Abstract
Purpose
Cavernous sinus invasion by pituitary adenomas is an important prognostic factor for evaluating the possibilities of complete remission and to guide patient management. A widely used Magnetic Resonance Imaging grading system, suggested by Knosp in 1993, has recently been revised by the same group. The aims of our study were to apply this revised grading system to our surgical series, to determine its association with surgical outcomes, gross-total resection (GTR) and endocrinological remission (ER), paying particular attention to grades 3A and 3B, which represent the novelty of this revised classification.
Methods
We included consecutive patients who underwent endoscopic endonasal surgery for a macroadenoma from September 2012 to December 2016. MRI images were reviewed and classified according to the revised Knosp classification. Surgical reports indicated the intra-operative CS invasion. GTR and ER were evaluated on 3-months post-operative MRI and endocrine evaluation.
Results
254 patients were included in this study. We found a total rate of cavernous sinus invasion of 18.4%. Different outcomes were observed for each grade, with an increased rate of cavernous sinus invasion with each grade. Per-operative rates of invasion were 61.5 and 78.6% in grades 3A and 3B respectively. GTR was negatively correlated with the grade, while rates were 55.8% and 30.0% for grades 3A and 3B respectively.
Conclusion
The revised Knosp radiological classification contributes to the prediction of surgical outcomes and early ER in pituitary adenomas. To manage, as precisely as possible, the risk of early recurrence in pituitary adenomas, clinicians should also consider other recognized prognostic factors, such as the proliferative status of the tumor.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pituitary adenomas are classically considered to be benign. However, a significant number of these tumors have an aggressive behavior. The prognosis of complete remission, tumor progression or invasive status of a pituitary adenomas can be evaluated using clinical, radiological, surgical and histopathological features [1,2,3,4,5,6] One of these predictive factors is the invasion of surrounding structures, particularly the sellar floor and parasellar extension, which is associated with a higher risk of subtotal tumor removal and therefore a higher risk of relapse [2, 7,8,9,10,11]. Even though invasiveness alone is not directly related to tumor aggressiveness, its evaluation by MRI, along with tumor dimensions and growth, forms part of the definition of aggressive pituitary tumors [12].
Cavernous sinus invasion is known to be particularly frequent, and is strongly associated with an increased risk of incomplete resection [13, 14]. Even though surgical inspection is the most effective technique for evaluation of this invasion [15, 16], a preoperative approach assessing the tumor’s invasive character has prognostic value and helps in designing an appropriate management of the patient before and after surgery [4, 11].
In order to preoperatively quantify the cavernous sinus invasion, in 1993 Knosp et al. suggested a now globally-used reference grading system of pre-operative magnetic resonance imaging (MRI), based on the comparison between detection of cavernous sinus invasion on MRI scans and surgical microscopic trans-sphenoidal observation [15, 17].
This classification has recently been re-evaluated and updated by the same group [18], taking into account the improvements in intra-operative observation due to improved endoscopic technologies, thus providing better rates of tumor removal and lower rates of complications [19]. This revised classification was evaluated using surgical findings, along with the gross-total resection (GTR) and endocrine remission (ER) in functioning pituitary adenomas. The novel aspect of this grading system is to differentiate superior or inferior cavernous sinus compartment invasion in grades 3A and 3B. Indeed, Micko et al. [18] observed different outcomes in these two grades, with a per-operative invasion rate of 26.5% for grade 3A and 70.6% for grade 3B, associated with a GTR of 85% and 64% and endocrine remission rates of 67% and 0% respectively.
The aim of our study was to validate the modified Knosp classification on our pituitary adenoma series, by comparing neuro-radiological pre-operative findings and cavernous sinus invasion observed in intra-operative endoscopic endonasal assessment. In order to determine the role of this pre-surgical MRI grading system in predicting GTR and early ER, we aimed to compare these findings with post-operative radiological, biochemical and clinical assessments.
Materials and methods
Study population
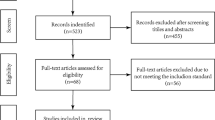
We retrospectively reviewed a series composed of all consecutive adult pituitary adenomas recorded in a local database (PITUICARE-Lyon, registered with the French data protection agency CNIL, 16-0.21, and clinicaltrials.org, NCT02854228), who were surgically treated for the first time (i.e. no recurrence) by a single expert neurosurgeon from September 2012 to December 2016, in the pituitary referral center of Lyon (Hospices Civils de Lyon). Only pathologically confirmed pituitary adenomas were included in this study. We excluded microadenomas, non-endoscopic endonasal surgeries, and surgeries performed for recurrent tumors. Cases lacking a proper pre-operative MRI, or without sufficient data were also excluded. One death occurred the day after surgery, and was also excluded. The inclusion and exclusion flow chart is shown in Fig. 1.
All patients were pre-operatively evaluated by a single expert neurosurgeon, and underwent complete pre-operative endocrine testing [gonadotropins (FSH-LH), prolactin (PRL), adrenocorticotropic hormone (ACTH), cortisol, thyroid stimulating hormone (TSH), Free-T4 (FT4) and growth hormone (GH)], in order to evaluate their functional status. At least one pre-operative MRI-scan was performed on each case, less than 6 months before surgery, to assess the tumor size and Knosp grade. All MRI-scans included T1-weighted coronal slices with and without contrast enhancement, and T2-weighted slices were available for most cases, as well as pre-operative navigation planning.
All patients underwent endonasal endoscopic surgery, with a complete or partial resection. Pathological examination and proliferation grading was performed on each resected pituitary adenomas, according to Trouillas et al. [3]. Each patient underwent MRI-scan with and without contrast enhancement, to assess the GTR at 3 months post-surgery. Endocrine remission (ER) was evaluated in those patients presenting with functioning pituitary adenomas 3 months post-operatively. ER was defined by normal IGF1 and suppressed GH (< 0.4 µg/L) after oral glucose tolerance test for somatotroph adenomas, normal 24 h- free urinary cortisol and suppressed 8am cortisol level after 1 mg Dexamethasone test for corticotroph adenomas; normal prolactin level for lactotroph adenomas and normal TSH and T4 for thyrotroph adenomas.
Data was collected by reviewing patients’ medical records.
Radiological evaluation and MRI-grading
A single radiologist, using the pre-operative MRI-scans, performed the neuro-radiological grading of each cavernous sinus. The grading was performed according to the modified Knosp grading system suggested by Micko et al. [18]. The radiologist was blinded for intra-operative observation results and for other data concerning the analyzed cases. According to the modified Knosp classification, the neuro-radiological evaluation was based on the tumor extension, either passing or not passing beyond medial and lateral tangents to the intra-cavernous and supra-cavernous internal carotid arteries (grade 0 to 4). The novel aspect of this modified classification is the difference between the tumor extension into the inferior and/or the superior cavernous sinus compartment (Grade 3A, 3B) [17]. Grade 4 corresponded to the total encasement of the intra-cavernous carotid artery. The tumor size in three dimensions was also collected.
Surgical technique and findings
Most cases followed the same operative scheme, using a uninostril trans-sphenoid endoscopic endonasal approach to the sella turcica, which allows maximal efficiency and minimal invasiveness in surgical and endocrine outcomes [20]. Invasiveness of each cavernous sinus and intra-operative observations were systematically reported on the operating record. The cavernous sinus was considered as showing no invasion if the medial wall was intact after tumor removal. The surgeon presumed the cavernous sinus to be invaded when the medial wall was found completely or partially opened, sometimes with direct vision of the internal carotid during surgery at the final exploration with angle endoscopes. Each questionable situation was classified as invasive. Sphenoid sinus invasion status was also reported. The extent of surgical tumor resection was considered as subtotal or complete, according to the surgeon’s judgment at the end of the surgery, and was systematically reported in surgical records. These results were also collected, and recorded as complete intra operative removal (CIOR).
Tumor characteristics and functional status
All cases consisted of macroadenomas (i.e. diameter ≥ 10 mm). Tumor diameter was reported with the radiological evaluation. We allocated the 254 pituitary adenomas into two subtypes: functioning and non-functioning pituitary adenomas (NFPAs). Functioning pituitary adenomas were ACTH, GH, TSH and PRL adenomas with secreting activity shown on the pre-operative evaluation. Where there was preoperative medical treatment in cases of functioning pituitary adenoma, this was reported. Pituitary adenomas were considered as non-functioning if they were either gonadotropinomas (FSH-LH), null-cell, or silent pituitary adenomas. These latter represented the ACTH, TSH, GH and PRL hormonal subtypes, that had no pre-operative endocrine secreting activity shown by pre-operative endocrine testing.
Pathology
Pituitary tumor samples were systematically collected intra-operatively and sent for pathological analysis. After confirmation of the nature of the tumor, the pathological subtype was determined by immuno-staining. Markers of the cell cycle (Ki67, mitotic figures) and P53 were evaluated to determine the proliferative status of the tumor, as the proliferative status is recognized to be a prognostic and predictive factor of remission and progression. According to a multicentric retrospective case–control study in 2013 [3], proliferation was histologically defined by the presence of at least 2 of the 3 following criteria: Ki67 > 3%, mitoses > 2/10 High Power Field (HPF), and P53 positive (> 10 strongly positive nuclei/10 HPF). Proliferative status was not available for four cases because of the samples being either too small to be analysed or the sample being necrotic.
Follow-up, gross total resection and endocrine remission
Each patient of this series was systematically followed 3 months post-operatively, with a 3 months post-operative MRI-scan with and without contrast enhancement. The MRI scans were evaluated additionally by the neurosurgeon who performed the surgery, to determine the presence of tumor residue. In case of doubt, the next follow-up MRI-scan was evaluated to determine the GTR. Two scans amongst all post-operative MRI-scans were not available.
Each patient treated for a functioning pituitary adenoma underwent endocrine testing in order to detect any persistent secretion to determine endocrine remission (ER). This result was unavailable for two cases.
Data analysis
The quantitative characteristics of the patients and tumors were described using the median, the minimum and the maximum values. Qualitative characteristics were described using the absolute and relative frequencies in each category. The sensitivity and specificity of the Knosp grading system (positive for grade ≥ 3A) and the modified Knosp grading system (positive for grade ≥ 3B) for the diagnosis of cavernous sinus invasion were estimated with their 95% confidence interval (CI) built using the method of Wilson. We compared the two grading systems by using the McNemar test, which allows comparison of paired proportions. The comparisons of proportions between independent groups were carried out using the Chi square test or the Fisher exact test. The homogeneity of the Ki67 marker staining according to the maximum grade of each tumor was examined using the test of the median. Statistical analyses were carried out using SPSS software (version 21.0) and R software (version 3.2.1).
Results
Our series included 254 patients. The male–female ratio was 1:0.85. The median age was 56.0 years (range 17–85). Clinical, radiological and histological characteristics are summarized in Table 1. The majority of cases were non functioning pituitary adenomas (NFPA) (n = 72), and most of these were gonadotroph tumors. Among the functioning pituitary adenomas (FPA) (n = 82), the majority were somatotroph (64.6%) followed by lactotroph (17.1%), corticotroph (12.2%) and thyrotroph (6.1%).
Twenty-eight cases received pre-operative treatment in order to reduce the tumor size before surgery and/or control the secretion and clinical symptoms. Three NFPA cases received a dopamine agonist pre-operatively for elevated serum prolactin, which were in fact due to pituitary stalk compression. Among treated cases, 10 were lactotroph, and 13 were somatotroph. The majority of medically pre-treated cases were surgically treated due to failure or incomplete response to treatment (20 cases) or treatment intolerance (1 case). Two cases were treated pre-operatively in order to reduce the tumor volume. All cases were macroadenomas. The average tumor diameter was 23.7 mm ± 8.2 mm.
Of the 254 cases, and using the revised Knosp classification by Micko et al, “Invasion of the cavernous sinus space in pituitary adenomas.”, we found 46 (18.1%) grade 0, 67 (26.4%) grade 1, 67 (26.4%) grade 2, 52 (20.5%) grade 3A, 12 (4.7%) grade 3B and 10 (3.9%) grade 4.
Among the 508 cavernous sinuses, 207 (41.4%) cavernous sinuses were grade 0, 132 (26.4%) were grade 1, 85 (17.0%) were grade 2, 52 (10.3%) were grade 3A, 14 (2.8%) were grade 3B, and 11 cavernous sinuses (2.2%) were classified as grade 4. Lastly, 7 could not be interpreted on the pre-operative MRI-scan (Table 1).
Invasion of the sphenoid sinus was present in 33 cases (12.9%), of which 4 (12.1%) were classified grade 0, 7 (21.2%) grade 1, 5 (15.1%) grade 2, 10 (30.3%) grade 3A, 1 (3.0%) grade 3B, and 6 (18.2) grade 4.
Surgically observed invasion
In total, intra-operative parasellar invasion was observed in 79 cases (31.1%). As thirteen patients had both cavernous sinuses invaded, we found in total 92 invaded cavernous sinuses (17.8%) (Table 2). Four grade 0 cavernous sinuses (1.9%) were found to be invasive. Parasellar extension Grades 1 and 2 were found to be invasive in respectively 11 (8.3%) and 20 (23.5%) cavernous sinuses. Among pituitary tumors with parasellar extension into the superior cavernous sinus compartment (Grade 3A), 32 (61.5%) were found to be invasive. As for the parasellar extension into the inferior compartment (Grade 3B), 11 (78.6%) were found to be invasive. All 11 parasellar extensions grade 4 were invasive (100%) (Table 2). The trend of these results was statistically significant.
Table 2 also shows results for sensitivities, specificities, positive predictive value (PPV) and negative predictive value (NPV) of tests considering cavernous sinus invaded beyond grade 3 (1993 Knosp classification [17]) or beyond grade 3A (2015 revised classification [18]). The differences between the two sensitivities, specificities, PPV and NPV were statistically significant (p < 0.001).
Parasellar extension and GTR results
Complete intra-operative tumor removal (CIOR) was achieved in 211 cases of our series (84.2%). As reported in Fig. 2, the proportion of complete resection decreased as the maximum grade for each pituitary tumor increased (p < 0.001). Almost all grade 0 (98%) were completely removed while no grade 4 could be peri-operatively considered as completely resected. Grades 1 and 2 showed similar results. We observed different surgical outcomes between grades 3A and 3B, which were completely removed in 37 (71.2%) and 4 (33.0%) of cases respectively.
Complete intraoperative removal (CIOR), gross total resection (GTR) and endocrine remission (ER) for each grade according to the revised Knosp classification grading system for the whole cohort (p < 0.05). (CIOR: Complete intra operative removal; GTR: Gross total resection; ER: Endocrine remission, for FPA)
The rate of GTR was negatively correlated with the tumor grade (p < 0.001). Grades 0, 1 and 2 were associated with similar high GTR rates. Lower GTR rates were found for grades 3A (n = 29; 56.0%), 3B (n = 3; 25.0%) and 4 (n = 1; 10%). GTR data were missing for two cases. The extent of resection and GTR results are shown in Fig. 2. Due to the limited number of cases, no statistically significant difference could be observed between post-operative results for grades 3A, 3B.
Parasellar extension and Endocrine remission
Results are described in Fig. 2. In total, 44 (55%) functioning adenomas were considered in endocrine remission at the three months post-operative evaluation. Thirteen functioning pituitary tumors (4 ACTH; 5 GH, 4 PRL) presented persistent hormonal hypersecretion without visible residual tumor. Moreover, one functional somatotroph tumor presented post-operative residual tumor without showing biochemical signs of hypersecretion (Normal IGF1 and GH suppression on oral glucose tolerance test).
Proliferation and Ki67
As shown in Table 3, the proportions of patients whose tumors showed histological proliferation were not statistically different according to the maximum grade of the tumor (p = 0.87). From grade 0 to grade 4, the proliferation rate varied from 14.3 to 24.3%. The results were approximately the same with the Ki67 proliferation marker, which was found to be greater than or equal to 3% for 14.2% to 30% of each grade. The Ki67 median values were not statistically different according to the maximum grade of each tumor (p = 0.75).
Discussion
We examined a large series of 254 cases of pituitary adenomas, and demonstrate the association between the revised Knosp grading system and the endoscopic endonasal surgical inspection. In our study, population and tumor characteristics were similar to those in the revised Knosp grading system [18]. As expected, the proportion showing parasellar invasion increased significantly as the maximum grade increased. The originality of the revised Knosp classification resides in the subdivision of grade 3 into grades 3A, passing into the superior compartment of the cavernous sinus, and 3B passing into the inferior compartment. Despite a similar number of 3A grades (49 vs. 52) the rate of per-operative invasion in Grade 3A was higher (61.5%) in our series compare to 26.5% found by Micko et al. One could argue that our intra-operative observation was more pessimistic thus overestimating true cavernous sinus invasion. Indeed, post-operative evaluation demonstrated a GTR in 71.0% of grade 3A however, no ER were noted in the 8 functioning adenomas compared to 85% and 67% respectively for grade 3A in the series of Micko et al. For Grade 3B adenomas, the rate of surgically observed invasion and ER showed a similar tendency (78.6 vs. 70.6%) and (20% vs. 0%) in both series but not GTR (33% in our series vs. 64%). This discrepancy indicated that intra-operative parasellar invasion for grade 3A remains a very subjective judgement, and therefore can be differently interpreted depending on the surgeon. We had the advantage in our series, to have a single neurosurgeon interpreting the invasiveness of the pituitary tumor in each cavernous sinus, thus limiting the interpretation bias [21, 22]. Moreover, the absence of ER in functioning pituitary adenomas underlines the limitation of this classification to predict invasion and the likelihood of cure after surgery. Ten functioning pituitary adenomas continued to show persistent hormone secretion, despite the absence of residual tumor on the 3 months post-operative MR-imaging. We chose to analyze the outcome at an early time-point in our series, but longer follow-up may have shown residual tumor images, temporally distant from surgery and imaging artefacts.
As a consequence of sellar floor and dural invasion, sphenoid sinus invasion can also be considered as a prognostic factor [8, 23]. Among all pituitary tumors, only two cases (one gonadotroph and one silent thyrotroph tumor) presenting intra-operative sphenoid sinus invasion only (i.e. no right or left CS invasion) presented post-operative residual tumor. Absence of ER was observed in 6 functioning pituitary tumors with both cavernous and sphenoid sinus invasion, and we were therefore unable to determine the role of each or both extensions in the absence of remission.
French guidelines for managing NFPAs recommend surgery based on several criteria, such as the natural evolution of NFPAs, proximity to the optic chiasma, and the risk of pituitary insufficiency [24, 25]. According to Chen et al., 12–46% of patients with operated NFPAs experience tumor regrowth [26]. Recent studies and meta-analysis report better early and long term outcomes in asymptomatic and small NFPAs [11, 26,27,28,29,30]. This revised Knosp grading system could additionally contribute to the prediction of surgical outcomes and an early surgical management of NFPAs. However, persistence of post-operative residual tumor is not always associated with progression during follow-up, indicating that additional risk factors must be considered [7].
GTR outcomes were better for null-cell and gonadotroph tumors, than for silent pituitary adenomas. The number of silent pituitary adenomas in our series was too few to associate the cavernous sinus invasion status and the MRI grade of each type of silent pituitary tumor. In the literature, some studies have reported an earlier and more aggressive recurrence for silent adenomas, especially silent corticotroph adenoma [31,32,33,34], though this remains controversial [35]. Yamada et al. and Langlois et al. [36, 37] reported, in large series of NFPAs, cavernous sinus invasion in 85% cases of silent corticotroph adenomas. Our results are different, with only 7 silent corticotroph adenomas (one grade 2, 3A and 3B and two grade 0 and 1), each considered to be in GTR at the 3 months evaluation. Additionally, 6 of the 7 silent somatotroph tumors of our series were found to be in GTR. Several studies have reported relapse rates in silent pituitary tumors similar to that of gonadotroph tumors. However, Langlois et al. [38, 39] have suggested that monohormonal tumors, expressing exclusively GH, are more aggressive compared to plurihormonal tumors (GH with staining for PRL and/or TSH).
As only a minority of our cases received medical treatment to reduce tumor volume before surgery, we could not predict the effect of pre-treatment on surgical outcome. Although Castinetti et al. showed somatostatin agonist pre-treatment in acromegaly reduced tumor volume in about 25% of cases and might reduce the rates of immediate post-surgical complications, no difference in surgical remission rates could be shown [40]. Surgery in macro-prolactinomas is recommended in the case of resistance to medical treatment, which was the case in most of our macro-prolactinoma cases [41, 42].
In our study no association was found between radiological grading and pathological proliferation grade or Ki67 expression. These findings underline the independence of these two tumor properties and the importance of combining several factors in predicting pituitary tumor behavior and aggressive status [3, 43, 44].
Conclusion
Our study validates the revised Knosp grading system as an important predictor of pituitary tumors in terms of cavernous sinus invasion, and its contribution to the prediction of GTR and ER. It underlines the importance of a careful follow-up in all pituitary tumor subtypes and a first-line surgical management in early stages of cavernous sinus extension, in order to increase the chances of complete remission. To be as effective as possible in predicting pituitary tumors outcomes and aggressive status, we suggest this radiological classification system to be taken into account along with other recognized predictive factors, such as pathological findings. Other risk factors must however be considered to aid in prediction of recurrence and progression.
References
Raverot G, Dantony E, Beauvy J et al (2017) Risk of recurrence in pituitary neuroendocrine tumors: a prospective study using a five-tiered classification. J Clin Endocrinol Metab 102:3368–3374. https://doi.org/10.1210/jc.2017-00773
Brochier S, Galland F, Kujas M et al (2010) Factors predicting relapse of nonfunctioning pituitary macroadenomas after neurosurgery: a study of 142 patients. Eur J Endocrinol 163:193–200. https://doi.org/10.1530/EJE-10-0255
Trouillas J, Roy P, Sturm N et al (2013) A new prognostic clinicopathological classification of pituitary adenomas: a multicentric case-control study of 410 patients with 8 years post-operative follow-up. Acta Neuropathol 126:123–135. https://doi.org/10.1007/s00401-013-1084-y
Bourdelot A, Coste J, Hazebroucq V et al (2004) Clinical, hormonal and magnetic resonance imaging (MRI) predictors of transsphenoidal surgery outcome in acromegaly. Eur J Endocrinol 150:763–771
Zaidi HA, Awad A-W, Bohl MA et al (2016) Comparison of outcomes between a less experienced surgeon using a fully endoscopic technique and a very experienced surgeon using a microscopic transsphenoidal technique for pituitary adenoma. J Neurosurg 124:596–604. https://doi.org/10.3171/2015.4.JNS15102
Aflorei ED, Korbonits M (2014) Epidemiology and etiopathogenesis of pituitary adenomas. J Neurooncol 117:379–394. https://doi.org/10.1007/s11060-013-1354-5
Raverot G, Vasiljevic A, Jouanneau E (2018) Prognostic factors of regrowth in nonfunctioning pituitary tumors. Pituitary 21:176–182. https://doi.org/10.1007/s11102-017-0861-3
Meij BP, Lopes M-BS, Ellegala DB et al (2002) The long-term significance of microscopic dural invasion in 354 patients with pituitary adenomas treated with transsphenoidal surgery. J Neurosurg 96:195–208. https://doi.org/10.3171/jns.2002.96.2.0195
Ahmadi J, North CM, Segall HD et al (1986) Cavernous sinus invasion by pituitary adenomas. AJR Am J Roentgenol 146:257–262. https://doi.org/10.2214/ajr.146.2.257
Cortet-Rudelli C, Bonneville J-F, Borson-Chazot F et al (2015) Post-surgical management of non-functioning pituitary adenoma. Ann Endocrinol 76:228–238. https://doi.org/10.1016/j.ando.2015.04.003
Roelfsema F, Biermasz NR, Pereira AM (2012) Clinical factors involved in the recurrence of pituitary adenomas after surgical remission: a structured review and meta-analysis. Pituitary 15:71–83. https://doi.org/10.1007/s11102-011-0347-7
Raverot G, Burman P, McCormack A et al (2018) European society of endocrinology clinical practice guidelines for the management of aggressive pituitary tumours and carcinomas. Eur J Endocrinol 178:G1–G24. https://doi.org/10.1530/EJE-17-0796
Juraschka K, Khan OH, Godoy BL et al (2014) Endoscopic endonasal transsphenoidal approach to large and giant pituitary adenomas: institutional experience and predictors of extent of resection. J Neurosurg 121:75–83. https://doi.org/10.3171/2014.3.JNS131679
Fahlbusch R, Buchfelder M (1988) Transsphenoidal surgery of parasellar pituitary adenomas. Acta Neurochir 92:93–99
Enseñat J, Ortega A, Topcewski T et al (2006) Predictive value of the Knosp classification in grading the surgical resection of invasive pituitary macroadenomas. A prospective study of 23 cases. Neurocir Astur Spain 17:519–526
Zoli M, Milanese L, Bonfatti R et al (2016) Cavernous sinus invasion by pituitary adenomas: role of endoscopic endonasal surgery. J Neurosurg Sci 60:485–494
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33:610–617 (Discussion 617–618)
Micko ASG, Wöhrer A, Wolfsberger S, Knosp E (2015) Invasion of the cavernous sinus space in pituitary adenomas: endoscopic verification and its correlation with an MRI-based classification. J Neurosurg 122:803–811. https://doi.org/10.3171/2014.12.JNS141083
Dehdashti AR, Ganna A, Karabatsou K, Gentili F (2008) Pure endoscopic endonasal approach for pituitary adenomas: early surgical results in 200 patients and comparison with previous microsurgical series. Neurosurgery 62:1006–1015. https://doi.org/10.1227/01.neu.0000325862.83961.12 (Discussion 1015–1017)
Berhouma M, Messerer M, Jouanneau E (2012) Occam’s razor in minimally invasive pituitary surgery: tailoring the endoscopic endonasal uninostril trans-sphenoidal approach to sella turcica. Acta Neurochir 154:2257–2265. https://doi.org/10.1007/s00701-012-1510-2
Lee S-H, Park J-S, Lee S et al (2016) Parasellar extension grades and surgical extent in endoscopic endonasal transsphenoidal surgery for pituitary adenomas: a single surgeon’s consecutive series with the aspects of reliability and clinical validity. J Korean Neurosurg Soc 59:577–583. https://doi.org/10.3340/jkns.2016.59.6.577
Shou X, Shen M, Zhang Q et al (2016) Endoscopic endonasal pituitary adenomas surgery: the surgical experience of 178 consecutive patients and learning curve of two neurosurgeons. BMC Neurol 16:247. https://doi.org/10.1186/s12883-016-0767-0
Selman WR, Laws ER, Scheithauer BW, Carpenter SM (1986) The occurrence of dural invasion in pituitary adenomas. J Neurosurg 64:402–407. https://doi.org/10.3171/jns.1986.64.3.0402
Castinetti F, Dufour H, Gaillard S, et al (2012) Consensus de la Société Française d’Endocrinologie sur les adénomes hypohysaires non fonctionnels
Galland F, Vantyghem M-C, Cazabat L et al (2015) Management of nonfunctioning pituitary incidentaloma. Ann Endocrinol 76:191–200. https://doi.org/10.1016/j.ando.2015.04.004
Chen Y, Wang CD, Su ZP et al (2012) Natural history of postoperative nonfunctioning pituitary adenomas: a systematic review and meta-analysis. Neuroendocrinology 96:333–342. https://doi.org/10.1159/000339823
Losa M, Donofrio CA, Barzaghi R, Mortini P (2013) Presentation and surgical results of incidentally discovered nonfunctioning pituitary adenomas: evidence for a better outcome independently of other patients’ characteristics. Eur J Endocrinol 169:735–742. https://doi.org/10.1530/EJE-13-0515
Murad MH, Fernández-Balsells MM, Barwise A et al (2010) Outcomes of surgical treatment for nonfunctioning pituitary adenomas: a systematic review and meta-analysis. Clin Endocrinol 73:777–791. https://doi.org/10.1111/j.1365-2265.2010.03875.x
Pereira AM, Biermasz NR (2012) Treatment of nonfunctioning pituitary adenomas: what were the contributions of the last 10 years? A critical view. Ann Endocrinol 73:111–116. https://doi.org/10.1016/j.ando.2012.04.002
Messerer M, Dubourg J, Raverot G et al (2013) Non-functioning pituitary macro-incidentalomas benefit from early surgery before becoming symptomatic. Clin Neurol Neurosurg 115:2514–2520. https://doi.org/10.1016/j.clineuro.2013.10.007
Bradley KJ, Wass JAH, Turner HE (2003) Non-functioning pituitary adenomas with positive immunoreactivity for ACTH behave more aggressively than ACTH immunonegative tumours but do not recur more frequently. Clin Endocrinol 58:59–64
Scheithauer BW, Jaap AJ, Horvath E et al (2000) Clinically silent corticotroph tumors of the pituitary gland. Neurosurgery 47:723–729 (Discussion 729–730)
Webb KM, Laurent JJ, Okonkwo DO et al (2003) Clinical characteristics of silent corticotrophic adenomas and creation of an internet-accessible database to facilitate their multi-institutional study. Neurosurgery 53:1076–1084 (Discussion 1084–1085)
Cooper O, Ben-Shlomo A, Bonert V et al (2010) Silent corticogonadotroph adenomas: clinical and cellular characteristics and long-term outcomes. Horm Cancer 1:80–92. https://doi.org/10.1007/s12672-010-0014-x
Fountas A, Lavrentaki A, Subramanian A et al (2018) Recurrence in silent corticotroph adenomas after primary treatment: a systematic review and meta-analysis. J Clin Endocrinol Metab 104:1039–1048. https://doi.org/10.1210/jc.2018-01956
Yamada S, Ohyama K, Taguchi M et al (2007) A study of the correlation between morphological findings and biological activities in clinically nonfunctioning pituitary adenomas. Neurosurgery 61:580–584. https://doi.org/10.1227/01.NEU.0000290906.53685.79
Langlois F, Lim DST, Yedinak CG et al (2018) Predictors of silent corticotroph adenoma recurrence; a large retrospective single center study and systematic literature review. Pituitary 21:32–40. https://doi.org/10.1007/s11102-017-0844-4
Langlois F, Woltjer R, Cetas JS, Fleseriu M (2018) Silent somatotroph pituitary adenomas: an update. Pituitary 21:194–202. https://doi.org/10.1007/s11102-017-0858-y
Langlois F, Lim DST, Varlamov E et al (2017) Clinical profile of silent growth hormone pituitary adenomas; higher recurrence rate compared to silent gonadotroph pituitary tumors, a large single center experience. Endocrine 58:528–534. https://doi.org/10.1007/s12020-017-1447-6
Castinetti F, Morange I, Dubois N et al (2009) Does first-line surgery still have its place in the treatment of acromegaly? Ann Endocrinol 70:107–112. https://doi.org/10.1016/j.ando.2009.03.002
Brue T, Delemer B, French Society of Endocrinology (SFE) work group on the consensus on hyperprolactinemia (2007) Diagnosis and management of hyperprolactinemia: expert consensus—French Society of Endocrinology. Ann Endocrinol 68:58–64. https://doi.org/10.1016/j.ando.2006.11.001
Casanueva FF, Molitch ME, Schlechte JA et al (2006) Guidelines of the pituitary society for the diagnosis and management of prolactinomas. Clin Endocrinol 65:265–273. https://doi.org/10.1111/j.1365-2265.2006.02562.x
Lelotte J, Mourin A, Fomekong E et al (2018) Both invasiveness and proliferation criteria predict recurrence of non-functioning pituitary macroadenomas after surgery: a retrospective analysis of a monocentric cohort of 120 patients. Eur J Endocrinol 178:237–246. https://doi.org/10.1530/EJE-17-0965
Asioli S, Righi A, Iommi M et al (2019) Validation of a clinicopathological score for the prediction of post-surgical evolution of pituitary adenoma: retrospective analysis on 566 patients from a tertiary care centre. Eur J Endocrinol 180:127–134. https://doi.org/10.1530/EJE-18-0749
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Buchy, M., Lapras, V., Rabilloud, M. et al. Predicting early post-operative remission in pituitary adenomas: evaluation of the modified knosp classification. Pituitary 22, 467–475 (2019). https://doi.org/10.1007/s11102-019-00976-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-019-00976-6