Abstract
Papillary thyroid carcinoma (PTC) has a good prognosis with a 10-yr survival greater than 90%. Recently, a micro-papillary pattern with hobnail appearance (MPHC) in PTC has been indicated as associated with poor prognosis, but this suggestion is based only on a few cases from geographical areas different from ours. Two-hundred ninety-nine consecutive PTC cases were collected between the years of 1992 and 2014 at our institution. The corresponding histologic sections (at least 6 for each case) were stained with hematoxylin and eosin and reviewed independently by two pathologists to reach a consensus on the identification and quantification of the MPHC. As done in other cohorts, parallel serial sections were stained with antisera for thyroglobulin, epithelial membrane antigen, thyroid-transcription-factor-1 and Ki 67. BRAF gene mutation at codon 600 and RET/PTC1 gene rearrangements were searched. A comparative statistical analysis was done between the present series and previously published series. Of the 295 PTC, 124 (42.5%) were follicular, 104 (35%) classic, 34 (11.5%) sclerosing, 15 (5%) tall cells, 10 (3.4%) Warthin-like, and 8 (2.7%) MPHC. Four MHPC cases (50%) harbored the BRAF V600E variant, while one was positive for RET/PTC1 rearrangement. Our rate of MPHC-PTC (2.7%) is 2X to 8X greater than those reported previously for cohorts from North America + North Italy, Korea and Mexico. MPHC prognosis appears to be better compared to other cohorts, probably due to not only to the lower rate of the vascular invasion, but also to the smaller size of the MPHC-PTC nodule.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Incidence rates of thyroid carcinomas have increased appreciably over the last few decades [1–3]. If recent trends are maintained, thyroid cancer may become the fourth most common cancer by 2030 in the United States [4]. Conversely, mortality rates have declined progressively in most areas of the world, likely due to improved diagnosis, management and treatment [5]. Among thyroid tumors, the papillary carcinoma (PTC) represents the most common histotype and the less malignant, which has a 5-year survival rate greater than 98% in Europe or North America [1–3]. However, unfavorable clinical and histologic features only occur in few patients, mainly in those affected by aggressive variants of PTC, such as diffuse the sclerosing, tall or columnar cell variant [6, 7]. Moreover in PTC, a newly described entity has been defined as micropapillary/hobnail variant (MPHC), which is characterized by the presence of small papillary clusters surrounded by lacunar spaces, hobnail features and high nuclear/cytoplasmic ratio [6, 7]. This MPHC subtype of PTC has been considered aggressive and rare, although its prevalence accounts for less than 1% of the whole PTC hystotype based on the few series reported in the literature [6–11].
In brief, we wished to ascertain the rate and main characteristics of the MHPC variant in our cohort of PTC from patients living in the southern most region of Italy (Sicily), and compare these indices with those of the few cohorts reported in the literature.
2 Materials and methods
A total of 295 consecutive PTC cases were collected between the years of 1992 and 2015. Of these, 242 (82%) were females and 53 (18%) males (F:M ratio = 4.7:1), with a mean age of 50 years (range 14–92). The corresponding histologic sections (at least 6 for each case) were stained with hematoxylin and eosin. Of the 295 PTC, 124 (42.5%) were codified as follicular variant, 104 (35%) as classic PTC, 34 (11.5%) as sclerosing, 15 (5%) as tall cells, 10 (3.4%) as Warthin-like, and 8 (2.6%) as micropapillary/hobnail variant. Stained sections of this last variant were reviewed independently by two pathologists in order to identify and quantify the MPHC pattern (that is, at least ≥30% of the tumor, as defined by Asioli et al. [6]). Consequently, a cohort of 8 MPHC was identified and selected for the present study. These 8 cases were from 8 patients who were thyroidectomized in the years 2010 to 2015.
Additional 4 μ-thick serial sections mounted on silane-coated slides were cut and stained with the following antisera: thyroglobulin (TG; Dako, Glostrup, Denmark, clone DAK-Tg6, working dilution: w.d. 1:200), epithelial membrane antigen (EMA; Dako, clone E29, w.d. 1:50), thyroid-transcription-factor-1 (TTF-1; Dako, clone 8G7G3/1, w.d. 1:200) and Ki 67 (Dako, clone MIB-1, w.d. 1:200). The nuclear counterstain was performed by Mayer’s hematoxylin. Immunoreactions were performed according to standardized procedures by an automated immunostainer (Dako autostainer Link48) and developed by En-Vision detection system (Dako), utilizing 3-3′diaminobenzidine tetrahydrochloride as chromogen as elsewhere suggested [12–15].
2.1 BRAF analysis
In order to evaluate BRAF mutational status, four 10 μm thick H&E-stained sections were microdissected by scalpel using an inverted microscope in order to collect only regions with the highest MPHC representation; distant nonneoplastic thyroid tissue was utilized as control. DNA extraction was performed by QIAamp DNA FFPE Tissue kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s recommendations and DNA quantified by fluorometry with the Qubit® platform (Life Technologies, CA, USA). DNA samples were subjected to BRAF mutational analysis utilizing the BRAF Codon 600 Mutation Analysis Kit II (EntroGen, Inc, CA, USA) that allows to identify five BRAF somatic mutations in codons 600 (V600D, V600E, V600K, V600M, V600R). The amplifications were carried out in a StepOnePlus™ Real-Time PCR system (Life Technologies) following the manufacturer’s procedures and the recommendations of both the Italian Association of Medical Oncology (AIOM) and the Italian Society of Pathology and Cytology (SIAPEC). Moreover, BRAF mutation negative cases were analyzed for RET/PTC gene rearrangement by the RNA extraction as elsewhere reported [16], followed by RT-PCR.
2.2 Statistical analysis
All data were analyzed by using the SPSS package version 6.1.3 (SPSS, Chicago, IL, USA). A comparative statistical analysis was done between our series and the series published previously [6–11]. A P value less than 0.05 was considered statistically significant, while a P value between 0.10 and 0.05 was considered borderline significant.
3 Results
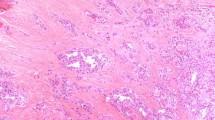
Upon reviewing the 295 PTC, the two pathologists agreed on the identification of six variants: 124 (42.5%) were follicular, 104 (35%) classic, 34 (11.5%) sclerosing, 15 (5%) tall cells, 10 (3.4%) Warthin-like and lastly 8 (2.7%) MPHC. Relevant data of the MHPC variant regarding age, sex, gross pathology, tumor stage and post-surgery radiotherapy are reported in Table 1. Mean age was 55 years, women outnumbered men by factor of 3, and the preferred localization was the left lobe (50% of cases). The maximum diameter, a proxy of tumor size, ranged from 10 to 32 mm (mean 17.75 mm). At surgery, two cases exhibited cervical lymph node metastases (cases no. 5 and 7). Neither recurrences nor distant metastases were recorded over a follow-up period that ranged from 39 to 60 months (mean 47.75) (Table 1). Histologically, the MPHC group showed a percentage of hobnail feature (Fig. 1a, b) varying from 30 to 70% (mean 41.87) characterized by a micropapillary architecture, the papillae with vascular cores were covered by neoplastic elements with abundant eosinophilic cytoplasm and high N/C ratio; occasionally, grooved nuclei with intranuclear holes were also observed. There was an evident direct correlation between the maximum diameter of the MPHC nodule and the percent MPHC pattern (r = 0.57, P = 0.14; data not shown). No atypical mitoses neither necrosis were encountered. Upon careful histological review of the 8 MHPC cases, the two pathologists agreed on the coexistence of two types of lesions, namely hyperplastic nodules (6/8 = 75%) or chronic lymphocytic thyroiditis (CLT; 2/8 = 25%).
By immunohistochemistry, all MPHC cases expressed thyroglobulin (cytoplasm) and TTF-1 (nucleus). EMA was positive in all cases with a peculiar usual membrane staining. The proliferative index measured by Ki67 antibody ranged from 2 to 20% (mean 6%) of the neoplastic elements (Fig. 2).
The rate of the BRAF p.V600E (c.1799T>A) mutation was 4/8 MHPC cases (50%) (Table 1). These 4 BRAF-mutated cases showed lymphatic invasion, with 2/4 presenting lymph node metastases at surgery. Among the 4 BRAF negative cases, only one exhibited RET/PTC1 gene rearrangement.
Table 2 summarizes the comparison of our 8 cases with the other 66 MPHC cases from six studies appeared in the English-language literature [6–11]. With two exceptions [7, 11], the F:M ratio is 3:1 and mean age comprised between 54 and 58 years. Our series stands out for the smallest diameter of the tumor (tied with the Korean series), lowest proportion of patients at stage IV (0%), and lowest proportion of patients with recurrent disease (0%). Though the follow-up of the Korean series was shorter compared to ours (9 to 28 months [median 12 months], vs. 39 to 60 months [mean 47.7], the Korean patients resemble our patients for the low rate of recurrence (10%), absence of distant metastases and high rate of disease-free survival (90%).
4 Discussion
A peculiar micropapillary neoplastic architectural pattern (MPC), firstly described in the mammary gland by Siriaunkgul & Tavassoli [17], was recognized as a distinct entity also in other organs, including the bladder, lung, pancreas, parotid gland, gallbladder, colorectum, ovary and stomach [17–21]. In most of these organs, studies have documented a frequent association of MPC with lymphovascular invasion, high propensity for lymph-node metastasis and consequent reduction in survival and increased mortality with poor clinical outcome [18–22].
In the thyroid literature, small-sized series of MPHC were reported, and they were characterized by a typical micropapillary growth pattern, hobnail appearance, inversion of cellular polarity, enlarged eosinophilic cytoplasm and nucleus/cytoplasm ratio [6–11].
Although MPHC has been only recently identified as a very rare PTC variant, its clinico-pathologic features suggested an aggressive behavior, as documented by larger volume, extrathyroidal extension, increased rate of spread to lymph nodes and distant organs [6–11]. Our rate of the MPHC variant is 2.7% of the whole PTC cohort. This frequency is 2X to 8X greater than those reported in previous series from South Korea (0.34%, P = 2.2 × 10−7) [11], North America/North Italy (0.7%, P = 0.0002) [8], North America (≈1%; exact denominator not given) [10] or Mexico (1.4%, P = 0.19) [7].
Another interesting finding of our MPHC series is represented by the smaller size of the neoplastic nodules compared to that of the other series, with the exception of the South Korea series [11]. Probably, the smaller size of MPHC at the time of thyroidectomy contributes to the best outcome of the disease in our MPHC cohort. This is similar to the data form of the Korean series [11], in which a mean maximum diameter of 16.3 mm was associated with a disease-free survival equal to 90%. Moreover, in our MPHC cohort, the mean Ki67 value was 6%, lower than values reported in other MPHC cohorts. [6, 7]. Consequently, taking into account the well-known direct correlation between expression of Ki67 and both metastatization and unfavorable prognosis in papillary thyroid microcarcinoma [11], the lower Ki67 percentage represents an additional explanation for the absent aggressive behavior in our MPHC cohort.
In all 4 MHPC patients having the intratumoral BRAFV600E mutation, the tumor was complicated by lymphatic invasion, and in 2/4 by the cervical lymph nodes spread. These two cases with lymph node involvement featured the highest proportion of MHPC pattern (60-70%), greater than the 30-40% range of the remaining 6 cases with no spread to lymph nodes. Of note, the two BRAF V600E-positive MHPC that had no cervical lymph node metastasis despite an evident lymphatic invasion, had a maximum diameter of 10 and 12 mm, while the two BRAF V600E-positive MPHC that spread to cervical lymph nodes had a maximum diameter of 14 and 32 mm. Of interest, within the 48–52 months of follow-up, these two patients had no disease recurrence. This favourable outcome could have been favored by the concurrent chronic lymphoctic thyroidits (CLT), the only two patients in whom this coexistence occurred. Indeed, CLT is known as an independent predictor for less aggressiveness in conventional PTC patients regardless of BRAF mutation status [23–27].
In PTC patients from several geographical areas, BRAFV600E mutation was significantly correlated with known clinicopathological poor prognostic factors, such as extrathyroidal extension, lymph node metastasis and advanced stage [23–32]. By contrast, this unfavorable correlation was not confirmed in other studies [33, 34]. Therefore, it is not surprising that interstudy differences may also apply to the prognostic significance of the BRAFV600E in the setting of the MPHC subtype. In any case, this BRAF mutation represents an early step during thyroid micropapillary carcinogenesis because it is detectable also in small size neoplasms, including those such as our cases, characterized by lymphatic or lymph node colonization. Moreover, the prognostic implications of BRAF mutation have been underlined in T1 papillary carcinomas, which are tumors of 2 cm or less in size; in this category, incidentally discovered papillary microcarcinomas measuring 1 cm or less have been encompassed [35–40]. Finally, among our BRAF negative cases, only one exhibited RET/PTC1 gene rearrangement.
In our MPHC series, we have noticed the coexistence of two thyroid lesions, hyperplastic nodules (75%) or chronic lymphocytic thyroiditis (25%). Since the coexistence of other lesions was disregarded by the other studies [6–11], we cannot contrast our data with the literature. Nevertheless, in recent studies, the presence of CLT in patients with PTC was reported to be associated with lower recurrence rate of PTC and improved survival rate [41–47]. Indeed, in the presence of CLT, PTC is associated with pathologic markers of decreased tumor aggressiveness, such as small tumor size and low stage, no extrathyroid extension, no lymph node metastases, better locoregional control, lower rates of recurrence and greater overall and disease-free survival [41–47]. A similar clinical behavior was found in our MPHC series, in which no recurrences, distant metastasis or cancer-related mortality were observed, even if two cases had metastatic lymph nodes.
In conclusion, our Sicilian MPHC series is the only one with the highest representativity (almost 3%) within the whole PTC hystotype. Because our cohort was collected in a recent period that spanned the years 2010 though 2015, we cannot have a long follow-up duration. However, within an average of 4 years after thyroidectomy, 100% of the patients were still alive with no disease. Unlike the generality of the literature, our MHPC cases feature a good prognosis.
References
La Vecchia C, Malvezzi M, Bosetti C, Garavello W, Bertuccio P, Levi F, et al. Thyroid cancer mortality and incidence: a global overview. Int J Cancer. 2015;136:2187–95.
AIRTUM Working Group, Busco S, Buzzoni C, Mallone S, Trama A, Castaing M, et al. Italian cancer figures--report 2015: the burden of rare cancers in Italy. Epidemiol Prev. 2016;40:83.
Kitahara CM, Sosa JA. The changing incidence of thyroid cancer. Nat Rev Endocrinol. 2016.
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913–21.
Bosetti C, Bertuccio P, Malvezzi M, Levi F, Chatenoud L, Negri E, et al. Cancer mortality in Europe, 2005–2009, and an overview of trends since 1980. Ann Oncol. 2013;24:2657–71.
Asioli S, Erickson LA, Sebo TJ, Zhang J, Jin L, Thompson GB, et al. Papillary thyroid carcinoma with prominent hobnail features: a new aggressive variant of moderately differentiated papillary carcinoma. A clinicopathologic, immunohistochemical, and molecular study of eight cases. Am J Surg Pathol. 2010;34:44–52.
Lino-Silva LS, Domínguez-Malagón HR, Caro-Sánchez CH, Salcedo-Hernández RA. Thyroid gland papillary carcinomas with “micropapillary pattern,” a recently recognized poor prognostic finding: clinicopathologic and survival analysis of 7 cases. Hum Pathol. 2012;43:1596–600.
Asioli S, Erickson LA, Righi A, Lloyd RV. Papillary thyroid carcinoma with hobnail features: histopathologic criteria to predict aggressive behavior. Hum Pathol. 2013;44:320–8.
Asioli S, Maletta F, Pagni F, Pacchioni D, Vanzati A, Mariani S, et al. Cytomorphologic and molecular features of hobnail variant of papillary thyroid carcinoma: case series and literature review. Diagn Cytopathol. 2014;42:78–84.
Lubitz CC, Economopoulos KP, Pawlak AC, Lynch K, Dias-Santagata D, Faquin WC, et al. Hobnail variant of papillary thyroid carcinoma: an institutional case series and molecular profile. Thyroid. 2014;24:958–65.
Lee YS, Kim Y, Jeon S, Bae JS, Jung SL, Jung CK. Cytologic, clinicopathologic, and molecular features of papillary thyroid carcinoma with prominent hobnail features: 10 case reports and systematic literature review. Int J Clin Exp Pathol. 2015;8:7988–97.
Ieni A, Barresi V, Branca G, Giuffrè G, Rosa MA, Tuccari G. Immunoexpression of lactoferrin in bone metastases and corresponding primary carcinomas. Oncol Lett. 2013;5:1536–40.
Ieni A, Barresi V, Caltabiano R, Cascone AM, Del Sordo R, Cabibi D, et al. Discordance rate of HER2 status in primary breast carcinomas versus synchronous axillary lymph node metastases: a multicenter retrospective investigation. Onco Targets Ther. 2014;7:1267–72.
Ieni A, Barresi V, Caltabiano R, Caleo A, Bonetti LR, Lanzafame S, et al. Discordance rate of HER2 status in primary gastric carcinomas and synchronous lymph node metastases: a multicenter retrospective analysis. Int J Mol Sci. 2014;15:22331–41.
Barresi V, Ieni A, Cardia R, Licata L, Vitarelli E, Reggiani Bonetti L, et al. HOXB13 as an immunohistochemical marker of prostatic origin in metastatic tumors. APMIS. 2016;124:188–93.
Salvatore G, Giannini R, Faviana P, Salvatore G, Giannini R, Faviana P, et al. Analysis of BRAF point mutation and RET/PTC rearrangement refines the fine-needle aspiration diagnosis of papillary thyroid carcinoma. J Clin Endocrinol Metab. 2004;89:5175–80.
Siriaunkgul S, Tavassoli FA. Invasive micropapillary carcinoma of the breast. Mod Pathol. 1993;6:660–2.
Amin MB, Ro JY, El-Sharkawy T, Lee KM, Troncoso P, Silva EG, et al. Micropapillary variant of transitional cell carcinoma of the urinary bladder. Histologic pattern resembling ovarian papillary serous carcinoma. Am J Surg Pathol. 1994;18:1224–32.
Barresi V, Vitarelli E, Grosso M, Tuccari G, Barresi G. Relationship between immunoexpression of mucin peptide cores MUC1 and MUC2 and Lauren’s histologic subtypes of gastric carcinomas. Eur J Histochem. 2006;50:301–9.
Barresi V, Branca G, Vitarelli E, Tuccari G. Micropapillary pattern and poorly differentiated clusters represent the same biological phenomenon in colorectal cancer: a proposal for a change in terminology. Am J Clin Pathol. 2014;142:375–83.
Barresi V, Branca G, Ieni A, Rigoli L, Tuccari G, Caruso RA. Phagocytosis (cannibalism) of apoptotic neutrophils by tumor cells in gastric micropapillary carcinomas. World J Gastroenterol. 2015;21:5548–54.
Cao Y, Zhu LZ, Jiang MJ, Yuan Y. Clinical impacts of a micropapillary pattern in lung adenocarcinoma: a review. Onco Targets Ther. 2015;9:149–58.
Kim SK, Woo JW, Lee JH, Park I, Choe JH, Kim JH, et al. Chronic lymphocytic thyroiditis and BRAF V600E in papillary thyroid carcinoma. Endocr Relat Cancer. 2016;23:27–34.
Kwak HY, Chae BJ, Eom YH, Hong YR, Seo JB, Lee SH, et al. Does papillary thyroid carcinoma have a better prognosis with or without Hashimoto thyroiditis? Int J Clin Oncol. 2015;20:463–73.
Lim JY, Hong SW, Lee YS, Kim BW, Park CS, Chang HS, et al. Clinicopathologic implications of the BRAF(V600E) mutation in papillary thyroid cancer: a subgroup analysis of 3130 cases in a single center. Thyroid. 2013;23:1423–30.
Marotta V, Guerra A, Zatelli MC, Uberti ED, Di Stasi V, Faggiano A, et al. BRAF mutation positive papillary thyroid carcinoma is less advanced when Hashimoto’s thyroiditis lymphocytic infiltration is present. Clin Endocrinol (Oxf). 2013;79:733–8.
Kim SK, Song KH, Lim SD, Lim YC, Yoo YB, Kim JS, et al. Clinical and pathological features and the BRAF(V600E) mutation in patients with papillary thyroid carcinoma with and without concurrent Hashimoto thyroiditis. Thyroid. 2009;19:137–41.
Benvenga S. Update on thyroid cancer. Horm Metab Res. 2008;40:323–8.
Benvenga S, Koch CA. Molecular pathways associated with aggressiveness of papillary thyroid cancer. Curr Genomics. 2014;15:162–70.
Murugan AK, Qasem E, Al-Hindi H, Shi Y. Alzahrani AS Classical V600E and other non-hotspot BRAF mutations in adult differentiated thyroid cancer. J Transl Med. 2016;14:204.
Fraser S, Go C, Aniss A, Sidhu S, Delbridge L, Learoyd D, et al. BRAF(V600E) mutation is associated with decreased disease-free survival in papillary thyroid cancer. World J Surg. 2016;40:1618–24.
Aragon Han P, Kim HS, Cho S, Fazeli R, Najafian A, Khawaja H, et al. Association of BRAF(V600E) mutation and microrna expression with central lymph node metastases in papillary thyroid cancer: a prospective study from four endocrine surgery centers. Thyroid. 2016;26:532–42.
Li F, Chen G, Sheng C, Gusdon AM, Huang Y, Lv Z, et al. BRAFV600E mutation in papillary thyroid microcarcinoma: a meta-analysis. Endocr Relat Cancer. 2015;22:159–68.
Ma YJ, Deng XL, Li HQ. BRAF(V600E) mutation and its association with clinicopathological features of papillary thyroid microcarcinoma: a meta-analysis. J Huazhong Univ Sci Technolog Med Sci. 2015;35:591–9.
Finkel A, Liba L, Simon E, Bick T, Prinz E, Sabo E, et al. Sub-clonality for BRAF mutation in papillary thyroid carcinoma is associated with earlier disease stage. J Clin Endocrinol Metab. 2016;101:1407–13.
Lee DY, Hwang SM, An JH, Son KR, Baek SK, Kim SG et al. Predicting extrathyroidal extension in patients with papillary thyroid microcarcinoma according to a BRAF mutation. Clin Exp Otorhinolaryngol. 2016.
Chen Y, Sadow PM, Suh H, Lee KE, Choi JY, Suh YJ, et al. BRAF(V600E) is correlated with recurrence of papillary thyroid microcarcinoma: a systematic review, multi-institutional primary data analysis, and meta-analysis. Thyroid. 2016;26:248–55.
Sun Y, Shi C, Shi T, Yu J, Li Z. Correlation between the BRAF(v600E) gene mutation and factors influencing the prognosis of papillary thyroid microcarcinoma. Int J Clin Exp Med. 2015;8(12):22525–8.
Bastos AU, Oler G, Nozima BH, Moysés RA, Cerutti JM. BRAF V600E and decreased NIS and TPO expression are associated with aggressiveness of a subgroup of papillary thyroid microcarcinoma. Eur J Endocrinol. 2015;173:525–40.
Tallini G, de Biase D, Durante C, Acquaviva G, Bisceglia M, Bruno R, et al. BRAF V600E and risk stratification of thyroid microcarcinoma: a multicenter pathological and clinical study. Mod Pathol. 2015;28:1343–59.
Kim EY, Kim WG, Kim WB, Kim TY, Kim JM, Ryu JS, et al. Coexistence of chronic lymphocytic thyroiditis is associated with lower recurrence rates in patients with papillary thyroid carcinoma. Clin Endocrinol (Oxf). 2009;7:581–6.
Yoon YH, Kim HJ, Lee JW, Kim JM, Koo BS. The clinicopathologic differences in papillary thyroid carcinoma with or without co-existing chronic lymphocytic thyroiditis. Eur Arch Otorhinolaryngol. 2012;269:1013–7.
Benvenga S. On the association between Hashimoto’s thyroiditis and papillary thyroid carcinoma: looking 100 years back, and hopefully, fewer years ahead to sort out this association. Thyroid. 2013;23:1180–1.
Latina A, Gullo D, Trimarchi F, Benvenga S, Trimarchi F, Benvenga S. Hashimoto’s thyroiditis: similar and dissimilar characteristics in neighboring areas. Possible implications for the epidemiology of thyroid cancer. PLoS One. 2013;8, e55450.
Lee JH, Kim Y, Choi JW, Kim YS. The association between papillary thyroid carcinoma and histologically proven Hashimoto’s thyroiditis: a meta-analysis. Eur J Endocrinol. 2013;168:343–9.
Lun Y, Wu X, Xia Q, Han Y, Zhang X, Liu Z, et al. Hashimoto’s thyroiditis as a risk factor of papillary thyroid cancer may improve cancer prognosis. Otolaryngol Head Neck Surg. 2013;148:396–402.
Dobrinja C, Bernardi S, Fabris B, Eramo R, et al. Surgical and pathological changes after radiofrequency ablation of thyroid nodules. 2015; 2015:576576.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Ieni, A., Barresi, V., Cardia, R. et al. The micropapillary/hobnail variant of papillary thyroid carcinoma: A review of series described in the literature compared to a series from one southern Italy pathology institution. Rev Endocr Metab Disord 17, 521–527 (2016). https://doi.org/10.1007/s11154-016-9398-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-016-9398-4