Abstract
Background and aims: Bariatric surgery is the most effective treatment in individuals with obesity to achieve remission of type 2 diabetes. Post-bariatric surgery hypoglycaemia occurs frequently, and management remains suboptimal, because of a poor understanding of the underlying pathophysiology. The glucoregulatory hormone responses to nutrients in individuals with and without post-bariatric surgery hypoglycaemia have not been systematically examined. Materials and methods: The study protocol was prospectively registered with PROSPERO. PubMed, EMBASE, Web of Science and the Cochrane databases were searched for publications between January 1990 and November 2021 using MeSH terms related to post-bariatric surgery hypoglycaemia. Studies were included if they evaluated individuals with post-bariatric surgery hypoglycaemia and included measurements of plasma glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), insulin, C-peptide and/or glucagon concentrations following an ingested nutrient load. Glycated haemoglobin (HbA1c) was also evaluated. A random-effects meta-analysis was performed, and Hedges’ g (standardised mean difference) and 95% confidence intervals were reported for all outcomes where sufficient studies were available. The τ2 estimate and I2 statistic were used as tests for heterogeneity and a funnel plot with the Egger regression-based test was used to evaluate for publication bias. Results: From 377 identified publications, 12 were included in the analysis. In all 12 studies, the type of bariatric surgery was Roux-en-Y gastric bypass (RYGB). Comparing individuals with and without post-bariatric surgery hypoglycaemia following an ingested nutrient load, the standardised mean difference in peak GLP-1 was 0.57 (95% CI, 0.32, 0.82), peak GIP 0.05 (-0.26, 0.36), peak insulin 0.84 (0.44, 1.23), peak C-peptide 0.69 (0.28, 1.1) and peak glucagon 0.05 (-0.26, 0.36). HbA1c was less in individuals with hypoglycaemia − 0.40 (-0.67, -0.12). There was no evidence of substantial heterogeneity in any outcome except for peak insulin: τ2 = 0.2, I2 = 54.3. No publication bias was evident. Conclusion: Following RYGB, postprandial peak plasma GLP-1, insulin and C-peptide concentrations are greater in individuals with post-bariatric surgery hypoglycaemia, while HbA1c is less. These observations support the concept that antagonism of GLP-1 would prove beneficial in the management of individuals with hypoglycaemia following RYGB.
PROSPERO Registration Number: CRD42021287515.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
1.1 Impact of bariatric surgery on obesity, type 2 diabetes and glucagon-like peptide-1 secretion
Bariatric surgery is the most effective treatment in the management of morbid obesity [1]. Roux-en-Y gastric bypass (RYGB) and sleeve gastrectomy are frequently performed procedures and, in general, have favorable short- and longer-term outcomes, including the remission of type 2 diabetes [2]. The exaggerated post-prandial secretion of glucagon-like peptide-1 (GLP-1) and consequent increase in insulin secretion/sensitivity is thought to be central to this effect [3]. The increase in GLP-1 is multifactorial and likely to reflect more rapid gastric pouch emptying and increased delivery of nutrients to the GLP-1-secreting L-cells in the distal small intestine, changes in the bile acid profile to stimulate GLP-1 release mediated via the G-protein-coupled bile acid receptor and alterations in the gut microbiota leading to stimulation of GLP-1 secretion via metabolites [4, 5]. An improvement in glucose tolerance is evident within a few days after surgery and precedes weight loss [3]. Although it is well-recognised that nutrient-induced GLP-1 secretion is greatly increased following some forms of bariatric surgery, it is not clear if individuals who experience hypoglycaemia post-bariatric surgery have a greater GLP-1 response than individuals without hypoglycaemia.
2 Prevalence of post-bariatric surgery hypoglycaemia
Post-bariatric surgery hypoglycaemia (PBH) is a complication of metabolic surgery and defined as having a low blood glucose level (< 3.0 mmol/L) associated with autonomic or neuroglycopenic symptoms, although there is no consensus for the threshold glucose level [6]. It occurs in about a quarter of patients following RYGB, characteristically 1–3 h following a meal [7, 8] and has also been described following sleeve gastrectomy [7]. PBH causes substantial morbidity [7, 9]. and, in extreme cases, is life-threatening [10]. The role of GLP-1 in PBH remains unclear with inconsistent observations [11, 12]. Clarification of this is important as GLP-1 receptor antagonists are being evaluated as a potential therapy in early clinical studies [13], where paradoxically, GLP-1 receptor agonists have also been suggested to have benefit in PBH [14]. There is currently no accepted standard medical treatment for PBH. Accordingly, this study has systematically reviewed the literature to evaluate if nutrient-induced peak GLP-1 concentrations are greater in individuals with PBH compared with individuals who have had bariatric surgery but do not have PBH.
2.1 Methods
2.1.1 Study design and registration
This systematic review and meta-analysis of cohort and case-control studies was designed in accordance with the latest methodological guidance [15, 16], and was reported in compliance with the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines [17]. Protocol details were prospectively registered on PROSPERO (CRD42021287515); there were no major protocol deviations.
2.2 Eligibility criteria
We included original research studies that reported a prognostic association between bariatric surgery (Roux-en-Y gastric bypass, sleeve gastrectomy or single anastomosis gastric bypass) and hypoglycaemia. We excluded abstracts and conference presentations, case reports, case series, editorials, expert opinions, publications with incompletely reported data, studies published in language other than English and non-human studies.
2.3 Search strategy
We searched PubMed, EMBASE, Web of Science and Cochrane Database of Systematic Reviews from inception to 27 Nov 2021. Our search strategy included a comprehensive set of relevant search terms (Supplemental 1) and was designed with the support of a professional librarian, experienced in systematic reviews [18].
2.4 Study selection
Two authors (R.J.J. and M.M.U.) independently screened titles and abstracts for potentially relevant studies. The full texts of shortlisted studies were extracted and were assessed against eligibility criteria independently and in duplicate. A third author (M.P.P.) adjudicated any disagreements. We also reviewed the reference and citation lists of included studies for additional potentially relevant studies.
2.5 Data extraction and management
Two authors (R.J.J. and M.M.U.) independently used standardised spreadsheets to extract data from included studies. Where reported, the following were recorded: study design, population baseline characteristics, operative details, diabetes status at the time of the study, test meal contents and definition of hypoglycaemia. The primary outcome was post-bariatric surgery nutrient stimulated peak plasma levels of GLP-1 in individuals with and without hypoglycaemia. Secondary outcomes included (i) post-bariatric surgery nutrient stimulated plasma levels of glucose dependent insulinotropic polypeptide (GIP), insulin and C-peptide in patients with and without hypoglycaemia, (ii) HbA1c, (iii) hypoglycaemic counter-regulatory hormones (cortisol, glucagon, adrenaline, noradrenaline) and (iv) gastric emptying data.
2.6 Risk of bias (quality) assessment
The same authors (R.J.J. and M.M.U.) independently assessed the risk of bias using the Newcastle-Ottawa Scale (NOS). Disagreements in assessment were discussed and consensus obtained.
2.7 Statistical analysis and data synthesis
In studies where outcomes of interest were presented in tables, but not reported in numerical form, the corresponding authors were contacted via email to request this information. No responses were received. The software PlotDigitizer.exe (Huwaldt JA. Plot Digitizer. Version 2.6.9, Free Software Foundation 2020) was used to extract peak concentrations of relevant enteropancreatic hormones and standardized deviations. We tabulated weighted mean differences (reported as Hedges’ g) and 95% confidence interval from each study and generated summary estimates using random effects modelling [19]. We performed separate meta-analyses for each outcome where reporting was sufficient across studies; otherwise, we performed qualitative analyses. Random-effects meta-analysis was used to account for potential between-study heterogeneity, and REML (restricted maximum likelihood) was used for the random effect estimation.Statistical heterogeneity is reported as the I2 statistic and \({\tau }^{2}\) estimate [20]. Where there were fewer than 10 included studies reporting on an outcome, publication bias was unable to be formally assessed [21]. All data analyses were performed in consultation with a professional biostatistician using STATA (StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC).
2.8 Search results
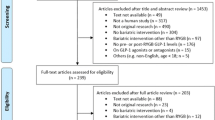
The search returned 376 results. No additional citations were identified from secondary searching of reference lists. After de-duplication, 334 studies underwent title and abstract screening. 104 potentially relevant studies underwent full-text review, from which 12 studies were included in this review. (Fig. 1). The majority of these 104 studies described an increase in GLP-1 following RYGB (which has been well established) but did not determine whether GLP-1 secretion is increased to a greater extent in individuals with PBH and were, therefore, excluded.
2.9 Description of included studies
Twelve studies involving 324 participants post-bariatric surgery were included. Detailed characteristics of included studies are presented in Table 1. (Table 1)
2.10 Methodological quality
The studies included were observational in design and of small sample sizes associated with large confidence intervals. The studies included were mostly at a low risk of bias as assessed by the Newcastle-Ottawa scale. Results are summarized in Table 2 below. (Table 2).
2.11 Outcomes
2.11.1 Peak postprandial GLP-1
In ten studies [22,23,24,25,26,27,28,29,30,31] (involving 264 individuals), there was an increase in peak postprandial GLP-1 in those with PBH compared to those without; Hedges’ g 0.57 (95% CI 0.32, 0.82) reported this outcome for both PBH (n = 132) and non-PBH (n = 132) cohorts (Fig. 2). There was no evidence of between-study heterogeneity (\({\tau }^{2}\)=0.01, \({I}^{2}\)= 3.36). Only two studies [23, 27] reported the outcome for GLP-1 AUC in both PBH and non-PBH cohorts, hence a meta-analysis for this outcome was not performed. The p value for Egger’s test was 0.306. There was no evidence of bias in the funnel plot. (Fig. 3)
2.12 Peak postprandial GIP
In 7 studies, involving 193 individuals, there was no difference between postprandial GIP in those with (n = 106) and without (n = 87) PBH [22, 25, 27,28,29,30,31]. (Fig. 4) Hedges’ g was 0.05 (95% CI -0.26, 0.36) and there was no evidence of between-study heterogeneity (\({\tau }^{2}\)=0.03, \({I}^{2}\)= 16.82). No study reported GIP AUC. As there were fewer than ten studies, we were unable to formally assess for publication bias.
2.13 Peak postprandial insulin
In 10 studies (involving 269 individuals), there was an increase in peak postprandial insulin in those with PBH (n = 151) compared to those without (n = 118) [22, 24, 25, 27,28,29,30,31,32,33]. Hedges’ g was 0.84 (95% CI 0.44, 1.23), however, there was some indication of between-study heterogeneity (\({\tau }^{2}\)=0.20, \({I}^{2}\)= 54.25, p=0.03). One study [24] reported the means, but not standard deviations, of the insulin AUC. This study was not included in the meta-analysis. (Fig. 5) The p value for Egger’s test was 0.632. There was no evidence of bias in the funnel plot. (Fig. 6)
2.14 Peak post-prandial C-peptide
In 6 studies (involving 191 individuals), there was an increase in peak postprandial C-peptide in those with PBH (n = 100) compared to those without (n = 91) [22, 25,26,27,28, 33]. Hedges’ g was 0.69 (95% CI 0.28, 1.10) and there was no evidence of between-study heterogeneity (\({\tau }^{2}\)=0.11, \({I}^{2}\)= 44.47) (Fig. 7). No study reported the C-peptide AUC. As there were fewer than ten studies, we were unable to formally assess for publication bias.
2.15 Peak glucagon
In 10 studies, involving 264 individuals, there was no difference between postprandial glucagon in those with (n = 132) and without (n = 132) PBH [22,23,24,25,26,27,28,29,30,31]. Hedges’ g was 0.05 (95% CI -0.26, 0.36) and there was no evidence of substantial between-study heterogeneity (\({\tau }^{2}\)=0.09, \({I}^{2}\)= 35.53) (Fig. 8). No study reported the glucagon AUC. The p value for Egger’s test was 0.015. There was no evidence of bias in the funnel plot. (Fig. 9)
2.16 HbA1c
In 7 studies, involving 208 individuals, HbA1c was lower in individuals with PBH (n = 104) compared with those without PBH (n = 104) [22, 23, 25,26,27,28, 31]. Hedges’ g was − 0.40 (95% CI -0.67, -0.12) and there was no evidence of between-study heterogeneity (\({\tau }^{2}\)=0.00, \({I}^{2}\)= 0.00). (Fig. 10) As there were fewer than ten studies, we were unable to formally assess for publication bias.
2.17 Other parameters
The studies were screened for other outcomes, including gastric emptying data (not reported in any study), catecholamines (not reported in any study), cortisol (not reported in any study) insulin secretion (reported in 1 study), disposition index (reported in 1 study) and HOMA-IR (not reported in any study), for which meta-analyses were, accordingly, unable to be performed.
3 Discussion
This is the first systematic review to evaluate associations between nutrient-stimulated enterohormones in individuals with postprandial hypoglycaemia following RYGB. Individuals who had post-bariatric surgery hypoglycaemia had greater postprandial GLP-1 and insulin concentrations, but GIP responses were not different, when compared with individuals who had the same type of bariatric surgery but who did not develop hypoglycaemia. Furthermore, in individuals with post-bariatric surgery hypoglycaemia, mean HbA1c was less. There was no substantial between-study heterogeneity in the outcomes analysed apart from peak insulin, where there was a strong association with PBH. The observed heterogeneity is likely due to reflect different frequencies of diabetes and remission as summarized in Table 1. Based on the funnel plots and Egger’s test, there was no evidence of publication bias, apart from the analysis of peak glucagon where the funnel plot was skewed to the right by the largest study [27]. Overall, the pooled results from all studies indicated that there was no association between peak glucagon and PBH. The majority of the studies (11/12) were well designed with a low risk of bias, as evaluated with the Newcastle-Ottawa Scale. Pre-specified sensitivity analyses of low risk of bias studies (included in Supplementary material) did not alter the results.
This meta-analysis of all studies has made it evident that a greater peak GLP-1 response is associated with PBH following RYGB while peak GIP is not. The latter is not surprising given that GIP-secreting K-cells are predominantly found in the duodenum/proximal small intestine which is bypassed following RYGB. There was only one study which found that a greater postprandial peak in GIP was associated with a lower risk of hypoglycaemia [22]. It should be appreciated that this study was published much earlier (2007) than the other studies, and the difference in GIP may be related to variations in surgical technique such as the length of proximal intestine bypassed, however, this information was not provided. Both peak insulin and C-peptide were predictably strongly associated with PBH, consistent with the concept that PBH is driven by exaggerated endogenous insulin secretion. In contrast, there was no evidence of an association between peak glucagon and PBH. We had hypothesized that, reflecting the capacity of GLP-1 to suppress glucagon, that a lower peak glucagon may be associated with PBH. It is possible that PBH is mainly driven by the exaggerated GLP-1 and insulin response rather than suppression of glucagon. As glucagon is released as a counter-regulatory response to hypoglycaemia [34], this may over-ride the suppressive effect of GLP-1 which occurs in a glucose-dependent manner. A lower HbA1c was associated with an increased risk of PBH. This may be explained by either the effect of hypoglycaemia to lower mean blood glucose or a protective effect against hypoglycaemia due to greater insulin resistance associated with higher HbA1c.
The underlying cause of PBH remains unclear, however, there are several putative mechanisms including increased beta cell mass [35], increased insulin-independent glucose uptake [36], increased secretion of GLP-1 [37], altered bile acid metabolism [38], altered gut microbiota [39] and increased fibroblast growth factor-19 [40]. Although GLP-1RA therapy in T2D without concomitant administration of insulin is not associated with hypoglycaemia, there are multiple physiologic changes that occur following RYGB that could explain why hypoglycaemia may occur in this group. RYGB is associated with gastric pouch emptying rates of up to 100 kcal/min [5] compared with gastric emptying rates of 1–4 kcal/min in health, which may lead to a transient marked postprandial glycaemic excursion [41]. The rapid and increased delivery of nutrients to the small intestine is thought to underlie the supraphysiologic levels of GLP-1 [5, 42]. GLP-1 has glucose-dependent effects to augment insulin and inhibit glucagon secretion [43], with a consequent marked glucose-lowering effect. RYGB also alters bile acid composition and the activation of intestinal farnesoid X receptor (FXR) by bile acids increases fibroblast growth factor-19 (FGF-19), an intestinally derived hormone, which reduces hepatic glucose production and increases peripheral glucose disposal independent of insulin [40, 44]. Interestingly, increased FGF-19 levels have been associated with PBH; moreover, the progressive increase in FGF-19 levels over time corresponds to the time course of PBH [40]. Changes to the gut microbiota occur also within 3 months post-RYGB, are sustained in the longer-term [45], and have been associated with lower postprandial glucose levels in rodent models [46]. While these changes are thought to be pivotal to remission of type 2 diabetes, it is likely that, in combination, they also contribute to PBH and in such individuals, we have now shown that GLP-1 responses are greater.
3.1 Limitations
Although all forms of bariatric surgery were included in the search criteria, only studies involving RYGB were identified. Thus, it is not known if there are similar associations between glucoregulatory hormones and PBH with other bariatric surgery procedures such as sleeve gastrectomy or the one anastomosis gastric bypass. Only English language articles were included, however, only 3 identified articles were non-English language. Only peak hormone levels were consistently reported among studies. Data were collected for area under the curve (AUC) hormone levels over time but due to varying time-frames and most studies not reporting this, a meta-analysis could not be performed. There is no standardized meal for the evaluation of PBH and accordingly, there was significant variation in the meals between studies. Similarly, there was varied definitions for hypoglycaemia between studies and we were unable to assess the relationship between severity of postprandial hypoglycaemia and GLP-1. Furthermore, we were unable to evaluate the temporal relationship between GLP-1 and hypoglycaemia/hyperinsulinaemia. The final number of studies included for GIP, C-peptide and HbA1c were too small to analyse for publication bias.
3.1.1 Implications and areas for further evaluation
The outcomes of this analysis support an association between the stimulation of GLP-1 with PBH. Early clinical trials have suggested that GLP-1 antagonism [37, 47] may represent a novel therapy for PBH. In a proof-of-principle double-blinded, placebo-controlled, crossover study, the intravenous infusion of exendin (9–39), a GLP-1 receptor antagonist, prevented hypoglycaemia in 8 out of 8 participants with PBH [37]. Our findings support the development of larger-scale clinical trials to evaluate the potential role of GLP-1 antagonism in PBH management. Recent trials have also suggested a role for GLP-1 receptor agonist (RA) treatment as an adjunct to increase rates of type 2 diabetes remission [48]. The effect of GLP-1RAs on PBH has not been established, however, two small, uncontrolled studies reported improvement of PBH following GLP-RA treatment [14, 49] but sample sizes were small (n = 5 and n = 13 respectively). Possible explanations to account for this paradox include slowing of intestinal transit, resulting in decreased stimulation of L-cells [50] and marked suppression of endogenous GLP-1 secretion [51].
Gastric emptying has been recognized as a major determinant of postprandial glycaemic excursions in individuals with and without type 2 diabetes [52] but was not evaluated in any of the included studies. There is no information about the role of gastric emptying/small intestinal transit in the pathogenesis of PBH and this would be of considerable interest. Additionally, there is evidence that an acceleration of gastric emptying is important in the counter-regulation of hypoglycaemia [53]. Catecholamines are an important counter-regulatory mechanism in hypoglycaemia and this response may be blunted following RYGB [54]. It is not known if individuals with PBH have a greater reduction in the catecholamine response to hypoglycaemia.
Finally, our findings suggest additional attention to PBH be given to individuals who have a lower HbA1c as they may be at greater risk.
4 Conclusion
Following RYGB, nutrient-induced peak concentrations of GLP-1, insulin and C-peptide are greater, while HbA1c is less in individuals with post-bariatric surgery hypoglycaemia. These observations suggest that antagonism of GLP-1 represents a rational intervention to prevent hypoglycaemia in patients who suffer from post-bariatric surgery hypoglycaemia after RYGB. Further evaluation of the role of gastric emptying and catecholamine response in this condition is warranted.
Abbreviations
- GIP:
-
Glucose dependent insulinotropic polypeptide
- GLP-1:
-
Glucagon-like peptide-1
- HbA1c:
-
Haemoglobin A1c
- MOOSE:
-
Meta-analysis Of Observational Studies in Epidemiology
- NOS:
-
Newcastle-Ottawa Scale
- PBH:
-
Post-bariatric surgery hypoglycaemia
- RA:
-
Receptor agonist
- RYGB:
-
Roux-en-Y gastric bypass
References
Buchwald H, Estok R, Fahrbach K, Banel D, Jensen MD, Pories WJ, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248–56.
Chang SH, Stoll CR, Song J, Varela JE, Eagon CJ, Colditz GA. The effectiveness and risks of bariatric surgery: an updated systematic review and meta-analysis, 2003–2012. JAMA Surg. 2014;149(3):275–87.
Holst JJ, Madsbad S, Bojsen-Moller KN, Svane MS, Jorgensen NB, Dirksen C, et al. Mechanisms in bariatric surgery: gut hormones, diabetes resolution, and weight loss. Surg Obes Relat Dis. 2018;14(5):708–14.
Olst N, Meiring S, Brauw M, Bergman J, Nieuwdorp M, Peet D et al. Small intestinal physiology relevant to bariatric and metabolic endoscopic therapies: Incretins, bile acid signaling, and gut microbiome. Tech Innovat Gastroi. 2020;22.
Nguyen NQ, Debreceni TL, Bambrick JE, Bellon M, Wishart J, Standfield S, et al. Rapid gastric and intestinal transit is a major determinant of changes in blood glucose, intestinal hormones, glucose absorption and postprandial symptoms after gastric bypass. Obes (Silver Spring). 2014;22(9):2003–9.
Salehi M, Vella A, McLaughlin T, Patti ME. Hypoglycemia after gastric bypass surgery: current concepts and controversies. J Clin Endocrinol Metab. 2018;103(8):2815–26.
Capristo E, Panunzi S, De Gaetano A, Spuntarelli V, Bellantone R, Giustacchini P, et al. Incidence of hypoglycemia after gastric bypass vs sleeve gastrectomy: a randomized trial. J Clin Endocrinol Metab. 2018;103(6):2136–46.
Brix JM, Kopp HP, Höllerl F, Schernthaner GH, Ludvik B, Schernthaner G. Frequency of hypoglycaemia after different bariatric surgical procedures. Obes Facts. 2019;12(4):397–406.
Emous M, Wolffenbuttel BHR, Totte E, van Beek AP. The short- to mid-term symptom prevalence of dumping syndrome after primary gastric-bypass surgery and its impact on health-related quality of life. Surg Obes Relat Dis. 2017;13(9):1489–500.
Marsk R, Jonas E, Rasmussen F, Naslund E. Nationwide cohort study of post-gastric bypass hypoglycaemia including 5,040 patients undergoing surgery for obesity in 1986–2006 in Sweden. Diabetologia. 2010;53(11):2307–11.
Vidal J, Nicolau J, Romero F, Casamitjana R, Momblan D, Conget I, et al. Long-term effects of roux-en-y gastric bypass surgery on plasma glucagon-like peptide-1 and islet function in morbidly obese subjects. J Clin Endocrinol Metab. 2009;94(3):884–91.
Tharakan G, Behary P, Albrechtsen NJW, Chahal H, Kenkre J, Miras AD, et al. Roles of increased glycaemic variability, GLP-1 and glucagon in hypoglycaemia after Roux-en-Y gastric bypass. Eur J Endocrinol. 2017;177(6):455–64.
Craig CM, Lawler HM, Lee CJE, Tan M, Davis DB, Tong J, et al. PREVENT: a randomized, placebo-controlled crossover trial of avexitide for treatment of post-bariatric hypoglycemia. J Clin Endocrinol Metab. 2021;106(8):3235–48.
Abrahamsson N, Engström BE, Sundbom M, Karlsson FA. GLP1 analogs as treatment of postprandial hypoglycemia following gastric bypass surgery: a potential new indication? Eur J Endocrinol. 2013;169(6):885–9.
Altman DG. Systematic reviews of evaluations of prognostic variables. BMJ. 2001;323(7306):224–8.
Moons KG, de Groot JA, Bouwmeester W, Vergouwe Y, Mallett S, Altman DG, et al. Critical appraisal and data extraction for systematic reviews of prediction modelling studies: the CHARMS checklist. PLoS Med. 2014;11(10):e1001744.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12.
Geersing GJ, Bouwmeester W, Zuithoff P, Spijker R, Leeflang M, Moons KG. Search filters for finding prognostic and diagnostic prediction studies in Medline to enhance systematic reviews. PLoS ONE. 2012;7(2):e32844.
Dettori JR, Norvell DC, Chapman JR. Fixed-effect vs Random-Effects Models for Meta-Analysis: 3 points to consider. Global Spine J. 2022;12(7):1624–6.
Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Smith G, Altman D, editors. Systematic reviews in health care. Wiley; 2001. pp. 285–312.
Page MJ, Higgins JPT, Sterne JAC. Chapter 13: Assessing risk of bias due to missing results in a synthesis. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editors. Cochrane handbook for systematic reviews of interventions version 6.3 (updated February 2022). Cochrane; 2022. Available from www.training.cochrane.org/handbook.
Goldfine AB, Mun EC, Devine E, Bernier R, Baz-Hecht M, Jones DB, et al. Patients with neuroglycopenia after gastric bypass surgery have exaggerated incretin and insulin secretory responses to a mixed meal. J Clin Endocrinol Metab. 2007;92(12):4678–85.
Guarino D, Moriconi D, Mari A, Rebelos E, Colligiani D, Baldi S, et al. Postprandial hypoglycaemia after Roux-en-Y gastric bypass in individuals with type 2 diabetes. Diabetologia. 2019;62(1):178–86.
Laurenius A, Werling M, Le Roux CW, Fändriks L, Olbers T. More symptoms but similar blood glucose curve after oral carbohydrate provocation in patients with a history of hypoglycemia-like symptoms compared to asymptomatic patients after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2014;10(6):1047–54.
Lobato CB, Pereira SS, Guimarães M, Hartmann B, Wewer Albrechtsen NJ, Hilsted L, et al. A potential role for endogenous glucagon in preventing post-bariatric hypoglycemia. Front Endocrinol (Lausanne). 2020;11:608248.
Poitou C, Bouaziz-Amar E, Genser L, Oppert JM, Lacorte JM, Le Beyec J. Fasting levels of glicentin are higher in Roux-en-Y gastric bypass patients exhibiting postprandial hypoglycemia during a meal test. Surg Obes Relat Dis. 2018;14(7):929–35.
Salehi M, Gastaldelli A, D’Alessio DA. Altered islet function and insulin clearance cause hyperinsulinemia in gastric bypass patients with symptoms of postprandial hypoglycemia. J Clin Endocrinol Metab. 2014;99(6):2008–17.
Salehi M, Gastaldelli A, D’Alessio DA. Blockade of glucagon-like peptide 1 receptor corrects postprandial hypoglycemia after gastric bypass. Gastroenterology. 2014;146(3):669–80. e2.
Salehi M, Gastaldelli A, D’Alessio DA. Role of vagal activation in postprandial glucose metabolism after gastric bypass in individuals with and without hypoglycaemia. Diabetes Obes Metab. 2019;21(6):1513–7.
Soeby M, Nielsen JB, Pedersen SB, Gribsholt SB, Holst JJ, Richelsen B. Relationship between biochemical and symptomatic hypoglycemia after RYGB. Responses to a mixed meal test: a case-control study. Surg Obes Relat Dis. 2020;16(9):1179–85.
Tharakan G, Behary P, Wewer Albrechtsen NJ, Chahal H, Kenkre J, Miras AD, et al. Roles of increased glycaemic variability, GLP-1 and glucagon in hypoglycaemia after Roux-en-Y gastric bypass. Eur J Endocrinol. 2017;177(6):455–64.
Kellogg TA, Bantle JP, Leslie DB, Redmond JB, Slusarek B, Swan T, et al. Postgastric bypass hyperinsulinemic hypoglycemia syndrome: characterization and response to a modified diet. Surg Obes Relat Dis. 2008;4(4):492–9.
Vaurs C, Brun JF, Bertrand M, Burcelin R, du Rieu MC, Anduze Y, et al. Post-prandial hypoglycemia results from a non-glucose-dependent inappropriate insulin secretion in Roux-en-Y gastric bypassed patients. Metabolism. 2016;65(3):18–26.
Marathe CS, Marathe JA, Rayner CK, Kar P, Jones KL, Horowitz M. Hypoglycaemia and gastric emptying. Diabetes Obes Metab. 2019;21(3):491–8.
Service GJ, Thompson GB, Service FJ, Andrews JC, Collazo-Clavell ML, Lloyd RV. Hyperinsulinemic hypoglycemia with nesidioblastosis after gastric-bypass surgery. N Engl J Med. 2005;353(3):249–54.
Patti ME, Li P, Goldfine AB. Insulin response to oral stimuli and glucose effectiveness increased in neuroglycopenia following gastric bypass. Obes (Silver Spring). 2015;23(4):798–807.
Craig CM, Liu LF, Deacon CF, Holst JJ, McLaughlin TL. Critical role for GLP-1 in symptomatic post-bariatric hypoglycaemia. Diabetologia. 2017;60(3):531–40.
van den Broek M, de Heide LJM, Sips FLP, Koehorst M, van Zutphen T, Emous M, et al. Altered bile acid kinetics contribute to postprandial hypoglycaemia after Roux-en-Y gastric bypass surgery. Int J Obes (Lond). 2021;45(3):619–30.
Zhou LY, Deng MQ, Xiao XH. Potential contribution of the gut microbiota to hypoglycemia after gastric bypass surgery. Chin Med J (Engl). 2020;133(15):1834–43.
Mulla CM, Goldfine AB, Dreyfuss JM, Houten S, Pan H, Pober DM, et al. Plasma FGF-19 levels are increased in patients with post-bariatric hypoglycemia. Obes Surg. 2019;29(7):2092–9.
Phillips LK, Deane AM, Jones KL, Rayner CK, Horowitz M. Gastric emptying and glycaemia in health and diabetes mellitus. Nat Rev Endocrinol. 2015;11(2):112–28.
Hutch CR, Sandoval D. The role of GLP-1 in the metabolic success of bariatric surgery. Endocrinology. 2017;158(12):4139–51.
Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27(4):740–56.
Wang W, Cheng Z, Wang Y, Dai Y, Zhang X, Hu S. Role of bile acids in bariatric surgery. Front Physiol. 2019;10:374.
Tremaroli V, Karlsson F, Werling M, Stahlman M, Kovatcheva-Datchary P, Olbers T, et al. Roux-en-Y gastric bypass and Vertical Banded Gastroplasty Induce Long-Term changes on the human gut Microbiome contributing to Fat Mass Regulation. Cell Metab. 2015;22(2):228–38.
Arora T, Seyfried F, Docherty NG, Tremaroli V, le Roux CW, Perkins R, et al. Diabetes-associated microbiota in fa/fa rats is modified by Roux-en-Y gastric bypass. ISME J. 2017;11(9):2035–46.
Craig CM, Liu LF, Nguyen T, Price C, Bingham J, McLaughlin TL. Efficacy and pharmacokinetics of subcutaneous exendin (9–39) in patients with post-bariatric hypoglycaemia. Diabetes Obes Metab. 2018;20(2):352–61.
Miras AD, Perez-Pevida B, Aldhwayan M, Kamocka A, McGlone ER, Al-Najim W, et al. Adjunctive liraglutide treatment in patients with persistent or recurrent type 2 diabetes after metabolic surgery (GRAVITAS): a randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2019;7(7):549–59.
de Heide LJ, Wouda SH, Peters VJ, Oosterwerff MM, Gerdes VA, Emous M et al. Medical and surgical treatment of postbariatric hypoglycemia: retrospective data from daily practice. Diabetes Obes Metab. 2022.
Thazhath SS, Marathe CS, Wu T, Chang J, Khoo J, Kuo P, et al. The glucagon-like peptide 1 receptor agonist exenatide inhibits small intestinal motility, flow, transit, and absorption of glucose in healthy subjects and patients with type 2 diabetes: a randomized controlled trial. Diabetes. 2016;65(1):269–75.
Kim SH, Abbasi F, Nachmanoff C, Stefanakis K, Kumar A, Kalra B, et al. Effect of the glucagon-like peptide-1 analogue liraglutide versus placebo treatment on circulating proglucagon-derived peptides that mediate improvements in body weight, insulin secretion and action: a randomized controlled trial. Diabetes Obes Metab. 2021;23(2):489–98.
Jalleh RJ, Jones KL, Rayner CK, Marathe CS, Wu T, Horowitz M. Normal and disordered gastric emptying in diabetes: recent insights into (patho)physiology, management and impact on glycaemic control. Diabetologia. 2022;65(12):1981–93.
Murthy TA, Grivell J, Hatzinikolas S, Chapple LS, Chapman MJ, Stevens JE, et al. Acceleration of gastric emptying by insulin-induced hypoglycemia is dependent on the degree of hypoglycemia. J Clin Endocrinol Metab. 2021;106(2):364–71.
Abrahamsson N, Börjesson JL, Sundbom M, Wiklund U, Karlsson FA, Eriksson JW. Gastric bypass reduces symptoms and hormonal responses in hypoglycemia. Diabetes. 2016;65(9):2667–75.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
K.L.J. has received research funding from Sanofi Aventis and AstraZeneca. M.H. has participated in advisory boards and/or symposia for Novo Nordisk, Sanofi-Aventis, Novartis, Eli Lilly, Merck Sharp & Dohme, Boehringer Ingelheim and AstraZeneca and received honoraria. The other authors report no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jalleh, R.J., Umapathysivam, M.M., Plummer, M.P. et al. Postprandial plasma GLP-1 levels are elevated in individuals with postprandial hypoglycaemia following Roux-en-Y gastric bypass – a systematic review. Rev Endocr Metab Disord 24, 1075–1088 (2023). https://doi.org/10.1007/s11154-023-09823-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-023-09823-3