., satellite tags). A major advantage of this method relative to AT and other tracking techniques was that it is typically relatively simple and cost-effective to sample more individuals from a targeted population (Dunlop et al. 2013). Sample sizes in animal tracking studies are relatively small—based on our literature review, the median number of individuals per species tagged in each AT study between 2010 and 2019 was 28. Therefore, the use of methods that enable more representative sampling of the target species can provide additional information that may not be attained using AT alone. For example, traditional fisheries sampling can be used to identify the population structure of a species within the study area via large-scale fishing activities, which may be of value when interpreting spawning migrations of individuals being tracked with AT (e.g., Andrews et al. 2020). Capturing study animals also inherently created opportunities to collaborate and engage with interested stakeholders, such as recreational, commercial, and Indigenous fisheries.
Biological sampling
Biological sampling included the measurement or examination of tissues, body parts, or digested remains of organisms to garner information about the biology of individuals or the population being studied (Table 1). Biological sampling typically consisted of physical (e.g., morphometrics, visual identification of gut contents), chemical (e.g., otolith microchemistry, stable isotopes, hormone levels), or DNA sampling. Non-lethal sampling was commonplace by analyzing tissues non-invasively such as fin clips, gill, muscle, blood, and feces, and was conducted in conjunction with animal tracking. Conversely, lethal sampling (e.g., gut content analysis) was also conducted to obtain information that would otherwise be difficult or impossible to gather, but its outcomes must be weighed against the costs of directly removing organisms from the environment (e.g., species with conservation concerns; Heupel et al. 2010). Numerous samples, each with different potential avenues to answer ecological questions, were obtained from individuals making biological sampling an efficient approach to supplement tracking technologies, which typically only provide one type of information (i.e., location data). However, when sampling multiple tissues or components of an individual non-lethally, prior experiments are needed to confirm sampling will not influence recovery, behaviour, and survival.
Visual observations
The application of visual observations provided observational data commonly relating to the presence or behaviour of animals (Table 1). Visual observations were conducted by researchers directly above (e.g., boat or drone survey) and below (e.g., scuba) the water surface, or indirectly via autonomous monitoring (e.g., video recording). Similar to the use of other methods, a subset of the population that is independent of the tracking component was generally sampled. As a result, unique or population-level behaviours were often identifiable from individuals not being tracked (e.g., Lowerre-Barbieri et al. 2019b). Advances in technology associated with aerial (e.g., drones) and underwater (e.g., baited remote underwater video system; BRUVS) surveys are increasingly being incorporated to complement tracking methods; however, observations are commonly limited by water visibility and in-person effort.
Other telemetry technologies
The implementation of other telemetry technologies provided presence and location data similar to that of AT using distinct technology (Table 1), often at variable spatial and temporal resolutions (Harcourt et al. 2019). Other types of telemetry were used in conjunction with AT for various reasons, such as to fill gaps in the monitoring period or study area, reduce costs, and use novel sensors (e.g., heart rate), among others. For example, satellite telemetry was sometimes used with AT to track an animal outside the spatial extent of the acoustic receiver array, since it is not limited by fixed monitoring stations (Strøm et al. 2017). On other occasions, infrastructure supporting telemetry types apart from AT (e.g., radio or PIT telemetry gates) already existed in the study area, providing additional opportunities to locate tagged animals, as well as validate AT findings. However, as with AT, logistical limitations of tracking technologies (e.g., size restrictions, equipment recovery) and the cost of equipment may limit applicability compared to other methods.
Hydroacoustics
The use of hydroacoustics consisted of using sonar technology or acoustic recordings to understand the physical or acoustic aspects of the aquatic environment, respectively (Table 1). Echosounders were sometimes used in addition to other tracking techniques to obtain a snapshot of organisms within the water column. Although species, such as fishes, can be difficult to identify, especially if they do not have air-filled swim bladders, and the area of coverage is often limited due to processing logistics, echosounders sample the water column uniformly and provide high spatial resolution information not always possible with tracking technologies. Like AT, acoustic recordings also autonomously sample the area near a hydrophone, which can relay presence or behaviour of vocal species.
Experimental approaches
Experimental approaches comprised measurements taken or observations made during an experiment in a controlled setting associated with the tracking approach (Table 1). Experimental procedures allowed for control over external factors to isolate the variable(s) of interest that was not otherwise possible in the field. For example, respirometry swim tunnels and visual assessments in experimental scenarios were used to identify behavioural and physiological aspects of an animal to help interpret its tracking data (e.g., Laskowski et al. 2016).
Outcomes of complementary methods
There were six broad categories identified regarding objectives or outcomes that complementary methods addressed (Table 2): space use (n = 172 occurrences, 37% of total occurrences), population biology (n = 119, 26%), reproduction (n = 51, 11%), trophic ecology (n = 47, 10%), health and survival (n = 41, 9%), and physiology (n = 33, 7%; Fig. 3). The following sections outline these outcomes and identify how they supplement goals of AT research. Note that complementary methods can contribute to multiple outcome categories.
Space use
Broadly, investigations of space use cover movement (or location) patterns of aquatic animals in space and time and associated drivers such as biology, environment, and resources, as well as their interactions (Table 2). The information garnered by space use research is typically location and timing data based on the presence (and absence) of organisms within a study area. Space use provides integral information about how and why animals select resources within their environment. As a result, this category delineates spatiotemporal behaviours such as migration and habitat selection (Strøm et al. 2017; Whitty et al. 2017) and helps us understand organismal or population responses to environmental or other perturbations (Raby et al. 2018). This knowledge is pertinent for management bodies to make effective spatial planning decisions, such as those related to marine protected areas (MPAs; Carlisle et al. 2019), fisheries closures (e.g., spawning; Adams et al. 2019), and movement corridors (Holbrook et al. 2015).
Space use data forms the basis of movement ecology research and can be acquired through various methods. Not surprisingly, when animal tracking technology such as radio, PIT, archival, and satellite telemetry are used with AT, it is to study space use patterns. Visual observations from animal-borne, mobile, or stationary equipment, or directly from humans (Khan et al. 2017; Papastamatiou et al. 2018a), hydroacoustic surveys (Bolland et al. 2019), and traditional fisheries sampling approaches such as direct capture and recapture (e.g., via fishing, Andrews and Quinn 2012; Bice et al. 2018) are primary alternatives that identify spatiotemporal patterns of target species (Fig. 3). For example, several studies that applied AT also incorporated an independent mark-recapture component, which often uses location data from a larger sample size of externally marked individuals (e.g., Zemeckis et al. 2017). Other non-telemetry approaches can reveal tagging bias, as demonstrated when blacktip reef sharks (Carcharhinus melanopterus) were observed using shallow forereefs at Palmyra Atoll with BRUVs but not AT (Papastamatiou et al. 2018b). By incorporating these additional techniques, the location of individuals (tagged or untagged) can be determined beyond the spatial extent of the telemetry approach and specific behaviours can be identified in-person, on video, or by other methods. Biological sampling can also be used to gain unique spatial resolution or verify consistency among findings from tracking methods through biomarkers such as stable isotopes (Doherty et al. 2010) and microchemistry (Baker et al. 2019).
Population biology
Population biology covers studies with a focus on the dynamic factors influencing the present or future status of a population within a study area (Table 2), helping researchers to better understand how populations change over time. The information provided by population biology research encompasses a broad range of aspects, such as species abundance, life history, and population structure. Traditional fisheries sampling such as direct capture and recapture and underwater visual observations are often used (Fig. 3) to provide estimates of abundance and distribution (e.g., catch per unit effort), which are integral for understanding density-dependent factors impacting vulnerability to overexploitation and ecosystem health (e.g., Rhodes et al. 2012). Similarly, accounting for life history traits (e.g., growth, age), often measured following capture, enables understanding of how the composition of a population varies (e.g., Crook et al. 2016). Furthermore, biological sampling is commonly used to assess genetic components of a population (i.e., population structure) such as identifying the ancestry of distinct fish stocks (e.g., Andrews et al. 2020). Hence, population biology research can be directly applied to myriad areas of knowledge that are beneficial to ecological understanding and management such as fishery stock assessments.
Given the broad level impacts that population biology has, there is significant potential for research pertaining to it to supplement AT. Without knowledge of the abundance, demography, and structure of a species within the study area, AT research is limited by only being representative of the individuals being tracked. Population biology provides context for AT findings. For example, Bice et al. (2018) found that juvenile recruitment of congolli (Pseudaphritis urvillii; quantified via direct capture) in the lower reaches of the River Murray, Australia increased 5–180 times relative to the previous year when female spawning migrations (tracked via AT) were obstructed due to closure of tidal barriers and diminished flow. By exploring recruitment in addition to adult movements, this study elucidated population-wide impacts of human-mediated connectivity barriers to better inform management pertaining to migration and population dynamics. Combining AT with traditional fisheries sampling (e.g., longline fishing data) has also been shown to improve mark-recapture models used to estimate demographic parameters, such as abundance, that inform fisheries management (e.g., stock assessments; Dudgeon et al. 2015). Technological innovations, such as high-resolution cameras, have also been successfully applied in conjunction with AT to further investigate population biology, such as quantifying fish densities using BRUVS (Fontes et al. 2014). Furthermore, several studies have incorporated DNA analysis with AT to address the genetic diversity or origin of a population (e.g., Raby et al. 2015; Andrews et al. 2020). In these studies, the use of genetic tools in combination with AT allowed for evaluation of potential differences in movement within populations and between distinct population segments.
Reproduction
Investigations into reproduction examine various aspects of life history as they relate to the continuation of genetic material through the production of offspring (Table 2). Reproduction outcomes from ecological studies provide information about reproductive status of individuals, as well as reproductive potential or success (e.g., egg density). Reproduction also consists of behavioural accounts such as reproductive site-selection and conspecific interactions (e.g., fish spawning aggregations). Understanding the conditions (biotic or abiotic) that mediate spawning or reproductive behaviours in fishes can serve as an essential tool to facilitate conservation goals to avoid overexploitation from fisheries (Johnston et al. 2013; van Overzee et al. 2015). Identifying the maturity levels of individuals or their reproductive output plays a significant role in evaluating the status and future potential of a population or stock and is often used in management to establish fishery quotas (Goodyear 1993). Similarly, knowing size-, age- or sex-specific reproductive windows plays a crucial part in designating extractive restrictions within fisheries (Brousseau and Armstrong 1987). Therefore, reproduction has an intrinsic role in understanding populations and optimizing resource use management.
Reproduction is one of the main biological drivers of aquatic animal movements, commonly resulting in seasonal or even diel shifts in distribution at both small (e.g., < 100 m) and large (e.g., > 1000 km) scales (Sadovy de Mitcheson and Colin 2012; Stevick et al. 2011). These movements have considerable implications at a species level, but also impact ecosystems broadly by structuring food webs and transporting nutrients from other locations (Deegan 1993). As a result, investigating reproduction in concert with movement ecology and animal tracking approaches provides fundamental, yet indispensable information regarding behaviour and population trends, as well as potential broader ecological impacts associated with reproductive drivers. Furthermore, sustainable resource use by humans, such as fishing at sustainable harvesting levels or delineating aquatic protected areas, is directly reliant on reproductive traits that are constrained spatially (e.g., aggregative spawning sites, Adams et al. 2019; Matley et al. 2020). Other human activities, such as habitat degradation/restoration (Marsden et al. 2016) and coastal development (Beatty et al. 2018) can similarly impact reproduction of aquatic animals at specific locations. Therefore, movement ecology and reproduction are often interlinked from a management context. For example, Lowerre-Barbieri et al. (2016) used a multidisciplinary approach representative of multiple spatial scales to integrate traditional stock assessment of red drum (Sciaenops ocellatus) with individual movements to better inform population spawning behaviour.
Trophic ecology
Broadly, trophic ecology involves investigations surrounding the structure of feeding (or trophic) relationships among organisms in an ecosystem (Table 2). Trophic ecology research provides information related to the structure of food webs, trophic positions, ecological niches, and diets of organisms in the study area. Trophic ecology also reveals predator–prey interactions and how they vary across different populations (of predator or prey), demographics, or geographic areas. Trophic ecology research can also determine variation in species dietary preferences over space and time. In turn, we can understand feeding patterns among groups of organisms and how they relate to the surrounding ecosystem and available resources. Knowledge of trophic ecology contributes to the management of aquatic communities; for example, stocking decisions are often made with the goals of promoting balanced ecosystems via knowledge of predator–prey interactions (Murry et al. 2010).
Trophic ecology can be investigated in combination with AT, generally using biological sampling methods such as stable isotope and gut content/feces analyses (Guzzo et al. 2016; Werry et al. 2011), the latter of which can be through traditional visual identification or more recent genetic approaches (e.g., Stamoulis et al. 2017), but also through visual observation, and the collection of prey species to estimate prey density (Fig. 3). The investigation of trophic ecology supplements AT by providing insight into movement patterns related to feeding relationships. Not surprisingly, food/energy acquisition and predator avoidance are major drivers influencing the movements of organisms; therefore, investigating trophic ecology can be a major contributor to help interpret AT findings. By exploring stable isotope values of snappers (Lutjanus spp.) in the Bahamas, Hammerschlag-Peyer and Layman (2010) found that δ13C values were associated with movement metrics, suggesting that movement patterns were indicative of distinct prey sources. These findings were independent of fish size and without the application of stable isotopes as a complementary method with AT, the main factor influencing movements may not have been found. Trophic ecology can also supplement AT by determining whether predator–prey interactions are driving movements and predict where fish will be based on prey availability (e.g., Rooker et al. 2018). This can assist with species management and marine protected area development to protect species’ foraging grounds or movement corridors to foraging grounds. Determining whether species are partitioning ecological niches based on space use and diet or are competing for limited resources helps understand fish community composition and species’ ability to coexist, which is relevant for fisheries management as well as ecosystem function (e.g., Guzzo et al. 2016; Matley et al. 2016). Fish that overlap in both space and diet may be more vulnerable to a decline in shared prey than other species, which could be further influenced by climate change, fisheries, pollution or other anthropogenic stressors.
Health and survival
Health and survival pertains to a broad range of studies that examine detrimental biological or chemical agents affecting the study animal and quantify mortality and sources of physical injury (Table 2). The information garnered by health and survival research is typically individual fish health status, assessed based on tissue sampling and infection assessments (parasite load, toxins), as well as captures/recaptures within the study area as a means of quantifying mortality (Fig. 3). Studies have also collected data on traditional fisheries sampling in contaminated areas for ecotoxicology research (Taylor et al. 2018). Health and survival studies can shed light on infectious disease dynamics and provide context for understanding the decline of wild populations (Teffer et al. 2018). These studies can also identify impacts of contaminant exposure and bioaccumulation patterns in mobile fishes (Taylor et al. 2018) and monitor fish welfare in fin-fish sea-cage aquaculture (Muñoz et al. 2020), and through other man-made structures like dams (Mensinger et al. 2021). Multiple stressors, such as rising temperatures due to climate change, anthropogenic impacts such as capture and handling in fisheries, movement through dams or other manmade structures (e.g., hydrokinetic devices), exposure to contaminants and toxins, and aquaculture can affect fish health and survival. In the wild, these factors can affect overall population productivity if individuals are dying before they reproduce or are too sick and injured to carry out spawning migrations (Cooke et al. 2006; Miller et al. 2011) and can impact fishery and aquaculture productivity (Muñoz et al. 2020). Population productivity estimates require an understanding of the mechanisms of mortality to provide reliable data for predictive modeling of loss, which is also pertinent to managers to effectively plan for seasonal fishery closures.
Multiple methods are used in conjunction with AT in health and survival studies, including physiological sampling (e.g., Beardsall et al. 2013; McLean et al. 2020), genomic testing (Miller et al. 2011), identification of infectious disease agents (Teffer et al. 2018; Mordecai et al. 2019), and other visual or tactile assessments of overall organismal wellness (e.g., Reflex Action Mortality Predictors, McLean et al. 2020). Assessing animal movements in relation to contaminated sites can help to explain patterns of bioaccumulation and aid managers in identifying regions of potential health risks associated with consumption of affected organisms (Taylor et al. 2018). At an industry level, identifying changes in fish movement and distribution in aquaculture can promote best practices for fish welfare and product quality (Muñoz et al. 2020), and identifying sources of mortality can aid in the development of regulations for successful fish passage in dammed rivers (Mensinger et al. 2021), or through regions slated for hydrokinetic energy devices (Lilly et al. 2021; Tsitrin et al. 2022). Together with AT, the collected information helps to robustly inform health and survival studies by providing insight into the drivers of movements and sources of mortality.
Physiology
Physiology represents the internal physiological state of an animal (Table 2) and is recognized as a key driver of its movement path in nature (Nathan et al. 2008; Wu and Seebacher 2022). Physiology encompasses numerous biological systems and processes that can interact with one another and the environment in complex (and often poorly understood) ways and be difficult to measure, especially in wild animals. The challenge is often taking hypotheses developed in laboratory experiments, where fine-scale behaviours in artificial arenas (e.g., mirror test, novel object test, thigmotaxis; Webster and Laland 2012; Cattelan et al. 2017) are measured in relation to physiology (e.g., stress hormones, metabolic rate), and using those hypotheses to make predictions about movements in the wild. Combined use of physiology and AT can bridge the gap between behaviours observed in a laboratory setting and movements of animals in the wild to further our understanding of the drivers of movement. For example, Miyoshi et al. (2014) conducted short-term lab experimentation to calibrate electromyogram radio transmitters against swimming performance and metabolic rate in masu salmon (Oncorhynchus masu) and chum salmon (Oncorhynchus keta) before releasing them to track their migration behaviour.
Combining movement data with physiology has typically involved tissue sampling (Fig. 3) such as biopsy of animals when tagging, which provides a ‘snapshot’ of the physiological state of the animal at that time, which can be compared with its spatial ecology. In Pacific salmon, a biopsy approach (gill, blood, fin sampling, and use of a microwave energy meter to assess somatic energy) was developed to investigate physiological conditions associated with successful migrations, confirmed using AT (Cooke et al. 2008). Often, physiology is investigated by exploring physiological responses of animals to various stressors specifically from AT tagging procedures (Caputo et al. 2009), which are paramount to differentiate between tagging effects and natural movement and behaviour. Stress responses resulting from interactions with humans, such as fishing capture and release, aquaculture, as well as passage through migratory barriers, are also commonplace in conjunction with AT (Roscoe et al. 2011; Bordeleau et al. 2018; McLean et al. 2020). In studies such as these, understanding the internal factors influencing movement can help evaluate the impact anthropogenic activities have on tracked animals. Similarly, physiological investigations into the migrations of diadromous fishes often provide insight into the metabolic, hormonal, or other physiological drivers affecting the onset, timing, and success of these movements, which have population-wide implications (Birnie-Gauvin et al. 2019; Hertz et al. 2019). The physiological state of organisms may also explain conspecific space use differences, as demonstrated by Rangel et al. (2022) when exploring the time tiger sharks (Galeocerdo Cuvier) spent at tourism provisioning sites in the Bahamas. Finally, important gains can be made towards understanding fish movements using physiology as researchers pair novel tools with animal tracking such as transcriptomics (and other ‘omics’) to assess physiological states of fishes (Jeffries et al. 2021).
Ancillary complementary methods and outcomes
Although not quantified in the systematic review of complementary methods, there were additional types of methods that were identified during the review process warranting discussion: habitat and environmental monitoring, social sciences, and citizen science.
Habitat and environmental monitoring
Characterizing aquatic habitat and environmental conditions is fundamental for understanding relationships between aquatic organisms and their environment for both basic ecology and applied management. To date, telemetry studies have included a range of concurrent environmental measures, quite commonly water depth and water temperature (Brownscombe et al. 2019). However, a wide range of conditions have been measured and are essential components of animal habitat, including dissolved oxygen, pH, contaminants, turbidity and light, water flow, substrate types, natural or artificial structures, and aquatic vegetation (de Kerckhove et al. 2008). These variables are often measured through in-person sampling (e.g., visual observations, benthic sampling), in situ biologgers that measure near-continuously (e.g., temperature, dissolved oxygen), or other means such as remote sensing (e.g., bathymetry, aquatic vegetation; Marcaccio et al. 2021). Animals equipped with tags incorporating integrated sensors can also serve as biologgers and environmental samplers (Donaldson et al. 2014).
Ultimately, characterizing environmental variables concurrent with telemetry studies is essential to elevate research beyond simple observational studies and adopt inferential approaches that describe the potential drivers of animal movement and space use (Brownscombe et al. 2022). The latter is increasingly important for adapting environmental management approaches proactively in our rapidly changing world. However, a major challenge is that environmental data must be measured at spatial and temporal scales that are congruent with animal tracking data. Acoustic telemetry encompasses a wide variety of approaches and therefore generates a range of data types, from fine-scale spatial positioning (e.g., < 1 m2 resolution; Espinoza et al. 2011) to broader positioning relative to the detection range of receivers (e.g., < 50 m to > 1 km; Kessel et al. 2014). Hence, the scale of animal positioning (related to technology, study design, and data analysis) needs to be matched with that of environmental variable measurement to effectively generate information on animal-habitat associations with telemetry studies.
Social sciences
There is increasing recognition of the importance of integrating social and ecological sciences to improve our understanding of the world, and in particular to improve the ongoing, long-term sustainable use of natural resources (Bennett et al. 2017; Moon et al. 2019). Interest is growing among movement ecologists to engage with social scientists and social research to better understand the implications of their research to society, promote evidence-based decisions and practices, as well as engage in a more holistic approach to a problem or research question. For example, Donaldson et al. (2013) combined biotelemetry, physiological assessments, and social surveys to illustrate effectiveness of and positive attitudes towards ‘recovery bags’ as means to facilitate recovery of sockeye salmon after recreational angling capture. Similarly, a study by Raby et al. (2014) combined biotelemetry and social surveys to explore solutions for bycatch mortality of an endangered population of coho salmon caught in a Indigenous beach seine fishery in British Columbia. The study showed willingness of fishers to employ and adapt handling practices informed by results from the biotelemetry study. The utility, value, and integration of movement ecology data in fisheries management and decisions have also been increasingly explored (e.g., Brooks et al. 2019; Nguyen et al. 2019, 2021). These studies have shown that researchers who engage, collaborate, and develop interpersonal relationships with end-users (whether fishers, fisheries managers, other stakeholders or rightsholders) are often more successful at supporting and informing decisions and practices. Still, there are few studies that integrate both social research and movement ecology.
Citizen science
Citizen science (also sometimes referred to as community, participatory, civic, or public science), defined here as the participation and engagement of the public or other non-scientific members of a community in a scientific project, is an effective tool to increase community engagement and awareness while contributing to scientific research (Kobori et al. 2016). Outcomes of citizen science have the potential to inform the community, scientists, managers, and policy makers about a diverse range of issues in the natural world, but also serve to create social linkages across these various groups. As discussed above, one of the most common forms of citizen science in movement ecology, particularly for AT, is engaging recreational, commercial, or other fishers, to act as community scientists. These groups can provide additional spatiotemporal recapture information of tagged animals that typically exceeds coverage provided by electronic tracking methods and with greater sampling effort. It is common practice to externally mark animals that are being electronically tracked; therefore, coordinated efforts to recapture individuals passively or actively are readily accomplishable. There are a multitude of cooperative tagging organizations across the world that manage long-term recapture databases and provide regimented guidelines to participants about tagging (external) and processing recaptures (Dunlop et al. 2013). For example, since its inception in 1984, the Oceanographic Research Institute’s Cooperative Fish Tagging Project (South Africa) has engaged 6000 members who have externally tagged 330,000 fish (from 368 species), resulting in 82 peer-reviewed journal articles (including the use of acoustic and satellite telemetry) and direct contribution to the management and sustainable use of resources in the Western Indian Ocean (Potts et al. 2021).
Citizen science has also been used to complement movement ecology research by using re-sightings as opposed to recaptures. Re-sightings often incorporate more conspicuous animals such as sea turtles, sharks, and marine mammals that are observed underwater (e.g., scuba), at the surface (e.g., snorkel, boating), or onshore (e.g., nesting sites, washed ashore). Animal tracking has helped validate citizen science monitoring (Vianna et al. 2014) but has also highlighted limitations of its use as an independent tool. For example, Cagua et al. (2015) showed that ecotourism observer bias relating to visibility of whale sharks (Rhincodon typus) at the surface led to conclusions that a population in Tanzania seasonally dispersed; however, AT showed they were present year-round but remained deeper and further offshore from where operators frequented. Still, the capacity for non-scientific groups to provide greater access to animal movement information is evident and with applicable technology readily available to them (e.g., GPS- and internet-enabled mobile devices), citizen science should be a consideration in project conceptualization, especially when societal-environmental relationships can be strengthened.
Future considerations
Although not widespread in the last decade, when complementary methods are used with AT, they help improve interpretation of research questions, often by elucidating drivers behind spatial patterns and placing AT findings in the context of the entire population, not simply a small subset of individuals. When planning animal tracking studies, we advise researchers to consider the strengths and limitations of the telemetry approach being used and whether there are additional ways to answer the questions being posed. Complementary methods should fit in with the specific needs or objectives of the study and can also be applied as new research questions develop throughout the study. Similarly, the use of multiple sampling techniques is dependent on the resources available to those conducting the research. Pertinent considerations, such as financial costs, logistical concerns, potential for social engagement, spatial and temporal resolution, and the impact of the method on the animals being studied (e.g., lethal vs non-lethal), are some of the many factors that will determine the impetus to include additional sampling techniques (Fig. 4). Nevertheless, combining multiple methodologies can help reduce the overall number of studies and animals impacted by research; it may also benefit from being viewed as prudent and cost-effective when seeking funding for research. With ongoing advancements in technology to facilitate monitoring of aquatic ecosystems (Heupel et al. 2018), we see no reason why the use of complementary approaches should not increase. In conjunction with these advances, there is an ongoing impetus to better address management issues (Brownscombe et al. 2022; Matley et al. 2022), and in many circumstances telemetry approaches alone have yielded new information only up to a point (e.g., Lowerre-Barbieri et al. 2016). Indeed, the most powerful way to answer scientific questions is to use multiple lines of evidence (Munafò and Smith, 2018). Therefore, as more refined questions and needs in research and management present themselves, optimizing multiple and related sampling methods should be increasingly expected to help generate more robust answers to research questions in telemetry studies. Examples of how AT research questions can be augmented through complementary methods are outlined in Table 3. Effective planning and clear objectives will, as always, be needed to ensure these methods are integrated optimally. Furthermore, with directed management goals in mind, ongoing collaborative efforts among academic, government, and other scientists can facilitate complementary sampling through the pooling of resources (e.g., equipment) and expertise.
Major considerations for the use of different specific complementary methods weighted on a scale from low (1) to high (5). In order from top to bottom, colours indicate the broad method categories as follows: visual observations, other telemetry, hydroacoustics, experimental, traditional fisheries sampling, biological sampling, and ancillary methods. Note that the rankings were based on first-hand experience from authors and are context-dependent across studies; therefore, this output is not representative of every scenario and is only meant as a general guide
There is also a need to incorporate more foundational complementary approaches to address the gaps that often exist between research output and management integration. Building interdisciplinary connections across groups of people is a major path to inclusive governance and social cohesion that has so far been underutilized in the field of movement ecology. Social science approaches that explore human dimensions affecting research and management are only just starting to be explored in combination with animal movement ecology and hold significant potential to guide future opportunities that bridge social, cultural, economic, and environmental views. Engagement with Indigenous communities has important value, not the least of which is the alternate viewpoints often garnered due to spiritual and reciprocal relationships with nature, and should be prioritized. For example, the application of Indigenous concepts or values such as Two-eyed Seeing (Etuaptmumk—Mi’kmaq) and ScIQ (science and Inuit Qaujimajatuqangit) should be used to strengthen movement ecology research, by shifting away from solely Western science perspectives and incorporating Indigenous ways of knowing to provide a more holistic view of ecosystems and human activities (Denny and Fanning, 2016; Pedersen et al. 2020).
Conclusion
Compatible methods that have accompanied animal tracking research are diverse, providing additional ways to help interpret the movement ecology of aquatic animals. Benefits of supplementing AT with other methods include expanding knowledge of the complex and assorted factors influencing movement patterns, gaining population- or ecosystem-level data on species being tracked, conducting cost-effective sampling with fewer individuals being impacted due to coordinated efforts, facilitating collaborations, and obtaining comprehensive ecological data to underpin management decisions. Greater strides should be made to design and carry out projects with foresight into how using additional methods can improve knowledge of species’ spatial patterns and other aspects of their ecology as well. By identifying the approaches that have been used in the last decade and the outcomes from using them, we hope to provide movement ecologists and others with practical resources to help plan future research.
Data availability
Review data are available from the corresponding author upon reasonable request.
References
Adams AJ, Shenker JM, Jud ZR, Lewis JP, Carey E, Danylchuk AJ (2019) Identifying pre-spawning aggregation sites for bonefish (Albula vulpes) in the Bahamas to inform habitat protection and species conservation. Environ Biol Fishes 102:159–173. https://doi.org/10.1007/s10641-018-0802-7
Andrews KS, Quinn TP (2012) Combining fishing and acoustic monitoring data to evaluate the distribution and movements of spotted ratfish Hydrolagus colliei. Mar Biol 159:769–782. https://doi.org/10.1007/s00227-011-1853-x
Andrews SN, Linnansaari T, Leblanc N, Pavey SA, Curry RA (2020) Interannual variation in spawning success of striped bass (Morone saxatilis) in the Saint John River, New Brunswick. River Res Appl 36:13–24. https://doi.org/10.1002/rra.3545
Baker R, Barnett A, Bradley M, Abrantes K, Sheaves M (2019) Contrasting seascape use by a coastal fish assemblage: a multi-methods approach. Estuaries Coasts 42:292–307. https://doi.org/10.1007/s12237-018-0455-y
Beardsall JW, McLean MF, Cooke SJ, Wilson BC, Dadswell MJ, Redden AM, Stokesbury MJW (2013) Consequences of incidental Otter Trawl capture on survival and physiological condition of threatened atlantic sturgeon. Trans Am Fish Soc 142(5):1202–1214. https://doi.org/10.1080/00028487.2013.806347
Beatty SJ, Tweedley JR, Cottingham A, Ryan T, Williams J, Lynch K, Morgan DL (2018) Entrapment of an estuarine fish associated with a coastal surge barrier can increase the risk of mass mortalities. Ecol Eng 122:229–240. https://doi.org/10.1016/j.ecoleng.2018.07.009
Bennett NJ, Roth R, Klain SC, Chan K, Christie P, Clark DA et al (2017) Conservation social science: understanding and integrating human dimensions to improve conservation. Biol Conserv 205:93–108. https://doi.org/10.1016/j.biocon.2016.10.006
Bice CM, Zampatti BP, Morrongiello JR (2018) Connectivity, migration and recruitment in a catadromous fish. Mar Freshw Res 69:1733–1745. https://doi.org/10.1071/MF17388
Birnie-Gauvin K, Flávio H, Kristensen ML, Walton-Rabideau S, Cooke SJ, Willmore WG, Koed A, Aarestrup K (2019) Cortisol predicts migration timing and success in both Atlantic salmon and sea trout kelts. Sci Rep 9:1–9. https://doi.org/10.1038/s41598-019-39153-x
Bolland JD, Murphy LA, Stanford RJ, Angelopoulos NV, Baker NJ, Wright RM et al (2019) Direct and indirect impacts of pumping station operation on downstream migration of critically endangered European eel. Fish Manag Ecol 26:76–85. https://doi.org/10.1111/fme.12312
Bordeleau X, Hatcher BG, Denny S, Fast MD, Whoriskey FG, Patterson DA, Crossin GT (2018) Consequences of captive breeding: fitness implications for wild-origin, hatchery-spawned Atlantic salmon kelts upon their return to the wild. Biol Conserv 225:144–153. https://doi.org/10.1016/j.biocon.2018.06.033
Brooks JL, Chapman JM, Barkley AN, Kessel ST, Hussey NE, Hinch SG et al (2019) Biotelemetry informing management: case studies exploring successful integration of biotelemetry data into fisheries and habitat management. Can J Fish Aquat Sci 76:1238–1252. https://doi.org/10.1139/cjfas-2017-0530
Brousseau CS, Armstrong ER (1987) The role of size limits in walleye management. Fisheries 12:2–5. https://doi.org/10.1577/1548-8446(1987)012%3c0002:TROSLI%3e2.0.CO;2
Brownscombe JW, Adams AJ, Young N, Griffin LP, Holder PE, Hunt J et al (2019) Bridging the knowledge-action gap: a case of research rapidly impacting recreational fisheries policy. Mar Policy 104:210–215. https://doi.org/10.1016/j.marpol.2019.02.021
Brownscombe JW, Griffin L, Brooks J, Danylchuk A, Cooke S, Midwood J (2022) Applications of telemetry to fish habitat science and management. Can J Fish Aquat Sci 99:1–13. https://doi.org/10.1139/cjfas-2021-0101
Cagua EF, Cochran JE, Rohner CA, Prebble CE, Sinclair-Taylor TH, Pierce SJ, Berumen ML (2015) Acoustic telemetry reveals cryptic residency of whale sharks. Biol Lett 11:20150092. https://doi.org/10.1098/rsbl.2015.0092
Caputo M, O’Connor CM, Hasler CT, Hanson KC, Cooke SJ (2009) Long-term effects of surgically implanted telemetry tags on the nutritional physiology and condition of wild freshwater fish. Dis Aquat Org 84:35–41. https://doi.org/10.3354/dao02025
Carlisle AB, Tickler D, Dale JJ, Ferretti F, Curnick DJ, Chapple TK et al (2019) Estimating space use of mobile fishes in a large marine protected area with methodological considerations in acoustic array design. Front Mar Sci 6:256. https://doi.org/10.3389/fmars.2019.00256
Cattelan S, Lucon-Xiccato T, Pilastro A, Griggio M (2017) Is the mirror test a valid measure of fish sociability? Anim Behav 127:109–116. https://doi.org/10.1016/j.anbehav.2017.03.009
Cooke SJ, Hinch SG, Crossin GT, Patterson DA, English KK, Shrimpton JM et al (2006) Physiology of individual late-run Fraser River sockeye salmon (Oncorhynchus nerka) sampled in the ocean correlates with fate during spawning migration. Can J Fish Aquat Sci 63:1469–1480. https://doi.org/10.1139/f06-042
Cooke SJ, Hinch SG, Farrell AP, Patterson DA, Miller-Saunders K, Welch DW et al (2008) Developing a mechanistic understanding of fish migrations by linking telemetry with physiology, behavior, genomics and experimental biology: an interdisciplinary case study on adult Fraser River sockeye salmon. Fisheries 33:321–339. https://doi.org/10.1577/1548-8446-33.7.321
Crook DA, Buckle DJ, Allsop Q, Baldwin W, Saunders TM, Kyne PM, Woodhead JD, Maas R, Roberts B, Douglas MM (2016) Use of otolith chemistry and acoustic telemetry to elucidate migratory contingents in barramundi Lates calcarifer. Mar Freshw Res 68:1554–1566. https://doi.org/10.1071/MF16177
Crossin GT, Heupel MR, Holbrook CM, Hussey NE, Lowerre-Barbieri SK, Nguyen VM et al (2017) Acoustic telemetry and fisheries management. Ecol Appl 27:1031–1049. https://doi.org/10.1002/eap.1533
De Kerckhove DT, Smokorowski KE, Randall RG (2008) A primer on fish habitat models. Can Tech Rep Fish Aquat Sci 2817:65
Deegan LA (1993) Nutrient and energy transport between estuaries and coastal marine ecosystems by fish migration. Can J Fish Aquat Sci 50:74–79. https://doi.org/10.1139/f93-009
Denny SK, Fanning LM (2016) A Mi’kmaw perspective on advancing salmon governance in Nova Scotia, Canada: setting the stage for collaborative co-existence. Int Indig Policy J. https://doi.org/10.18584/iipj.2016.7.3.4
Doherty CA, Curry RA, Munkittrick KR (2010) Spatial and temporal movements of white sucker: implications for use as a sentinel species. Trans Am Fish Soc 139:1818–1827. https://doi.org/10.1577/T09-172.1
Donaldson MR, Raby GD, Nguyen VN, Hinch SG, Patterson DA, Farrell AP et al (2013) Evaluation of a simple technique for recovering fish from capture stress: integrating physiology, biotelemetry, and social science to solve a conservation problem. Can J Fish Aquat Sci 70:90–100. https://doi.org/10.1139/cjfas-2012-0218
Donaldson MR, Hinch SG, Suski CD, Fisk AT, Heupel MR, Cooke SJ (2014) Making connections in aquatic ecosystems with acoustic telemetry monitoring. Front Ecol Environ 12:565–573. https://doi.org/10.1890/130283
Dudgeon CL, Pollock KH, Braccini JM, Semmens JM, Barnett A (2015) Integrating acoustic telemetry into mark–recapture models to improve the precision of apparent survival and abundance estimates. Oecologia 178:761–772. https://doi.org/10.1007/s00442-015-3280-z
Dunlop SW, Mann BQ, Van der Elst RP (2013) A review of the oceanographic research Institute’s Cooperative Fish Tagging Project: 27 years down the line. Afr J Mar Sci 35:209–221. https://doi.org/10.2989/1814232X.2013.769909
Espinoza M, Farrugia TJ, Webber DM, Smith F, Lowe CG (2011) Testing a new acoustic telemetry technique to quantify long-term, fine-scale movements of aquatic animals. Fish Res 108:364–371. https://doi.org/10.1016/j.fishres.2011.01.011
Fontes J, Schmiing M, Afonso P (2014) Permanent aggregations of a pelagic predator at shallow seamounts. Mar Biol 161:1349–1360. https://doi.org/10.1007/s00227-014-2423-9
Goodyear CP (1993) Spawning stock biomass per recruit in fisheries management: foundation and current use. In: Smith SJ, Hunt JJ, Rivard D (eds) Risk descriptive evaluation and biological reference points for fisheries management. Canadian Special Publication in Fisheries and Aquatic Sciences No. 120. National Research Council of Canada, Ottawa, pp 67–81
Guzzo MM, Blanchfield PJ, Chapelsky AJ, Cott PA (2016) Resource partitioning among top-level piscivores in a sub-Arctic lake during thermal stratification. J Great Lakes Res 42:276–285. https://doi.org/10.1016/j.jglr.2015.05.014
Hammerschlag-Peyer CM, Layman CA (2010) Intrapopulation variation in habitat use by two abundant coastal fish species. Mar Ecol Prog Ser 415:211–220. https://doi.org/10.3354/meps08714
Harcourt R, Sequeira AM, Zhang X, Roquet F, Komatsu K, Heupel M et al (2019) Animal-borne telemetry: an integral component of the ocean observing toolkit. Front Mar Sci 6:326. https://doi.org/10.3389/fmars.2019.00326
Hays GC, Bailey H, Bograd SJ, Bowen WD, Campagna C, Carmichael RH et al (2019) Translating marine animal tracking data into conservation policy and management. Trends Ecol Evol 34:459–473. https://doi.org/10.1016/j.tree.2019.01.009
Hertz M, Jensen LF, Pertoldi C, Aarestrup K, Thomsen SN, Alstrup AK, Asmus H, Madsen SS, Svendsen JC (2019) Investigating fish migration, mortality, and physiology to improve conservation planning of anadromous salmonids: a case study on the endangered North Sea houting (Coregonus oxyrinchus). Can J Zool 97:1126–1136. https://doi.org/10.1139/cjz-2019-0045
Heupel MR, Simpfendorfer CA (2010) Science or slaughter: need for lethal sampling of sharks. Conserv Biol 24:1212–1218. https://doi.org/10.1111/j.1523-1739.2010.01491.x
Heupel MR, Kessel ST, Matley JK, Simpfendorfer CA (2018) Acoustic telemetry. In: Carrier JC, Heithaus MR, Simpfendorfer CA (eds) Shark Research: emerging technologies and applications for the field and laboratory. CRC Press, Boca Raton, FL, pp 133–156
Holbrook CM, Bergstedt R, Adams NS, Hatton TW, McLaughlin RL (2015) Fine-scale pathways used by adult sea lampreys during riverine spawning migrations. Trans Am Fish Soc 144:549–562. https://doi.org/10.1080/00028487.2015.1017657
Jeffries KM, Teffer A, Michaleski S, Bernier NJ, Heath DD, Miller KM (2021) The use of non-lethal sampling for transcriptomics to assess the physiological status of wild fishes. Comp Biochem Physiol B Biochem Mol Biol 256:110629. https://doi.org/10.1016/j.cbpb.2021.110629
Johnston FD, Arlinghaus R, Dieckmann U (2013) Fish life history, angler behaviour and optimal management of recreational fisheries. Fish Fish 14:554–579. https://doi.org/10.1111/j.1467-2979.2012.00487.x
Kessel ST, Cooke SJ, Heupel MR, Hussey NE, Simpfendorfer CA, Vagle S, Fisk AT (2014) A review of detection range testing in aquatic passive acoustic telemetry studies. Rev Fish Biol Fish 24:199–218. https://doi.org/10.1007/s11160-013-9328-4
Khan JA, Goatley CH, Brandl SJ, Tebbett SB, Bellwood DR (2017) Shelter use by large reef fishes: long-term occupancy and the impacts of disturbance. Coral Reefs 36:1123–1132. https://doi.org/10.1007/s00338-017-1604-7
Kobori H, Dickinson JL, Washitani I, Sakurai R, Amano T, Komatsu N et al (2016) Citizen science: a new approach to advance ecology, education, and conservation. Ecol Res 31:1–19. https://doi.org/10.1007/s11284-015-1314-y
Laskowski KL, Monk CT, Polverino G, Alós J, Nakayama S, Staaks G et al (2016) Behaviour in a standardized assay, but not metabolic or growth rate, predicts behavioural variation in an adult aquatic top predator Esox lucius in the wild. J Fish Biol 88:1544–1563. https://doi.org/10.1111/jfb.12933
Lilly J, Dadswell MJ, McLean MF, Avery TS, Comolli PD, Stokesbury MJ (2021) Atlantic sturgeon presence in a designated marine hydrokinetic test site prior to turbine deployment: a baseline study. J Appl Ichthyol 37:826–834. https://doi.org/10.1111/jai.14274
Lowerre-Barbieri SK, Walters Burnsed SL, Bickford JW (2016) Assessing reproductive behavior important to fisheries management: a case study with red drum, Sciaenops ocellatus. Ecol Appl 26:979–995. https://doi.org/10.1016/j.ecoleng.2018.07.009
Lowerre-Barbieri SK, Kays R, Thorson JT, Wikelski M (2019a) The ocean’s movescape: fisheries management in the bio-logging decade (2018–2028). ICES J Mar Sci 76:477–488. https://doi.org/10.1093/icesjms/fsy211
Lowerre-Barbieri SK, Tringali MD, Shea CP, Walters Burnsed S, Bickford J, Murphy M, Porch C (2019b) Assessing red drum spawning aggregations and abundance in the Eastern Gulf of Mexico: a multidisciplinary approach. ICES J Mar Sci 76:516–529. https://doi.org/10.1093/icesjms/fsy173
Marcaccio JV, Gardner Costa J, Midwood JD (2021) Potential use of remote sensing to support the management of freshwater fish habitat in Canada. Canadian Technical Report of Fisheries and Aquatic Sciences 3197 vii + 57 p
Marsden JE, Binder TR, Johnson J, He J, Dingledine N, Adams J et al (2016) Five-year evaluation of habitat remediation in Thunder Bay, Lake Huron: comparison of constructed reef characteristics that attract spawning lake trout. Fish Res 183:275–286. https://doi.org/10.1016/j.fishres.2016.06.012
Matley JK, Heupel MR, Fisk AT, Simpfendorfer CA, Tobin AJ (2016) Measuring niche overlap between co-occurring Plectropomus spp. using acoustic telemetry and stable isotopes. Mar Freshw Res 68:1468–1478. https://doi.org/10.1071/MF16120
Matley JK, Faust MD, Raby GD, Zhao Y, Robinson J, MacDougall T et al (2020) Seasonal habitat-use differences among Lake Erie’s walleye stocks. J Great Lakes Res 46:609–621. https://doi.org/10.1016/j.jglr.2020.03.014
Matley JK, Klinard NV, Martins APB, Aarestrup K, Aspillaga E, Cooke SJ et al (2022) Global trends in aquatic animal tracking with acoustic telemetry. Trends Ecol Evol 37:79–94. https://doi.org/10.1016/j.tree.2021.09.001
McLean MF, Litvak MK, Stoddard EM, Cooke SJ, Patterson DA, Hinch SG et al (2020) Linking environmental factors with reflex action mortality predictors, physiological stress, and post-release movement behaviour to evaluate the response of white sturgeon (Acipenser transmontanus Richardson, 1836) to catch-and-release angling. Comp Biochem Physiol A Mol Integr Physiol 240:110618. https://doi.org/10.1016/j.cbpa.2019.110618
Mensinger MA, Blomberg EJ, Zydlewski JD (2021) The consequences of dam passage for downstream-migrating American eel in the Penobscot River, Maine. Can J Fish Aquat Sci 78:1181–1192. https://doi.org/10.1139/cjfas-2020-0402
Miller KM, Li S, Kaukinen KH, Ginther N, Hammill E, Curtis JM et al (2011) Genomic signatures predict migration and spawning failure in wild Canadian salmon. Science 331:214–217. https://doi.org/10.1126/science.1196901
Miyoshi K, Hayashida K, Sakashita T, Fujii M, Nii H, Nakao K, Ueda H (2014) Comparison of the swimming ability and upstream-migration behavior between chum salmon and masu salmon. Can J Fish Aquat Sci 71:217–225. https://doi.org/10.1139/cjfas-2013-0480
Moon K, Blackman DA, Adams VM, Colvin RM, Davila F, Evans MC et al (2019) Expanding the role of social science in conservation through an engagement with philosophy, methodology, and methods. Methods Ecol Evol 10:294–302. https://doi.org/10.1111/2041-210X.13126
Mordecai GJ, Miller KM, Di Cicco E, Schulze AD, Kaukinen KH, Ming TJ et al (2019) Endangered wild salmon infected by newly discovered viruses. Elife 8:e47615. https://doi.org/10.7554/eLife.47615
Munafò MR, Davey Smith G (2018) Robust research needs many lines of evidence. Nature 553:399–401. https://doi.org/10.1038/d41586-018-01023-3
Muñoz L, Aspillaga E, Palmer M, Saraiva JL, Arechavala-Lopez P (2020) Acoustic telemetry: a tool to monitor fish swimming behavior in sea-cage aquaculture. Front Mar Sci. https://doi.org/10.3389/fmars.2020.00645
Murry BA, Connerton MJ, O’Gorman R, Stewart DJ, Ringler NH (2010) Lakewide estimates of alewife biomass and Chinook salmon abundance and consumption in Lake Ontario, 1989–2005: implications for prey fish sustainability. Trans Am Fish Soc 139:223–240. https://doi.org/10.1577/T08-216.1
Nathan R, Getz WM, Revilla E, Holyoak M, Kadmon R, Saltz D, Smouse PE (2008) A movement ecology paradigm for unifying organismal movement research. Proc Natl Acad Sci 105:19052–19059. https://doi.org/10.1073/pnas.0800375105
Nguyen VM, Young N, Brownscombe JW, Cooke SJ (2019) Collaboration and engagement produce more actionable science: quantitatively analyzing uptake of fish tracking studies. Ecol Appl 29:e01943. https://doi.org/10.1002/eap.1943
Nguyen VM, Delle Palme C, Pentz B, Vandergoot CS, Krueger CC, Young N, Cooke SJ (2021) Overcoming barriers to transfer of scientific knowledge: integrating biotelemetry into fisheries management in the Laurentian Great Lakes. Socio-Ecol Pract Res 3:17–36. https://doi.org/10.1007/s42532-020-00069-w
Papastamatiou YP, Watanabe YY, Demšar U, Leos-Barajas V, Bradley D, Langrock R et al (2018a) Activity seascapes highlight central place foraging strategies in marine predators that never stop swimming. Mov Ecol 6:1–15. https://doi.org/10.1186/s40462-018-0127-3
Papastamatiou YP, Bodey TW, Friedlander AM, Lowe CG, Bradley D, Weng K et al (2018b) Spatial separation without territoriality in shark communities. Oikos 127:767–779. https://doi.org/10.1111/oik.04289
Pedersen C, Otokiak M, Koonoo I, Milton J, Maktar E, Anaviapik A et al (2020) ScIQ: an invitation and recommendations to combine science and Inuit Qaujimajatuqangit for meaningful engagement of Inuit communities in research. Arct Sci 6:326–339. https://doi.org/10.1139/as-2020-0015
Potts WM, Mann-Lang JB, Mann BQ, Griffiths CL, Attwood CG, de Blocq AD et al (2021) South African marine citizen science–benefits, challenges and future directions. Afr J Mar Sci 43:353–366. https://doi.org/10.2989/1814232X.2021.1960890
Raby GD, Donaldson MR, Nguyen VM, Taylor MK, Sopinka NM, Cook KV et al (2014) Bycatch mortality of endangered coho salmon: impacts, solutions, and aboriginal perspectives. Ecol Appl 24:1803–1819. https://doi.org/10.1890/13-1885.1
Raby GD, Hinch SG, Patterson DA, Hills JA, Thompson LA, Cooke SJ (2015) Mechanisms to explain purse seine bycatch mortality of coho salmon. Ecol Appl 25:1757–1775. https://doi.org/10.1890/14-0798.1
Raby GD, Vandergoot CS, Hayden TA, Faust MD, Kraus RT, Dettmers JM et al (2018) Does behavioural thermoregulation underlie seasonal movements in Lake Erie walleye? Can J Fish Aquat Sci 75:488–496. https://doi.org/10.1139/cjfas-2017-0145
Rangel BS, Moreira RG, Rider MJ, Sulikowski JA, Gallagher AJ, Heithaus MR et al (2022) Physiological state predicts space use of sharks at a tourism provisioning site. Anim Behav 191:149–163. https://doi.org/10.1016/j.anbehav.2022.07.004
Rhodes KL, McIlwain J, Joseph E, Nemeth RS (2012) Reproductive movement, residency and fisheries vulnerability of brown-marbled grouper, Epinephelus fuscoguttatus (Forsskål, 1775). Coral Reefs 31:443–453. https://doi.org/10.1007/s00338-012-0875-2
Rooker JR, Dance MA, Wells RD, Quigg A, Hill RL, Appeldoorn RS et al (2018) Seascape connectivity and the influence of predation risk on the movement of fishes inhabiting a back-reef ecosystem. Ecosphere 9:e02200. https://doi.org/10.1002/ecs2.2200
Roscoe DW, Hinch SG, Cooke SJ, Patterson DA (2011) Fishway passage and post-passage mortality of up-river migrating sockeye salmon in the Seton River, British Columbia. River Res Appl 27:693–705. https://doi.org/10.1002/rra.1384
Sadovy de Mitcheson Y, Colin PL (eds.) (2012) Reef fish spawning aggregations: biology, research and management, Vol. 35. Fish Fish Ser. New York, New York: Springer
Sequeira AM, O’Toole M, Keates TR, McDonnell LH, Braun CD, Hoenner X et al (2021) A standardisation framework for bio-logging data to advance Ecol Res and conservation. Methods Ecol Evol 12:996–1007. https://doi.org/10.1111/2041-210X.13593
Smith RV, Densmore LD, Lener EF (2016) Graduate research: a guide for students in the sciences, 4th edn. Academic Press, Cambridge, MA
Stamoulis KA, Friedlander AM, Meyer CG, Fernandez-Silva I, Toonen RJ (2017) Coral reef grazer-benthos dynamics complicated by invasive algae in a small marine reserve. Sci Rep 7:1–12. https://doi.org/10.1038/srep43819
Stevick PT, Neves MC, Johansen F, Engel MH, Allen J, Marcondes MC, Carlson C (2011) A quarter of a world away: female humpback whale moves 10 000 km between breeding areas. Biol Lett 7:299–302. https://doi.org/10.1098/rsbl.2010.0717
Strøm JF, Thorstad EB, Chafe G, Sørbye SH, Righton D, Rikardsen AH, Carr J (2017) Ocean migration of pop-up satellite archival tagged Atlantic salmon from the Miramichi River in Canada. ICES J Mar Sci 74:1356–1370. https://doi.org/10.1093/icesjms/fsw220
Taylor MD, van der Meulen DE, Brodie S, Cadiou G, Knott NA (2018) Applying acoustic telemetry to understand contaminant exposure and bioaccumulation patterns in mobile fishes. Sci Total Environ 625:344–354. https://doi.org/10.1016/j.scitotenv.2017.12.177
Teffer AK, Bass AL, Miller KM, Patterson DA, Juanes F, Hinch SG (2018) Infections, fisheries capture, temperature, and host responses: multistressor influences on survival and behaviour of adult Chinook salmon. Can J Fish Aquat Sci 75:2069–2083. https://doi.org/10.1139/cjfas-2017-0491
Tsitrin E, Sanderson BG, McLean MF, Gibson AJF, Hardie DC, Stokesbury MJ (2022) Migration and apparent survival of post-spawning alewife (Alosa pseudoharengus) in Minas Basin, Bay of Fundy. Anim Biotelemetry 10:1–17. https://doi.org/10.1186/s40317-022-00277-z
van Overzee HM, Rijnsdorp AD (2015) Effects of fishing during the spawning period: implications for sustainable management. Rev Fish Biol Fish 25:65–83. https://doi.org/10.1007/s11160-014-9370-x
Vianna GM, Meekan MG, Bornovski TH, Meeuwig JJ (2014) Acoustic telemetry validates a citizen science approach for monitoring sharks on coral reefs. PLoS ONE 9:e95565. https://doi.org/10.1371/journal.pone.0095565
Webster MM, Laland KN (2012) Social information, conformity and the opportunity costs paid by foraging fish. Behav Ecol Sociobiol 66:797–809. https://doi.org/10.1007/s00265-012-1328-1
Werry JM, Lee SY, Otway NM, Hu Y, Sumpton W (2011) A multi-faceted approach for quantifying the estuarine–nearshore transition in the life cycle of the bull shark, Carcharhinus leucas. Mar Freshw Res 62:1421–1431. https://doi.org/10.1071/MF11136
Whitty JM, Keleher J, Ebner BC, Gleiss AC, Simpfendorfer CA, Morgan DL (2017) Habitat use of a Critically Endangered elasmobranch, the largetooth sawfish Pristis pristis, in an intermittently flowing riverine nursery. Endanger Species Res 34:211–227. https://doi.org/10.3354/esr00837
Wu NC, Seebacher F (2022) Physiology can predict animal activity, exploration, and dispersal. Commun Biol 5:1–11. https://doi.org/10.1038/s42003-022-03055-y
Zemeckis DR, Liu C, Cowles GW, Dean MJ, Hoffman WS, Martins D, Cadrin SX (2017) Seasonal movements and connectivity of an Atlantic cod (Gadus morhua) spawning component in the western Gulf of Maine. ICES J Mar Sci 74:1780–1796. https://doi.org/10.1093/icesjms/fsw190
Acknowledgements
The authors thank A. Fisk for initial discussion regarding this review.
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing or financial interests to declare that are relevant to the content of this article.
Consent to participate
All the authors agree with the contents of the manuscript and give their consent to submit.
Consent for publication
This is original research carried out by the authors and all agree with its publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matley, J.K., Klinard, N.V., Larocque, S.M. et al. Making the most of aquatic animal tracking: a review of complementary methods to bolster acoustic telemetry. Rev Fish Biol Fisheries 33, 35–54 (2023). https://doi.org/10.1007/s11160-022-09738-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-022-09738-3