Abstract
Aims
Salt stress presents a significant impediment to crop growth and development. However, the effects of indigenous arbuscular mycorrhizal fungi (AMF) and the addition of exogenous AMF on soybean growth strategies under salt stress remain poorly understood. The purpose of this study was to examine the impact of different AMF sources on soybean growth strategies under salt stress conditions.
Methods
In this study, we established three different salt stress gradients (1, 2, and 4 g NaCl kg−1 soil) along with two AMF treatments (indigenous AMF and added exogenous AMF) to evaluate soybean growth parameters, enzymes, and soil indicators.
Results
Under salt stress, exogenous AMF significantly increased mycorrhizal colonization in soybean, resulting in a notable enhancement in phosphorus (P) and potassium (K) concentration while reducing nitrogen (N) absorption. Additionally, the addition of exogenous AMF demonstrated the capacity to enhance soybean salt tolerance by lowering soybean sodium (Na) and malondialdehyde (MDA) content, catalase (CAT) activity, and increasing K+/Na+ ratio and acid phosphatase (A-Pase) activity. In contrast, in the indigenous AMF treatment, rhizosphere A-Pase activity in soybean exhibited predominantly positive correlations with each trait, and the K+/Na+ ratio relied more on mycorrhizal colonization and CAT activity. Soybean biomass was influenced both directly and indirectly, with the K+/Na+ ratio serving as a crucial pivot in the indirect pathway.
Conclusions
The addition of exogenous AMF can enhance soybean salt tolerance by regulating nutrient and sodium absorption, enzyme activity, and MDA content. Meanwhile, indigenous AMF promotes salt tolerance in soybeans by global regulating trait associations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The salinization of land poses a significant challenge to agricultural development leading to soil structure degradation, reduced soil porosity, and compromised soil aeration and water retention capacity (Paeida & Das 2005). Consequently, these factors affect root respiration and water uptake efficiency. Elevated salt levels in the soil solution or irrigation water induce osmotic and ionic stress in plants, adversely affecting various physiological and biochemical processes (Allah et al. 2015; Hashem et al. 2015). Among the detrimental effects of high salinity is the disruption of critical cell structures, such as membranes, proteins, lipids, and nucleic acids. Salinity-induced membrane peroxidation compromises membrane integrity, leading to the leakage of essential cellular components and hindering plant growth (Ahanger et al. 2014). Under salt stress, the rapid generation of harmful radicals like superoxide, hydrogen peroxide, and peroxides under salt stress triggers chain reactions, resulting in an excessive production of peroxyl radicals. These radicals disrupt normal plant growth processes. Furthermore, the detrimental impacts of salt stress on plant growth and metabolism are linked to changes in the endogenous levels of phytohormones (Khan et al. 2015). Secondary salinization not only reduces crop productivity but also negatively affects soil microbial diversity and disrupts ecosystem equilibrium, thereby diminishing the soil's capacity to self-regenerate.
Arbuscular mycorrhizal fungi (AMF) can establish symbiotic associations with the majority of terrestrial plants, playing a crucial ecological role in enhancing plant growth. Typically found in the rhizosphere of saline soils, AMF extends delicate hyphae that increase root surface area (Anderson & Cairney 2007), aiding host plants in nutrient uptake (Allen 1982), ion balance regulation, enhancing water use efficiency (Auge et al. 2014; Klinsukon et al. 2021), and boosting antioxidant activity (Feng et al. 2002; Chang et al. 2018). This symbiosis involes a proficient reactive oxygen species (ROS) scavenging mechanism, bolstering plants' early defense and growth capabilities under salt stress (Evelin et al. 2009; Chandrasekaran et al. 2014). AMF contribute to enzymatic and non-enzymatic antioxidant defense systems (Wu et al. 2014; Ahmad et al. 2015), induce acquired systemic tolerance (Hashem et al. 2015), and mitigate lipid peroxidation (Allah et al. 2015). Moreover, AMF improve nutrient uptake, particularly phosphorus (Pu et al. 2023), by extending mycelium extends beyond the rhizosphere, connecting roots with surrounding soil microhabitats, thus expanding the root zone for enhanced nutrient absorption. This extensive hyphal network efficiently transport water and nutrients for plant absorption (Anderson & Cairney 2007). Presently, research primarily focuses on the effects of exogenous AMF addition on plants, leaving the mechanisms by which indigenous AMF influence plant responses to salt stress largely unexplored.
Soybean (Glycine max) is a vital crop within the legume family, playing a pivotal role as a global oilseed crop and protein source, thus holding considerable significance in the food and agricultural sectors. Under salt stress, soybean plant growth, nodulation, nitrogen fixation, number of pods, and yield were reduced (Egamberdieva et al. 2016). Inoculating legumes with AMF also improves salt stress tolerance, plant growth, nodulation, and nitrogen fixation in soybean under saline conditions (Hashem et al. 2019). However, the influence of indigenous AMF and adding exogenous AMF on soybean growth responses under salt stress remains uncertain.
In this study, we comprehensively analyzed soybean physiological parameters, enzymes, osmotic regulatory substances, and soil indicators by subjecting the plants to three levels of salt stress and two different AMF treatments involving indigenous AMF and added exogenous AMF. The primary aim of this research is to determine potential differences in the effects of indigenous and added exogenous AMF on soybean growth strategies under varying salt stress levels. Additionally, we aim to explore any potential correlations between the presence AMF and shoot/root traits in soybeans.
Materials and methods
Soil, seeds and strains
Soil samples were collected from the Agronomy Experiment Station (117°09’E, 36°20’N) at Shandong Agricultural University, located in the Daiyue district of Taian City, Shandong Province, China. They were air-dried, sieved through a 2-mm mesh, and thoroughly mixed. Two kilograms of dry soil were then transferred to each plastic flowerpot, measuring 13.5 cm in height and 18.5 cm in diameter. The soil properties were as follows: pH 7.52, electrical conductivity 300 µs cm−1, total nitrogen 1.64 g kg−1, available phosphorus 51.0 mg kg−1, and available potassium 223.0 mg kg−1.
The soybean cultivar utilized in this study was "Wiliams 82" which is known to be salt-sensitive. The arbuscular mycorrhizal fungi (AMF) employed were Rhizophagus irregularis (R. irregularis) and Rhizophagus intraradices (R. intraradices) (‘Jin Bei Lai’ Limited Co., Beijing, China). The AMF was screened in the saline-alkali land used for coastal corn cultivation. Compared with the dominant AMF community in experimental soil, the abundance of this AMF is less than 1%, so it has been selected as an exogenous AMF. The fungus was cultivated in a greenhouse using a 5:1 (w/w) mixture of zeolite and silica sand with maize plants. There are 70 spores per gram.
Experimental design
This study was conducted in the greenhouse facilities of Shandong Agricultural University, located in Taian City, Shandong Province (117°12′E, 36°16′N). A randomized complete block design was used consisting of three salt treatments (1 g kg−1—S1, 2 g kg−1—S2, and 4 g kg−1—S4) applied as NaCl solution and two AMF treatments (indigenous AMF and addition of mixed AMF inocula of R. irregularis and R. intraradices. Each treatment was replicated three times. The soil was mixed well following being sifted through a sieve and filled into plastic pots (2 kg of dry soil per pot). The pot size was 13.5 cm height and 18.5 cm in diameter. The pots were arranged in a randomized complete block design, with plants within each block being re-randomized weekly to minimize the influence of plant location within blocks.
The experiment used NaCl to simulate the saline condition instead of using the natural coastal neutral saline soil to eliminate the effect of other forms of salt ions, such as SO42−, CO32−, HCO3−, which are often presented in the natural soils. The three salt stress concentrations were determined based on the salt tolerance of the experimental soybean variety (S1: initial salt stress concentration; S2: moderate salt stress level; S4: highest salt stress level) (Liu et al. 2020). Soil salt stress was adjusted 7 days before the experiment. A consistent amount of base fertilizer was added to each treatment: N = 0.521 g, P2O5 = 0.172 g, and K2O = 0.285 g per kg of soil. In the added exogenous AMF treatment, 5 g (dosage of each pot) of R. irregularis and R. intraradices microbial inoculants were applied 1 cm below the seeds in the soil before sowing. In order to ensure that only indigenous AMF is present in the soil, only the 5 g of inactivated microbial inoculants were applied in the indigenous AMF treatment.
High-quality soybean seeds were selected, washed with deionized water, and surface-sterilized for 6 h in a 0.3% (v/v) H2O2 solution before rinsing with deionized water. These seeds were germinated in a Petri dish lined with wet absorbent cotton at 25℃ in the dark. When roots reached 3–5 cm, 5 plantlets were transplanted into each pot. After emergence and thinning, 3 healthy plants remained in each pot. To minimize the impact of the growth environment on exogenous AMF, all plants grew in the same environmental conditions. During the plant growth period, soil moisture was kept at 18%–20% (w/w, i.e., 70% of water holding capacity) determined by weighing method. The ambient room temperature was maintained between 22–27 °C, with relative humidity kept at 45%-55%. The experiment commenced on November 26, 2023, and plants were harvested on January 25, 2024, providing a total growth period of 60 days.
Photosynthetic index, root traits and mycorrhizal colonization
The net photosynthetic rate (Pn) and transpiration rate (Tr) of the third leaves from the main stem of soybean were measured using a portable photosynthetic apparatus (Li-6800) from 9:00 to 11:00 a.m. under sunny weather conditions at harvest. Simultaneously, the chlorophyll content (SPAD value) of the leaves was determined using a portable chlorophyll analyzer (SPAD-502).Root samples were cleaned and dispersed in water within a transparent array measuring 30 × 20 × 3 cm. They were then scanned using an EPSON scanner at a resolution of 400 dpi (Epson Expression 1600 pro, Model EU-35, Japan). Root morphological traits such as length and diameter were analyzed from the scanned images using WinRHIZO Pro software (Regent Instruments Inc, Quebec, Canada).
Root samples were treated with 10% (w/v) KOH solution in a 90 °C water bath for 20 min, rinsed with water, acidified with 2% (v/v) HCl for 5 min at room temperature to make it transparent, and then stained with 0.05% (w/v) nonvital Trypan blue in a 90 °C water bath for 30 min. Stained root fragments were placed in a lactic acid-glycerolwater (v/v/v, 1:1:1) solution overnight to remove excess stain. For each sample, 30 stained root fragments of second-order roots with an average length of 1 cm were selected randomly and observed with light microscope after mounted on two slides. Colonization by AMF (%) was assessed using the method described by Trouvelot et al. (1986).
Acid phosphatase activity
Acid phosphatase (A-Pase) is primarily derived from plant roots and microorganisms (Spohn and Kuzyakov 2013). A-Pase had maximal activities under acidic conditions which is often a feature of farmland soils (Tazisong et al. 2015). So we measured the A-Pase activity. Roots were transferred into a 200-mL vials containing a measured amount of 0.2 mmol L−1 CaCl2 solution depending on root volume (Pearse et al. 2007). Roots were repeatedly dunked (about 60 s in this study) into solution until as much rhizosphere soil as possible was removed. Care was taken to minimize root damage. Two 0.5-mL aliquots of soil suspension were transferred into 2-mL centrifuge tubes for measurement of acid phosphatase activity (Neumann 2006; Wen et al. 2017).
Plant traits
For malondialdehyde (MDA), 100 mg of material was extracted in 1 ml 0.1% (w/v) TCA solution. The homogenates were centrifuged at 12,000 × g for 15 min at 4 °C. Five hundred microliters of supernatant was then mixed with 1 ml 0.5% TBA solution (500 mg TBA dissolved in 100 ml of 20% TCA). Then the reaction mixture was placed in a boiling water bath for 1 h and immediately kept on ice for 5 min. The absorption of the samples was measured at 532 nm, after centrifugation at 10,000 × g for 10 min. The data were calculated from the extinction coefficient of MDA (155 mM-1 cm-1) after subtracting the value of non-specific absorption at 600 nm. (Baryla et al. 2000).
For catalase (CAT) analysis, 1 g of shoot material was ground with a small amount of phosphoric acid buffer solution at pH 7.8 to form a homogenate. The homogenate was transferred to a 10-mL graduated test tube, and the mortar was rinsed with the buffer solution, which was then transferred into the same graduated test tube. After centrifugation at 4,000 rpm for 15 min using the same buffer solution, the absorbance of the supernatant was measured at 240-nm (Nakano & Asada 1981).
Both shoots and roots were collected and subsequently dried at 75 °C until reaching a constant weight. The phosphorus (P) content in the plants was determined using the standard vanado-molybdate method (Murphy & Riley 1962), following the digestion of samples in an H2SO4-H2O2 mixture at 360 °C for 2 h. Plant total nitrogen (N) was determined using the semi-micro Kjeldahl method. The potassium (K) and sodium (Na) content in the plants were determined by a flame photometer (Bao 2000).
Calculation of plant traits
Data analysis
We utilized a Two-way ANOVA method with a randomized block design to assess the effects of salt stress and AMF treatment, as well as the interactions between these two main factors. Significant differences among means were determined using the t-test (P < 0.05). For multiple comparisons between treatments, we employed the Tukey test (P < 0.05). Principal component analysis (PCA) was conducted to investigate the relationship between plant traits and soil indices. Sperman's correlation analysis was used to evaluate pairwise relationships among root traits. A multiple regression model was utilized to examine the relationship between soybean traits and mycorrhizal colonization. To determine the tradeoff relationship between the K+/Na+ ratio of soybean in MDA, CAT, and mycorrhizal colonization under different treatments, we conducted a 3D model analysis. All statistical analyses were performed using SPSS 20.0 statistical software (IBM Corp., Armonk, NY, USA), and the figures were generated using Origin 2024 (OriginLab, Northampton, MA, USA).
Furthermore, structural equation models were constructed to analyze the relationships between biomass, plant traits, mycorrhizal colonization, and K+/Na+ under different AMF treatments. The structural equation model plots were created using Amos 26.0 software developed by IBM Corp. in Armonk, NY, USA.
Results
Salt stress and AMF influence on shoot traits and nutrient indices
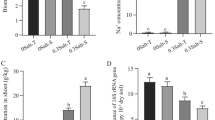
Mycorrhizal colonization was significantly influenced by the interaction between salt stress and AMF treatment (Table 1). Under various salt stress, the addition of exogenous AMF treatment increased mycorrhizal colonization in soybean. However, as soil salt stress increased, the enhancement of mycorrhizal colonization decreased (Fig. 1).
The responses of mycorrhizal colonization to salt stress, and AMF treatment. Each value is the mean (± SE) of three replicates. Lowercase letters indicate significant differences between different microbial treatments under the same salt stress and AMF treatment (based on Tukey’s post hoc analysis, P < 0.05). Abbreviations: S1, 1 g kg−1 NaCl; S2, 2 g kg−1 NaCl; S4, 4 g kg−1 NaCl; Exogenous AMF, the addition of exogenous AMF
Two-way ANOVA revealed that shoot biomass, root biomass, transpiration, and photosynthetic rates of soybean were not significantly affected by the interaction between salt stress and AMF treatment (Table 1). The increase in soil salt stress significantly inhibited soybean growth, but the addition of exogenous AMF alleviated this phenomenon and increased the SPAD value of soybean (Fig. S1c and S1d).
Soybean N concentration and K concentration (both shoot and root) were influenced by salt stress and AMF treatments (Table 1). As depicted in Fig. 2a, with increasing salt stress, the shoot N concentration of soybean significantly decreased, and the addition of exogenous AMF exacerbated this phenomenon. Similarly, as soil salinity increased, the root N concentration of soybeans also significantly decreased. Root N concentration was significantly higher in AMF inoculation treatment under S2 salt stress, while higher in indigenous AMF treatment under S1 salt stress (Fig. 2b). Adding exogenous AMF significantly improved phosphorus (P) concentration in soybean. Under S2 salt stress, both shoot and root P concentrations of soybean increased most significantly with the addition of exogenous AMF (Fig. 2c and 2d). Under S1 salt stress, the addition of exogenous AMF significantly decreased shoot K concentration in soybean, but increased root K concentration. However, with increasing salt stress, the addition of exogenous AMF only significantly increased shoot K concentration in soybean. The most significant increase was observed under S4 salt stress (Fig. 2e and 2f).
The responses of plant N concentration (a-b), plant P concentration (c-d) and plant K concentration (e–f) to soil salt stress and AMF. Each value is the mean (± SE) of three replicates. Letter indicates significant difference between different inoculant treatments (based on Tukey’s post hoc analysis, P < 0.05). Abbreviations: S1, 1 g kg−1 NaCl; S2, 2 g kg−1 NaCl; S4, 4 g kg−1 NaCl; Exogenous AMF, the addition of exogenous AMF
Salt stress and AMF influence on Na content
The salt stress significantly elevated the sodium (Na) content of soybean, while the addition of exogenous AMF treatment notably decreased the Na content of soybean. Under S2 salt stress, there was a significant reduction in the Na transport coefficient of soybean, resulting in a higher retention of Na+ in the root system of soybean (Fig. 3a and 3b). Under S1 and S2 salt stress, the addition of exogenous AMF significantly increased the K+/Na+ ratio of soybean, with this effect being more pronounced at lower salt stress (Fig. 3c).
The responses of plant Na content (a), Na transfer coefficient (b) and K+/Na+ (c) to soil salt stress and AMF. Each value is the mean (± SE) of three replicates. Letter indicates significant difference between different inoculant treatments (based on Tukey’s post hoc analysis, P < 0.05). Abbreviations: S1, 1 g kg−1 NaCl; S2, 2 g kg−1 NaCl; S4, 4 g kg−1 NaCl
Salt stress and AMF influence on A-Pase activity, MDA and CAT
Two-way ANOVA revealed that the activity of A-Pase, as well as the levels of MDA and CAT in soybean, were significantly influenced by the interaction between salt stress and AMF treatment (Table 1). The addition of exogenous AMF treatment significantly increased the A-Pase activity in the rhizosphere under various salt stress, with the greatest increase observed under S2 and S4 salt stress conditions (Fig. 4a). Both MDA and CAT levels indicated that the addition of exogenous AMF treatment had no significant effect on these substances under S1 salt stress. However, under moderate and high salt stress, the addition of exogenous AMF treatment significantly reduced the levels of these substances (Fig. 4b and 4c).
The responses of Acid Pase activity (a), MDA (b) and CAT (c) to soil salt stress and AMF. Each value is the mean (± SE) of three replicates. Letter indicates significant difference between different inoculant treatments (based on Tukey’s post hoc analysis, P < 0.05). Abbreviations: S1, 1 g kg−1 NaCl; S2, 2 g kg−1 NaCl; S4, 4 g kg−1 NaCl; Exogenous AMF, the addition of exogenous AMF
Principal component analysis of plant traits in different salt stress
Principal component analysis (PCA) was employed to perform multivariate ordination of the soil and plant indices for peanuts under different salt stress (Fig. 5). In the three salt stress treatment, principal axis (principal component 1 (PC1) and PC2) contributed more than 75% (Fig. 5a-c). Plant traits exhibited high scores on the first axis under three salt stress treatment. But root P concentration (S1 salt stress), root diameter (S2 salt stress), root length and root K concentration (S4 salt stress) scored high on the second axis.
Principal component analysis (PCA) of soil indices and plant indices in response to the S1 stress (a), S2 stress (b) and S4 stress (c). Abbreviations: Transpiration rate (Tr); photosynthetic rate (Ph); shoot biomass (ShB); root biomass (RB); shoot N concentration (ShN); root N concentration (RN); shoot P concentration (ShP); root P concentration (RP); shoot K concentration (ShK); root K concentration (RN); Na content (NaC); K+/Na.+ (K/Na); Na translocation factor (TFNa); root length (RL); root diameter (RD); rhizosphere acid phosphatase activity (A-Pase activity); Propylene glycol (MDA); Catalase (CAT); mycorrhizal colonization (MC)
Correlation analysis between soil and plant traits in different AMF treatments
The correlation results revealed that, compared to the indigenous AMF treatment, the addition of exogenous AMF treatment increased the correlation between above-ground potassium content and other indices of soybean under salt stress (Fig. 6). Under the indigenous AMF treatment, there was no correlation between the Na transport coefficient of soybean and other indices. However, under the exogenous AMF treatment, the Na transport coefficient was positively correlated with shoot P concentration and negatively correlated with MDA and CAT. Additionally, there was a negative correlation between A-Pase activity and other indices under the indigenous AMF treatment. Conversely, under the exogenous AMF treatment, A-Pase activity was positively correlated with other indices.
Correlation analysis of soil indices and plant indices. On the lower left is for indigenous AMF and on the upper right is for adding exogenous AMF. Red represents the positive correlation, blue represents the negative correlation, and the depth of the color represents the strength of the correlation. The value is the correlation coefficient P, when P < 0.05, the two were significantly correlated. Abbreviations: Transpiration rate (Tr); photosynthetic rate (Ph); shoot biomass (ShB); root biomass (RB); shoot N concentration (ShN); root N concentration (RN); shoot P concentration (ShP); root P concentration (RP); shoot K concentration (ShK); root K concentration (RN); Na content (NaC); K+/Na+ (K/Na); Na translocation factor (TFNa); root length (RL); root diameter (RD); rhizosphere acid phosphatase activity (A-Pase activity); Propylene glycol (MDA); Catalase (CAT); mycorrhizal colonization (MC)
Correlation between plant traits and mycorrhizal colonization responsiveness
Across soybean, functional traits associated with biomass, root morphology, and exudates showed covariation with mycorrhizal colonization (Fig. 7). Mycorrhizal colonization was positively correlated with shoot biomass (P < 0.001, R2 = 0.742; Fig. 7a), root diameter (P < 0.001, R2 = 0.648; Fig. 7b), root length (P < 0.001, R2 = 0.531; Fig. 7c), root diameter (P < 0.001, R2 = 0.799; Fig. 7d), P concentration (P < 0.001, R2 = 0.707; Fig. 7f), K concentration (P = 0.011, R2 = 0.344; Fig. 7g), and K+/Na+ (P < 0.001, R2 = 0.677; Fig. 7i).
Mycorrhizal colonization was negatively correlated with Na content (P < 0.001, R2 = 0.648; Fig. 7h), MDA (P < 0.001, R2 = 0.558; Fig. 7j), and CAT (P < 0.001, R2 = 0.671; Fig. 7k). However, mycorrhizal colonization was almost unrelated to N concentration (P = 0.076, R2 = 0.183; Fig. 7e) and A-Pase activity (P = 0.154, R2 = 0.123; Fig. 7l).
Strategy analysis between mycorrhizal colonization, K+/Na+, MDA and CAT
The strategy analysis revealed that soybeans with added exogenous AMF exhibited a higher dependency on MDA and CAT for K+/Na+ regulation. Conversely, soybeans with indigenous AMF showed a higher reliance on mycorrhizal colonization and CAT (Fig. 8). Furthermore, as salt stress increased, soybeans with added exogenous AMF shifted their dependence from CAT to MDA.
Pathway analysis of plant traits and biomass
The direct effects of root length, root diameter, MDA, CAT, A-Pase activity, and mycorrhizal colonization on K+/Na+ and biomass were analyzed using a structural equation model for soybeans with indigenous AMF or those with added exogenous AMF (Fig. 9a and 9b).
Mycorrhizal colonization, rhizosphere acid phosphatase activity in the rhizosphere (A-Pase), root diameter, root length, CAT, MDA and K+/Na.+ in path analysis for biomass. a, indigenous AMF; b adding exogenous AMF. Red arrows indicate positive relationship, and green arrows indicate negative relationship. Line arrow indicates significant relationships (P < 0.05)
Under saline soil conditions, indigenous AMF enhanced the correlation between soybean traits and K+/Na+ as well as biomass (Fig. 9a). Root diameter, CAT, mycorrhizal colonization, and K+/Na+ exhibited direct and significant effects on biomass. A decrease of 0.327 units in biomass was observed for each unit increase in root diameter, while an increase of 0.292 units in CAT resulted in a decrement of 0.389 units in biomass. Conversely, an increase of 0.389 units in mycorrhizal colonization led to an increase of 0.843 units in biomass. Additionally, for every unit increase in K+/Na+, there was an improvement of 0.843 units in biomass. Root length, A-Pase activity, and MDA indirectly affected soybean biomass through K+/Na+. Each unit increase in MDA led to a decline of 0.484 units in K+/Na+, while each unit increase in A-Pase activity resulted in an increase of 0.389 units in K+/Na+. Similarly, for every unit increase in root length, there was an improvement of 0.285 units in K+/Na+.
However, the addition of exogenous AMF diminished the impact of soybean traits on biomass (Fig. 9b). Only root diameter directly influenced biomass, with root length indirectly affecting biomass accumulation through root diameter. Notably, A-Pase activity and soybean K+/Na+ exhibited negative regulation in the presence of added exogenous AMF treatment.
Discussion
Soybean growth and nutrition traits response to variation in salt and AMF
We observed that the addition of exogenous AMF treatment significantly enhanced soybean mycorrhizal colonization under salt-alkali stress but markedly suppressed soybean mycorrhizal colonization as soil salinity increased (Fig. 1). This suggests that high salt stress is unfavorable for mycorrhizal colonization in soybean roots (Hashem et al. 2019).
The addition of exogenous AMF did not facilitate the absorption of shoot N in soybean (Fig. 2a and 2b). There might be a competition for nitrogen between AMF and host plants, as demonstrated in grassland ecosystems (Helgason & Fitter 2009; Puschel et al. 2016). Therefore, adding exogenous AMF may exacerbate this competition, leading to reduced nitrogen uptake by soybean shoots. The increase in nitrogen content in the subsurface under S2 salt stress further supports this notion (Fig. 2a).
Shoot P concentration serves as a vital indicator for assessing P nutrition in plants and can also indicate if crop yield is hindered by stress (Barry & Miller 1989; Bollons & Barraclough 1999). Under moderate and high salt stress, the shoot P concentration of soybean was inhibited; however, the addition of exogenous AMF significantly increased the shoot P concentration of soybean under S2 salt stress (Fig. 2c and 2d). This suggests that adding exogenous AMF can effectively alleviate the decrease in P concentration caused by salt stress, consistent with previous findings (Allen 1982; Smith et al. 2011). AMF mycelium can provide mineral nutrients for plants, and plants nutrients absorbed through mycelium (Allen 1982; Smith et al. 2011). Moreover, the addition of exogenous AMF treatment notably enhanced rhizosphere A-Pase activity under various salt stresses (Fig. 4a). A-Pase activity plays a crucial role in mobilizing bound P in the soil, thereby facilitating plant uptake of soil P (Hinsinger 2001; Ryan et al. 2001; Lambers et al. 2008; Shen et al. 2011).
The K concentration in soybeans exhibited varied patterns under different salt stresses (Fig. 2e and 2f). Specifically, under low salt stress, root K concentration significantly increased with the addition of exogenous AMF treatment, while soybean shoots demonstrated a notable increase in K concentration with added exogenous AMF treatment under moderate and high salt stress. This phenomenon is intriguing, as previous studies have indicated that AMF can enhance K uptake in plants (Smith et al. 1994). However, there is a gap in the literature regarding the distinct mechanisms through which potassium enrichment occurs under different salt stresses. The onset of salt stress in plants is primarily attributed to the high osmotic potential in saline environments, resulting in the initial accumulation of Na+ and disrupting the balance between Na+ and K+ within plant cells (Evelin et al. 2009; Chandrasekaran et al. 2014). To maintain a harmonized Na+ and K+ equilibrium, plants adjust their K distribution strategy in response to salt stress (Nejad et al. 2021; Santander et al. 2021).
Effect of AMF on salt tolerance of soybean
Our study demonstrated that adding exogenous AMF effectively reduces Na absorption in soybeans under salt stress (Fig. 3a). Moreover, adding exogenous AMF significantly elevated the K+/Na+ ratio in soybean under both S1 and S4 salt stress (Fig. 3c). Alterations in the K+ and Na+ uptake balance in plants, favoring a higher K+/Na+ ratio in roots and shoots, may represent a key mechanism for coping with high salt stress (Auge et al. 2014; Klinsukon et al. 2021; Wang et al. 2021; Huang et al. 2023). Therefore, the increased K+/Na+ ratio in AMF-inoculated plant tissues under salt stress helps alleviate the impacts of high salinity, thus reducing osmotic stress caused by salt stress.
Furthermore, our findings revealed that adding exogenous AMF could significantly decrease the Na transport coefficient and enhance Na accumulation in soybean roots under S2 salt stress (Fig. 3b). This strategic response of soybean to different salt stress conditions involves increasing local salt ion concentrations to reduce ion stress in roots and enhance water uptake to directly confront the saline environment.
Under salt stress, plants tend to generate excessive reactive oxygen species (ROS) to combat the challenges posed by salt stress (Evelin et al. 2009; Chandrasekaran et al. 2014). However, an overabundance of ROS can lead to oxidative damage in plant tissues, resulting in oxidative stress (Azmat & Moin 2019; Wu et al. 2019; Yilmaz et al. 2023). Our results indicated significant reductions in MDA and CAT contents at moderate and high salt concentrations (Fig. 4b and 4c). Interestingly, these findings differ from those of Pan et al. (2020), who reported in a meta-analysis that AMF can mitigate salt stress-induced damage by enhancing CAT activity in mycorrhizal plants and reducing MDA levels. The variation in CAT outcomes could be attributed to the cumulative effects of ROS. In our experiment, the adding exogenous AMF treatment mitigated the oxidative stress induced by salt stress in soybeans, thereby reducing ion stress and diminishing the overall ROS accumulation, leading to a decline in CAT activity. This observation can be elucidated by the significant negative correlation between CAT activity and mycorrhizal infection rate (Fig. 7k).
Correlation of soybean index and effect of AMF on soybean growth strategy under salt stress
The study revealed a positive correlation between the Na transport coefficient and shoot P concentration under adding exogenous AMF treatment (Fig. 6). This finding is intriguing as it suggests that adding exogenous AMF treatment can enhance Na + enrichment in the root system of soybeans by improving P absorption. Salt stress typically results in P loss in plants, yet AMF could enhance mineral P absorption (Kakabouki et al. 2023; Pu et al. 2023). This phenomenon also indicates a P ion absorption strategy that enhances salt tolerance in soybeans (Xue et al. 2023). Adjusting the K + /Na + ratio mitigates ion stress, and the increase in P3 + inhibits the transfer of Na + above ground, thereby safeguarding the shoot and photosynthetic systems of soybeans (Zong et al. 2023; Han et al. 2024). A-Pase activity under indigenous AMF treatment showed a significantly positive correlation with soybean traits, suggesting that soybeans employ varied nutrient absorption strategies under salt stress. Both modification of root morphology and the association with AMF are P scavenging strategies, and they perform in a complementary manner to increase the exploration of soil for P (Yao et al. 2001a, b). By contrast, release of P-mobilizing exudates (e.g. carboxylates) represents a P-mining strategy, enhancing soil P availability through mobilization by ligand exchange or chelating cations from precipitated phosphates (Hinsinger 2001; Ryan et al. 2001; Shen et al. 2011). The results showed that under indigenous AMF treatment, soybean relies more on the "P-mining" strategy.
PCA was utilized for dimension reduction of soybean traits under varying salt stresses. Results indicated that most parameters aligned along axis 1 under all salt stress, suggesting that soybeans tend to gravitate towards axis 1 to alleviate salt stress (Fig. 5). This observation may signify a growth strategy adopted by soybeans under salt stress. The strategy analysis revealed that K+/Na+ of soybean with adding exogenous AMF exhibited a higher dependence on MDA and CAT. However, K+/Na+ of soybean with indigenous AMF exhibited a higher dependence on mycorrhizal colonization and CAT (Fig. 8). The results of the correlation analysis mutually confirm each other and further indicate that different growth strategies exist for soybeans in varying salt stress.
Under indigenous AMF treatment, soybean exhibited both direct and indirect pathways influencing biomass accumulation. Results identified the tissue K+/Na+ ratio as a key hub of indirect influence on soybean biomass accumulation (Fig. 9a). Maintaining a balanced K+/Na+ ratio promotes water and nutrient absorption in plants, thereby facilitating biomass accumulation (Auge et al. 2014; Klinsukon et al. 2021; Huang et al. 2023). Thus, the indirect influence pathway centered on the K+/Na+ ratio may be crucial in regulating salt stress in soybean. However, the influence of soybean parameters on biomass accumulation was diminished under external AMF treatment (Fig. 9b). Unlike microbial activators (Sun & Shahrajabian 2023), the application of adding exogenous AMF might have led to microbial competition and reduced the impact of indigenous AMF (Martignoni et al. 2020; Yu & He 2022). Although the mycorrhizal colonization increased, the effectiveness of other soil microorganisms decreased, suggesting that an excessive presence of microorganisms in field experiments could disrupt indigenous microbial dominance.
Conclusion
Our results revealed notable differences in the impact of indigenous AMF and the addition of exogenous AMF on soybean growth strategies under varying levels of salt stress. Specifically, we found that the addition of exogenous AMF increased mycorrhizal colonization in soybean, resulting in enhanced concentration of P and K, while significantly reducing N and Na absorption. This enhancement in salt tolerance was attributed to an increased K+/Na+ ratio in plant tissues. Conversely, with indigenous AMF, we observed a regulated relationship among soybean traits, particularly with the K+/Na+ ratio being influenced by mycorrhizal colonization and CAT activity. Soybean biomass was both directly and indirectly affected, with the K+/Na+ ratio serving as a pivotal point in the indirect pathway. Notable, the addition of exogenous AMF weakened this relationship. Therefore, under salt stress conditions, the addition of exogenous AMF primarily enhances soybean nutrient uptake and enzyme activity, reduces sodium absorption, and balances K+ and Na+ levels in soybean plants, ultimately improving soybean salt tolerance. In contrast, indigenous AMF enhances soybean salt tolerance by improving the correlation among soybean traits, altering regulatory strategies, and strengthening the influence pathway. Given the complexity of the soil-soybean system, a comprehensive evaluation of the impacts of indigenous AMF and the addition of exogenous AMF on the soil-soybean ecosystem is essential. Future investigations will include the natural soils from various saline-alkali origins with different saline-alkali level.
Data availability
The data that supports the findings of this study are available in the supplementary material of this article.
References
Ahanger M A, Hashem A, Abd Allah E F, Ahmad P. (2014) Arbuscular mycorrhiza in crop improvement under environmental stress. In: Ahmad, P. (Ed.), Emerging Technologies and Management of Crop Stress Tolerance, vol. 2. Elsevier Inc.
Ahmad P, Hashem A, Abd Allah EF, Alqarawi AA, John R, Egamberdieva D, Gucel S (2015) Role of Trichoderma harzianum in mitigating NaCl stress in Indian mustard (Brassica juncea L) through antioxidative defense system. Front Plant Sci 6:868. https://doi.org/10.3389/fpls.2015.00868
Allah EFA, Hashem A, Alqarawi AA, Bahkali AH, Alwhibi MS (2015) Enhancing growth performance and systemic acquired resistance of medicinal plant Sesbania sesban (L.) Merr using arbuscular mycorrhizal fungi under salt stress. Saudi J Biol Sci 22:274–283. https://doi.org/10.1016/j.sjbs.2015.03.004
Allen MF (1982) Influence of vesicular-arbuscular mycorrhizae on water movement through bouteloua gracilis (H.B.K.) lag ex steud. New Phytol 91:683–693
Anderson IC, Cairney JWG (2007) Ectomycorrhizal fungi: exploring the mycelial frontier. Fems Microbiol Rev 31:388–406. https://doi.org/10.1111/j.1574-6976.2007.00073.x
Auge RM, Toler HD, Saxton AM (2014) Arbuscular mycorrhizal symbiosis and osmotic adjustment in response to NaCl stress: a meta-analysis. Front Plant Sci 5:562. https://doi.org/10.3389/fpls.2014.00562
Azmat R, Moin S (2019) The remediation of drought stress under VAM inoculation through proline chemical transformation action. J Photochem Photobiol B-Biol 193:155–161. https://doi.org/10.1016/j.jphotobiol.2019.03.002
Bao S D. (2000) Soil agrochemical analysis third edition. China Agriculture Press: Beijing
Barry D, Miller M (1989) Phosphorus nutritional requirement of maize seedlings for maximum yield. Agron J 81:95–99
Baryla A, Laborde C, Montillet JL, Triantaphylides C, Chagvardieff P (2000) Evaluation of lipid peroxidation as a toxicity bioassay for plants exposed to copper. Environ Pollut 109:131–135. https://doi.org/10.1016/S0269-7491(99)00232-8
Bollons HM, Barraclough PB (1999) Assessing the phosphorus status of winter wheat crops: inorganic orthophosphate in whole shoots. J Agr Sci Cambridge 133:285–295
Chandrasekaran M, Boughattas S, Hu SJ, Oh SH, Sa TM (2014) A meta-analysis of arbuscular mycorrhizal effects on plants grown under salt stress. Mycorrhiza 24:611–625. https://doi.org/10.1007/s00572-014-0582-7
Chang W, Sui X, Fan XX, Jia TT, Song FQ (2018) Arbuscular mycorrhizal symbiosis modulates antioxidant response and ion distribution in salt-stressed Elaeagnus angustifolia seedlings. Front Microbiol 9:652. https://doi.org/10.3389/fmicb.2018.00652
Egamberdieva D, Jabborova D, Berg G (2016) Synergistic interactions between Bradyrhizobium japonicum and the endophyte Stenotrophomonas rhizophila and their effects on growth, and nodulation of soybean under salt stress. Plant Soil 405:35–45. https://doi.org/10.1007/s11104-015-2661-8
Evelin H, Kapoor R, Giri B (2009) Arbuscular mycorrhizal fungi in alleviation of salt stress: a review. Ann Bot 104:1263–1280. https://doi.org/10.1093/aob/mcp251
Feng G, Su YB, Li XL, Wang H, Zhang FS, Tang CX, Rengel Z (2002) Histochemical visualization of phosphatase released by arbuscular mycorrhizal fungi in soil. J Plant Nutr 25:969–980
Han S, Cheng Y, Wu GQ, He XW, Zhao GZ (2024) Enhancing salt tolerance in poplar seedlings through arbuscular mycorrhizal fungi symbiosis. Plants-Basel 13:233. https://doi.org/10.3390/plants13020233
Hashem A, Abd Allah EF, Alqarawi AA, Aldubise A, Egamberdieva D (2015) Arbuscular mycorrhizal fungi enhances salinity tolerance of Panicum turgidum Forssk by altering photosynthetic and antioxidant pathways. J Plant Interact 10:230–242. https://doi.org/10.1080/17429145.2015.1052025
Hashem A, Abd Allah EF, Alqarawi AA, Wirth S, Egamberdieva D (2019) Comparing symbiotic performance and physiological responses of two soybean cultivars to arbuscular mycorrhizal fungi under salt stress. Saudi J Biol Sci 26:38–48. https://doi.org/10.1016/j.sjbs.2016.11.015
Helgason T, Fitter AH (2009) Natural selection and the evolutionary ecology of the arbuscular mycorrhizal fungi (Phylum Glomeromycota). J Exp Bot 60:2465–2480. https://doi.org/10.1093/jxb/erp144
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195. https://doi.org/10.1023/A:1013351617532
Huang SC, Gill S, Ramzan M, Ahmad MZ, Danish S, Huang P, Al Obaid S, Alharbi SA (2023) Uncovering the impact of AM fungi on wheat nutrient uptake, ion homeostasis, oxidative stress, and antioxidant defense under salinity stress. Sci Rep 13:8249. https://doi.org/10.1038/s41598-023-35148-x
Kakabouki I, Stavropoulos P, Roussis I, Mavroeidis A, Bilalis D (2023) Contribution of arbuscular mycorrhizal fungi (AMF) in improving the growth and yield performances of flax (Linum usitatissimum L.) to salinity stress. Agronomy-Basel 13:2416. https://doi.org/10.3390/agronomy13092416
Khan AL, Waqas M, Lee IJ (2015) Resilience of Penicillium resedanum LK6 and exogenous gibberellin in improving Capsicum annuum growth under abiotic stresses. J Plant Res 128:259–268. https://doi.org/10.1007/s10265-014-0688-1
Klinsukon C, Lumyong S, Kuyper TW, Boonlue S (2021) Colonization by arbuscular mycorrhizal fungi improves salinity tolerance of eucalyptus (Eucalyptus camaldulensis) seedlings. Sci Rep 11:4362. https://doi.org/10.1038/s41598-021-84002-5
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103. https://doi.org/10.1016/j.tree.2007.10.008
Liu X X, Chang R Z, Guan R X, Qu L J. (2020) Establishment of screening method for salt tolerant soybean at emergence stage and screening of tolerant germplasm. ACTA Agron Sin., 46, 1–8 (China). http://kns.cnki.net/kcms/detail/11.1809.S.20190903.1632.008.html
Martignoni MM, Garnier J, Hart MM, Tyson RC (2020) Investigating the impact of the mycorrhizal inoculum on the resident fungal community and on plant growth. Ecol Model 438:109321. https://doi.org/10.1016/j.ecolmodel.2020.109321
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Agr Ecosyst Environ 26:678–681
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Nejad RAH, Kafi M, Jari SK, Mozafari H, Motesharezadeh B (2021) Arbuscular mycorrhizal fungi improve growth, physiological status and nutrients accumulation of Ailanthus altissima seedlings under cadmium pollution and salinity. Russ J Plant Physiol 68:266–273. https://doi.org/10.1134/S102144372102014X
Neumann G (2006) Quantitative determination of acid phosphatase activity in the rhizosphere and on the root surface. In: Luster J, Finlay R (eds) Handbook of methods used in rhizosphere research–online edition. http//www.rhizo.at/handbook
Paeida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Pan J, Peng F, Tedeschi A, Xue X, Wang T, Liao J, Zhang WJ, Huang CH (2020) Do halophytes and glycophytes difer in their interactions with arbuscular mycorrhizal fungi under salt stress? A Meta-Analysis. Bot Stud 61:13. https://doi.org/10.1186/s40529-020-00290-6
Pearse SJ, Veneklaas EJ, Cawthray G, Bolland MD, Lambers H (2007) Carboxylate composition of root exudates does not relate consistently to a crop species’ ability to use phosphorus from aluminium, iron or calcium phosphate sources. New Phytol 173:181–190. https://doi.org/10.1111/j.1469-8137.2006.01897.x
Pu ZT, Zhang RF, Wang H, Li QY, Zhang JQ, Wang XX (2023) Root morphological and physiological traits and arbuscular mycorrhizal fungi shape phosphorus-acquisition strategies of 12 vegetable species. Front Plant Sci 14:1150832. https://doi.org/10.3389/fpls.2023.1150832
Puschel D, Janouskova M, Hujslova M, Slavikova R, Gryndlerova H, Jansa J (2016) Plant-fungus competition for nitrogen erases mycorrhizal growth benefits of Andropogon gerardii under limited nitrogen supply. Ecol Evol 6:4332–4346. https://doi.org/10.1002/ece3.2207
Ryan PR, Delhaize E, Jones DL (2001) Function and mechanism of organic anion exudation from plant roots. Annu Rev Plant Biol 52:527–560
Santander C, Aroca R, Cartes P, Vidal G, Cornejo P (2021) Aquaporins and cation transporters are differentially regulated by two arbuscular mycorrhizal fungi strains in lettuce cultivars growing under salinity conditions. Plant Physiol Biochem 158:396–409. https://doi.org/10.1016/j.plaphy.2020.11.025
Shen JB, Yuan LX, Zhang JL, Li HG, Bai ZH, Chen XP, Zhang WF, Zhang FS (2011) Phosphorus dynamics: from soil to plant. Plant Physiol 156:997–1005. https://doi.org/10.1104/pp.111.175232
Smith SE, Gianinazzi-Pearson V, Koide R, Cairney JWG (1994) Nutrient transport in mycorrhizas: structure, physiology and consequences for efficiency of the symbiosis. Plant Soil 159:103–113
Smith SE, Jakobsen I, Gronlund M, Smith FA (2011) Roles of arbuscular mycorrhizas in plant phosphorus nutrition: interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiol 156:1050–1057. https://doi.org/10.1104/pp.111.174581
Spohn M, Kuzyakov Y (2013) Distribution of microbial- and root-derived phosphatase activities in the rhizosphere depending on P availability and C allocation–Coupling soil zymography with 14C imaging. Soil Biol Biochem 67:106–113. https://doi.org/10.1016/j.soilbio.2013.08.015
Sun WL, Shahrajabian MH (2023) The application of arbuscular mycorrhizal fungi as microbial biostimulant, sustainable approaches in modern agriculture. Plants-Basel 12:3101. https://doi.org/10.3390/plants12173101
Tazisong IA, Senwo ZN, He Z (2015) Phosphatase hydrolysis of organic phosphorus compounds. Adv Enzyme Res 3:39–51. https://doi.org/10.4236/aer.2015.32005
Trouvelot A, Kough, Gianiazzi-Pearson V. (1986) Mesure du taux de mycorrhization VA dun systeme radiculaire. Recherche de methode destimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V and Gianinazzi S (Eds.). Physiological Genetical Aspects of Mycorrhizae. INRA Press, Paris, France.
Wang H, An T, Huang D, Liu R, Xu B, Zhang S, Deng X, Siddique KHM, Chen Y (2021) Arbuscular mycorrhizal symbioses alleviating salt stress in maize is associated with a decline in root-to-leaf gradient of Na+/K+ ratio. BMC Plant Biol 21:457. https://doi.org/10.1186/s12870-021-03237-6
Wen ZH, Li HG, Shen JB, Rengel Z (2017) Maize responds to low shoot P concentration by altering root morphology rather than increasing root exudation. Plant Soil 416:377–389. https://doi.org/10.1007/s11104-017-3214-0
Wu QS, He JD, Srivastava AK, Zou YN, Kuca K (2019) Mycorrhizas enhance drought tolerance of citrus by altering root fatty acid compositions and their saturation levels. Tree Physiol 39:1149–1158. https://doi.org/10.1093/treephys/tpz039
Wu Q, Zou Y N, Abd Allah E F. (2014) Mycorrhizal association and ROS in plants. In: Ahmad, P. (Ed.), Oxidative Damage to Plants. Elsevier Inc.
Xue L, Liu P, Wu AP, Dong LJ, Wu QQ, Zhao MS, Liu H, Li Y, Zhang NL, Wang YH (2023) Resistance of mycorrhizal Cinnamomum camphora seedlings to salt spray depends on K+ and P uptake. J Fungi 9:964. https://doi.org/10.3390/jof9100964
Yao Q, Li X, Christie P (2001a) Factors affecting arbuscular mycorrhizal dependency of wheat genotypes with different phosphorus efficiencies. J Plant Nutr 24:1409–1419
Yao Q, Li X, Feng G, Christie P (2001b) Influence of extramatrical hyphae on mycorrhizal dependency of wheat genotypes. Commun Soil Sci Plan 32:3307–3317
Yilmaz A, Yildirim E, Yilmaz H, Soydemir HE, Guler E, Ciftci V, Yaman M (2023) Use of arbuscular mycorrhizal fungi for boosting antioxidant enzyme metabolism and mitigating saline stress in sweet basil (Ocimum basilicum L.). Sustainability 15:5982. https://doi.org/10.3390/su15075982
Yu HW, He WM (2022) Arbuscular mycorrhizal fungi compete asymmetrically for amino acids with native and invasive Solidago. Microb Ecol 84:131–140. https://doi.org/10.1007/s00248-021-01841-5
Zong JW, Zhang ZL, Huang PL, Yang YH (2023) Arbuscular mycorrhizal fungi alleviates salt stress in Xanthoceras sorbifolium through improved osmotic tolerance, antioxidant activity, and photosynthesis. Front Microbiol 14:1138771. https://doi.org/10.3389/fmicb.2023.1138771
Acknowledgements
This study was funded by the Key R&D program of Shandong Province (2021CXGC010804) and National Natural Science Foundation of China (32270065). YC was supported by Australian Research Council (FT210100902).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Responsible Editor: Ricardo Aroca.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pu, Z., Hu, R., Wang, D. et al. The impact of arbuscular mycorrhizal fungi on soybean growth strategies in response to salt stress. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06901-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06901-6