Abstract
Both epidemiological investigation and animal experiments demonstrated that pre-/postnatal exposure to perfluorooctane sulfonic acid (PFOS) could induce neurodevelopmental disorders. Previous studies showed that astrocyte was involved in PFOS-induced neurotoxicity, while little information is available. In the present study, the role of astrocyte-derived calmodulin-dependent protein kinase II (CaMKII)-phosphorylated discs large homolog 1 (DLG1) signaling in PFOS eliciting cytotoxicity in neuron was explored with primary cultured hippocampal astrocyte and neuron. The application of PFOS showed a decreased cell viability, synapse length and glutamate transporter 1 (GLT-1) expression, but an increased CaMKII, DLG1 and cyclic AMP response element binding protein (CREB) expression in primary cultured astrocyte. With 2-(2-hydroxyethylamino)-6-aminohexylcarbamic acid tert-butyl ester-9-isopropylpurine (CK59), the CaMKII inhibitor, the disturbed cell viability and molecules induced by PFOS could be alleviated (CREB expression was excluded) in astrocytes. The cytotoxic effect of neuron exposed to astrocyte conditional medium collected from PFOS (PFOS-ACM) pretreated with CK59 was also decreased. These results indicated that PFOS mediated GLT-1 expression through astrocyte-derived CaMKII-DLG signaling, which might be associated with injuries on neurons. The present study gave an insight in further exploration of mechanism in PFOS-induced neurotoxicity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Perfluorooctane sulfonic acid (PFOS) is the typical chemical for perfluorinated compounds (PFCs), which had been catalogued as one of the Persistent Organic Pollutants by the World Health Organization (WHO/IPCS, 2002). PFOS has been added into a variety of commercial and industrial uses, such as surfactants, refrigerants, lubricants, adhesives and fire retardants [1]. Because of extensive application, PFOS could be detected in biotic and abiotic system, even in human specimen, such as serum, cord blood and breast milk [2, 3]. Except for occupational exposure, dietary intake is the main way for PFOS exposure to human [4]. The average half-life for PFOS in human is about 5.4 years [5]. van Asselt et al. reported that PFOS elimination rate is low, though almost all PFOS is eliminated through the urinary route [6]. Hence, PFOS posed a risk of health-threating effects on human.
Both epidemiological studies and animal experiments revealed that PFOS presented neurotoxicity. Wang et al. suggested that prenatal exposure to perfluoroalkyl substances was associated with decreased IQ test scores [7]. Long et al. reported that PFOS could impair spatial learning and memory ability in mice after three months’ exposure [8]. Chen et al. also demonstrated that chronic exposure to PFOS could cause obvious neurotoxicity and behavior defects in nematodes [9]. These studies were in accordance with the findings that PFOS could cross the blood–brain barrier and exert the toxic effect on the nerve system through biomolecules in brain [10, 11]. Different mode of actions were proposed to explain the neurotoxicity induced by PFOS, such as thyroid system, calcium homeostasis, protein kinase C, synaptic plasticity and cellular differentiation [12]. However, the mechanism is still unclear.
Astrocyte is involved in clearance of neurotransmitters, release of gliotransmitters, and regulation of ion balance in maintaining synapse [13]. It was reported that astrocyte-derived factors exhibited different roles for the fate of neurons in the stress environment [14]. In context of PFOS, secretion of excessive levels of pro-inflammatory cytokines by astrocyte that initiated injuries in neuron were reported [15, 16]. Previous studies in our lab also found that astrocytes-derived d-serine was involved in PFOS-induced neurotoxicity through the N-methyl-d-aspartate receptors (NMDAR) in the rat primary hippocampal neurons [17]. Excitatory glutamatergic neurotransmission via NMDAR, one subgroup of ligand-gated ionotropic glutamate receptors (iGluRs), is critical for synaptic plasticity and survival of neurons. The concentration of glutamate in the synaptic cleft is subject to strict regulation by glutamate uptake/transporter system under the physiological condition [18]. Hence, disturbance in glutamate uptake/transporter system is neurotoxic to the central nervous system [19]. PFOS could decrease the expression of glutamate transporters, including glutamate transporter 1 (GLT-1) and glutamate aspartate transporter (GLAST) in astrocytes, resulting in the disturbance of glutamate-glutamine system between astrocyte and neuron [20]. However, little is known about the upstream mediator in GLT-1 induced by PFOS.
Multiple mechanisms and pathways were reported to mediate GLT-1 expression in astrocyte [21, 22], calcium overload in astrocyte was also included [23]. Ca2+ influx could activate its sensitive downstream signaling molecular CaMKII, and involved in biological function, such as inflammation, apoptosis, and mitochondrial damage [24, 25]. Therefore, we evaluated the factors of astrocyte-derived CaMKII-discs large homolog 1 (DLG1) signaling in modulating GLT-1 in PFOS-induced toxic effect on neuron. Then the CaMKII inhibitor, 2-(2-hydroxyethylamino)-6-aminohexylcarbamic acid tert-butyl ester-9-isopropylpurine (CK59) was added to explore the associated mechanism. The results showed that PFOS could disturb GLT-1 expression via CaMKII-DLG1 signaling in astrocyte, leading to toxic effect on neuron.
Materials and Methods
Chemicals
PFOS (purity > 98%, CAS: 2795-39-3) was supplied by Sigma in America. Dulbecco’s Modified Eagle Medium (DMEM)/F12 medium was purchased from Gibco (New York, USA). Penicillin, and streptomycin were the products of Hyclone (Beijing, China). Trypsin was purchased from Invitrogen (Grand Island, USA). Antibody for p-CaMKII (Santa, sc-32289) was purchased from Santa Cruz Biotecnology. Antibody for GLT-1 (Protein tech, 22515–1-AP), DLG1 (Protein tech, 55085–1-AP),
Glyceraldehyde-3-phosphate dehydrogenase mouse monoclonal antibody (Protein tech, 60004–1-lg), anti-rabbit HRP-conjugated secondary antibody (Protein tech, SA00001-2) and anti-mouse HRP-conjugated secondary antibody (Protein tech, SA00001-1) were purchased from Protein Tech Biotecnology (Chicago, USA). Cyclic AMP response element binding protein (CREB) rabbit monoclonal antibody (Beyotime, AF1018) was purchased from Beyotime Biotecnology (Shanghai, China). 2-(2-hydroxyethylamino)-6-aminohexylcarbamic acid tert-butyl ester-9-isopropylpurine (CK59; 208922-5MG) was purchased from millipore Biotecnology (Darmstadt, Germany). TransScript® All-in-One First-Strand cDNA Synthesis SuperMix for qPCR (One-Step gDNA Removal) kit and TransStart® Top Green qPCR SuperMix kit were supplied by Transgen (Beijing, China). All the other chemicals were analytical grade, purchased from Solarbio (Beijing, China).
Isolation of Rat Primary Neuron and Astrocytes
The protocol for animal use was in accordance with the Animal Guideline of the Dalian Medical University and has been approved by the Ethics Committee of Dalian Medical University. Timed-pregnant Wistar rats were provided by the Laboratory Animal Research Center (SPF) of the Dalian Medical University. Rat primary hippocampal neurons were isolated and maintained according to the published protocols by Chen et al. [16] with some modifications. Briefly, male pups in postnatal day 0 from Wistar rats were decapitated rapidly, and hippocampi were separated under the dissecting microscope and immediately dissected into small pieces, followed by incubation in a solution of 0.25% trypsin (pH 7.8) in a humidified atmosphere (37 °C, 5% CO2) for 10 min. To terminate the digestion by trypsin, the tissues were added to Dulbecco’s Modified Eagle Medium (DMEM)/F12 medium containing 10% fetal bovine serum (FBS) and passed through a 200 mesh strainer. The cell suspension was then centrifuged for 5 min at 174 g and resuspended in DMEM/F12 medium with 10% FBS, 2% B-27 supplement, and 100 u/ml penicillin–streptomycin stock solutions. The cells were diluted in to 1 × 106 cells/ml, and then distributed to the 35 mm poly-l-lysine-coated 6-well culture plates. After 24 h, the medium was fully replaced with the same volume of the Neurobasal® A medium containing 2% B27 (without FBS). After 48 h, a half of the medium was exchanged with the Neurobasal® A medium containing 2% B27 (without FBS) on alternate days. Cells were collected for experimentation after culturing for 7–10 days.
Primary rat astrocytes were prepared from neonatal male rats (1 to 2 days after birth), as described elsewhere [26]. Briefly, the cortex was dissociated, incubated with 0.125% trypsin at 37 °C for 20 min and transferred into DMEM/F12 with 10% (v/v) FBS and 1% (w/v) penicillin–streptomycin. Then the mixture was centrifuged at 174 g for 3 min and resuspended in DMEM/F12 supplemented with 10% FBS. The cell suspension was passed through stainless steel (200 meshes, Solarbio, Beijing). After differential attachment for 30 min to remove fibroblasts, cells were plated in the culture dishes precoated with poly-L-lysine at 37 °C in a 5% CO2 and 100% humidified atmosphere. The culture media were refreshed every three days. On days 10–11, the confluent cultures were shaken overnight to minimize glia contamination. The purity of astrocytes isolated from the rat brain is > 95%, as determined by activated glial fibrillary acidic protein [27].
Astrocyte Conditional Media (ACM) Preparation and Treatment
To evaluate the effect of ACM (with or without PFOS) on neuron, ACM was collected. According to the reference [28], astrocyte cultures were thoroughly washed to eliminate residual serum after reaching confluence. The cultures were maintained in DMEM/F12 with 1% (w/v) penicillin–streptomycin without fetal bovine serum for 1 day. Then, the medium was collected and centrifuged (3000 × g for 15 min) to remove cellular debris. The mudium was used as astrocyte conditioned medium (ACM). To analyze the effect of ACM on neuronal survival, neuronal cultures were maintained in ACM with presence or absence of PFOS.
Cell Viability
The 3-(4, 5-Dimethyl-thiazol-2-yl)-2, 5-diphenyl-tetrazolium bromide (MTT) assay is based on the enzymatic conversion of MTT in mitochondria. Briefly, cells were plated in 96-well culture plates at a density of 5 × 104 cells/cm2 for astrocytes in 100 μL medium. After treatment with PFOS for 12, 18 or 24 h at concentrations range from 0 to 100 μM, MTT with 100 μL (0.5 mg/mL) was added to each well. Following incubation at 5% CO2 and 37 °C for 2 h, and 100 μL dimethyl sulfoxide (DMSO) was added to dissolve the formazan blue formed by mitochondria reducing in the cell. The cell viability was determined by measuring the absorbency of the DMSO-dissolved solution at 595 nm with ELISA Reader (DG-I, China).
Western Blot Analysis
After PFOS exposure (15, 25, 50, 75, 100 μM) [20], equal amounts of proteins, 40 μg from each sample, were loaded and separated by 10% SDS-PAGE and transferred to a PVDF membrane. Then the membrane was incubated for 2 h with 5% skim milk to block non-specific binding. After blocking, membranes were incubated with primary antibody that diluted in 0.1% (V/V) Tween 20 (TBST) (mouse monoclonal anti-CaMKII antibody, 1:1500; rabbit polyclonal anti-DLG-1, 1:1500; rabbit polyclonal anti-GLT-1, 1:3000; mouse monoclonal GAPDH, 1:1500) overnight at 4 °C. The membranes were washed three times, and incubated with second antibodies (anti-rabbit IgG conjugated with goat-horseradish peroxidase that diluted in 0.1% (V/V) Tween 20 (TBST) for CaMKII, 1:1500; anti-rabbit conjugated to horseradish peroxidase at a dilution of 1:1500 for DLG-1; anti-rabbit conjugated to horseradish peroxidase at a dilution of 1:3000 for GLT-1; anti-mouse IgG conjugated with mouse-horseradish peroxidase diluted in 0.1% (V/V) Tween 20 (TBST) for GAPDH, 1:1500). Bands were visualized using ECL detection reagents and values were normalized to GAPDH.
RNA Extraction, Reverse Transcription and Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
Total RNA was isolated from the astrocytes using the TransZolTM Up (Transgen Biotech, Beijing). Eight hundred nanogram of each sample was converted to cDNA in 20 μl reaction volume using the kit of TransScript® All-in-One First-Strand cDNA Synthesis SuperMix for qPCR (One-Step gDNA Removal) (Transgen Biotech, Beijing) according to the manufacturer’s instruction. Briefly, 5 μl TransScript All-in-One First-Strand cDNA Synthesis SuperMix with 14 μl RNase-free water and 1 μl RNA was added into the reaction. The reaction was started by incubating at 42 °C for 15 min and then stopped by heating at 85 °C for 5 s. qRT-PCR was performed using the TransStart® Top Green qPCR SuperMix kit (Transgen Biotech, Beijing). Primers were synthesized in TakaRa (TakaRa Bio, Dalian) for GAPDH, GLT-1b as shown in Table 1. The PCR procedures consisted of an initial denaturation at 95 °C for 30 s, followed by 40 cycles at 95 °C for 5 s and at 60 °C for 30 s with Thermal Cycler Dice® Real Time System (TaKaRa Code: TP800). At the end of the cycles, melting temperatures of the PCR products were determined to be between 57 and 95 °C. GAPDH mRNA was used as an internal control to measure the relative quantitation of the expression of the target genes [29]. Data were analyzed by comparative threshold cycle method (2-ΔΔCT). Then, the relative expressions of genes were normalized against the expression of GAPDH.
Immunofluorescence
The experiment was applied according to the protocols [30, 31]. After PFOS-ACM treatment, the cells were fixed with 4% (W/V) paraformaldehyde in PBS, permeabilized with 0.1% Triton X-100 in PBS for 5 min, and then blocked with 5% BSA. After incubated with primary antibody (microtubule associated protein-2 (MAP-2), 17490–1-AP; NeuN, D3S3I), the cells were washed three times by PBS and incubated with fluorescein-conjugated secondary antibodies. Finally, the cells were counterstained with 4’, 6-diamidino-2-phenylindole (DAPI) to visualize the nuclear morphology. Confocal images were captured using the Olympus upright fluorescence microscope (Olympus Corporation, Japan). After image acquisition, analysis was performed by manual tracing of synaptic length on MAP-2 immunostained images. The synaptic length was expressed as length of MAP-2 positive cells minus length of DAPI positive cells. The number of surviving neuronal cells in NeuN immunostained images were counted using Image J software (1.4.3.67). The data were obtained from 3 independent experiments for each group. The data are expressed as the average cell number and synaptic length in µm.
Statistical Analysis
All results were expressed as mean ± standard error, and statistical analysis was performed by one-way analysis of variance (ANOVA), followed by LSD and Dunnett’s T3 test using SPSS 13.0 (SPSS Inc, Chicago, IL). Differences were considered statistically significant at p < 0.05.
Results
PFOS Exerts Toxic Effect on Neurons Through Astrocyte
To observe the role of astrocyte in PFOS-induced toxic effect on neuron, we first evaluated the effect of PFOS exposure on astrocytes. The viability of astrocyte was explored by MTT after PFOS exposure for 12, 18 and 24 h. As shown in Fig. 1A, PFOS with 75 or 100 μM decreased the viability of astrocytes significantly (p < 0.05) after 12 or 18 h exposure, compared with that of respective control. The MTT result for PFOS exposure on astrocyte for 24 h could be found in our previous study [20]. Lower concentrations of PFOS (15, 25 and 50 μM) did not affect the viability of astrocytes, the concentrations at 75 and 100 μM significantly decreased the viability of astrocytes, about 71% and 47% of the control, respectively (p < 0.05) [20]. Changed lactate dehydrogenase (LDH) and morphology could also be found in astrocyte exposed to PFOS [20]. Together with the results in our present study, it showed that PFOS had a toxic effect on astrocytes.
Cytotoxic effect of astrocytes or neurons. A The cell viability was decreased in the rat primary astrocytes exposed to PFOS for 12 or 18 h. B The cellular morphology of neurons exposed to PFOS-ACM (10×). C The surving neurons exposed to PFOS-ACM (40×). D Quantification of surving neurons exposed to PFOS-ACM. E The changes in synapse length of neurons exposed to PFOS-ACM (40×). F Quantification of synapse length in neurons exposed to PFOS-ACM. Each bar represented mean values ± SEM of at least three independent biological replicates. *p < 0.05, compared with the control group in 12 h; # p < 0.05, compared with the control group in 18 h; ^p < 0.05, compared with the control group in 24 h; ^^p < 0.01, compared with the control group in 24 h; ^^^p < 0.001, compared with the control group in 24 h
In response to injuries, astrocyte-derived molecules triggered the status of neurons during development and adulthood [32, 33]. Hence, the ACM induced by PFOS was collected to investigate the effect of PFOS-ACM on neurons. Consistent with the data reported by our previous study [17], the cellular morphology in Fig. 1B showed that injuries in neuron could be induced by PFOS-ACM directly. Immunofluorescence was also performed to observe the changes in synapse length (Fig. 1E). For neurons in 0-ACM, the soma was obvious. Though the cells had small length of neurite, axon growths were obvious. In PFOS-ACM group, length of neurite was smaller with the concentration of PFOS-ACM increased. Some neurite disappeared (Fig. 1C, D), and the neurite outgrowth was decreased (Fig. 1E, F) in PFOS-ACM groups. The data in immunofluorescence also showed that 25, 50 or 75 μM PFOS-ACM could decreased the neurite outgrowth significantly (p < 0.05 or p < 0.01 or p < 0.001) (Fig. 1E). Our results suggested that PFOS exerted toxic effect on neurons through astrocytes mediation.
PFOS Induced Changes of GLT-1b, CaMKII, DLG1 and CREB in Astrocyte
Glutamate homeostasis could be disturbed in astrocyte under PFOS exposure [20], and was involved in PFOS-induced adverse neurological outcomes [34]. More than 90% of total glutamate uptake is dependent on GLT-1 [35]. In the present study, the changes in the expression of GLT-1b gene and protein were evaluated in primary cultured astrocytes exposed to PFOS for 24 h. As shown in Fig. 2A, PFOS with 50, 75 or 100 μM significantly decreased the GLT-1b gene expression (p < 0.05) in a dose dependent way, about 63%, 33% and 26% of the control, respectively. Accordingly, the GLT-1 protein expression (Fig. 2B) was also significantly decreased in astrocytes exposed to PFOS with 50, 75 or 100 μM, about 75%, 45% or 19% of the control, respectively (p < 0.05). The results of gene and protein expression of GLT-1 in astrocyte membrane induced by PFOS indicated a lost in clearing extracellular accumulated glutamate.
Effect of PFOS on GLT-1 and related proteins in astrocytes. A GLT-1b gene expression. B GLT-1 protein expression. C CaMKII protein expression. D DLG1 protein expression. E CREB protein expression. Each bar represented mean values ± SEM of at least three independent biological replicates. *p < 0.05, compared with the control group; **p < 0.01, compared with the control group
CaMKII-DLG1 signaling was involved in mediating GLT-1b protein expression of astrocyte [23]. Therefore, we investigated the impact of PFOS exposure on CaMKII and DLG1. The astrocytes exposed to PFOS (25, 50, 75 or 100 μM) for 24 h disclosed a significant dose-dependent increase in the expression of CaMKII, about 26%, 25%, 51% or 74% increase than the control (p < 0.01) (Fig. 2C). In Fig. 2D, the astrocytes exposed to PFOS (15, 25, 50, 75 or 100 μM) disclosed a significant dose-dependent increase in the expression of p-DLG1 protein (p < 0.01), about 123%, 127%, 246%, 242% and 339% increase over the control, respectively. Hence, disturbed GLT-1b protein expression in membrane of astrocyte might be associated with changes in CaMKII-DLG1 signaling induced by PFOS. It was reported that more than 95% of extracellular glutamate was regulated by GLT-1 and GLAST [36]. Li et al. showed that PFOS induced an increase in extracellular level of glutamate at a dose-dependent manner [20]. In this context, the function expression levels of GLT-1 protein alteration mediated by CaMKII-DLG1 signaling in astrocyte exposed to PFOS might lose the uptake of glutamate, leading to injuries in neuron.
In addition, CaMKII activation could lead to the phosphorylation and subsequent activation of the transcription factor cyclic AMP response element binding protein (CREB) in the nucleus [37]. Hence, CREB was also evaluated in astrocyte in the present study. The results in Fig. 2E showed that CREB protein was increased in a dose-dependent manner in astrocytes exposed to PFOS (25, 50, 75 or 100 μM). And changed CREB indicated that CREB might be also involved in disordered GLT-1 gene expression in CaMKII-DLG1 signaling in astrocyte exposed to PFOS.
CK59 Improved the Injuries in Astrocyte and Neuron
In order to explore the role of astrocyte-derived CaMKII-DLG1 signaling in PFOS exerting injuries on neuron, CK59 was applied in the present study. CK59 is a specific inhibitor for CaMKII activation [38]. According to the reference, astrocyte was pretreated with 100 μM CK59 for 30 min. Then, CK59 was removed before PFOS was added.
Compared with PFOS exposure alone, CK59 with 100 μM pretreatment could alleviate the cell loss in astrocytes exposed to 75 or 100 µM PFOS significantly (p < 0.05) (Fig. 3A). The effect of PFOS-ACM on the viability of neuron could be found in Fig. 3B. Compared with 0-ACM exposure group, 75-ACM exposure group decreased the viability of neuron significantly (p < 0.05), about decrease by 17% (Fig. 3C). Compared with the 75-ACM exposure group, CK59 with 100 µM pretreatment increased the cell viability of neuron significantly (p < 0.05), about increase by 28% (Fig. 3C).
Role of CK59 in cytotoxic effect of astrocytes or neurons. A The decreased cell viability induced by PFOS for 24 h was alleviated in the rat primary astrocytes by CK59. B The cellular morphology of neurons exposed to PFOS-ACM for 24 h pretreated with CK59. C The cell viability of neurons exposed to PFOS-ACM for 24 h pretreated with CK59. *p < 0.05, compared with the control group (CK59 + 0 μM PFOS); #p < 0.05, compared with the control group (0 μM PFOS); ¥p < 0.05, compared with PFOS group
CK59 Improved the Expression of GLT-1, CaMKII, DLG1 and CREB in Astrocyte
The expression of GLT-1, CaMKII and DLG1 in astrocyte pretreated with CK59 was also analyzed in the present study. As shown in Fig. 4, GLT-1b gene and protein expression in CK59 pretreated astrocytes were improved significantly. The results in gene expression showed that CK59 pretreatment exerted a 100% or 120% increase of GLT-1b gene expression in the astrocytes exposed to PFOS alone (p < 0.01) (Fig. 4A). Furthermore, quantitative analysis for protein showed an 86% or 172% increase of GLT-1 protein expression in astrocytes pretreated with CK59, compared with the group exposed to PFOS alone (p < 0.01) (Fig. 4B). Compared with the group exposed to 75 or 100 μM PFOS alone, the results revealed a 17% or 12% decrease of CaMKII expression in the primary astrocytes pretreatment with CK59 (p < 0.01) (Fig. 4C). Compared with the group exposed to 75 or 100 μM PFOS alone, the results in Fig. 4D revealed a 66% or 70% decrease of DLG1 expression in the rat primary astrocytes pretreatment with CK59 (p < 0.01). Figure 4E showed that CK59 with 100 μM didn’t alleviate CREB protein increase induced by PFOS in astrocyte. Together with the results, the improved expression of mentioned proteins in astrocyte pretreated with CK59 indicated that CaMKII-DLG signaling was involved in GLT-1 changes induced by PFOS.
Role of CK59 in effect of PFOS on GLT-1 and related proteins in astrocytes. A GLT-1b gene expression. B GLT-1 protein expression. C CaMKII protein expression. D DLG1 protein expression. E CREB protein expression. Each bar represented mean values ± SEM of at least three independent biological replicates. *p < 0.05, compared with the control group; **p < 0.01, compared with the control group; ##p < 0.05, compared with 75 μM PFOS group; ##p < 0.01, compared with 75 μM PFOS group; ¥¥p < 0.01, compared with 100 μM PFOS group
Discussion
Previous studies showed that astrocyte was a potential target for PFOS-induced neurotoxicity [15, 16]. Astrocyte could be activated by PFOS, then it exerted injuries in neuron through the cytokines release [39]. In the present study, we observed that astrocyte-derived CaMKII-DLG1 signaling mediating GLT-1 expression took part in PFOS-induced injuries in neuron.
Consistent with the decreased cell viability in astrocytes exposed to PFOS [20, 40], the present study also showed that PFOS exhibited a cytotoxic effect on astrocytes after 12, 18 h exposure. Previous study in our lab and others’ reported that death of astrocyte could be found after PFOS exposure (≥ 75 μM) for 24 h [20, 41]. According to the present study, the injury in astrocyte induced by PFOS might be earlier than 24 h. It has been revealed that some glia-derived factors mediated the effect of glia on synaptic plasticity, such as glutamate [42], tumor necrosis factor alpha (TNF-α) [43] and ATP [44]. We have previously shown that the time peak for extracellular glutamate of astrocyte exposed to PFOS was 24 h, resulting in decreased microtubule associated protein-2 (MAP-2) protein expression in co-cultured neuron [20]. Decreased synapse length in PFOS-ACM (50 μM) group also supported that the injuries in astrocyte exposed to PFOS affected neurons in the present study. Hence, the injuries in astrocyte might affect the pre- and post-synaptic neuronal components in synapse, though the significant death of astrocyte affected by PFOS (≥ 75 μM) was not observed. The concentration in the present study was designed on the report by Dong et al. [40]. According to the results from Austin et al. [45], the dose was extrapolated to 0–53822 ng/mL. The dose was in the range of that might be found in the serum of occupational workers. Fu et al. found that level of PFOS could be up to 118,000 ng/ml (236 μM) in serum from occupational workers [46]. Thus, the changes in molecular observed in our study may have implications for the possible dysfunction in occupational people.
Previous study in our lab showed that both glutamate outside the astrocytes and calcium inside the astrocytes was increased under the similar condition of PFOS exposure [20]. Whether calcium inside the astrocyte took part in mediating glutamate outside the astrocytes was unkonwn. As we know, excitatory amino acid transporters (EAATs) maintain the physiologic quantity of glutamate on membrane of astrocytes. Of these EAATs, EAAT2 (GLT-1) is the main regulator for the majority of glutamate uptake by astrocytes from the synaptic cleft. Modulation of GLT-1 in glutamate spillover could also be induced in excitotoxic mechanisms at astrocyte level [36, 47]. The dysfunction in glutamate transporter might occur at both RNA and protein levels, and produce excessive levels of extracellular glutamate [48]. Both gene and protein analysis in the present study demonstrated that PFOS (≥ 50 μM) decreased the expression of GLT-1. Among indicators observed in exogenous compounds exerting excitotoxicity via glutamate, GLT-1 appeared to be more sensitive [49,50,51]. The results were in agreement with our previous study, which showed that disturbed glutamate transporters resulted in overload glutamate and then lead to excitotoxicity observed in neurons [20].
Generally speaking, certain proteins that regulate cellular and membrane trafficking can target and activate glutamate transporters. Among these proteins, glutamate transporter-associated protein (GTRAP) 3–18 (EAAC1 interactor), GTRAP 41 or GTRAP 48 (EAAT4 interactor), and DLG1 (GLT-1 interactor) have been identified [52,53,54]. GLT-1 is the most abundant and predominant glutamate transporters in astrocytes. Hence, DLG1, the upstream protein associated GLT-1 was evaluated in the present study. DLG1 is not only included in postsynaptic markers, but also in scaffolding proteins. It has been strongly implicated in the trafficking and localization of both NMDA and AMPA-type glutamate receptors [55]. In the present study, DLG1 protein was increased in the rat primary astrocytes exposed to PFOS (≥ 15 μM). The results illuminated the findings observed by Underhill et al. that shRNA knockdown of DLG1 expression decreased surface GLT-1 in cultured astrocytes [53]. Underhill et al. also disclosed the regulation of two glutamate transporter GLT-1 by DLG1 and CaMKII [53]. CaMKII signaling is important in synaptic plasticity and memory formation, which could be activated by calcium. Our earlier studies and others’ showed that PFOS induced increased CaMKII protein expression in hippocampus of rats exposed to PFOS in vivo and in vitro [56,57,58]. Consistently, the current study demonstrated that not only DLG1, but also the expression of CaMKII protein could be increased in model built with the rat primary astrocytes exposed to PFOS (≥ 25 μM). The results indicated the possibility of CaMKII signaling in PFOS-mediating GLT-1 via DLG1 in astrocytes. Hence, the inhibitor of CaMKII (CK59) was added for further exploration. It was interested to find that CK59 could alleviate the cell loss in 75 and 100 µM PFOS-treated astrocytes. And after PFOS exposure, improved of CaMKII, GLT-1 and DLG1 protein could be observed in the rat primary astrocytes pretreated with CK59. These results demonstrated our hypothesis that PFOS disturbed the expression of GLT-1 via CaMKII-DLG1 signaling in astrocyte. In addition, the detection of PFOS inside the cell indicated that PFOS might interact with the bilayer by incorporation [58]. Under the condition, PFOS might increase calcium concentration by mediating the proteins or the iron channel on the membrane of astrocytes directly or indirectly. Specially, changed GLT-1 gene expression could also be found in astrocyte exposed to PFOS or CK59. The regulation might be attributed to the role of CREB, a downstream molecule of CaMKII in the nucleus, that could regulate gene transcription through binding to CRE [59]. Huang et al. found that mRNA of GLT-1 could be induced by 1, 2-dichloroethane through CaMKII-CREB pathway [60]. However, GLT-1 gene expression on the membrane of astrocytes was inconsistent with CREB expression in astrocyte exposed to PFOS or CK59 in the present study. The results might dependent on the location we observed for GLT-1 gene. In addition, many other signaling cascades were also involved in GLT-1 mRNA regulation [61, 62]. Hence, the changed GLT-1 gene expression on membrane was mediated paryly by CaMKII-CREB pathway, mainly through astrocyte-derived CaMKII-DLG1 signaling. However, further study is need to explore the role of CREB in regulation GLT-1 expression in astrocyte exposed to PFOS.
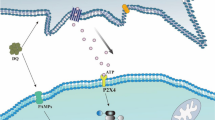
In summary, PFOS could decrease GLT-1 expression via DLG1 mediated by CaMKII in the primary cultured astrocytes, which had injuries in neuron, and then induce neuronal damages. PFOS might interact with the bilayer in astrocyte, and increase the calcium. Then, calcium activated CaMKII, caused cascade in phosphorylating DLG1, and dissociated GLT-1b from DLG1 in astrocytes exposed to PFOS. The instability of GLT-1 in cell surface localization would lead to glutamate uptake loss [20], overload in synaptic cleft, and excitotoxicity in subsequent of PFOS exposure (Fig. 5). Hence, CaMKII-DLG1 might, at least in partially, be the mechanism of PFOS mediating GLT-1 in neuronal damages. Our findings would give insight to the potential therapeutic target for PFOS induced neuronal damages.
Possible role of CaMKII-DLG1 signaling in astrocyte exposed to PFOS: PFOS decreased GLT-1 expression via DLG1 mediated by CaMKII in the rat primary astrocytes (lost the ability of glutamate uptake [20]), then exerted injuries on neurons.
Data Availability
If be asked, we would apply.
References
Ge JB, Wang C, Nie XK, Yang JB, Lu HJ, Song XJ, Su K, Li T, Han JL, Zhang Y, Mao JM, Gu YY, Zhao JY, Jiang SY, Wu QY (2016) ROS-mediated apoptosis of HAPI microglia through p53 signaling following PFOS exposure. Environ Toxicol Phar 46:9–16
Salgado R, Lopez-Doval S, Pereiro N, Lafuente A (2016) Perfluorooctane sulfonate (PFOS) exposure could modify the dopaminergic system in several limbic brain regions. Toxicol Lett 240:226–235
Choi GH, Lee DY, Jeong DK, Kuppusamy S, Lee YB, Park BJ, Kim JH (2017) Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) concentrations in the South Korean agricultural environment: a national survey. J Integr Agr 16:1841–1851
Augustsson A, Lennqvist T, Osbeck CMG, Tibblin P, Glynn A, Nguyen MA, Westberg E, Vestergren R (2021) Consumption of freshwater fish: a variable but significant risk factor for PFOS exposure. Environ Res 192:110284
Olsen GW, Burris JM, Ehresman DJ, Froehlich JW, Seacat AM, Butenhoff JL, Zobel LR (2007) Half-life of serum elimination of perfluorooctanesulfonate, perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ Health Persp 115:1298–1305
van Asselt ED, Kowalczyk J, van Eijkeren JCH, Zeilmaker MJ, Ehlers S, Furst P, Lahrssen-Wiederholt M, van der Fels-Klerx HJ (2013) Transfer of perfluorooctane sulfonic acid (PFOS) from contaminated feed to dairy milk. Food Chem 141:1489–1495
Wang Y, Rogan WJ, Chen HY, Chen PC, Su PH, Chen HY, Wang SL (2015) Prenatal exposure to perfluroalkyl substances and children’s IQ: The Taiwan maternal and infant cohort study. Int J Hyg Envir Heal 218:639–644
Long Y, Wang YB, Ji GX, Yan LF, Hu F, Gu AH (2013) Neurotoxicity of perfluorooctane sulfonate to hippocampal cells in adult mice. PLoS ONE 8:e54176
Chen N, Li J, Li D, Yang YS, He DF (2014) Chronic exposure to perfluorooctane sulfonate induces behavior defects and neurotoxicity through oxidative damages, in vivo and in vitro. PLoS ONE 9:e113453
Wang X, Li B, Zhao WD, Liu YJ, Shang DS, Fang WG, Chen YH (2011) Perfluorooctane sulfonate triggers tight junction “opening” in brain endothelial cells via phosphatidylinositol 3-kinase. Biochem Bioph Res Co 410:258–263
Greaves AK, Letcher RJ, Sonne C, Dietz R (2013) Brain region distribution and patterns of bioaccumulative perfluoroalkyl carboxylates and sulfonates in east greenland polar bears (Ursus maritimus). Environ Toxicol Chem 32:713–722
Mariussen E (2012) Neurotoxic effects of perfluoroalkylated compounds: mechanisms of action and environmental relevance. Arch Toxicol 86:1349–1367
Liu X, Ying J, Wang X, Zheng Q, Zhao T, Yoon S, Yu W, Yang D, Fang Y, Hua F (2021) Astrocytes in neural circuits: key factors in synaptic regulation and potential targets for neurodevelopmental disorders. Front Mol Neurosci 14:729273
Kwon HS, Koh SH (2020) Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener 9:42
Chen X, Nie X, Mao J, Zhang Y, Yin K, Sun P, Luo J, Liu Y, Jiang S, Sun L (2018) Perfluorooctane sulfonate mediates secretion of IL-1beta through PI3K/AKT NF-small ka, CyrillicB pathway in astrocytes. Neurotoxicol Teratol 67:65–75
Chen X, Nie X, Mao J, Zhang Y, Yin K, Jiang S (2018) Per fl uorooctanesulfonate induces neuroinflammation through the secretion of TNF-alpha mediated by the JAK2/STAT3 pathway. Neurotoxicology 66:32–42
Wang R, Wang R, Niu X, Cheng Y, Shang X, Li Y, Li S, Liu X, Shao J (2019) Role of astrocytes-derived d-serine in PFOS-induced neurotoxicity through NMDARs in the rat primary hippocampal neurons. Toxicology 422:14–24
Wang R, Reddy PH (2017) Role of Glutamate and NMDA Receptors in Alzheimer’s Disease. J Alzheimers Dis 57:1041–1048
Lau A, Tymianski M (2010) Glutamate receptors, neurotoxicity and neurodegeneration. Pflugers Arch 460:525–542
Li Z, Liu Q, Liu C, Li C, Li Y, Li S, Liu X, Shao J (2017) Evaluation of PFOS-mediated neurotoxicity in rat primary neurons and astrocytes cultured separately or in co-culture. Toxicol In Vitro 38:77–90
Lee E, Sidoryk-Wegrzynowicz M, Wang N, Webb A, Son DS, Lee K, Aschner M (2012) GPR30 regulates glutamate transporter GLT-1 expression in rat primary astrocytes. J Biol Chem 287:26817–26828
Zhang B, Su D, Song Y, Li H, Chen C, Liao L, Zhang H, Luo J, Yang M, Zhu G, Ai Z (2023) Yueju volatile oil plays an integral role in the antidepressant effect by up-regulating ERK/AKT-mediated GLT-1 expression to clear glutamate. Fitoterapia 169:105583
Ibanez I, Bartolome-Martin D, Piniella D, Gimenez C, Zafra F (2019) Activity dependent internalization of the glutamate transporter GLT-1 requires calcium entry through the NCX sodium/calcium exchanger. Neurochem Int 123:125–132
Shah D, Gsell W, Wahis J, Luckett ES, Jamoulle T, Vermaercke B, Preman P, Moechars D, Hendrickx V, Jaspers T, Craessaerts K, Horre K, Wolfs L, Fiers M, Holt M, Thal DR, Callaerts-Vegh Z, D’Hooge R, Vandenberghe R, Himmelreich U, Bonin V, De Strooper B (2022) Astrocyte calcium dysfunction causes early network hyperactivity in Alzheimer’s disease. Cell Rep 40:111280
Li GZ, Hu YH, Lu YN, Yang QY, Fu D, Chen F, Li YM (2023) CaMKII and Ca(V)3.2 T-type calcium channel mediate connexin-43-dependent inflammation by activating astrocytes in vincristine-induced neuropathic pain. Cell Biol Toxicol 39:679–702
Lee ESY, Yin ZB, Milatovic D, Jiang HY, Aschner M (2009) Estrogen and tamoxifen protect against mn-induced toxicity in rat cortical primary cultures of neurons and astrocytes. Toxicol Sci 110:156–167
Cho WS, Messing A (2009) Properties of astrocytes cultured from GFAP over-expressing and GFAP mutant mice. Exp Cell Res 315:1260–1272
Buosi AS, Matias I, Araujo APB, Batista C, Gomes FCA (2018) Heterogeneity in synaptogenic profile of astrocytes from different brain regions. Mol Neurobiol 55:751–762
Yang QL, Wang WS, Liu C, Wang Y, Sun K (2016) Effect of PFOS on glucocorticoid-induced changes in human decidual stromal cells in the first trimester of pregnancy. Reprod Toxicol 63:142–150
Popova D, Jacobsson SOP (2014) A fluorescence microplate screen assay for the detection of neurite outgrowth and neurotoxicity using an antibody against βIII-tubulin. Toxicol in Vitro 28:411–418
Sultan N, Amin LE, Zaher AR, Grawish ME, Scheven B (2020) Neurotrophic effects of dental pulp stem cells on trigeminal neuronal cells. Sci Rep-Uk. https://doi.org/10.1038/s41598-020-76684-0
Tang J, Bair M, Descalzi G (2021) Reactive astrocytes: critical players in the development of chronic pain. Front Psychiatry 12:682056
Shan L, Zhang T, Fan K, Cai W, Liu H (2021) Astrocyte-neuron signaling in synaptogenesis. Front Cell Dev Biol 9:680301
Brown-Leung JM, Cannon JR (2022) Neurotransmission targets of per- and polyfluoroalkyl substance neurotoxicity: mechanisms and potential implications for adverse neurological Outcomes. Chem Res Toxicol 35:1312–1333
Bajrektarevic D, Nistri A (2017) Ceftriaxone-mediated upregulation of the glutamate transporter GLT-1 contrasts neurotoxicity evoked by kainate in rat organotypic spinal cord cultures. Neurotoxicology 60:34–41
Castañeda-Cabral JL, López-Ortega JG, Fajardo-Fregoso BF, Beas-Zárate C, Ureña-Guerrero ME (2020) Glutamate induced neonatal excitotoxicity modifies the expression level of EAAT1 (GLAST) and EAAT2 (GLT-1) proteins in various brain regions of the adult rat. Neurosci Lett 735:135237
Paschou M, Liaropoulou D, Kalaitzaki V, Efthimiopoulos S, Papazafiri P (2023) Knockdown of amyloid precursor protein increases ion channel expression and alters Ca signaling pathways. Int J Mol Sci 24:2302
Wei Y, Han CS, Wang YJ, Wu BB, Su T, Liu Y, He RQ (2015) Ribosylation triggering Alzheimer’s disease-like Tau hyperphosphorylation via activation of CaMKII. Aging Cell 14:754–763
Zheng Y, Ding W, Zhang T, Zhao Z, Wang R, Li Z, Yu S, Li J, Zhao X, Wu Q (2021) Antimony-induced astrocyte activation via mitogen-activated protein kinase activation-dependent CREB phosphorylation. Toxicol Lett 352:9–16
Dong L, Yang XY, Gu W, Zhao KF, Ge HZ, Zhou JJ, Bai XT (2015) Connexin 43 mediates PFOS-induced apoptosis in astrocytes. Chemosphere 132:8–16
Alharthy SA, Hardej D (2021) The role of transcription factor Nrf2 in the toxicity of perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) in C57BL/6 mouse astrocytes. Environ Toxicol Pharmacol 86:103652
Parpura V, Haydon PG (2000) Physiological astrocytic calcium levels stimulate glutamate release to modulate adjacent neurons. Proc Natl Acad Sci USA 97:8629–8634
Beattie EC, Stellwagen D, Morishita W, Bresnahan JC, Ha BK, Von Zastrow M, Beattie MS, Malenka RC (2002) Control of synaptic strength by glial TNFalpha. Science 295:2282–2285
Koizumi S, Fujishita K, Tsuda M, Shigemoto-Mogami Y, Inoue K (2003) Dynamic inhibition of excitatory synaptic transmission by astrocyte-derived ATP in hippocampal cultures. Proc Natl Acad Sci USA 100:11023–11028
Austin ME, Kasturi BS, Barber M, Kannan K, MohanKumar PS, MohanKumar SMJ (2003) Neuroendocrine effects of perfluorooctane sulfonate in rats. Environ Health Persp 111:1485–1489
Fu JJ, Gao Y, Cui L, Wang T, Liang Y, Qu GB, Yuan B, Wang YW, Zhang AQ, Jiang GB (2016) Occurrence, temporal trends, and half-lives of perfluoroalkyl acids (PFAAs) in occupational workers in China. Sci Rep-Uk. https://doi.org/10.1038/srep38039
Neves D, Salazar IL, Almeida RD, Silva RM (2023) Molecular mechanisms of ischemia and glutamate excitotoxicity. Life Sci 328:121814
Maragakis NJ, Rothstein JD (2004) Glutamate transporters: animal models to neurologic disease. Neurobiol Dis 15:461–473
Zhao FH, Liao YJ, Jin YP, Li GX, Lv XQ, Sun GF (2012) Effects of arsenite on glutamate metabolism in primary cultured astrocytes. Toxicol in Vitro 26:24–31
Feng S, Xu ZF, Liu W, Li YH, Deng Y, Xu B (2014) Preventive effects of dextromethorphan on methylmercury-induced glutamate dyshomeostasis and oxidative damage in rat cerebral cortex. Biol Trace Elem Res 159:332–345
Huo TG, Li WK, Zhang YH, Yuan J, Gao LY, Yuan Y, Yang HL, Jiang H, Sun GF (2015) Excitotoxicity induced by realgar in the rat hippocampus: the involvement of learning memory injury, dysfunction of glutamate metabolism and NMDA receptors. Mol Neurobiol 51:980–994
Struzynska L (2009) A glutamatergic component of lead toxicity in adult brain: the role of astrocytic glutamate transporters. Neurochem Int 55:151–156
Underhill SM, Wheeler DS, Amara SG (2015) Differential regulation of two isoforms of the glial glutamate transporter EAAT2 by DLG1 and CaMKII. J Neurosci 35:5260–5270
Robinson MB, Jackson JG (2016) Astroglial glutamate transporters coordinate excitatory signaling and brain energetics. Neurochem Int 98:56–71
Fourie C, Li D, Montgomery JM (2014) The anchoring protein SAP97 influences the trafficking and localisation of multiple membrane channels. Bba-Biomembranes 1838:589–594
Johansson N, Eriksson P, Viberg H (2009) Neonatal exposure to PFOS and PFOA in mice results in changes in proteins which are important for neuronal growth and synaptogenesis in the developing brain. Toxicol Sci 108:412–418
Liu XH, Liu W, Jin YH, Yu WG, Liu L, Yu HY (2010) Effects of subchronic perfluorooctane sulfonate exposure of rats on calcium-dependent signaling molecules in the brain tissue. Arch Toxicol 84:471–479
Liu XH, Jin YH, Liu W, Wang FQ, Hao S (2011) Possible mechanism of perfluorooctane sulfonate and perfluorooctanoate on the release of calcium ion from calcium stores in primary cultures of rat hippocampal neurons. Toxicol in Vitro 25:1294–1301
Kaldun JC, Sprecher SG (2019) Initiated by CREB: resolving gene regulatory programs in learning and memory switch in cofactors and transcription regulators between memory consolidation and maintenance network. BioEssays 41:e1900045
Huang WY, Wang ZJ, Wang GY, Li KY, Jin YP, Zhao FH (2022) Disturbance of glutamate metabolism and inhibition of CaM-CaMKII-CREB signaling pathway in the hippocampus of mice induced by 1,2-dichloroethane exposure. Environ Pollut 310:119813
Wang Z, Ma W, Chabot JG, Quirion R (2010) Calcitonin gene-related peptide as a regulator of neuronal CaMKII-CREB, microglial p38-NFkappaB and astroglial ERK-Stat1/3 cascades mediating the development of tolerance to morphine-induced analgesia. Pain 151:194–205
Matos M, Shen HY, Augusto E, Wang Y, Wei CJ, Wang YT, Agostinho P, Boison D, Cunha RA, Chen JF (2015) Deletion of adenosine A2A receptors from astrocytes disrupts glutamate homeostasis leading to psychomotor and cognitive impairment: relevance to schizophrenia. Biol Psychiatry 78:763–774
Funding
This work was supported by Liaoning Province Educational Project (LJKMZ20221263), Natural Science Foundation of Liaoning Province (2021-MS-284), the Dalian Tech Stars Program (2016RQ044 to XL) and the China’s Post-doctoral Science Fund (2016M591438 to XL).
Author information
Authors and Affiliations
Contributions
Jiawei Yang, Ying Wang: Methodology, Investigation, Formal analysis. Yuyan Xia, Yajie Ren: Formal analysis, Investigation, Software, Validation. Jing Shao: Formal analysis, Data curation. Xiaohui Liu: Conceptualization, Formal analysis, Writing-review & editing, Validation, Supervision, Funding acquisition, Project administration, Resources.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical Approval
Specific pathogen-free (SPF) grade Wistar rats were provided by animal center of Dalian Medical University (Ethics number No. AEE18060).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, J., Wang, Y., Xia, Y. et al. PFOS Elicits Cytotoxicity in Neuron Through Astrocyte-Derived CaMKII-DLG1 Signaling In Vitro Rat Hippocampal Model. Neurochem Res 49, 1226–1238 (2024). https://doi.org/10.1007/s11064-024-04109-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-024-04109-9