Abstract
Lévy noise plays a significant role in jump-noise processes found in neurons. We perform numerical simulations of synaptically coupled Izhikevich networks under the effect of general non-Gaussian Lévy noise. Firing dynamics of an all-to-all coupled Izhikevich network and two excitatory coupled Izhikevich networks with differing adaptation properties are studied in response to applied Lévy noise. Whole parameter space of Lévy noise is investigated by changing its characteristic exponent, skewness, scale and mean parameters. The simulation results show that the noise generated from \(\alpha \)-stable Lévy distribution causes significant dynamical changes in the firing pattern of neuronal network. Different firing patterns exhibited by Izhikevich neurons such as tonic spiking, chattering, intrinsic bursting, low threshold spiking and fast spiking are studied in the network level with and without Lévy noise. In all cases, neurons are found to be irregularly spiking when Lévy noise with characteristic exponent \(\alpha \) equal to 0.5 is applied. Lévy noise with this particular value of \(\alpha \) as 0.5 causes the Izhikevich network to have a dynamical behaviour independent of topology and heterogeneity of the network. At this value of characteristic exponent, density of the stable Lévy distribution in standard parameterisation is maximum. Other parameters of Lévy noise do not influence the way in which a particular neuron is firing. The effects of Lévy noise on the correlation between individual neurons and on the networks are investigated using statistical measures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
In recent times, the spiking neuron models have been subjected to intense research in the field of neuronal science owing to the fact that neurons carry information among each other using their firing rates [1]. Examples of commonly investigated spiking neuron models are Hodgkin–Huxley-type model, FitzHugh–Nagumo model and Hindmarsh–Rose model [2]. Also, several hybrid neuron models have been introduced which combine continuous spike-generation mechanisms and discontinuous resetting process after spiking. An example of nonlinear hybrid neuron model is the Izhikevich neuron model which can generate different types of bifurcation and is capable of reproducing most of the spiking activities observed in the actual neural systems by tuning a few parameters. This two-dimensional model has been shown to capture a wide range of cellular firing characteristics in various parts of mammalian brain. It is a biologically plausible neuron model and can reproduce spiking and bursting behaviour of known types of neocortical and thalamic neurons [3, 4].
It is well known that the brain can produce fluctuations with complex scaling properties. Using the statistical properties of these fluctuations, evolution of the brain dynamics can be studied [5]. The fluctuations during a brain activity can be considered as a random motion. In the continuous limit, these fluctuations can also be approximated as the motion of a diffusing macroscopic particle [6, 7]. This framework can be well modelled by a generalised Langevin differential equation consisting of Lévy noise with deterministic and stochastic components [8, 9].
Though the stochastic effect is often regarded as unwanted as it damages the extraction of useful information, in certain nonlinear systems it is realised that the presence of internal or external noise can enhance the response of the dynamics of the signal [10]. The noise in dynamical systems has wide applications in various fields of physics, biology, geology and neurology [11,12,13,14,15,16]. Usually, the random excitations of noise are assumed to be of Gaussian white noise kind which can only describe random small fluctuations without big jumps. But it has been investigated that small disturbances are present in the biological and neurological activities and those disturbances are always combined with discontinuous unpredictable jumps of random nature. Also, the statistical characteristics of the random noise significantly deviate from the Gaussian distribution [17,18,19]. Therefore, it is necessary to consider the non-Gaussian disturbance in the studies of biological systems. Lévy noise can model such non-Gaussian noise having both small and big random fluctuations [20, 21]. Since the paths of Lévy processes may contain random jump discontinuities of arbitrary size occurring at arbitrary random times, Lévy processes form a rich class of stochastic processes having applications in many different areas of science [22].
Another advantage of Lévy noise model is that it includes not only pure-diffusion and pure-jump models but also jump-diffusion models [23, 24], and it is much better than a pure-diffusion neuron model with additive Gaussian noise in describing the evolution of membrane potential of a neuron. Pure-diffusion neuron models require large number of synapses with small membrane effects due to the small coupling coefficients or the synaptic weights [25, 26]. On the contrary in real scenarios, often fewer synaptic inputs are produced near the postsynaptic neuron’s trigger zone, and these inputs produce impulses in noise amplitudes [27, 28]. Thus, standard Gaussian noise does not faithfully represent the dynamics of realistic neuron models but Lévy noise does. The general Lévy model is favourable in engineering applications as well [29,30,31]. Lévy noise can also effectively represent the random noises due to external radiation-induced magnetic flux of electrical fields when applied on neuron networks [32,33,34].
Lévy noise can greatly describe the complex biological and neuronal environment [35, 36]. One of the simplest creatures Escherichia coli has been studied based on biological fluctuation framework using the Lévy walks model [37]. Dubkov and Kharcheva [38] have examined the steady- state time characteristics of anomalous diffusion in the form of Lévy flights in a symmetric bistable quadratic potential. Xu et al. have investigated stochastic resonance phenomenon in a FitzHugh– Nagumo system induced by an additive Lévy noise [39]. Wu et al. [40] have investigated the electrical activities in an improved Hindmarsh–Rose model excited by the external electromagnetic radiation of Lévy noise. They have observed that decreasing of the characteristic exponent of Lévy noise improves the firing of Hindmarsh–Rose neuron. They have chosen arbitrary values for characteristic exponent and skewness parameter of the noise. In this paper, complete parameter space of Lévy noise is investigated, and the Lévy noise with particular characteristic exponent is found to increase the firing activity of neurons. Cai et al. [41] have considered the dynamics of escape in the stochastic FitzHugh–Nagumo (FHN) neuronal model driven by symmetric \(\alpha \)-stable Lévy noise. Zhang et al. [42] have done a study on low-dimensional reduction for a slow–fast data assimilation system with non-Gaussian \(\alpha \)-stable Lévy noise via stochastic averaging. Zhan and Liu [43] have studied dynamical response of Morris–Lecar system with electromagnetic induction (EMI) and Gaussian white noise. Jin et al. [44] have explored how noise can affect the synchronisation and stability of a neuronal network. Transition of electrical activities in neurons induced by electromagnetic radiation has been also explored by researchers [45, 46]. But the influence of Lévy noise on the networks of Izhikevich neurons models has not been investigated. Therefore, the study of effect of non-Gaussian Lévy noise on the neurons of such model is relevant.
2 Neuron model and method
2.1 Izhikevich neuron model
The Izhikevich model consists of two parts: a two-dimensional system of ordinary differential equations and a reset mechanism. The differential equations model the subthreshold behaviour of the system and generate the upstroke of the action potential, while the reset mechanism is responsible for producing the downstroke of the action potential. V and U are the model variables. V is the membrane potential. U is the current which represents the effect of the slow currents on spike generation and allows for spike- frequency adaptation [3, 4]. The model is represented as
While \(V<V_\mathrm{peak}\):
When V crosses a certain value called \(V_\mathrm{peak}\) from below, V and U are updated as
C is the membrane capacitance, \(V_\mathrm{R}\) is the resting membrane potential, \(I_\mathrm{ext}\) is the applied current (the effect of externally injected and synaptic currents), \(V_\mathrm{T}\) is the voltage threshold when \(b=0\) and \(I_\mathrm{ext}=0\), \(V_\mathrm{peak}\) is the spike cut-off value, k is a scaling factor that affects the value of \(\hbox {d}V/\hbox {d}t\) and the spike width, a is the time constant of the adaptation current U, b describes the sensitivity of the adaptation current to subthreshold fluctuations of the membrane potential V, c is the voltage reset value, d describes the difference between outward and inward currents activated during the spike which affects the after-spike behaviour.
When neuron models of Izhikevich kind are coupled together in large networks, they show bifurcations that isolated neuron model cannot generate [47]. A feasible method of analysing large networks is through population density equations. In population density methods, a time-varying population density function is defined initially whose evolution is determined by a conservation law. Population density equation is simplified using moment closure methods and a sequence of approximations as described in the literature [48, 49]. Finally, a system of ODEs is achieved which can be analysed numerically. Equations (2.1)–(2.3) are rewritten as [48]:
for \(i = 1, 2, \ldots N\). The parameter values are chosen as follows. \(C=250\) pF, \(k=2.5\) nS/mV, \(V_\mathrm{R}=-\,65\) mV, \(V_\mathrm{T}=V_\mathrm{R}+40-\frac{\eta }{k}=41.7\) mV, \(V_\mathrm{peak}=30\) mV, \(V_\mathrm{reset}=-\,55\) mV, \(U_\mathrm{jump}=200\) pA, \(\tau _{U}=100\) ms, \(\eta =-\,1\) nS, \(s_\mathrm{jump}=0.8, \tau _\mathrm{syn}=2\) ms.
2.2 The Lévy noise
Let \(\xi \) denotes the time-dependent Lévy noise and obeys the probability density function \(L_{\alpha ,\beta }(\xi ;\,\sigma ,\,\mu )\), whose characteristic function is [50]:
Therefore, for \(\alpha \,\in \,(0,1)\,\bigcup \,(1,2]\),
and for \(\alpha =1\),
here \(\alpha {{\in }}(0,2]\) is known as the characteristic exponent, and it denotes the stability index that describes an asymptotic power law of the Lévy distribution. The constant \(\beta \,\,(\beta \,\in [-\,1,1])\) is the asymmetry or skewness parameter. \(\sigma \,\,(\sigma \,\in \,(0,\infty ))\) is the scale parameter, and \(\mu \,(\mu \in R)\) is the mean parameter. The noise intensity is denoted as \(D=\sigma ^{\alpha }\), and hence, the Lévy process can also be represented as \(L_{\alpha ,\beta }(\xi ;\,D,\,\mu )\). When \(\alpha =0.5\), the stable distribution is called \(\alpha \)-stable Lévy distribution.
We analyse the effect of Lévy noise on Izhikevich neurons using the approach mentioned in Sect. 2.1. The paper is organised as follows: we consider two neural networks that are deterministic and homogeneous. The first example, studied in Sect. 2.3, is a homogeneous all-to-all coupled Izhikevich network. Section 2.4 considers two Izhikevich networks that are all-to-all coupled both internally and externally and each network is split into two populations, one strongly adapting and the other weakly adapting. The parameters are chosen so as to match the neuronal experimental results from research papers [47, 48].
2.3 The single all-to-all coupled Izhikevich network with Lévy noise
We consider a network of N identical neurons of the Izhikevich model [Eqs. (2.4)–(2.6)], with all-to-all coupling. The coupling is modelled as synaptic [51]. The postsynaptic neuron generates a postsynaptic potential by integrating the inputs from the neighbouring presynaptic neuron. The synaptic current in the jth neuron is modelled by
where \(g_\mathrm{syn}\) is the maximal synaptic conductance and \(E_\mathrm{r}\) is the synaptic reversal potential. \(s_{j,i}\) is the gating variable of the synapse from the ith neuron to the jth, and it is not allowed to exceed 1.
When \(V_{i}\) crosses 0 from below:
where \(S_{\max }\) is a synaptic update parameter and is chosen to be 0.8.
Otherwise:
Also, the total effect of current in the neuronal system is taken as
Therefore, dimensionless form of the single all-to-all coupled Izhikevich network with synaptic coupling and Lévy noise is given by
for \(i = 1, \ldots , N\), where \(v_{i}=1+\frac{Vi}{|V_\mathrm{R}|},\,u_{i}=\frac{U_{i}}{k|V_\mathrm{R}|^{2}}\). The dimensionless parameters are given as follows:
\(\alpha =1+\frac{V_\mathrm{T}}{\left| V_\mathrm{R}\right| }=0.62\), \(b=\frac{\eta }{k\left| V_\mathrm{R}\right| }=0.006\), \(I=\frac{I_\mathrm{app}}{k\left| V_\mathrm{R}\right| ^{2}}=0.14\), \(g=\frac{g_\mathrm{syn}}{k\left| V_\mathrm{R}\right| }=1.23\), \(a=\left( \frac{C}{\tau _{U}k\left| V_\mathrm{R}\right| }\right) =0.015\), \(\tau _{s}=\frac{\tau _\mathrm{sync}k\left| V_\mathrm{R}\right| }{C}=1.3\), \(u_\mathrm{jump}=\frac{U_\mathrm{jump}}{k\left| V_\mathrm{R}\right| ^{2}}=0.0189, I_\mathrm{app}=1500\) pA, \(g_\mathrm{syn}=300\) nS, \(N=1000\).
2.4 Excitatory coupled pair of Izhikevich networks with different adaptation properties with Lévy noise
Neuronal adaptation refers to the response profile when a stimulus is applied to a neuron. It indicates the ability of a neuron to adapt to a particular situation by changing its threshold. Strongly adapting neurons quickly modify their firing rate in response to a stimulus. The adaptation levels of the networks are determined and controlled by \(\tau _{U,\mathrm{SA}}\), \(\tau _{U,\mathrm{WA}}\), \(I_\mathrm{app,SA}\), \(I_\mathrm{app,WA}\) and synaptic gating variables \(s_{j}\). We consider a pair of networks which are excitatory in nature, and they are all-to-all coupled. Each network consists of two distinct homogeneous populations: one is strongly adapting and the other is weakly adapting. Biological networks present samples for such cases. For example, pyramidal neurons in hippocampal area CA3 can be grouped into strongly adapting, weakly adapting and intrinsically bursting populations based on the amount of spike-frequency adaptation they exhibited [53]. Since there are two populations, four maximal synaptic conductances \(g_{mj}\) and two synaptic gating variables \(s_{j}\) are needed. Here j denotes the presynaptic network and m denotes the postsynaptic network, with \(j =\) SA indicating the subnetwork with strong adaptation and \(j =\) WA is the subnetwork with weak adaptation, and similarly for m.
The equations for the network are [48],
for \(i=1,\ldots N_{m}\). Note that \(p=N_\mathrm{SA}/(N_\mathrm{SA}+N_\mathrm{WA})\) is the proportion of strongly adapting neurons in the network.
Equations (2.19)–(2.22) are converted into dimensionless form. There are separate population density equations for strongly and weakly adapting populations. Then moment closure reduced population density equations are coupled together by the two coupling variables and two adaptation variables [48]. Lévy noise is applied to the voltage variable of each neuron of Izhikevich population. The other parameter values are \(\tau _{U,\mathrm{SA}}=100\) ms, \(\tau _{U,\mathrm{WA}}=10\) ms, \(I_\mathrm{app,SA}=\) 1000–2000 pA, \(I_\mathrm{app,WA}=1200\) pA, \(g_{\mathrm{syn},mj}=200\) nS, \(N_\mathrm{SA}=800\) and \(N_\mathrm{WA}=200\).
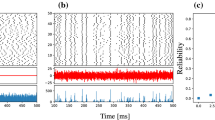
Simulation of a neuron in the network of 1000 tonic spiking Izhikevich neurons with Lévy noise (\(\alpha =0.5,\beta =1,\sigma =1,\mu =0\)). Membrane potential V and synaptic conductance g(t) exhibit irregular and random spiking behaviour. As time evolves, the adaptation current U also behaves randomly. Lévy noise with \(\alpha =0.5\) causes additional spike resettings in the neurons of the network
3 Results
3.1 Dynamics of single all-to-all network with Lévy noise
For analysing the time series of the Izhikevich model network, Euler method is used. The whole parameter space of Lévy noise is investigated. It is found that when characteristic exponent \(\alpha \) becomes 0.5, Lévy noise causes the neuronal firing to be irregular in different types of firing activities shown by Izhikevich network such as tonic spiking, intrinsic bursting, chattering, low threshold spiking and fast spiking (refer Figs. 1, 2, 3, 4, 7, 8, 9, 10, 11, 12, 13, 14). Other parameter values of Lévy noise are varied in their respective ranges, but a significant change in neuronal firing is not observed. According to Nolan, density takes maximum value when \(\alpha \) becomes 0.5 in the standard parameterisation of the stable Lévy distribution [52]. This property may be considered as the reason why Lévy noise with \(\alpha \) equal to 0.5 triggers irregular firing in the neuronal network. Figure 15 shows the probability density function f(x) of stable Lévy distribution for different values of \(\alpha \). Other parameters of Lévy distribution are chosen to be \(\beta =0, \sigma =1, \mu =0\). This figure is reproduced using the program stable.exe with permission from the website http://fs2.american.edu/jpnolan/www/stable/. When \(\alpha =\) 0.5, the stable density is found to be maximum. Therefore, maximum influence due to the Lévy noise on the neurons occurs when Lévy noise has the characteristic exponent \(\alpha =\) 0.5. Noise with other values of \(\alpha \) has minimal effect on the neuronal firings.
In Figs. 1 and 2, membrane potential, adaptation current and synaptic conductance of a randomly selected tonic spiking neuron of all-to-all Izhikevich network are plotted without and with Lévy noise, respectively. The parameters of Lévy noise are chosen as \(\alpha =0.5,\beta =1,\sigma =1,\mu =0\). The spiking activities are found to be different with and without Lévy noise. It has been observed that only for the Lévy noise characterised by the \(\alpha \)-stable distribution \(L_{\alpha ,\beta }(\xi ;\,\sigma ,\,\mu ) = L_{0.5,\beta }(\xi ;\,1,\,0)\), the activity modes show deviation from the noise-free case. The tonic spiking mode of the membrane potential has become aperiodic and irregular. It can be seen that the neurons which produced 2 spikes in a particular time interval have been excited to yield 3, 4 or more spikes in the same interval under the influence of Lévy noise with \(\alpha =0.5\). The spiking pattern of membrane voltage shows transition into an irregular state. The adaptation current also exhibits a deviation from the fluctuation pattern shown when noise is absent. It can also be seen that synaptic conductance goes into a completely random and irregular pattern. For all other cases of parameter values of Lévy noise, even if they are varied within their respective limits, no significant difference in spiking activities is noticed. Figures 3 and 4 represent the cases where noise does not alter the spiking behaviour of the neurons and retains the dynamics of noise-free network. The phase space plots of the network without and with Lévy noise are represented in Figs. 5 and 6.
Also, the effect of Lévy noise is studied for other firing types shown by Izhikevich neuronal network, namely intrinsic bursting, chattering, fast spiking and low threshold spiking (Figs. 7, 8, 9, 10, 11, 12, 13, 14). In all cases, it is found that when \(\alpha \) parameterof Lévy noise is 0.5, firings become random and irregular.
3.2 Dynamics of excitatory coupled pair of networks with Lévy noise
To study the response of excitatory coupled pair of Izhikevich networks towards the applied noise, neurons are randomly chosen from strongly and weakly adapting populations. Figure 16 represents the neuronal fluctuations without noise. Neuronal fluctuations for strongly adapting neuron are marked in red colour, while those for weakly adapting neuron are in green colour. The strongly adapting neuron population is bursting, while the weakly adapting population has an oscillatory non-bursting firing pattern. Figure 17 denotes the dynamics of the networks with Lévy noise having parameters \(\alpha =0.5,\,\beta =1,\sigma =1,\mu =0\). Spikes have become irregular in nature. Completely random fluctuations appear in the membrane potential and in the synaptic conductance variables of strongly adapting population. The spiking patterns of membrane potential of weakly adapting population have also undergone a change with a loss in periodicity. But the current of weakly adapting population is not showing a change in the firing pattern. The noise is found to have no effect on the overall behaviour of the networks for any other \(\alpha \) parameter value of the Lévy noise. It is interesting to note that the strongly adapting neuronal population in coupled pair of heterogeneous networks under Lévy noise with \(\alpha =0.5\) (Fig. 17) shows spiking patterns similar to those exhibited by the single homogenous network under the Lévy noise with same \(\alpha \) (Fig. 2). It shows that Lévy noise with particular value of \(\alpha \) equal to 0.5 causes the Izhikevich network to have a dynamical behaviour independent of topology and heterogeneity of the network. In cases where the networks are either noise-free or has Lévy noise with \(\alpha \ne 0.5\) (Figs. 18, 19), spiking dynamics depend on the topology of the networks.
Simulation of a network of Izhikevich neurons without Lévy noise. The network consists of two distinct populations: 800 strongly adapting neurons and 200 weakly adapting neurons. Dynamics of strongly adapting neuron are shown in red, while that of weakly adapting neuron is in green. Approximately regular bursting dynamics can be seen for both kinds of neurons
Simulation of Izhikevich network with 800 strongly adapting neurons and 200 weakly adapting neurons. Lévy noise (\(\alpha =0.5,\beta =1,\sigma =1,\mu =0\)) is applied. The regularity of the dynamics has been lost. In the noise-free case, strongly and weakly adapting neurons had undergone bursting simultaneously. But when Lévy noise (\(\alpha =0.5,\beta =1,\sigma =1,\mu =0\)), no such synchronisation is seen between neurons with different adaptations. Quiescent states in the firing patterns have undergone a resetting into spiking states. The whole dynamical behaviour of coupled pair of these heterogeneous networks under Lévy noise (\(\alpha =0.5\)) matches with that of a single homogenous network under the same Lévy noise (refer Fig. 2)
Simulation of a network of Izhikevich neurons. The network consists of two distinct populations: 800 strongly adapting neurons and 200 weakly adapting neurons. Lévy noise (\(\alpha =1.7,\beta =0,\sigma =1,\mu =0\)) is applied. No change in the dynamical behaviour is brought about by the introduction of this noise
3.3 Statistical analysis
The irregularity and spiking variability of the neuronal fluctuations of the networks with Lévy noise \((\alpha =0.5)\) are further analysed using statistical methods. Analyses show that clustered neuronal networks behave in a random manner. Single all-to-all coupled Izhikevich network and the excitatory coupled pair of Izhikevich networks produce similar graphs for spike rate and variation measures. The analyses are carried out for tonic spiking neuron network for a time duration of 1000 ms.
3.3.1 Spiking rate
Spike rate is a measure of neuronal activity over a duration of time measured in Hz. They are commonly used to quantify a neural response over a given duration of time T. When a neuron’s spiking activity is represented as a vector of spike times \(t_{1},t_{2},t_{3}\ldots t_{N}\), the rate is defined by
where N is the total number of spikes over a given duration T. Spike rates can be computed as a function of single neuron across trials, or over a network, thus generating a distribution of rates with variability [54].
The activities of the 100 neurons randomly chosen from the Izhikevich population are plotted as a function of their firing rate. Without noise, neurons fire approximately at a constant rate of 0.162 (Fig. 20). It is observed that the population activity is significantly increased to 0.36–0.365 when the Lévy noise is applied (Fig. 21). Such increase is seen only when Lévy noise with the parameter \(\alpha =0.5\) is applied. This result is in agreement with the increased spiking behaviour of both single homogeneous network (Fig. 2) and coupled pair of heterogeneous networks (Fig. 17) with Lévy noise having \(\alpha =0.5\). When the \(\alpha \) value of Lévy noise is changed, spiking rate decreases to its value in the noise-free case.
3.3.2 Inter-spike intervals (ISIs)
An ISI measures the latency \(\Delta \)t between precise spike times. ISIs can be computed for every single spike (minus one) or a percentage of spikes. When a neuron’s spiking activity is arranged in a vector of spike times \(t_{1},t_{2},t_{3}\ldots t_{N}\), the i4th ISI is defined by
where t represents a given spike time. Intervals are then counted, generating a distribution of ISIs [54].
A bifurcation plot of ISIs is drawn against varying neuronal currents of the network under the Lévy noise with \(\alpha =0.5\) (Fig. 22). Noise is imposed on the neuronal network from time t=0 onwards. It is observed that chaotic nature is introduced from the beginning of time when the applied Lévy noise is having characteristic index \(\alpha \) as 0.5.
3.3.3 Coefficient of variation (Cv)
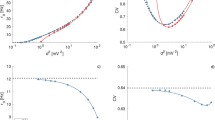
The Cv is a measure of the dispersion in the ISI distribution. Using the vector of spike times \(t_{1},t_{2},t_{3}\ldots t_{N}\), then Cv is defined by
where N is the total number of spikes, \(\Delta t\) is a given ISI, and \(\bar{\Delta t}\) is defined by
A Poisson process produces distributions with \(Cv=1\). When \(Cv>1\), it implies that a given spike train is less regular than a Poisson process with the same firing rate. Cv detects a global variability of a whole ISI sequence and is sensitive to the firing rate fluctuation which may occur in natural conditions. Cases where either noise is absent or Lévy noise \((\alpha \ne 0.5)\) is applied, neurons in the network show a Cv value much less than 1 (Fig. 23). For Izhikevich networks having Lévy noise (\(\alpha =0.5\)), the coefficient of variation of each neuron has a value greater than 1 which indicates the irregularity in the spiking nature of the neurons introduced by the particular \(\alpha \)- stable Lévy noise (Fig. 24).
Coefficient of variation graph of neurons when Lévy noise is absent. Cv values of the neurons lie in the range of 0.01–0.016. It is much less than 1. The Izhikevich neurons behave in a more or less periodic spiking manner in the absence of Lévy noise. Neurons produce similar graph with low values of Cv when Lévy noise \((\alpha \ne 0.5)\) is applied
3.3.4 Autocorrelation
Autocorrelation gives a measure of how well a signal is correlated with itself. Using an autocorrelation, we quantify the self-similarity of a neuron signal as a function of the time lag \(\tau \) [55]. Autocorrelation plot helps to check randomness in a data set. This randomness is ascertained by computing autocorrelations for data values at varying time lags. If random, such autocorrelations should be near zero for any and all time-lag separations. If non-random, then one or more of the autocorrelations will be significantly nonzero.
The neuronal fluctuations of Izhikevich networks with Lévy noise \((\alpha =0.5)\) show the autocorrelation diagram as in Fig. 26. They have self-similarity only when \(\tau =\) 0. It occurs when the Lévy noise with \(\alpha =0.5\) is added to the action potentials of the neurons in the network. When noise distribution takes other parameter values, such irregularity is lost (Fig. 25).
4 Discussion
The present work investigates the dimensionless stochastic Izhikevich model under the effect of non-Gaussian Lévy noise and examines the electrical activities of neurons. It is observed that the noise generated from Lévy noise distribution can cause the mode transition of electrical activities both in single Izhikevich network and in excitatory coupled pair of Izhikevich networks. When the noise produced from the Lévy distribution with parameter \(\alpha =0.5\) is applied to neuronal networks, the rest states of the electrical activities of neurons are excited to be firing states. The spiking states in a periodic electrical activity can be induced to be spiking states with high randomness. Also, the spiking states can be changed into a sort of bursting state. Irregularity brought about by the Lévy noise depends only on the value taken by the characteristic exponent \(\alpha \). Even a slight deviation in the \(\alpha \) parameter value of Lévy noise causes the neurons to revert to their more or less periodically spiking state. It is also noted that the strongly adapting neuronal population in coupled pair of heterogeneous networks under Lévy noise with \(\alpha =0.5\) and the single homogenous network under the Lévy noise with same \(\alpha \) show similar spiking patterns. It shows that Lévy noise with particular value of \(\alpha \) as 0.5 causes the Izhikevich network to have a dynamical behaviour independent of topology and heterogeneity of the network. When the networks are either noise-free or have Lévy noise with \(\alpha \ne 0.5\), spiking dynamics depend on the topology of the networks. The statistical measure also shows the increased firings of neurons under the Lévy noise with \(\alpha =0.5\).
Wu et al. [40] have studied the influence of Lévy noise on Hindmarsh–Rose (HR) model under electromagnetic induction. They have investigated that decrease in characteristic exponent from its upper boundary value 2 and increase in intensity of Lévy noise can heighten neuronal firings in HR neuron. Also the upward or downward skewing of the jumps of Lévy noise can alter HR neuron’s firing dynamics. In this paper, we have explored that in the case of Izhikevich network an increase in neuronal firings is observed when characteristic exponent is set at \(\alpha =0.5\). But intensity, skewness and mean parameters of Lévy noise do not influence the dynamics of Izhikevich network.
In general, our studies establish that \(\alpha \)-stable Lévy noise could improve electrical activity in a neuron individually and on the network level. This study can be extended to investigate the mean field approximation of the present network and to study the cumulative effect of electromagnetic radiation and Lévy noise on the network.
References
Rabinovich, M.I., Varona, P., Selverston, A.I., Abarbanel, H.D.I.: Dynamical principles in neuroscience. Rev. Mod. Phys. 78, 1213–1265 (2006)
Thottil, S.K., Ignatius, R.P.: Nonlinear feedback coupling in Hindmarsh–Rose neurons. Nonlinear Dyn. 87, 1879–1899 (2016)
Izhikevich, E.M.: Simple model of spiking neurons. IEEE Trans. Neural Netw. 14, 1569–1572 (2003)
Izhikevich, E.M.: Which model to use for cortical spiking neurons? IEEE Trans. Neural Netw. 15, 1063–1070 (2004)
Papo, D.: Functional significance of complex fluctuations in brain activity: from resting state to cognitive neuroscience. Front. Syst. Neurosci. 8, 112 (2014)
Gerstein, G.L., Mandelbrot, B.: Random walk models for the spike activity of a single neuron. Biophys. J. 4(1 Pt 1), 41–68 (1964)
Capocelli, R., Ricciardi, L.: Diffusion approximation and first passage time problem for a model neuron. Kybernetik 8(6), 214–223 (1971)
Linaro, D., Storace, M., Giugliano, M.: Accurate and fast simulation of channel noise in conductance-based model neurons by diffusion approximation. PLoS Comput Biol. 7(3), e1001102 (2011)
West, B.J.: Fractal physiology and the fractional calculus: a perspective. Front. Physiol. (2010). https://doi.org/10.3389/fphys.2010.00012
Benzi, R., Sutera, A., Vulpiani, A.: The mechanism of stochastic resonance. J. Phys. A Math. Gen. (1981). https://doi.org/10.1088/0305-4470/14/11/006
Wiesenfeld, K., Moss, F.: Stochastic resonance and the benefits of noise: from ice ages to crayfish and SQUIDs. Nature 373, 33–36 (1995)
Douglass, J.K., Wilkens, L., Pantazelou, E., et al.: Noise enhancement of information transfer in crayfish mechanoreceptors by stochastic resonance. Nature 365, 337–340 (1993)
Zhang, X.F., Hu, N.Q., Hu, L., et al.: Multi-scale bistable stochastic resonance array: a novel weak signal detection method and application in machine fault diagnosis. Sci. China Technol. Sci. 56(9), 2115–2123 (2013)
Xu, Y., Wu, J., Zhang, H.Q., Ma, S.J.: Stochastic resonance phenomenon in an underdamped bistable system driven by weak asymmetric dichotomous noise. Nonlinear Dyn. 70(1), 531–539 (2012)
Zhang, H., Yang, T., Xu, Y., Xu, W.: Parameter dependence of stochastic resonance in the FitzHugh-Nagumo neuron model driven by trichotomous noise. Eur. Phys. J. B (2015). https://doi.org/10.1140/epjb/e2015-50865-3
He, Z.Y., Zhou, Y.R.: Vibrational and stochastic resonance in the FitzHugh-Nagumo neural model with multiplicative and additive noise. Chin. Phys. Lett. (2011). https://doi.org/10.4028/www.scientific.net/AMM.117-119.685
Xu, Y., Li, J.J., Feng, J., et al.: Lévy noise-induced stochastic resonance in a bistable system. Eur. Phys. J. B 86, 1–7 (2013)
Li, X.L., Ning, L.J.: Stochastic resonance in FizHugh–Nagumo model driven by multiplicative signal and non-Gaussian noise. Ind. J. Phys. 89(2), 189–194 (2015)
Sun, X.J., Lu, Q.S.: Non-Gaussian colored noise optimized spatial coherence of a Hodgkin–Huxley neuronal network. Chin. Phys. Lett. (2014). DOI: https://doi.org/10.1088/0256-307X/31/2/020502.
Xu, Y., Feng, J., Li, J.J., Zhang, H.Q.: Lévy noise induced switch in the gene transcriptional regulatory system. Chaos 23, 013110 (2013)
Xu, Y., Feng, J., Li, J.J., et al.: Stochastic bifurcation for a tumor–immune system with symmetric Lévy noise. Phys. A 392, 4739–4748 (2013)
Applebaum, D.: Lévy Processes and Stochastic Calculus. Cambridge University Press, Cambridge (2004)
Hohn, N., Burkitt, A.N.: Shot noise in leaky integrate-and-fire neuron. Phys. Rev. E 63(3 Pt 1), 031902 (2001)
Sacerdote, L., Sirovich, R.: Multimodality of the interspike interval distribution in a simple jump-diffusion model. Sci. Math. Jpn. Online 58, 307–322 (2003)
Gerstner, W., Kistler, W.M.: Spiking Neuron Models: Single Neurons, Populations, Plasticity. Cambridge University Press, Cambridge (2002)
Kohn, A.F.: Dendritic transformations on random synaptic inputs as measured from neuron’s spike train-modeling and simulation. IEEE Trans. Biomed. Eng. 36(1), 44–54 (1989)
Giraudo, M.T., Sacerdote, L.: Jump-diffusion processes as models of neuronal activity. BioSystems 40, 75–82 (1997)
Kandel, E.R., Schwartz, J.H., Thomas, M.J.: Principles of Neuroscience, 4th edn. McGraw-Hill, New York (2000)
Mead, C.: Analog VLSI and Neural Systems. Addison-Wesley, Reading (1989)
Grigoriu, M.: Applied Non-Gaussian Processes: Examples, Theory, Simulation, Linear Random Vibration, and MATLAB Solutions. Prentice-Hall, Englewood Cliffs (1995)
Nikias, C.L., Shao, M.: Signal Processing with Alpha-Stable Distributions and Applications. Wiley, New York (1995)
Yi, G., Wang, J., Tsang, K.M., Wei, X., Deng, B., Han, C.: Spike-frequency adaptation of a two-compartment neuron modulated by extracellular electric fields. Biol. Cybern. 109, 287–306 (2015)
Reato, D., Rahman, A., Bikson, M., Parra, L.C.: Low-intensity electrical stimulation affects network dynamics by modulating population rate and spike timing. J. Neurosci. 30, 15067–79 (2010)
Akiyama, H., Shimizu, Y., Miyakawa, H., Inoue, M.: Extracellular DC electric fields induce nonuniform membrane polarization in rat hippocampal CA1 pyramidal neurons. Brain Res. 1383, 22–35 (2011)
Dubkov, A.A., Spagnolo, B., Uchaikin, V.V.: Lévy flight superdiffusion: an introduction. Int. J. Bifurc. Chaos 18, 2649–72 (2008)
Xu, Y., Li, Y., Zhang, H., Li, X., Kurths, J.: The switch in a genetic toggle system with Lévy Noise. Sci. Rep. 6, 31505 (2016)
Nurzaman, S.G., Matsumoto, Y., Nakamura, Y., Shirai, K., Koizumi, S., Ishiguro, H.: From Lévy to Brownian: a computational model based on biological fluctuation. PLoS ONE 6, e16168 (2011)
Dubkov, A.A., Kharcheva, A.A.: Features of barrier crossing event for Lévy flights. Europhys. Lett. 113, 30009 (2016)
Wang, Z., Xu, Y., Yang, H.: Lévy noise induced stochastic resonance in an FHN model. Sci. China Technol. Sci. 59(3), 371–375 (2016)
Wu, J., Xu, Y., Ma, J.: Lévy noise improves the electrical activity in a neuron under electromagnetic radiation. PLoS ONE 12(3), e0174330 (2017)
Cai, R., Chen, X., Duan, J., Kurths, J., Li, X.: Lévy noise-induced escape in an excitable system. J. Stat. Mech. Theory Exp. (2017). https://doi.org/10.1088/1742-5468/aa727c
Zhang, Y., Cheng, Z., Zhang, X., Chen, X., Duan, J., Li, X.: Data assimilation and parameter estimation for a multiscale stochastic system with \(\alpha \)-stable Lévy noise. J. Stat. Mech. Theory Exp. (2017). https://doi.org/10.1088/1742-5468/aa9343
Zhan, F., Liu, S.: Response of electrical activity in an improved neuron model under electromagnetic radiation and noise. Front. Comput. Neurosci. 11, 107 (2017). https://doi.org/10.3389/fncom.2017.00107
Jin, W., Lin, Q., Wang, A., Wang, C.: Computer simulation of noise effects of the neighborhood of stimulus threshold for a mathematical model of homeostatic regulation of sleep-wake cycles. Complexity 2017, 4797545 (2017)
Lv, M., Ma, J.: Multiple modes of electrical activities in a new neuron model under electromagnetic radiation. Neurocomputing 205, 375–381 (2016)
Wang, Y., Ma, J., Xu, Y., et al.: The electrical activity of neurons subject to electromagnetic induction and Gaussian white noise. Int. J. Bifurcat. Chaos 27, 1750030 (2017)
Dur-e-Ahmad, M., Nicola, W., Campbell, S.A., Skinner, F.: Network bursting using experimentally constrained single compartment CA3 hippocampal neuron models with adaptation. J. Comput. Neurosci. 33(1), 21–40 (2012)
Nicola, W., Campbell, S.A.: Bifurcations of large networks of two-dimensional integrate and fire neurons. J. Comput. Neurosci. 35(1), 87–108 (2013)
Abbott, L.F., van Vreeswijk, C.: Asynchronous states in networks of pulse-coupled oscillators. Phys. Rev. E 48(2), 1483–1490 (1993)
Janicki, A., Weron, A.: Simulation and Chaotic Behavior of Alpha-Stable Stochastic Processes. Marcel Dekker, New York (1994)
Destexhe, A., Mainen, Z.F., Sejnowski, T.J.: Kinetic models of synaptic transmission. In: Koch, C., Segev, I. (eds.) Methods in Neuronal Modeling: From Synapses to Networks. MIT Press, Cambridge (1998)
Nolan, J. P.: Stable distributions—models for heavy tailed data. Boston: Birkhauser (2018) (In progress, Chapter 1 online at http://fs2.american.edu/jpnolan/www/stable/stable.html)
Hemond, P., Epstein, D., Boley, A., Migliore, M., Ascoli, G., Jaffe, D.: Distinct classes of pyramidal cells exhibit mutually exclusive firing patterns in hippocampal area CA3b. Hippocampus 18(4), 411–424 (2008)
Kuebler, E.S., Thivierge, J.P.: Spiking variability: theory, measures and implementation in Matlab. Quant. Methods Psychol. 24(10), 131–142 (2014)
Reike, F., Warland, D., van Steveninck, R.R., Bialek, W.: Spikes: Exploring the Neural Code. MIT Press, Cambridge (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vinaya, M., Ignatius, R.P. Effect of Lévy noise on the networks of Izhikevich neurons. Nonlinear Dyn 94, 1133–1150 (2018). https://doi.org/10.1007/s11071-018-4414-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-018-4414-8