Abstract
The photosynthetic oxygen evolution rate, Hill reaction activity of seedlings and photosynthetic parameter, Pn–Ci curve and some source-sink metabolism-related enzyme activities, and substance content of flag leaves were measured by using two wheat near isogenic lines with significant differences in the photosynthetic rate of the 154 (high photosynthetic rate) and 212 (low photosynthetic rate) lines as materials. The results showed that the maximal carboxylation efficiency (Vcmax) and Hill reaction activity were higher in line 154 than that of line 212. The Pn in flag leaves of line 154 was significantly higher than that of line 212 during the anthesis to grain-filling stage. Higher leaf sucrose phosphate synthase activity, grain sucrose synthase activity, and grain ADPG pyrophosphorylase activity ensured that the photosynthate of line 154 could be transported to grains and translated into starch in a timely and effective manner, which also contributed to the maintenance of its high photosynthetic rate. Eventually, all of these factors of line 154 resulted in its higher grain yield compared with the low photosynthetic rate of line 212.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wheat is a globally dominant food crop, essential to food security and is closely related to human life (Araus et al. 2007; Braun et al. 2010; Reynolds et al. 2011, 2012; Shewry 2009). As the global demand for wheat has grown, most countries have focused on increasing the yield of wheat per unit area (Hawkesford et al. 2013; He et al. 2011). It is predicted that the annual global growth demand for wheat from now to 2030 will be 1.6 %, while the yield growth from 1982 to 2008 was 1.3 % (Dixon 2009). By 2050, the demand for wheat in developed countries will have increased by 60 %, while climate change will have reduced wheat yields by 29 % in developing countries (Rosegrant and Agcaoili 2010). Given that production levels continue to increase and the maximum yield remains stagnant, many scientists endeavor to increase the yield potential of wheat through a second “green revolution” by improving its photosynthetic performance and promoting light utilization (Araus et al. 2008; Evans 2013; Long et al. 2006; Reynolds et al. 2009; Van Camp 2005; Zhu et al. 2008, 2010). Over the years, the relationship between the leaf photosynthetic rate and crop yield has been controversial. Some scientists believe that there is a positive correlation between the photosynthetic rate and crop yield (Blum 1990; Fischer et al. 1998; Murchie et al. 2009; Parry et al. 2010; Raines 2011; Zheng et al. 2011; Zhu et al. 2008, 2010), while others believe that there is a negative correlation between the two (Evans 1993; Moll et al. 1994). Xu (1999) suggested that a positive correlation is an accurate reflection of the relationship between the leaf photosynthetic rate and crop yield.
Wheat yield and grain weight depend on the capacity of source tissues (especially flag leaves) to produce photoassimilates during the grain-filling process, as well as on the ability of sink tissues to convert this photoassimilates into dry matter such as starch (Tuncel and Okita 2013; Xiong et al. 1994; Zhang et al. 2001).Therefore, the plant’s production capacity of photoassimilates (sources) and the grain’s conversion and storage capacity of photoassimilates (sinks) after flowering have become important factors limiting wheat production (Miralles and Slafer 2007; Wang and He 1999). The key period for wheat grain formation occurs from the flowering period to the mature period. During this period, leaf photoassimilates are formed and transported mainly in the form of sucrose (Lalonde et al. 2004; Lbraheem et al. 2008; Lemoine 2000; Reynolds et al. 2012; Xiong et al. 1994), therefore maintaining a high leaf photosynthetic capacity and adequate supply of sugar is advantageous for improving yield. The synthesis and accumulation of starch is closely related to the crop yield as starch accounts for approximately 70 % of the total dry weight of grains (Awika 2011; Jiang et al. 2002a, b, c).
Photosynthesis is a comprehensive physiological process that is mutually connected and restricted by the light and dark reactions, and the photosynthetic rate is an important indicator of photosynthesis. Past research on photosynthetic rates in wheat was mainly conducted on different genotype varieties and between the parental and offspring generations (Cheng et al. 2009; Jiang et al. 2002a, b, c; Liu et al. 2002; Yang et al. 2006, 2007, Zivcak et al. 2013). The different genetic backgrounds used in the study of the photosynthetic rate increase the difficulty of further studying the key control process and site of the photosynthetic rate in wheat. There are few reports about the relationships between flag leaf photosynthesis, sucrose synthesis in flag leaves, and sucrose degradation and starch synthesis in grains. In this study, we used two wheat near isogenic lines (line 154 and line 212) with significant differences in the photosynthetic rate to explore the relationship between the leaf photosynthetic rate and source-sink adjustments. Our results indicate that higher activity of leaf sucrose phosphate synthase (SPS), grain sucrose synthase (SS), and grain ADPG pyrophosphorylase (AGP) enhanced the photosynthate of line 154 to be transported to grains and translated into starch in a timely and effective manner, which also contributed to the maintenance of its high photosynthetic rate.
Materials and methods
Plant materials
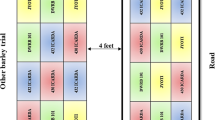
The NILs (line 154 and line 212) of wheat were selected from crossing winter wheat Xiaoyan 54 × 8602 using the net photosynthetic rate (Pn) as a physiological selection index from the F6 progeny, with line 154 as the high photosynthetic rate line. After six generations, they were genetically stable and showed no separation phenomenon. Seeds were grown in one of two conditions: in a greenhouse or in the field. For the hydroponic seedlings, plump seeds were germinated on filter paper moistened with water for 12 h at 25 ± 1 °C in an illumination incubator after being sterilized with 0.2 % sodium hypochlorite. The seeds were then grown in plastic pots with soil at the appropriate density containing Hoagland solution in a greenhouse at 25 °C under a 16 h/8 h day/night photoperiod (300–400 μmol m−2 s−1) until the eight-leaf stage. For the field experiment, during the 2013 and 2014 growing seasons, plump seeds were grown in the fields of the Experimental Station of Shandong Agricultural University. Each line had six 4 m2 interspersed plots with 25 cm spaced rows and 5 cm spaced lines, and conventional agricultural management was maintained during the growing seasons of the plants. Flag leaves in field and hydroponic seedling leaves were used for the analyses.
Measurement of the photosynthetic oxygen evolution rate of seedlings
Leaf disks of wheat seedlings at the eight-leaf stage with a 0.55-cm diameter were collected and then fixed in a reaction cup containing 50 mmol/L sodium bicarbonate with 800 μmol m−2 s−1 light intensity. The oxygen evolution rate was determined using a Chlorolab- 2 liquid oxygen electrode (Hansatech, UK) at room temperature.
Measurement of the Hill reaction activity of the chloroplast
Chloroplast isolation and Hill reaction activity assays were conducted as described by Zhao et al. (2007), who modified the procedure published by Ye and Qian (1985).
Measurement of the photosynthetic gas exchange parameters of flag leaves
Intact wheat flag leaves in the field were used to measure the net photosynthetic rate (Pn), intercellular CO2 concentration (Ci), and stomatal conductance (Gs) with a portable photosynthetic system (CIRAS-2, PP Systems, Hitchin, UK) in the morning between 9:00 and 11:00. Measurements were made under atmospheric CO2 concentrations with saturating photosynthetic photo flux densities at 1400 μmol m−2 s−1 PPFD, the temperature inside leaf chamber was controlled at 25 °C, and ambient humidity approximate 70 % was used. To obtain the maximal carboxylation efficiency (Vcmax), the Pn was measured under a PPFD of 1400 μmol m−2 s−1 and under CO2 concentrations in a sequence of 1400, 1200, 1000, 700, 400, 300, 250, 200, 150, 100, and 50 μmol m−2 s−1. To allow the leaves to reach steady-state photosynthesis, the leaves were kept under each level of CO2 concentration for 5 min before the Pn was recorded. The slope of the linear correlation of the Pn–Ci response was calculated as Vcmax (Von Caemmerer and Farquhar 1981; Farquhar et al. 1980).
Measurement of the sucrose content
Grain samples of 50 mg of dry weight were extracted twice using 4 ml of 80 % boiling ethanol for 40 min. The supernatant was collected and decolorized with 10 mg of activated carbon at 80 °C for 30 min, fixed to a constant volume at 10 ml, and purified by filtering. Two-hundred microliters of 2 mol/L sodium hydroxide was added to the 0.4 ml filtrate, boiled at 100 °C for 5 min, then 0.8 ml of 0.1 % resorcinol was added and the reaction mixture was boiled at 80 °C for 10 min. Once cooled, the starch content of the reaction mixture was determined by measuring the OD value at 480 nm.
Measurement of the starch content
Samples of 0.10–0.30 g of dry weight were extracted twice using 5–10 ml of boiling water for 30 min. Fractions were collected and purified by filtering. The fractions were boiled in 20 ml of water for 15 min. Then, 2 ml of 9.2 mol/L perchloric acid was added to each fraction and extracted for 15 min, filtered, and the volume of filtrate adjusted to 50 ml. Next, 0.5 ml of the diluted filtrate and 1.5 ml distilled water were mixed together in a 20 ml tube. To this mixture, 0.5 ml of anthrone ethyl acetate and 5 ml of concentrated sulfuric acid were added and the mixture immediately boiled at 100 °C for 1 min. Once cooled, the sucrose content of the reaction mixture was determined by measuring the OD value at 630 nm.
Measurement of the activity of SPS and SS
The enzymatic activities of SPS and SS were determined using a kit from Nanjing Jiancheng Bioengineering Institute.
Measurement of the activity of AGPase
The activity of AGPase was determined using a kit from Beijing Solarbio technology limited company.
Statistical analysis
The data were preprocessed using Excel 2013 (Microsoft, Redmond, WA), and the figures were generated using Sigma-Plot 10.0 (Systat Software, Erkrath, Germany). Significant differences among the data were determined through an analysis of variance using SAS (version 8.1; SAS Institute, Cary, NC), followed by the Tukey test for multiple comparisons.
Results
Photosynthetic oxygen evolution rate and Hill reaction activity of seedlings
As shown in Fig. 1a, there was a significant difference (P < 0.01) in the photosynthetic oxygen evolution rate of the two NILs wheat seedlings; line 154 had a 24.1 % higher rate compared with line 212. This suggests that a significant difference in the photosynthetic rate between the two NILs exists at the seedling stage. Hill reaction activity is an important index for the photochemical activity of leaf photosynthesis during the light reaction stage and reflects the structure, functional integrity, and electron transfer efficiency of PSII. As shown in Fig. 1b, line 154 had a 28.6 % higher activity compared with line 212 (P < 0.01), which suggests that line 154 had a higher photochemical activity of leaf photosynthesis during the light reaction stage.
Comparison of the photosynthetic oxygen evolution rate (a) and the Hill reaction activity (b) between the two wheat NILs seedlings. Each bar represents the mean ± SE of three independent experiments. 154 represents line 154; 212 represents line 212. Double asterisk indicates significant differences at 1 % level via Tukey’s test
Changes in the photosynthetic characteristics of flag leaves during the anthesis-filling stage
The net photosynthetic rate (Pn) of the two wheat NILs rose after anthesis and peaked on the 10th day after anthesis. The Pn of line 154 was significantly higher than the Pn of line 212 during the whole stage (Fig. 2a).
Stoma, channels for water and CO2, regulates photosynthesis and transpiration in the plant. Stomatal conductance (Gs) is an index of stomatal aperture. Similar to the results in the photosynthetic rate, the Gs of both NILs peaked on the 10th day after anthesis. Line 154 had significantly higher stomatal conductance than line 212 during the early and late grain-filling stages (Fig. 2b).
The intercellular CO2 concentration (Ci) of flag leaves decreased until 10 days after anthesis, at which point it increased (Fig. 2c). During the entire measurement time period, the Ci of line 154, the line with a high photosynthetic rate, was relatively low.
The stomatal limitation value (Ls) of flag leaves showed a low–high-low trend after anthesis, peaking on the 10th day. During the entire measurement time period, line 154 had a significantly higher Ci than line 212.
Ci and Ls are the two indexes for determining the cause of the decline of Pn. When Pn and Ci decline and Ls rises, then the decline of Pn is caused by stomatal limitation. When Pn and Ls decline and Ci rises, then the decline of Pn is mainly caused by non-stomatal limitation (Farquhar and Sharkey 1982). Our results (Figs. 2a, c, d) indicate that the difference in Pn of the two wheat NILs was caused by non-stomatal limitation.
Comparison of the Pn–Ci curves of flag leaves
Pn–Ci curves were investigated under saturated light intensity (PPFD = 1400 µmol m−2 s−1) to explore the cause of the difference in photosynthetic rate between the two wheat NILs. As shown in Fig. 3a, the Pn of the two lines increased with increasing Ci and the rate of line 154 was always higher than line 212. Vcmax (maximal carboxylation efficiency), as calculated from the curve, was 15.6 % higher in line 154 than in line 212. Significant differences of Vcmax were found between the two lines (P < 0.05) and are most likely one of the reasons for the difference in Pn of the two wheat NILs.
Pn–Ci curve of flag leaves of the two wheat NILs (a). The maximal rate of carboxylation (Vcmax) was calculated as the slope of the linear correlation in the relatively low range of Ci for each line (b). Each bar represents the mean ± SE of three independent experiments. 154 represents line 154; 212 represents line 212
Changes in the SPS, SS activities, and sucrose content of flag leaves in the two NILs
Sucrose phosphate synthetase (SPS) is an important regulatory enzyme in sucrose synthesis that catalyzes the reaction: UDP-glucose + fructose-6-P ↔ UDP + sucrose-6-P. Higher SPS activity translates to increased phosphorylation and transport of sucrose. As shown in Fig. 4a, the activity of SPS in flag leaves in both NILs increased after anthesis and peaked on the 18th day after anthesis before declining significantly thereafter. The activity of SPS in line 154 was always higher than line 212 during the whole period, which suggests that line 154 had a higher sucrose supply capacity.
Sucrose synthase (SS) is a cytosolic enzyme that is essential to sucrose metabolism and catalyzes the reversible reaction: UDPG + Fru ↔ Suc + UDP (Lu et al. 2005). As shown in Fig. 4b, the activity of SS in flag leaves started to increase and peaked 14 days after anthesis in line 154 and 18 days after anthesis in line 212. The activity of SS in flag leaves began to decline after the 18th day post anthesis. For the duration of experiment, the SS activity in flag leaves from line 154 was significantly higher than that of line 212, suggesting that line 154 had an increased ability to synthesize sucrose.
Sucrose is the main synthesized and transported form of photoassimilates. The key period for wheat grain yield formation occurs from the flowering to the mature period and the synthesis of sucrose in flag leaves is a reflection of the ability of source tissues to supply photoassimilates during this stage. As shown in Fig. 4c, the sucrose content in flag leaves showed a low–high-low trend. One possible reason for the low level of sucrose content in the early filling stage may be that the grain was at the construction stage, so the storage capacity was low, which restricted the transfer of sucrose. The highest content of sucrose appeared on the 10th day after anthesis in the middle of the grain-filling stage, at which point the Pn and sucrose synthesis activity were high. The decreased sucrose content of flag leaves during the late filling stage may be associated with the declined photosynthetic rate, the slow pace of sucrose synthesis, the reduced sucrose outward transport capacity, and other factors. The sucrose content of flag leaves in line 154 was higher than in line 212 during the whole period, which suggests that line 154 had a higher photoassimilates synthesis ability and leaf sucrose supply capacity.
Changes in the SS activity and sucrose content of grains in two wheat NILs
Sucrose synthase (SS) catalyzes both the synthesis and degradation of sucrose. In wheat grains, its main role is to provide substrates for the synthesis of starch by catalyzing the degradation of sucrose to fructose and uridine diphosphate glucose (UDPG) (Keeling et al. 1988). Therefore, SS activity in wheat grains directly reflects the ability of the grains to degrade and utilize sucrose for the synthesis of starch. When SS activity is high, the substrates for starch synthesis are sufficient (Li et al. 2001). As shown in Fig. 5a, the SS activity of the two wheat NILs increased after anthesis and reached peak activity on the 18th day in line 154 and the 25th day in line 212, respectively. Thereafter, the SS activity decreased. Throughout the grain-filling stage, the grain SS activity of line 154 was higher than line 212, which suggests that line 154 had higher sucrose degradation and bioavailability.
After anthesis, sucrose is constantly degraded in the cytoplasm of wheat grain endosperm cells to UDPG for the synthesis of starch and other carbohydrates (Keeling et al. 1988). As shown in Fig. 5b, the sucrose content of wheat grains declined gradually, beginning on the 3rd day after anthesis. The grain sucrose content of line 154 was higher than line 212 during the whole period. This suggests that the level of grain sucrose supply in line 154 was adequate and available for starch synthesis.
Changes in the AGPase activity and starch content of grains in two wheat NILs
ADPG pyrophosphorylase (AGPase) catalyzes the first step in starch synthesis that takes place in amyloplasts and is considered to be the rate-limiting enzyme for starch synthesis (Jiang et al. 2002a, b, c; Nakamura et al. 1989; Preiss and Sivak 1996; Preiss et al. 1988; Tuncel and Okita 2013). As shown in Fig. 6a, the grain AGPase activity changed in a single-peak curve, with maximal activity on the 25th and 18th day after anthesis in line 154 and line 212, respectively. The AGPase activity of grains was higher in line 154 than line 212 throughout the grain-filling stage, which indicates that line 154 was able to produce more ADPG than line 212.
Starch is the main component of wheat grains, accounts for approximately 70 % of the dry weight of grains at the mature stage, and is synthesized and accumulated during the grain-filling process (Bhullar and Jenner 1985; Dale and Housley 1986; Hurkman et al. 2003; Yang et al. 2004). Therefore, the starch accumulation level during this period directly affects the grain yield. As shown in Fig. 6b, the total grain starch content increased with the grain-filling process. The greatest accumulation rate of grain starch occurred from the 7th–14th day after anthesis. Thereafter, the grain starch accumulation rates slowed and starch accumulation reached a maximal level at the maturity stage. The grain starch content in line 154 was higher than in line 212 during the whole period, indicating that line 154 had higher grain starch synthesis ability.
Comparison of some agronomic traits in the two wheat NILs
As shown in Table 1, there were significant differences between the two wheat NILs in effective panicle number, grain weight per spike, 1000-grain weight, and yield. The yield of line 154 at 5488.89 kg/hm2 was 18.64 % higher than line 212. The yield corresponds to the results of grain starch content as it accounts for approximately 70 % of the grain dry weight.
Discussion
In the last half century, remarkable increases in the potential and realized yields of crops have been made worldwide. These increases mainly resulted from the adoption of high-yielding varieties of major crops such as rice, wheat, and maize, accompanied by the use of nitrogen fertilizer and improvements in water irrigation and agricultural techniques (Conway and Toenniessen 1999; Edgerton 2009; Evans 1997, 2013; Huang et al. 2002; Khush 2003; Sakamoto and Matsuoka 2004; Sayre et al. 1997; Shearman et al. 2005; Zhu et al. 2010). However, the capacity to further increase production levels and maximize yield is limited. Increasingly, research on how to break through the yield potential has been the focus both at home and abroad. Many scientists believe in the concepts of the second “Green Revolution,” improvement of photosynthesis and ascension of photosynthetic utilization as ways to increase the yield potential (Long et al. 2006; Zhu et al. 2008). There is an ongoing debate about whether enhancing leaf photosynthesis can raise the yield potential given the many steps between leaf photosynthesis and the final yield (Fischer and Edmeades 2010). In addition, some researchers put forward that the crop yield is sink-limited rather than source-limited (Borrás et al. 2004), leading some to conclude that an improvement in photosynthesis is unlikely to increase crop yields (Sinclair et al. 2004). However, two lines of evidence contradict this. First, free-air CO2 enrichment studies have shown that CO2-induced increases in leaf photosynthesis generally lead to increased crop yield. Second, C4 plants have greater rates of photosynthesis and produce more biomass per unit of intercepted sunlight than C3 plants (Sheehy et al. 2007). In this current work, there were significant differences in Pn between the two wheat NILs, whether it was at the seedling stage or the flowering grain-filling stage (Figs. 1, 2a). Higher photosynthetic rates are beneficial for crops to make more photoassimilates that can be used for grain filling, which ultimately increases yields.
Wheat is a saccharophyllous plant. From the flowering to the mature period, source leaves export sucrose long distances through the veins to non-photosynthetic tissues (net carbohydrate importers, called sinks), such as roots, stems, flowers, fruits, and seeds (Ludewig and Flügge 2013; Turgeon 1989). In sink tissues, sucrose is degraded into materials for starch synthesis. Therefore, the synthesis of sucrose in flag leaves reflects the source supply ability, while the degradation of sucrose in grains reflects the sink strength (Pan et al. 2002). The concept of source strength and sink strength is introduced to measure the ability of the source tissues to produce, and the sink tissues to accept assimilates. Source strength refers to the ability of the source tissues (leaves, sheaths, and stems) to synthesize and output photoassimilates. The photosynthetic rate is a strong intuitive indicator for the source strength. The rate of sucrose synthesis is the limiting factor for the export of photoassimilates from mesophyll cells. Therefore, sucrose synthesis can be used as an indicator of source strength. According to sucrose synthesis metabolic pathways, SPS is an important regulatory enzyme in the process (Champigny 1995; Huber and Huber 1996; Stitt 1989; Wang et al. 2013; Winter and Huber 2000; Worrell et al. 1991). SPS activity reflects the ability of flag leaves to convert photosynthate into sucrose; therefore, it is also an indicator for the source strength (Champigny 1995; Okita 1992). Sink strength refers to the ability of the sink tissues (spike) to accept and convert photoassimilates. The degradation of sucrose in sink tissues is mainly catalyzed by SS, so some researchers regard SS activity as the indicator for sink strength (Baroja-Fernández et al. 2009; Delmer and Haigler 2002; Schaffer and Petreikov 1997; Thévenot et al. 2005; Wang et al. 1993; 1999; Zrenner et al. 1995).
In this study, there were significant differences in the SPS activity, SS activity and sucrose content of flag leaves and SS activity, AGPase activity, sucrose and starch content of grains between the two wheat NILs (Figs. 4, 5, 6). Higher leaf SPS activity during the grain-filling period ensured that line 154 had a sufficient leaf sucrose content to be transported for starch synthesis. Besides this, higher SPS activity was also available for the phosphorylation and transport of sucrose (Li and Wang 2013). Little sucrose content was observed in flag leaves at the beginning of the grain-filling period as the plant was at the sink construction stage, so the sink capacity was low, which also corresponded with the low SS activity in grain at this period (Figs. 4c, 5a). SS can both synthesize and degrade sucrose, and the field is currently in disagreement as to the main role of SS in sucrose metabolism. It is generally believed that, in the photosynthetic apparatus, SS catalyzes sucrose synthesis, while in grains it catalyzes sucrose degradation. In this study, the SS activity of flag leaves exhibited a similar trend to that of SPS during the whole grain-filling period. SS activity in flag leaves was lower compared with the SPS activity in flag leaves and SS activity in grain (Figs. 4a, b, 5a). These results provide evidence that SS catalyzes sucrose degradation in grains (Zamski and Schaffer 1996). With synthesized sucrose being transported from flag leaves through the phloem to grains, the SS activity in grain gradually increased and led to the gradual decline of grain sucrose content (Fig. 5). The products of sucrose degradation by SS in grains were converted to glucose-1-phosphate (G1P) or glucose-6-phosphate (G6P) and were subsequently converted to ADPG, the initial substrate for starch synthesis, by AGPase (Ballicora et al. 2004). Because the activity of AGPase is positively correlated with starch accumulation rate and grain-filling rate, AGPase is a key rate-limiting enzyme for starch synthesis in rice, wheat, corn, potato, and other crops (Jiang et al. 2002a, b, c; Nakamura et al. 1989; Preiss and Sivak 1996; Preiss et al. 1988; Tuncel and Okita 2013). A number of studies have shown that increasing AGPase activity enhances crop starch biosynthesis and yield (Li et al. 2011; Sakulsingharoj et al. 2004; Smidansky et al. 2002, 2003, 2007). In this study, the activity of AGPase increased gradually along with grain filling, which led to the increase of the grain starch content. After 18 days post anthesis, the plants went into senescence and the AGPase activity and plants’ photosynthetic performance began to decrease, which caused the starch accumulation in grains to proceed more steadily. During the whole grain-filling period, the grain starch content and grain starch accumulation rate of line 154 were always higher than line 212 due to its higher Pn and AGPase activity (Figs. 2a, 6a, b). As grain starch accounts for approximately 70 % of grain dry weight, the yield of line 154 was 18.64 % higher than that of line 212 (Table 1). Significant differences were found in the effective panicle number, grain weight per spike, and 1000-grain weight through comparing of some agronomic traits between the two wheat NILs (Table 1), which were also beneficial to the improvement of the grain yield of line 154.
In conclusion, the Pn in flag leaves of line 154 was significantly higher than that of line 212 during the anthesis to grain-filling stage. Higher leaf sucrose phosphate synthase (SPS) activity, grain sucrose synthase (SS) activity, and grain ADPG pyrophosphorylase (AGPase) activity ensured that the photosynthate of line 154 could be transported from leaves to grains and translated into starch in a timely and effective manner, thus relieving any end-product inhibition on leaf photosynthesis (Paul and Pellny 2003; Winder et al. 1998), which also contributed to the maintenance of its high photosynthetic rate. Eventually, all of these factors of line 154 resulted in its higher grain yield compared with the low photosynthetic rate of line 212.
Abbreviations
- AGPase:
-
Adenosine diphosphate glucose pyrophosphorylase
- Ci:
-
Intercellular CO2 concentration
- Gs:
-
Stomatal conductance
- Ls:
-
Stomatal limitation value
- NILs:
-
Near isogenic lines
- Pn:
-
Net photosynthetic rate
- SPS:
-
Sucrose phosphate synthase
- SS:
-
Sucrose synthase
- Vcmax:
-
Maximal carboxylation efficiency
References
Araus JL, Ferrio JP, Buxo R, Voltas J (2007) The historical perspective of dryland agriculture: lessons learned from 10,000 years of wheat cultivation. J Exp Bot 58:131–145
Araus JL, Slafer GA, Royo C, Serret MD (2008) Breeding for yield potential and stress adaptation in cereals. Crit Rev Plant Sci 7:377–412
Awika JM (2011) Major cereal grains production and use around the world. In advances in cereal science: implications to food processing and health promotion., ACS symposium seriesAmerican Chemical Society, Washington DC, pp 1–13
Ballicora MA, Iglesias AA, Preiss J (2004) ADP-glucose pyrophosphorylase: a regulatory enzyme for plant starch synthesis. Photosynth Res 79:1–24
Baroja-Fernández E, Muñoz FJ, Montero M, Etxeberria E, Sesma MT, Ovecka M, Pozueta-Romero J (2009) Enhancing sucrose synthase activity in transgenic potato (Solanum tuberosum L.) tubers results in increased levels of starch, ADPglucose and UDPglucose and total yield. Plant Cell Physiol 50:1651–1662
Bhullar SS, Jenner CF (1985) Differential responses to high temperatures of starch and nitrogen accumulation in the grain of four cultivars of wheat. Funct Plant Biol 12:363–375
Blum A (1990) Variation among wheat cultivars in the response of leaf gas exchange to light. J Agric Sci 115:305–311
Borrás L, Slafer GA, Otegui ME (2004) Seed dry weight response to source-sink manipulations in wheat, maize and soy-bean: a quantitative reappraisal. Field Crops Res 86:131–146
Braun HJ, Atlin G, Payne T (2010) Multi-location testing as a tool to identify plant response to global climate change. Climate change and crop production. CABI Publishers, Wallingford, pp 115–138
Champigny ML (1995) Integration of photosynthetic carbon and nitrogen metabolism in higher plants. Photosynth Res 46:117–127
Cheng JF, Ma WM, Chen GY, Hu MJ, Shen YG, Li ZS, Dong YP, Li B, Li HW (2009) Dynamic changes of photosynthetic characteristics in Xiaoyan 54, Jing 411, and the stable selected superior strains of their hybrid progenies. Acta Agronomica Sinica 35:1051–1058
Conway G, Toenniessen G (1999) Feeding the world in the twenty-first century. Nature 402:C55–C58
Dale EMD, Housley TL (1986) Sucrose synthase activity in developing wheat endosperms differing in maximum weight. Plant Physiol 82:7–10
Delmer DP, Haigler CH (2002) The regulation of metabolic flux to cellulose, a major sink for carbon in plants. Metab Eng 4:22–28
Dixon JM (2009) Wheat facts and futures 2009. CIMMYT, Mexico City
Edgerton MD (2009) Increasing crop productivity to meet global needs for feed, food, and fuel. Plant Physiol 149:7–13
Eli Zamski, Schaffer AA (1996) Photoassimilate distribution in plants and crops: source-sink relationships. Marcel Dekker, New York
Evans LT (1993) Crop evolution, adaptation and yield. Cambridge University Press, New York
Evans LT (1997) Adapting and improving crops: the endless task. Philosophical transactions of the Royal Society of London. Ser B: Biol Sci 352:901–906
Evans JR (2013) Improving photosynthesis. Plant Physiol 162:1780–1793
Farquhar GD, Sharkey TD (1982) Stomatal conductance and photosynthesis. Annu Rev Plant Physiol 33:317–345
Farquhar GD, von Caemmerer S, Berry JA (1980) A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149:78–90
Fischer RA, Edmeades GO (2010) Breeding and cereal yield progress. Crop Sci 50:S85–S98
Fischer RA, Rees D, Sayre KD, Lu ZM, Condon AG, Saavedra AL (1998) Wheat yield progress associated with higher stomatal conductance and photosynthetic rate, and cooler canopies. Crop Sci 38:1467–1475
Hawkesford MJ, Araus JL, Park R, Calderini D, Miralles D, Shen T, Zhang JP, Parry MAJ (2013) Prospects of doubling global wheat yields. Food Energy Secur 2:34–48
He ZH, Xia XC, Chen XM, Zhuang QS (2011) Progress of wheat breeding in china and the future perspective. Acta Agronomica Sinica 37:202–215
Huang J, Pray C, Rozelle S (2002) Enhancing the crops to feed the poor. Nature 418:678–684
Huber SC, Huber JL (1996) Role and regulation of sucrose-phosphate synthase in higher plants. Ann Rev Plant Biol 47:431–444
Hurkman WJ, McCue KF, Altenbach SB, Korn A, Tanaka CK, Kothari KM, Johnson EL, Bechtel DB, Wilson JD, Anderson OD (2003) Effect of temperature on expression of genes encoding enzymes for starch biosynthesis in developing wheat endosperm. Plant Sci 164:873–881
Jiang D, Yu ZW, Li YG, Yu SL (2002a) Dynamic changes of enzyme activities involving in starch synthesis in superior and inferior grains of high-yield winter wheat. Scientia Agricultura Sinica 35:378–383
Jiang D, Yu ZW, Li YG, Yu SL (2002b) Effects of different nitrogen application levels on changes of sucrose content in leaf, culm, grain and photosynthate distribution and grain starch accumulation of winter wheat. Scientia Agricultura Sinica 35:157–162
Jiang H, Wang HW, Su JH, Shi XB, Shen YG, Li ZS, Wei QK, Li B, Li M, Zhang JJ (2002c) Photosynthesis in offspring of hybridization between two wheat cultivars. Acta Agronomica Sinica 28:451–454
Keeling PL, Wood JR, Tyson RH, Bridges IG (1988) Starch biosynthesis in developing wheat grain. Plant Physiol 87:311–319
Khush G (2003) Productivity improvements in rice. Nutr Rev 61:S114–S116
Lalonde S, Wipf D, Frommer WB (2004) Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Ann Rev Plant Biol 55:341–372
Lbraheem O, Hove RM, Bradley G (2008) Sucrose assimilation and the role of sucrose transporters in plant wound response. Afr J Biotechnol 7:4850–4855
Lemoine R (2000) Sucrose transporters in plants: update on function and structure. Biochimica et Biophysica Acta (BBA)-Biomembranes 1465:246–262
Li X, Wang C (2013) Physiological and metabolic enzymes activity changes in transgenic rice plants with increased phosphoenolpyruvate carboxylase activity during the flowering stage. Acta Physiologiae Plantarum 35:1503–1512
Li YG, Yu ZW, Jiang D, Yu SL (2001) Studies on the dynamic changes of the synthesis of sucrose in the flag leaf and starch in the grain and related enzymes of high-yielding wheat. Acta Agronomica Sinica 27:658–664
Li N, Zhang S, Zhao Y, Li B, Zhang J (2011) Over-expression of AGPase genes enhances seed weight and starch content in transgenic maize. Planta 233:241–250
Liu HQ, Jiang GM, Zhang QD, Sun JZ (2002) Changes of gas exchanges in leaves of different cultivars of winter wheat released in different years. Acta Botanica Sinica 44:913–919
Long SP, Zhu XG, Naidu SL, Ort DR (2006) Can improved photosynthesis increase crop yields? Plant, Cell Environ 29:315–330
Lu HQ, Shen FF, Liu LX, Su WF (2005) Recent advances in study on plant sucrose synthase. Chin Agric Sci Bull 21:34–37
Ludewig F, Flügge UI (2013) Role of metabolite transporters in source-sink carbon allocation. Front Plant Sci 4:231
Miralles DJ, Slafer GA (2007) Sink limitations to yield in wheat: how could it be reduced? J Agric Sci, Cambridge 145:139–150
Moll RH, Jackson WA, Mikkelsen RL (1994) Recurrent selection for maize grain yield: dry matter and nitrogen accumulation and partitioning changes. Crop Sci 34:874–881
Murchie EH, Pinto M, Horton P (2009) Agriculture and the new challenges for photosynthesis research. New Phytol 18:532–552
Nakamura Yasunori, Yuki Kazuhiro, Park Shin-Young, Ohya Toshihide (1989) Carbohydrate metabolism in the developing endosperm of rice grains. Plant Cell Physiol 30:833–839
Okita TW (1992) Is there an alternative pathway for starch synthesis? Plant Physiol 100:560–564
Pan QM, Yu ZW, Wang YF, Yu SL (2002) Effects of nitrogen applying stage on both sucrose synthesis in flag leaves and cleavage in grains of wheat. Scientia Agricultura Sinica 35:771–776
Parry MAJ, Reynolds M, Salvucci ME, Raines C, Andralojc PJ, Zhu XG, Furbank RT (2010) Raising yield potential of wheat. II. Increasing photosynthetic capacity and efficiency. J Exp Bot 62:453–467
Paul MJ, Pellny TK (2003) Carbon metabolite feedback regulation of leaf photosynthesis and development. J Exp Bot 54:539–547
Preiss J, Sivak M (1996) Starch synthesis in sinks and sources. In: Photoassimilate Distribution in Plants and Crops, E Zamski (ed), Source-Sink Relationships, Marcel Dekker, New York, NY, USA, pp 139–168
Preiss J, Ball K, Smith-White B (1988) Biosynthesis of starch and its regulation. Biochem Plants 14:181–254
Raines CA (2011) Increasing photosynthetic carbon assimilation in C3 plants to improve crop yield: current and future strategies. Plant Physiol 155:36–42
Reynolds M, Foulkes MJ, Slafer GA, Berry P, Parry MAJ, Snape JW, Angus WJ (2009) Raising yield potential in wheat. J Exp Bot 60:1899–1918
Reynolds M, Bonnett D, Chapman SC, Furbank RT, Manes Y, Mather DE, Parry MAJ (2011) Raising yield potential of wheat I. Overview of a consortium approach and breeding strategies. J Exp Bot 62:439–453
Reynolds M, Foulkes J, Furbank R, Griffiths S, King J, Murchie E, Parry M, Slafer G (2012) Achieving yield gains in wheat. Plant, Cell Environ 35:1799–1823
Rosegrant MW, Agcaoili M (2010) Global food demand, supply, and price prospects to 2010. International Food Policy Research Institute, Washington, DC
Sakamoto T, Matsuoka M (2004) Generating high-yielding varieties by genetic manipulation of plant architecture. Curr Opin Biotechnol 15:144–147
Sakulsingharoj C, Choi SB, Hwang SK, Edwards GE, Bork J, Meyer CR, Okita TW (2004) Engineering starch biosynthesis for increasing rice seed weight: the role of the cytoplasmic ADP-glucose pyrophosphorylase. Plant Sci 167:1323–1333
Sayre KD, Rajaram S, Fischer RA (1997) Yield potential progress in short bread wheats in northwest Mexico. Crop Sci 37:36–42
Schaffer AA, Petreikov M (1997) Sucrose-starch metabolism in tomato fruit undergoing transient starch accumulation. Plant Physiol 113:739–746
Shearman VJ, Sylvester-Bradley R, Scott RK, Foulkes MJ (2005) Physiological processes associated with wheat yield progress in the UK. Crop Sci 45:175–185
Sheehy JE, Ferrer AB, Mitchell PL, Elmido-Mabilangan A, Pablico P, Dionora MJA (2007) How the rice crop works and why it needs a new engine. In: Sheehy JE, Mitchell PL, Hardy B (eds) Charting new pathways to C4 rice. International Rice Research Institute, Los Baños, pp 3–26
Shewry PR (2009) Wheat. J Exp Bot 60:1537–1553
Sinclair TR, Purcell LC, Sneller CH (2004) Crop transformation and the challenge to increase yield potential. Trends Plant Sci 9:70–75
Smidansky ED, Clancy M, Meyer FD, Lanning SP, Blake NK, Talbert LE, Giroux MJ (2002) Enhanced ADP-glucose pyrophosphorylase activity in wheat endosperm increases seed yield. Proc Nat Acad Sci 99:1724–1729
Smidansky ED, Martin JM, Hannah CL, Fischer AM, Giroux MJ (2003) Seed yield and plant biomass increases in rice are conferred by deregulation of endosperm ADP-glucose pyrophosphorylase. Planta 216:656–664
Smidansky ED, Meyer FD, Blakeslee B, Weglarz TE, Greene TW, Giroux MJ (2007) Expression of a modified ADP-glucose pyrophosphorylase large subunit in wheat seeds stimulates photosynthesis and carbon metabolism. Planta 225:965–976
Stitt M (1989) Control analysis of photosynthetic sucrose synthesis: assignment of elasticity coefficients and flux-control coefficients to the cytosolic fructose-1, 6-bisphosphatase and sucrose phosphate synthase. Philos Trans Royal Society of London B: Biol Sci 323:327–338
Thévenot C, Simond-Côte E, Reyss A, Manicacci D, Trouverie J, Le Guilloux M, Prioul JL (2005) QTLs for enzyme activities and soluble carbohydrates involved in starch accumulation during grain filling in maize. J Exp Bot 56:945–958
Tuncel A, Okita TW (2013) Improving starch yield in cereals by over-expression of ADPglucose pyrophosphorylase: expectations and unanticipated outcomes. Plant Sci 211:52–60
Turgeon R (1989) The sink-source transition in leaves. Ann Rev Plant Physiol Plant Mol Biol 40:119–138
Van Camp W (2005) Yield enhancement genes: seeds for growth. Curr Opin Biotechnol 16:147–153
Von Caemmerer S, Farquhar GD (1981) Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 153:376–387
Wang ZL, He MR (1999) Effects of source sink manipulation on production and distribution of photosynthate after flowering in irrigated and rainfed wheat. Acta Agronomica Sinica 25:162–168
Wang F, Sanz A, Brenner ML, Smith AG (1993) Sucrose synthease, starch accumulation, and tomato fruit sink strength. Plant Physiol 101:321–327
Wang AY, Kao MH, Yang WH, Sayion Yiyang, Liu LF, Lee PD, Su JC (1999) Differentially and developmentally regulated expression of three rice sucrose synthase genes. Plant Cell Physiol 40:800–807
Wang L, Cui N, Zhang KY, Fan HY, Li TL (2013) Research Advance of Sucrose Phosphate Synthase (SPS) in Higher Plant. Intern J Agric Biol 15:1221–1226
Winder TL, Sun J, Okita TW, Edwards GE (1998) Evidence for the occurrence of feedback inhibition of photosynthesis in rice. Plant Cell Physiol 39:813–820
Winter H, Huber SC (2000) Regulation of sucrose metabolism in higher plants: localization and regulation of activity of key enzymes. Crit Rev Plant Sci 19:31–67
Worrell AC, Bruneau JM, Summerfelt K, Boersig M, Voelker TA (1991) Expression of a maize sucrose phosphate synthase in tomato alters leaf carbonhydrate partitioning. Plant Cell 3:1121–1130
Xiong FS, Gao YZ, Zhan YC (1994) Relationship between leaf sucrose and starch content and their degradative enzymes activities in crop plants. Acta Agronomica Sinica 20:52–58
Xu DQ (1999) Photosynthetic rate, photosynthetic efficiency and crop yield. Bull Biol 34:8–10
Yang JC, Zhang JH, Wang ZQ, Xu GW, Zhu QS (2004) Activities of key enzymes in sucrose-to-starch conversion in wheat grains subjected to water deficit during grain filling. Plant Physiol 135:1621–1629
Yang XH, Chen XY, Ge QY, Li B, Tong YP, Zhang AM, Li ZS, Kuang TY, Lu CM (2006) Tolerance of photosynthesis to photoinhibition, high temperature and drought stress in flag leaves of wheat: a comparison between a hybridization line and its parents grown under field conditions. Plant Sci 171:389–397
Yang XH, Chen XY, Ge QY, Li B, Tong YP, Zhang AM, Li ZS, Kuang TY, Lu CM (2007) Characterization of photosynthesis of flag leaves in wheat hybrid and its parents grown under field conditions. J Plant Physiol 164:318–326
Ye JY, Qian YQ (1985) Detection of Hill reaction with spectrophotometer. In: Xue YL, Xia ZA (eds) Plant physiology experiment handbook. Shanghai Science and Technology Press, Shanghai, pp 104–107
Zhang QD, Jiang GM, Zhu XG, Wang Q, Lu CM, Bai KZ, Kuang TY (2001) Photosynthetic capability of 12 genotypes of Triticum aestivum. Acta Phytoecologica Sinica 25:532–536
Zhao XX, Ma QQ, Liang C, Fang Y, Wang YQ, Wang W (2007) Effect of glycinebetaine on function of thylakoid membranes in wheat flag leaves under drought stress. Biol Plant 51:584–588
Zheng TC, Zhang XK, Yin GH, Wang LN, Han YL, Chen L, He ZH (2011) Genetic gains in grain yield, net photosynthesis and stomatal conductance achieved in Henan Province of China between 1981 and 2008. Field Crops Res 122:225–233
Zhu XG, Long SP, Ort DR (2008) What is the maximum efficiency with which photosynthesis can convert solar energy into biomass? Curr Opin Biotechnol 19:153–159
Zhu XG, Long SP, Ort DR (2010) Improving photosynthetic efficiency for greater yield. Ann Rev Plant Biol 61:235–261
Zivcak M, Brestic M, Balatova Z, Drevenakova P, Olsovska K, Kalaji HM, Yang XH, Allakhverdiev SI (2013) Photosynthetic electron transport and specific photoprotective responses in wheat leaves under drought stress. Photosynth Res 117:529–546
Zrenner R, Salanoubat M, Willmitzer L, Sonnewald U (1995) Evidence of the crucial role of sucrose synthase for sink strength using transgenic potato plants (Solanum tuberosum L.). Plant J 7:97–107
Acknowledgments
This work was supported by the National Natural Sciences Foundation of China (31271634), the National High Technology Research and Development Program of China (2012AA10A309), and the State Key Basic Research and Development Plan of China (2015CB150105).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, B., Ma, M., Lu, H. et al. Photosynthesis, sucrose metabolism, and starch accumulation in two NILs of winter wheat. Photosynth Res 126, 363–373 (2015). https://doi.org/10.1007/s11120-015-0126-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-015-0126-9