Abstract
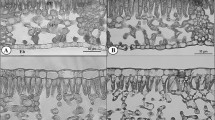
Morphological, anatomical, and physiological leaf traits of Corylus avellana plants growing in different light conditions within the natural reserve “Siro Negri” (Italy) were analyzed. The results highlighted the capability of C. avellana to grow both in sun and shade conditions throughout several adaptations at leaf level. In particular, the more than 100% higher specific leaf area in shade is associated to a 44% lower palisade to spongy parenchyma thickness ratio compared with that in sun. Moreover, the chlorophyll (Chl) a to Chl b ratio decreased in response to the 97% decrease in photosynthetic photon flux density. The results highlighted the decrease in the ratio of Chl to carotenoid content, the maximum PSII photochemical efficiency, and the actual PSII photochemical efficiency (ΦPSII) associated with the increase in the ratio of photorespiration to net photosynthesis (P N) in sun. Chl a/b ratio was the most significant variable explaining P N variations in shade. In sun, P N was most influenced by the ratio between the fraction of electron transport rate (ETR) used for CO2 assimilation and ETR used for photorespiration, by ΦPSII, nitrogen content per leaf area, and by total Chl content per leaf area. The high phenotypic plasticity of C. avellana (PI = 0.33) shows its responsiveness to light variations. In particular, a greater plasticity of morphological (PIm = 0.41) than of physiological (PIp = 0.36) and anatomical traits (PIa = 0.24) attests to the shade tolerance of the species.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- C:
-
soil organic carbon content
- C/N:
-
ratio between carbon and nitrogen content
- Chl:
-
chlorophyll
- Car:
-
carotenoid content
- C i :
-
substomatal CO2 concentration
- DM:
-
dry mass
- E :
-
transpiration rate
- ETR:
-
electron transport rate
- ETRA :
-
fraction of ETR used for CO2 assimilation
- ETRP :
-
fraction of ETR used for photorespiration
- F0 :
-
minimal fluorescence yield of dark-adapted state
- F0′:
-
minimal fluorescence yield of the light-adapted state
- Fm :
-
maximal fluorescence yield of the dark-adapted state
- Fm′:
-
maximal fluorescence yield of the light-adapted state
- Fs :
-
steady-state fluorescence yield
- Fv/Fm :
-
maximal quantum yield of PSII photochemistry
- g m :
-
mesophyll conductance
- g s :
-
stomatal conductance
- LA:
-
leaf area
- L:
-
total leaf thickness
- N:
-
total soil nitrogen content
- Na :
-
leaf nitrogen content
- PI:
-
mean plasticity index
- PIa :
-
anatomical plasticity index
- PIm :
-
morphological plasticity index
- PIp :
-
physiological plasticity index
- P N :
-
net photosynthetic rate
- PNUE:
-
photosynthetic nitrogen use efficiency
- P r :
-
photorespiration rates
- R D :
-
respiration rate
- RH:
-
relative air humidity
- SLA:
-
specific leaf area
- SOM:
-
soil organic matter content
- SWC:
-
soil water content
- T a :
-
air temperature
- T l :
-
leaf temperature
- T m :
-
mean air temperature
- T max :
-
mean maximum air temperature
- T min :
-
mean minimum air temperature
- ΦPSII :
-
effective quantum yield of PSII photochemistry
References
Ackerly D.D., Dudley S.A., Sultan S.E. et al.: The evolution of plant ecophysiological traits: recent advances and future directions. — BioScience 50: 979–995, 2000.
Adiloglu A., Adiloglu S.: An investigation on nutritional status of hazelnut (Corylus avellana L.) grown in acid soils of Turkey. — Arch. Agron. Soil Sci. 50: 617–622, 2004.
Amthor J.S.: The McCree-de Wit-Penning de Vries Thornley respiration paradigms: 30 years later. — Ann. Bot-London 86: 1–20, 2000.
Asada K.: The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. — Annu. Rev. Plant Phys. 50: 601–639, 1999.
Atkin O.K., Evans J.R., Ball M.C., Siebke K.: Relationships between the inhibition of leaf respiration by light and enhancement of leaf dark respiration following light treatment. — Aust. J. Plant Physiol. 25: 437–443, 1998.
Bassil N.V., Botta R., Mehlenbacher S.A.: Microsatellite markers in hazelnut: isolation, characterization, and crossspecies amplification. — J. Am. Soc. Hortic. Sci. 130: 543–549, 2005.
Bailey S., Horton P., Walters R.G.: Acclimation of Arabidopsis thaliana to the light environment: the relationship between photosynthetic function and chloroplast composition. — Planta 218: 793–802, 2004.
Bernacchi C.J., Portis A.R., Nakano H. et al.: Temperature response of mesophyll conductance. Implications for the determination of rubisco enzyme kinetics and for limitations to photosynthesis in vivo. — Plant Physiol. 130: 1992–1998, 2002.
Björkman O., Demmig B.: Photon yield of O2 evolution and chlorophyll fluorescence at 77k among vascular plants free-air carbon dioxide enrichment affect photochemical of diverse origins. — Planta 170: 489–504, 1987.
Björkman O., Demmig-Adams B.: Regulation of photosynthetic light energy capture, conversion, and dissipation in leaves of higher plants. — In: Schulze E.D., Caldwell M.M. (ed.): Ecophysiology of Photosynthesis. Pp 17–47. Springer-Verlag, Berlin 1994.
Boccacci P., Botta R., Rovira M.: Genetic diversity of hazelnut (Corylus avellana L.) germplasm in northeastern Spain. — HortScience 43: 667–672, 2008.
Cai Z.Q., Slot M., Fan Z.X.: Leaf development and photosynthetic properties of three tropical tree species with delayed greening. — Photosynthetica 43: 91–98, 2005.
Castagneri D., Garbarino M., Nola P.: Host preference and growth patterns of ivy (Hedera helix L.) in a temperate alluvial forest. — Plant Ecol. 214: 1–9, 2013.
Catoni R., Gratani L., Varone L.: Physiological, morphological and anatomical trait variations between winter and summer leaves of Cistus species. — Flora 207: 442–449, 2012.
Chabot B.F., Chabot J.F.: Effects of light and temperature on leaf anatomy and photosynthesis in Fragaria vesca. — Oecologia 26: 363–377, 1977.
de Lucia E.H., Whitehead D., Clearwater M.J.: The relative limitation of photosynthesis by mesophyll conductance in cooccurring species in a temperate rainforest dominated by the conifer Dacrydium cupressinum. — Funct. Plant Biol. 30: 1197–1204, 2003.
Demmig-Adams B., Adams W.W. III.: Xanthophyll cycle and light stress in nature: Uniform response to excess direct sunlight among higher plants species. — Planta 198: 460–470, 1996.
Demmig-Adams B., Adams W.W. III, Barker D.H. et al.: Using chlorophyll fluorescence to assess the fraction of absorbed light allocated to thermal dissipation of excess excitation. — Physiol. Plantarum 98: 253–264, 1996.
Dorn L.A., Pyle E.H., Schmitt J.: Plasticity to light cues and resources in Arabidopsis thaliana: testing for adaptive value and costs. — Evolution 54: 1982–1994, 2000.
Duan B., Lü Y., Yin C., Li C.: Morphological and physiological plasticity of woody plant in response to high light and low light. — Chin. J. Appl. Environ. Biol. 11: 238–245, 2005.
Epron D., Godard D., Cornic G., Genty B.: Limitations of net CO2 assimilation rate by internal reistances to CO, transfer in leaves of two tree species (Fagus sylvatica L. and Castanea sativa Mill.). — Plant Cell Environ. 18: 43–51, 1995.
Evans J.R., Poorter H.: Photosynthetic acclimation of plants to growth irradiance: the relative importance of specific leaf area and nitrogen partitioning in maximizing carbon gain. — Plant Cell Environ. 24: 755–767, 2001.
FAO Production Yearbook: http://faostat.fao.org/site/339/default.aspx. 2011.
FAOSTAT: Food and Agricultural Organization of the United Nations. Production, Crops. http://faostat.fao.org/site/567/default.aspx#ancor. 2012.
FAOSTAT: Agriculture Data. http://faostat3.fao.org/home/index.html. 2013.
Genty B., Briantais J.M., Baker N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. — Biochim. Biophys. Acta 990: 87–92, 1989.
Givnish T.J.: Adaptation to sun and shade: A whole plant perspective. — Aust. J. Plant Physiol. 15: 63–92, 1988.
Grassi G., Magnani F.: Stomatal, mesophyll conductance and biochemical limitations to photosynthesis as affected by drought and leaf ontogeny in ash and oak trees. — Plant Cell Environ. 28: 834–849, 2005.
Gratani L., Foti I.: Estimating forest structure and shade tolerance of the species in a mixed deciduous broad-leaved forest in Abruzzo, Italy. — Ann. Bot. Fenn. 35: 75–83, 1998.
Gratani L., Meneghini M., Pesoli P., Crescente M.F.: Structural and functional plasticity of Quercus ilex seedlings of different provenances in Italy. — Trees 17: 515–521, 2003.
Hampson C.R., Azarenko A.N., Potter J.R.: Photosynthetic rate, flowering, and yield component alteration in hazelnut in response to different light environments. — J. Amer. Soc. Hort. Sci. 121: 1103–1111, 1996.
Harley P.C., Loreto F., di Marco G., Sharkey T.D.: Theoretical consideration when estimating the mesophyll conductance to CO2 flux by analysis of the response of photosynthesis to CO2. — Plant Physiol. 98: 1429–1436, 1992.
Herr-Turoff A., Zedler J.B.: Does morphological plasticity of the Phalaris arundinacea canopy increase invasiveness? — Plant Ecol. 193: 265–277, 2007.
Holm G.: Chlorophyll mutations in barley. — Acta Agr. Scand. 4: 457–471, 1954.
Huang D., Wu L., Chen J.R., Dong L.: Morphological plasticity, photosynthesis and chlorophyll fluorescence of Athyrium pachyphlebium at different shade levels. — Photosynthetica 49: 611–618, 2011.
Iglesias-Prieto R., Trench R.K.: Acclimation and adaptation to irradiance in symbiotic dino flagellates. II. Response of chlorophyll-protein complexes to different photon-flux densities. — Mar. Biol. 130: 23–33, 1997.
Johnson G.N., Young A.J., Scholes J.D., Horton P.: The dissipation of excess excitation energy in British plant species. — Plant Cell Environ. 16: 673–679, 1993.
Kasapligil B.: A bibliography on Corylus (Betulaceae) with annotations. — Annu. Rep. North. Nut. Growers Assoc. 63: 107–162, 1972.
Koike T., Kitao M., Maruyama Y. et al.: Leaf morphology and photosynthetic adjustments among deciduous broad-leaved trees within the vertical canopy profile. — Tree Physiol. 21: 951–958, 2001.
Krall J.P., Edwards G.E.: Relationship between photosystem II activity and CO2 fixation in leaves. — Physiol. Plantarum 86: 180–187, 1992.
Kull O., Niinemets Ü.: Variations in leaf morphometry and nitrogen concentration in Betula pendula Roth., Corylus avellana L. and Lonicera xylosteum L. — Tree Physiol. 12: 311–318, 1993.
Lam E., Oritz W., Mayfield S., Malkin R.: Isolation and characterization of a light-harvesting chlorophyll a/b protein complex associated with photosystem I. — Plant Physiol. 74: 650–655, 1984.
Larcher W.: Physiological Plant Ecology. 4th ed. Pp. 514. Springer, Berlin, 2003.
Loveys B.R., Scheurwater I., Pons T.L. et al.: Growth temperature influences the underlying components of relative growth rate: an investigation using inherently fast- and slow-growing plant species. — Plant Cell Environ. 25: 975–988, 2002.
Maclachlan S., Zalik S.: Plastid structure, chlorophyll concentration, and free aminoacid composition of a chlorophyll mutant of barley. — Can. J. Bot. 41: 1053–1062, 1963.
Matos F.S., Wolfgramm R., Gonçalves F.V. et al.: Phenotypic plasticity in response to light in the coffee tree. — Environ. Exp. Bot. 67: 421–427, 2009.
Matos F.S., de Oliveria L.R., de Freitas R.G. et al.: Physiological characterization of leaf senescence of Jatropha curcas L. populations. — Biomass Bioenerg. 45: 57–64, 2012.
Mendes M.M., Gazarini L.C., Rodrigues M.L.: Acclimation of Myrtus communis to contrasting Mediterranean light environments-effects on structure and chemical composition of foliage and plant water relations. — Environ. Exp. Bot. 45: 165–178, 2001.
Moraes G.A.B.K., Chaves A.R.M., Martins S.C.V. et al.: Why is it better to produce coffee seedlings in full sunlight than in the shade? A morphophysiological approach. — Photosynthetica 48: 199–207, 2010.
Motta R., Nola P., Berretti R.: The rise and fall of the black locust (Robinia pseudoacacia L.) in the “Siro Negri” Forest Reserve (Lombardy, Italy): lessons learned and future uncertainties. — Ann. For. Sci. 66: 410–419, 2009.
Navas M.L., Garnier E.: Plasticity of whole plant and leaf traits in Rubia peregrina in response to light, nutrient and water availability. — Acta Oecol. 23: 375–383, 2002.
Niinemets Ü.: Role of foliar nitrogen in light harvesting and shade tolerance of four temperate deciduous woody species. — Funct. Ecol. 11: 518–531, 1997.
Niinemets Ü., Kull O., Tenhunen J.D.: An analysis of light effects on foliar morphology, physiology, and light interception in temperate deciduous woody species of contrasting shade tolerance. — Tree Physiol. 18: 681–696, 1998.
Niinemets Ü., Valladares F.: Photosynthetic acclimation to simultaneous and interacting environmental stresses along natural light gradients: optimality and constraints. — Plant Biol. 6: 254–268, 2004.
Ort D.R., Baker N.R.: A photoprotective role for O2 as an alternative electronic sink in photosynthesis? — Curr. Opin. Plant Biol. 5: 193–98, 2002.
Pearcy R.W., Sims D.A.: Photosynthetic acclimation to changing light environments: scaling from the leaf to the whole plant. — In: Roy J., Caldwell M.M., Pearcy R.W. (ed.): Exploitation of Environmental Heterogeneity by Plants. Ecophysiological Processes Above- and Belowground. Physiological Ecology. Pp. 145–174. Academic Press, San Diego 1994.
Percerisa J., Rafecas M., Castellote A.I. et al.: Influence of variety and geographical origin on the lipid fraction of halzenuts (Corylus avellana L.) from Spain: III. Oil stability, tocopherol content and some mineral contents. — Food Chem. 53: 71–74, 1995.
Pershern A.S., Breene W.M., Lulai E.C.: Analysis of factors influencing lipid oxidation in hazelnuts (Corylus sp.). — J. Food Process. Pres. 19: 9–26, 1995.
Persson H., Widén B., Andersson S., Svensson L.: Allozyme diversity and genetic structure of marginal and central populations of Corylus avellana L. (Betulaceae) in Europe. — Plant Syst. Evol. 244: 157–179, 2004.
Pignatti S.: [Flora of Italy.] Vol. 1, Pp. 112. Bologna Edagricole, 1982. [In Italian]
Pintado A., Valladares F., Sancho L.G.: Exploring phenotypic plasticity in the lichen Ramalina capitata: morphology, water relations and chlorophyll content in north- and south-facing populations. — Ann. Bot.-London 80: 345–353, 1997.
Poorter L., Kwant R., Hernández R. et al.: Leaf optical properties in Venezuelan cloud forest trees. — Tree Physiol. 20: 519–526, 2000.
Reich P.B., Kloeppel B.D., Ellsworth D.S., Walters M.B.: Different photosynthesis-nitrogen relations in deciduous hardwood and evergreen coniferous tree species. — Oecologia 104: 24–30, 1995.
Sack L., Melcher P.J., Liu W.H. et al.: How strong is intracanopy leaf plasticity in temperate deciduous trees? — Am. J. Bot. 93: 829–839, 2006.
Sartori F.: [Alluvial forests of the lower Ticino valley (Northern Italy)]. — Colloq. Phytosiol. 9: 201–216, 1984. [In French].
Schlichting C.D.: The evolution of phenotypic plasticity in plants. — Annu. Rev. Ecol. Syst. 17: 667–693, 1986.
Schulze E.D., Küppers M.: Short-term and long-term effects of plant water deficits on stomatal response to humidity in Corylus avellana L. — Planta 146: 319–326, 1979.
Shahidi F., Alasalvar C., Liyana-Pathirana C.M.: Antioxidant phytochemicals in hazelnut kernel (Corylus avellana L.) and hazelnut byproducts. — J. Agric. Food Chem. 55: 1212–1220, 2007.
Sims J.R., Haby V.A.: Simplified colorimetric determination of soil organic matter. — Soil Sci. 112: 137–141, 1971.
Soolanayakanahally R.Y., Guy R.D., Silim S.N. et al.: Enhanced assimilation rate and water use efficiency with latitude through increased photosynthetic capacity and internal conductance in balsam poplar (Populus balsamifera L.). — Plant Cell Environ. 32: 1821–1832, 2009.
Sultan S.E.: Phenotypic plasticity and plant adaptation. — Acta Bot. Neerl. 44: 363–383, 1995.
Sultan S.E.: Phenotypic plasticity for plant development, function and life history. — Trends Plant Sci. 5: 537–542, 2000.
Thiele A., Krause G.H., Winter K.: In situ study of photoinhibition of photosynthesis and xanthophyll cycle activity in plants growing in natural gaps of the tropical forest. — Aust. J. Plant Physiol. 25: 189–195, 1998.
Valentini R., Epron D., de Angelis P. et al.: In situ estimation of net CO2 assimilation, photosynthetic electron flow and photorespiration in Turkey oak (Q. cerris L.) leaves: diurnal cycles under different levels of water supply. — Plant Cell Environ. 18: 631–640, 1995.
Valentini N., Calizzano F., Boccacci P., Botta R.: Investigation on clonal variants within the hazelnut (Corylus avellana L.) cultivar ‘Tonda Gentile delle Langhe’. — Sci. Hortic.-Amsterdam 165: 303–310, 2014.
Valladares F., Wright S.J., Lasso E. et al.: Plastic phenotypic response to light of 16 congeneric shrubs from a Panamanian rainforest. — Ecology 81: 1925–1936, 2000.
Valladares F., Chico J.M., Aranda I. et al.: The greater seedling high-light tolerance of Quercus robur over Fagus sylvatica is linked to a greater physiological plasticity. — Trees 16: 395–403, 2002.
Valladares F., Arrieta S., Aranda I. et al.: Shade tolerance, photoinhibition sensitivity and phenotypic plasticity of Ilex aquifolium in continental-Mediterranean sites. — Tree Physiol. 25: 1041–1052, 2005.
Violante P.: [Chemical Methods of Soil Analysis.] Pp. 60. Franco Angeli Editore, Milan 2000. [In Italian]
Walters M.B., Reich P.B.: Are shade tolerance, survival, and growth linked? Light and nitrogen effects on hardwood seedlings. — Ecology 77: 841–853, 1996.
Walters M.B., Reich P.B.: Low-light carbon balance and shade tolerance in the seedlings of woody plants: do winter deciduous and broad-leaved evergreen species differ? — New Phytol. 143: 143–154, 1999.
Whitcher I.N., Wen J.: Phylogeny and biogeography of Corylus (Betulaceae): inference from ITS sequences. — Syst. Bot. 26: 283–298, 2001.
Yamashita N., Ishida A., Kushima H., Tanaka N.: Acclimation to sudden increase in light favouring an invasive over native trees in subtropical islands, Japan. — Oecologia 125: 412–419, 2000.
Zunzunegui M., Ain-Lhout F., Díaz Barradas M.C. et al.: Physiological, morphological and allocation plasticity of a semi-deciduous shrub. — Acta Oecol. 35: 370–379, 2009.
Zunzunegui M., Díaz Barradas M.C., Ain-Lhout F. et al.: Seasonal physiological plasticity and recovery capacity after summer stress in Mediterranean scrub communities. — Plant Ecol. 212: 127–142, 2011.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: This work was supported by grants from the Department of Earth and Environmental Sciences, University of Pavia, for the project of “Caratterizzazioni funzionali e strutturali delle specie presenti nella Riserva Naturale Integrale Bosco Siro Negri”.
Rights and permissions
About this article
Cite this article
Catoni, R., Granata, M.U., Sartori, F. et al. Corylus avellana responsiveness to light variations: morphological, anatomical, and physiological leaf trait plasticity. Photosynthetica 53, 35–46 (2015). https://doi.org/10.1007/s11099-015-0078-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-015-0078-5