Abstract
The central nervous system (CNS) is the most injury-prone part of the mammalian body. Any acute or chronic, central or peripheral neurological disorder is related to abnormal biochemical and electrical signals in the brain cells. As a result, ion channels and receptors that are abundant in the nervous system and control the electrical and biochemical environment of the CNS play a vital role in neurological disease. The N-methyl-d-aspartate receptor, 2-amino-3-(5-methyl-3-oxo-1,2-oxazol-4-yl) propanoic acid receptor, kainate receptor, acetylcholine receptor, serotonin receptor, α2-adrenoreceptor, and acid-sensing ion channels are among the major channels and receptors known to be key components of pathophysiological events in the CNS. The primary amine agmatine, a neuromodulator synthesized in the brain by decarboxylation of l-arginine, can regulate ion channel cascades and receptors that are related to the major CNS disorders. In our previous studies, we established that agmatine was related to the regulation of cell differentiation, nitric oxide synthesis, and murine brain endothelial cell migration, relief of chronic pain, cerebral edema, and apoptotic cell death in experimental CNS disorders. In this review, we will focus on the pathophysiological aspects of the neurological disorders regulated by these ion channels and receptors, and their interaction with agmatine in CNS injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ion channels and receptors are the macromolecular membrane pores that traffic a series of ions (Na+, K+, Ca2+, Cl−) and chemicals (neurotransmitters, hormones etc.) in or out of the cells and propagate the biochemical and electrical signals. Functional initiation of ion channels requires stimuli such as ligand binding, chemical/mechanical changes, and altered membrane potentials. Therefore, any iatrogenic, autoimmune, toxic, or genetic dysfunction can cause ion channel-related diseases termed channelopathies, which overlap with acute and chronic neurodegenerative disorders such as ischemic stroke, traumatic injury, epilepsy, Alzheimer’s disease (AD), schizophrenia, and Huntington’s disease (HD) [1]. The majority of neurodegenerative diseases are related to cell death caused by disrupted ion channels and receptors or metabolic functions of neuronal cells [2,3,4,5]. In the healthy brain, ionic homeostasis is a major component of neuronal information transmission via action potentials, which are also related to synaptic transmission between presynaptic and postsynaptic neurons. Loss of synaptic function due to low energy supply in neurodegenerative disorders leads to attenuation of ion homeostasis [6, 7]. In the diseased brain, ion homeostasis is maintained by a series of ion channels in brain cells, predominantly in neuron, astrocyte and microglia, and a myriad of ions that are transported across the cell membrane via those channels (Table 1). Among the range of associated ions, potassium, sodium, chloride, and calcium collectively play a pivotal role in cellular damage [1, 8]. Following central nervous system (CNS) injury, the well-orchestrated ion exchange across the membrane via ion channels or receptors is disrupted and brain cells lose their normal function, leading to death.
Agmatine, an arginine-derived primary amino acid found in the nerve cell body and synaptic terminals, acts as a neuromodulator that mimics the functional properties of other neurotransmitters [9, 10]. The enzyme arginine decarboxylase (ADC) synthesizes agmatine by decarboxylation of l-arginine [11]. On the other hand, studies suggested that in the mammal lacking ADC, decarboxylation of l-arginine is catalyzed by the ornithine decarboxylase enzyme. Agmatine-expressing cells have been found in all regions of the brain such as the hypothalamus, frontal cortex, striatum, medulla, hippocampus, and locus coeruleus along with measurable ADC activity [9, 10, 12,13,14,15,16]. However, the highest number of these cells was observed in the paraventricular (PVN) and supraoptic (SON) nuclei of the hypothalamus, which also exhibited the highest ADC activity. Diverse mechanisms of neuroprotection by agmatine in response to neurodegenerative diseases have been reported by many research groups, such as blocking of harmful ion channels, suppressing harmful reactive oxygen species (ROS), promoting neurogenesis, angiogenesis, reducing glial scars etc [17,18,19,20]. In a recent review, Laube and Bernstein preciously reviewed the recent studies about the agmatine metabolism and clinical application including nervous system [21]. In acute brain diseases, both exogenous and endogenous (via overexpression of ADC or down regulation of agmatinase experiments etc.) agmatine were reported to be effective in reducing hypoxic brain tissue damage [17, 20, 22,23,24,25,26]. The exogenous agmatine is also reported to be an anti-proliferative agent which reduces the biosynthesis and increases the degradation of the cell growth and proliferative polyamines such as putrescine, spermidine and spermine in different cancer cell lines [27, 28]. Exogenous agmatine treatment also can inhibit advanced glycation end products formation in diabetic kidney [29]. Over the last few decades, a significant number of studies have been performed regarding the interacting targets of both endogenous and exogenous agmatine in the brain and other systems. Agmatine interacts with the different receptors and ion channels of the brain cells which are responsible for different neurodegenerative diseases. Here, we will discuss the association of the different ion channels and receptors responsible for various neurodegenerative diseases and their possible relationship with agmatine.
Medicinal Chemistry of Agmatine
The primary amine agmatine is produced from the l-arginine by the enzyme ADC. This decarboxylated arginine, agmatine can be found naturally in herring sperm, octopus muscle, ergot fungi, ragweed pollen, and also in mammalian brain [11]. Structurally agmatine has a protonated guanidine group and an amino group. At the physiological pH, agmatine can act as a divalent cation due to these protonated groups. On the other hand, due to the presence of the additional carboxylate group, arginine acts as a net monovalent cation [30]. Agmatine is also suggested to behave as monovalent when it interacts with the microscopic channels and divalent as a macroscopic charge transfer [30].
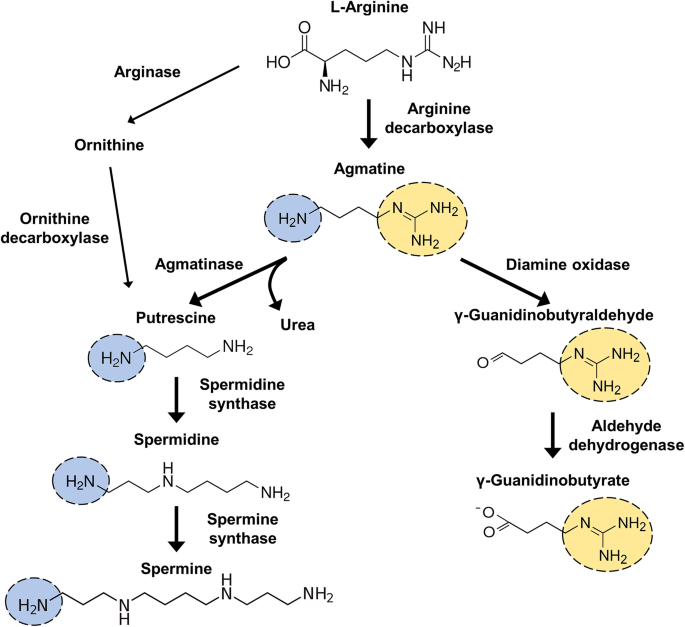
Agmatine can be metabolized into putrescine by the enzyme agmatinase or oxidized into γ-guanidinobutyraldehyde by diamine oxidase (Fig. 1). Agmatinase belongs to the family of hydrolases, those acting on carbon–nitrogen bonds other than peptide bonds, specifically in linear amidines, then synthesizes putrescine, one of polyamines. Putrescine is metabolized further into the polyamines, spermidine and spermine [31]. On the other hand, diamine oxidase deaminates oxidatively diamines to produce aldehydes, ammonia and hydrogen peroxide. Therefore, the amino group of agmatine is metabolized by the action of diamine oxidase to become γ-guanidinobutylbutylic acidaldehyde, and is further metabolized to γ-guanidinobutyric acid by aldehyde dehydrogenase (Fig. 1). Agmatine has two reactive groups, which increased its possibility of various chemical reactions in vivo. With this potentiality for biochemical reactions, agmatine has been used as an experimental and investigational drug in different neurological disorders. However, the precise mechanism of action for its potential clinical indications has not been identified yet.
Synthesis and metabolism of agmatine. Agmatine is synthesized from the l-arginine by arginine decarboxylase (ADC). Agmatine structurally contains two protonated groups, a guanidine group and an amino group, and thus can act as a divalent cation in the physiological pH. The guanidine group of agmatine is metabolized by diamine oxidaze and the amino group is metabolized by agmatinase to γ-guanidinobutyraldehyde and putrescine respectively
Target Ion Receptors/Channels of Agmatine
Electrical and chemical signals in neurons are orchestrated by the neurotransmitters, ion channels, receptors, and the electrochemical gradient. In this manner, neurons can communicate with each other and other cells in the brain, which is essential for normal brain function. In the healthy brain, agmatine is known to be a neuromodulator that regulates multiple neurotransmitters and signaling pathways. It has also been demonstrated to exert neuroprotective effects, which are likely due to the interaction between the membrane receptors/channels and agmatine, in various neuronal pathologies. However, the mechanism underlying the interaction between agmatine and the membrane receptors/channels remains unknown in both the healthy and diseased brain. Therefore, we will discuss the major ion channels and receptors associated with various brain functions and disorders, and their association with agmatine.
N-Methyl-d-Aspartate Receptor (NMDAR)
NMDARs, the glutamate-gated channels permeable to calcium, sodium, and potassium, are crucial for CNS development, cognitive function, locomotion, and breathing. NMDARs are activated by the excitatory neurotransmitter glutamate and are also a key component of many CNS related major acute and chronic pathological conditions such as stroke, TBI, PD, AD, pain, depression, schizophrenia etc. [21, 32, 33]. A typical heteromeric NMDAR consists of four subunits, two NMDAR1 and two NMDAR2 (A–D), and may also contain a less common subunit, NMDAR3 (A, B) [34]. NMDAR3 is expressed in neurons of various different regions of the brain according to the developmental age. Among the seven distinct subunits, NMDAR2 determines the functional heterogeneity of the NMDAR. Each NMDAR subunit has a large extracellular amino-terminal domain, which includes the ligand binding site, connected to four helical transmembrane domains that form the ion channel, and the transmembrane domain connects to the short intracellular carboxyl-terminal domain [34]. NMDAR can be found in both neuronal and glial cells. In a neuron, NMDARs are synaptic or extra-synaptic according to their localization and the majority of the synaptic NMDARs are located post-synaptically. NMDARs are associated with both survival and death mechanisms of neurons in neurodegenerative diseases. In the diseased brain, the extracellular glutamate increase over-activates the NMDARs, resulting in elevated Ca2+ and Na+ influx into the cell which triggers NMDAR-mediated neuronal excitotoxicity (Fig. 2a) [35]. After ischemic injury, NMDAR over-activation also produces NO by activating nitric oxide synthase (NOS), which is a major mediator of neuronal death [36, 37]. As a result, controlling neuronal excitotoxicity and inhibition of NO production via modulation of NMDAR function is a first line treatment choice for ion channel-related brain disorders. The primary amine agmatine has been reported to be a neuroprotective agent by modulating NMDARs, the NO pathway, and oxidative stress in various neurodegenerative diseases [18, 23, 38, 39]. Agmatine treatment in the different neuronal cultures have been demonstrated to be neuroprotective against excitotoxicity by blocking NMDAR and inhibiting the increase in cellular calcium levels (Fig. 2b) [18, 40, 41]. Agmatine inhibits NMDA excitotoxicity-induced cell death, but not by intercellular Ca2+ or protein kinase blockade [41]. Differential studies on synaptic and extrasynaptic NMDARs have suggested that synaptic NMDARs were more neuroprotective, whereas the extrasynaptic NMDARs induced cell death, and increase in neuronal NO [18, 42,43,44]. In various neuropathologies, neuronal death by NO synthesis was also attenuated by the NMDA antagonist agmatine via inhibition of NOS [45]. Previous studies also reported that endothelial NOS was attenuated by agmatine in cerebral ischemia in rats [23, 38]. Although a number of studies have explored the relationship between NMDAR and agmatine, determination of the full molecular interaction between these two could be an important finding in the treatment of various neurological disorders.
Agmatine inhibits NMDAR activation. a at resting stage the pore of the NMDAR is blocked by magnesium ion. b In brain injury excessive glutamate release over-activate the NMDAR and transports the ion in and outside of the cell which causes Ca2+ excitotoxicity. c NMDA antagonist agmatine inhibits ionic transport via NMDAR and attenuates the Ca2+ excitotoxicity
Non-NMDA Receptors
According to the agonist preference, the non-NMDA receptors are divided into two subclasses; α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors (AMPARs) and kainite receptors (KARs). Among all ionotropic and glutamate receptors, AMPARs are known for fast excitatory synaptic transmission and are densely dispersed in the mammalian brain, whereas KARs are generally copious in the least abundant NMDAR pathways. Both AMPAR and KAR are tetrameric and comprise GluA (1–4) and GluK (1–5) subunits, respectively [46, 47]. A high proportion of AMPARs and few KARs are impermeable to Ca2+. However, Ca2+-permeable AMPARs and KARs can regulate neurological disease processes in a similar manner to NMDARs [48, 49]. Ca2+-permeable AMPARs are typically expressed in hippocampal CA1 regional pyramidal neurons, and the increase in the number of AMPARs in the diseased brain indicates that they are of marked importance. A high number of AMPARs can decrease the vulnerability of the ischemic neurons, but a low number of AMPARs increases the vulnerability [50, 51]. Due to the higher excitotoxicity of NMDARs and AMPARs, the role of the KARs in excitotoxic cell death have received less focus. Of the five subunits of the KAR, GluK4 and GluK5 exhibit high agonist-affinity, and GluK4 is co-expressed pre-and post-synaptically with GluK2 in the CA3 region of the hippocampus, whereas the remaining subunits are expressed throughout the CNS [52, 53]. Both AMPARs and KARs follow the same intracellular Ca2+ loading mechanism of cell death during ischemia, but it remains unclear whether Ca2+ overload is the sole reason for cell death. Koh et al. demonstrated that the divalent cation, Zn2+, to which AMPARs are permeable, contributes to neuronal death via mechanisms such as poly ADP ribose polymerase activation, generation of ROS, and enzyme induction [54]. However, the pro-apoptotic c-Jun N-terminal kinase signaling cascade can be activated by over activation of the KAR subunit GluK2 in the ischemic brain [55]. The effect of agmatine on AMPARs and KARs has not been explored to the same extent as that on NMDAR. However, Neis et al. reported that agmatine has potential antidepressant properties via activation of AMPARs [56]. In their study, they found that the pretreatment of the agmatine (0.1 mg/kg oral) reduced the immobility time than the AMPAR antagonist treated mice in tail suspension tests. The pretreatment of the agmatine also increased the synaptic GluA1 and PSD95 protein expression and activated the PI3K/Akt/mTOR pathway. They suggested that the antidepressant effects of agmatine resemble the antidepressant effects of ketamine.
α2-Adrenoreceptor/Imidazoline Receptor
Both the imidazoline receptor and the α2-adrenoreceptor are well-known components of the sympathetic nervous system, and their agonist have been used as a common drug to treat hypertension, pain and panic disorders, addiction, depression, and other behavioral disorders. However, due to difficulties regarding distinction of the functional properties of the α2-adrenoreceptor and the imidazoline receptor, researchers focused on the agents or endogenous ligands that could selectively identify the receptors and bind to them. The α2-adrenoreceptors are a family of G protein-coupled receptors (GPCRs) that can be found in both the CNS and the PNS and are pharmacologically divided into three subclasses: α-2A (CNS, sympatholytic), α-2B (blood vessels, vasodepressor), and α-2C (CNS, sympatholytic) [57]. Structurally, the α2-adrenoreceptor has three extracellular domains (with small amino termini) and three intracellular domains (with large carboxy-termini), and the hydrophobic transmembrane domain consists of seven well-conserved helices (Fig. 3a) [58]. Henderson et al. suggested that these membrane spanning transmembrane domains form a pocket-like structure, which serves as the ligand binding site for the adrenoreceptors [58, 59]. The notion of imidazoline receptors evolved while searching for the anti-hypertensive function of clonidine, an imidazoline compound, which was first thought to be a peripheral α2-adrenoreceptor agonist. However, recent studies suggested that the anti-hypertensive function of clonidine and other imidazole-possessing ligands occurred via interaction with an imidazoline receptor rather than binding to the α2-adrenoreceptor [60]. Several imidazoline-like drugs have been developed, which selectively bind to the imidazoline receptors but have no affinity for α2-adrenoreceptors. The imidazoline receptors have been identified as three distinct classes: I-1, I-2 (I-2A, I-2B), and I3. The I-1 receptors regulate the sympathetic inhibitory functions of the sympathetic nervous system and regulate the systolic and diastolic blood pressure by reducing the peripheral resistance, and have been well-studied. The functions of the I-2 receptors are yet to be determined, but are suggested to be an important therapeutic target for pain and stroke [61, 62]. The I-3 receptors induce insulin secretion from pancreatic β cells. Agmatine was first purified as a clonidine displacing substance (CDS) from the α2-adrenoreceptors and named CDS by Atlas et al. [63]. Others later found that it also displaced imidazoline binding from I-1 and I-2 receptors [12, 64, 65]. Li et al. first reported the notion that the previously identified CDS was agmatine and could be formed in mammalian tissues [12]. Agmatine is considered to have therapeutic potential for treating pain, ischemic injury, and seizures. It has been suggested to regulate morphine-induced analgesia in SCI via a neuronal NOS-dependent mechanism involving the site-specific imidazoline receptors and α2-adrenoreceptors [42, 66, 67]. In mixed I-1/α2 agonists study showed that the anticompulsive-like effects of agmatine in OCD rodents were significantly related to imidazoline binding sites [68]. The anti-depressant, anti-convulsant, and memory retrieval effects of agmatine have been found to be associated with α2-adrenoreceptors in addition to NMDAR and NO [69,70,71]. The imidazoline receptors I-1/I-2 also interact with agmatine and reduce ethanol-induced anxiolysis, opioid withdrawal anxiety, and depression in mammals. Agmatine has greater potential to reduce morphine dependency than do other I-1-specific ligands such as moxonidine and rilmenidine [72,73,74]. Giusepponi et al. reported that agmatine has higher affinity and greater anti-addiction potential in the triple interaction among the imidazoline receptors I-1 and I-2 and α2-adrenoreceptors than in the double interaction of imidazoline receptors I-1 and I-2 only [75].
Acetylcholine Receptor
Acetylcholine receptors (AChRs) are divided into two groups according to their agonist; muscarinic (mAChR) and nicotinic (nAChR). Most AChRs are the associated with the parasympathetic nervous system. mAChRs are members of the α-branch of class A GPCRs and have five subtypes (M1–5) [76]. Although signal transduction via mAChRs is slow, these receptors were reviewed to be key components in several mammalian physiological processes such as smooth muscle contraction, heart rate regulation, glandular secretion, and many CNS functions [77, 78]. All mAChR subtypes are found in the CNS with higher expression in the cortex, hippocampus, and thalamus, but M1–M4 can be found in various tissues throughout the body. The extracellular domain of the mAChR binds with their activator (ACh), the intracellular carboxyl-terminal domain of the M1, M3, and M5 subtypes couples with the Gq-G protein, and that of the M2 and M4 subtypes couples with Gi/Go-G proteins [79]. The diseases and their pathophysiology related to the particular mAChRs are still poorly known due to the absence of specific small-molecule ligands. However, recent studies reviewed that the mAChRs are related to diseases such as AD, PD, Sjögren’s disease, schizophrenia, Chagas’ disease, various smooth muscle disorders such as overactive bladder, and chronic obstructive pulmonary disease [80,81,82]. The ionotropic nAChRs are ligand-gated ion channels found in both the CNS and PNS. Each nAChR is formed by homomeric or heteromeric pentamers from a group of sixteen subunits (α1–α7, α9–10, β1–β4, γ, δ, ε) [83]. nAChRs are also a member of the cys-loop receptor superfamily, which includes 5-hydroxytryptamine (5-HT), γ-aminobutyric acid, and glycine, and shares structural similarities such as two extracellular N- and C-terminal domains, four transmembrane domains, and a large cytoplasmic domain. The quaternary ammonium affinity-labeling test revealed that the quaternary ammonium reagent could only label the α-subunits, which identified these subunits as the primary binding sites for the agonists [84]. nAChRs are expressed in the neuromuscular junction, autonomic ganglia, and synapses of the brain and spinal cord, and can functionally regulate classical neurotransmission (post-synaptic), neurotransmitter release (presynaptic), and second messengers via Ca2+ signaling [83, 85]. The neurotransmitter release function of the nAChRs allows them to participate in mammalian cognitive functions such as learning, memory, and attention [86]. Numerous neurological pathologies, such as AD, PD, epilepsy, schizophrenia, and dysautonomia, are associated with nAChRs [87, 88]. In addition to chronic neurological diseases, the central cholinergic system was reviewed to be associated with increased cortical perfusion in cerebral ischemia via impaired ACh synthesis, and mAChRs in particular are associated with alteration of cerebral blood flow via vasodilation [89]. The neuromodulator agmatine can act on both of the AChR subtypes. However, until now, the efficacy of agmatine on nAChRs was more apparent than that on mAChRs. Loring et al. reported that agmatine can act as both a cation and a neuronal receptor antagonist [90]. Nicotine and other psychoactive drugs act on central nAChRs and mediate conditioned place preference, addiction, depression, and anxiety. Even ethanol and morphine withdrawal syndrome has been reported to be attenuated by agmatine [91,92,93,94,95,96]. Studies suggested that agmatine modulates neuropeptide Y-mediated neurotransmission in the brain to regulate nAChR-related anxiolytic function [97, 98]. Scopolamine-induced learning and memory impairment have been reported to be reversed by exogenous agmatine treatment [99]. Thus, further study of agmatine and AChR function warrants more attention.
Voltage-Dependent Calcium Channel
As a type of voltage-gated ion channel, voltage-dependent calcium channels (VDCCs) are activated by cell membrane depolarization and allow the densely-concentrated extracellular calcium ions to flow into the cell and act as the second messenger of the electrical signals that transduce the membrane potential in various excitable and non-excitable tissues such as cardiac and smooth muscle, neurons, and endocrine tissues. The fundamental activity of VDCCs is to couple the cell surface electrical signals with physiological intracellular processes such as calcium-dependent enzyme and protein modulation, contraction, gene expression regulation, and synaptic transmission. In a review Tsien et al., suggested that VDCCs are divided into two major groups; low-voltage activated (LVA) and high-voltage activated (HVA), according to their activation in response to the membrane potential [100]. Considering the cellular distribution, pharmacology, kinetics, and single channel conductance, the HVAs are classified into L, P/Q, N, and R-types, whereas the LVA has only the T-type transient channel [101, 102]. VDCCs are reviewed to be heteromultimeric and comprise multiple subunits, such as the pore-forming common principal subunit Cavα1 and other ancillary subunits Cavα2δ1–4, Cavβ1–4, and Cavγ1–8, but the LVA channels are devoid of ancillary subunits [103]. Each Cavα1 subunit contains four transmembrane domains of six membrane-spanning helices each, S1–S6. Of these, the positively charged amino acids (lysine/arginine) of S4 regulate the voltage-dependent activation of the VDCCs [104]. Cavα1 forms the selective pore for the ions and comprises the drug or ligand binding site. The large carboxy- and short amino-terminal of the Cavα1 subunit are located intracellularly. The different subtypes of VDCCs are associated with different neurological and non-neurological diseases. Neurological diseases related to the VDCC subtypes and drug development targeting those subtypes have been reported previously, including PD (L, T-types), AD (L-type) epilepsy (T, R-types), pain (L, N, R, T, and N-types), anxiety/dependency (N-types), and febrile seizures (L, T-types) [105,106,107]. Wang et al., suggested that, although the neuromodulator agmatine does not exert any effect on Na2+ and K+ currents, it might have some physiological and pharmacological effects on the Ca2+ current via VDCC blockade in rat hippocampal neurons [108]. They also suggested that the mechanism underlying VDCC blockade involves reversible blocking of the L-type channel and few other subtypes, and is voltage-dependent [108, 109]. Wang et al. suggested that the presence of agmatine in the presynaptic region of the hypothalamic magnocellular neurons of the SON nuclei and PVN nuclei also regulates VDCCs to modulate neurotransmitter release [110]. The N-type Ca2+ channels present in the sympathetic nerve terminals are reported to be inhibited by agmatine treatment, which reduced the intracellular Ca2+ and noradrenaline release by the I-2 receptor and eventually reduced the sympathetic vascular tone [111].
Acid-Sensing Ion Channel
The extracellular acidic environment-sensing channels belong to a proton-gated ion channel family known as epithelial Na+ channel/degenerin (ENaC/DEG) and termed acid-sensing ion channels (ASICs). ASICs and the other ENaC/DEG super family members share the same topological structures, which comprise a properly organized large extracellular domain rich in cysteine, two hydrophobic transmembrane domains (TM1 and TM2), and small intracellular N and C-termini [112, 113]. The extracellular loop, which spans the TM domains, harbors a pocket-like structure known as an “acidic pocket”, and is responsible for the acid-dependent channel gating, desensitization, and response to extracellular regulators [114]. To date, four genes (ACCN1-4) have been found that encode six different types of ASIC subunits. The various ASICs function in a range of extracellular pH levels and can be located in both the CNS and PNS, such as ASIC1a (pH 5.8–6.8, CNS, PNS), ASIC1b (pH 6.1–6.2, PNS), ASIC2a (pH 4.5–4.9 CNS, PNS), ASICb (N/A, CNS, PNS), ASIC3 (pH 6.4–6.8, PNS), and ASIC4 (N/A, CNS, PNS) [115, 116]. Structurally, ASICs are trimers and can be both homomeric and heteromeric, e.g., homomeric ASIC1a and heteromeric ASIC1a/2b. ASICs are selectively permeable to Na+ but low amounts of other cations (Ca2+, K+, H+, and Li+) can also diffuse through ASICs. Pathophysiological events such as Inflammation, ischemia, hematoma, exercise etc. that cause the pH to fall below 7 and induce pain by stimulating nociceptive neurons are suggested to be regulated by ASICs [117]. However, the mechanism related to pain processing via ASICs is yet to be clarified. In addition to nociceptive functions, ASICs are also associated with other acute and chronic neurological diseases such as ischemic stroke, SCI, multiple sclerosis, HD, PD, migraine, glioblastoma, and epilepsy, and also with processes related to synaptic plasticity, learning, and memory [118,119,120]. Therefore, the study of modulators of ASICs has received much attention with regard to the treatment of such neurological diseases. Li et al. reported that agmatine might act as an extracellular non-proton ligand for ASIC3 [121]. They demonstrated that agmatine and its analog, arcaine, can activate both homomeric and heteromeric ASIC3 channels even in neutral pH conditions, and that the mechanism of activation is not via Ca2+ chelation but rather by non-proton ligand-sensitive domain interaction [117, 121].
Serotonin Receptors
Serotonin, also known 5-HT, is the one of the oldest neurotransmitters and receptors and is thought to have appeared approximately 700–800 million years ago in single cell eukaryotes [122]. The serotonin receptors are known to control emotional and psychological events in various natural conditions. In 1957, Gaddum and Picarelli proposed that the 5-HT receptors were of two kinds; “M” receptors, which were likely found in nervous tissue, and “D” receptors, which were likely found in muscle. However, based on the pharmacological properties, the modern classification divides 5-HT receptors into seven classes: 5-HT (1–7). All 5-HTs belong to the seven transmembrane domain-containing GPCR family with the exception of 5-HT3, which is a ligand-gated ion channel. To date, several subtypes of 5-HT1 (A, B, D–F), 5-HT2 (A–C), and 5-HT5 (A and B) have been identified [123]. Structurally, 5-HT receptors, as class A GPCRs, have seven transmembrane α-helices connected by large extracellular amino-termini and short intracellular carboxy-termini. They also have an intra-membrane helix (H8), which is connected via three extracellular/intracellular loops (EL/IL). The binding pocket, which is partially covered by EL2, is located in TM3, 5–7, and EL2 and EL3 [124, 125]. Most of the 5-HT receptors are found in the CNS and regulate animal and human behavioral responses e.g. anxiety, depression, locomotor activity, aggression, and other psychiatric conditions [126]. Agmatine does not exert any direct effect on serotonin or serotonin release in PC12 neurons [127]. However, the antidepressant effect of selective serotonin reuptake inhibitors is suggested to be associated with agmatine-induced imidazoline receptor modulation [74]. Zomkowski et al. demonstrated that agmatine exerts an anti-depressant-like function involving the 5-HT1A/1B and 5-HT2 receptors in mice, as assessed by the forced swim test [128]. The 5-HT3 receptors can be inhibited by agmatine, which is about 4 ± 3%, lowest among all imidazoline drugs [129]. In a recent study, researchers found that the antidepressant effect of agmatine was not mediated through serotonergic mechanism but via glutamatergic mechanism [130]. Agmatine was also reported to exert neuroprotective effects in response to corticosterone-mediated injury, via α2-adrenergic and 5-HT2A receptor-regulated Nrf2 induction [131]. However the mechanism of GPCRs blockage by agmatine are yet to discover (Fig. 3b).
Role of Agmatine in CNS Disorders
The neuroprotective effects of agmatine were first reported by Gilad in 1996 [132]. Since then, these effects have been demonstrated by numerous studies of neurological diseases [18, 26, 132,133,134,135,136]. In most studies agmatine has been administered via intravenous, intraperitoneal or oral route in different dosages (in vitro ranging 10 nM–100 µM and in vivo from 20–100 mg/kg). In the following section, we will discuss the role of agmatine in various CNS disorders.
Ischemic Stroke
Ischemic stroke, via occlusion of the cerebral artery(ies), results in energy depletion and subsequent death of cells in the vascular territory. This condition is a cause of long-term disability and ranks as the third most frequent cause of death following heart disease and cancer; yet, despite the high prevalence, the number of approved therapies remains low [137]. Agmatine has been effective in ameliorating neuropathological damage in in vivo and in vitro models of ischemic stroke. Previous studies have reported that agmatine led to a reduction in the size of ischemic infarctions or the loss of neurons under excitotoxic conditions in various stroke models in rodents [39, 41, 138]. Agmatine (100 µM) also improved the survival rate of neurons and astrocytes in vitro following ischemic and ischemia-like insults [139]. Additionally, Kim et al. reported that agmatine (100 mg/kg i.v.) mitigated the severe ischemia-induced neuronal damage in a cat model, which was used to mimic the clinical situation of hyperacute ischemic stroke [140], and in mice model agmatine (100 mg/kg, i.p.) was able to attenuate brain edema via regulation of aquaporin-1 expression in endothelial cells after experimental stroke [141]. Furthermore, agmatine treatment has been demonstrated to regulate neuroinflammation by decreasing the expression of proinflammatory factors such as nuclear factor kappa B (NF-κB) and matrix metalloproteinases in experimental stroke models [139, 142]. Therefore, the investigation of agmatine has clear clinical implication in terms of potential neuroprotective therapies for ischemic stroke and related conditions.
Traumatic Brain Injury (TBI) and Spinal Cord Injury (SCI)
TBI is a complex injury that occurs when an external force traumatically injures the brain. In the primary process, apoptotic cell death and brain edema occur immediately after the insult. After a period of hours to days, TBI leads to brain edema and hemorrhage, which trigger the inflammatory response. Following TBI, agmatine treatment decreased necrosis, blood–brain barrier disruption, and brain edema by reducing the phosphorylation of mitogen-activated protein kinases, the expression of aquaporins, and the promotion of NF-κB nuclear translocation [143]. Agmatine (50 mg/kg, i.p) was also able to improve the cortical lesion size, neurobehavioral outcome, and neuronal vitality, and reduce apoptosis, gliosis, increased hippocampal levels of glutamate, nitric oxide (NO), lactate-to pyruvate ratio, glycerol levels, and intracranial hypertension induced by TBI [20, 144]. Another condition associated with traumatic damage is spinal cord injury (SCI), which results in permanent disability or loss of movement and sensation below the site of injury, leading to paraplegia (thoracic level injury) or tetraplegia (cervical level injury). SCI causes neuronal and glial cell death, induces glial scar formation, and inhibits axonal regeneration and remyelination. Goracke-Postle et al. reported that agmatine was transported into spinal cord-derived nerve terminals in a concentration- and temperature-dependent manner [145]. Furthermore, agmatine (100 mg/kg, i.p.) administration was able to accelerate the recovery of neurological function and prevent the loss of motoneurons in an SCI rat model of spinal cord ischemia [133]. Park et al. demonstrated that transplantation of human mesenchymal stromal cells transfected with the ADC gene improved locomotor function and the viability of neurons and oligodendrocytes after SCI [146]. Following SCI, agmatine can promote remyelination, increase neuronal viability, and interrupt glial scar formation, which are related to increased bone morphogenetic protein (BMP) 2/7 expression in neurons, oligodendrocytes, and astrocytes. Furthermore, after complete spinal cord transection, agmatine can reduce collagen scar formation and enhance functional recovery associated with decreased tumor growth factor beta-2 and increased BMP-7 expression [19, 147].
Alzheimer’s Disease (AD)
AD is a well-known degenerative brain disease characterized by the formation of amyloid-beta (Aβ)-containing plaques and intraneuronal deposits of neurofibrillary tangles [148]. It is the most common cause of dementia. Agmatine treatment improved the cognitive performance of rodents as assessed by the inhibitory avoidance task and the Morris water maze test [149, 150], and prevented morphine-induced memory impairment in mice as assessed by the step-down inhibitory avoidance test [151]. In addition, agmatine pre-treatment reversed hippocampal extracellular-signal-regulated kinase and protein kinase B inactivation induced by scopolamine, suggesting that this endogenous substance may be a candidate treatment for amnesia [99, 152]. Our recent study demonstrated that agmatine (100 mg/kg i.p.) interrupted hippocampal Aβ accumulation, prevented cognitive decline as assessed by the Morris water maze, and attenuated apoptosis and expression of nuclear factor (erythroid derived 2)-like 2 (Nrf2)-mediated anti-oxidant signaling in a streptozotocin-induced AD rat model [153]. Agmatine (40 mg/kg i.p.) was also capable of protecting against Aβ25-35-induced neuronal toxicity and memory deficits as assessed by behavioral tests, such as the elevated plus maze, open field, memory version of the water maze task, and object recognition memory task [154]. In the mouse brain, agmatine suppressed the accumulation of Aβ and phosphorylated-tau, which may contribute to reduce the cognitive decline in mice subjected to high-fat diet [148].
Parkinson’s Disease (PD)
Agmatine elicited neuroprotective effects in experimental models of PD, which is a chronic progressive disease characterized by the degeneration of dopaminergic neurons in the substantia nigra [155,156,157]. Daily agmatine treatment attenuated the dopaminergic neurotoxicity in the mouse 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) model of PD [157]. A recent study reported that agmatine (30 mg/kg i.p.) repeatedly attenuated short-term memory and motor impairments induced by intranasal administration of MPTP in 15-month-old mice, which also causes a decrease in hippocampal glutamate uptake. In the human-derived dopaminergic neuroblastoma cell line (SHSY5Y), agmatine (10–500 nM) was capable of preventing cell damage caused by exposure to rotenone in a PD model [156]. It’s beneficial effect could be associated with blocking of NF-κB nuclear translocation, suppression of ROS levels, and interruption of the apoptosis signaling cascade [155]. These results are associated with reducing oxidative damage and apoptotic cell death by agmatine in experimental models of PD.
Epilepsy
Epilepsy is a neurological disorder characterized by epileptic seizures, which increases with age. Indeed, agmatine appears to play an anti-epileptic role. It plays an anti-seizure role against maximal electroshock seizures (MES), an experimental model for generalized tonic–clonic seizures, in both mice and rats [158]. Agmatine (100 mg/kg i.p.) also improves the anticonvulsant function of phenobarbital and valproate in the MES model [159], and enhances the anticonvulsant effect of morphine or lithium chloride in mice via modulation of α2-adrenoceptors [160, 161]. In addition, agmatine mimicked the anticonvulsant effects of melatonin on the pentylenetetrazole (PTZ)-induced seizure threshold in mice [162], and had strong anticonvulsant effects in MES- and glutamate-induced seizure models in mice. These effects were likely related to N-methyl-d-aspartate receptor (NMDAR) antagonism [163]. In the hippocampi of PTZ-induced seizure model mice, a high dose of agmatine (20–80 mg/kg i.p.) reduced astrocytic hyperplasia and neuronal damage, indicating a reduction in the expression of the NR1 subunit of the NMDAR by agmatine [164]. This finding supports the involvement of the glutamatergic system in the anticonvulsant effects of agmatine. Based on these studies, agmatine is effective as an antiepileptic agent, and its effects are likely related to the l-arginine-NO pathway.
Other Neuropsychiatric Disorders
The therapeutic effects of agmatine also have been studied in different other neuropsychiatric disorders such as autism, schizophrenia, obsessive–compulsive disorder (OCD), depression and anxiety-like behaviours. Autism, also known as autism spectrum disorder (ASD), is a neurobehavioral condition lacks in social interaction and developmental impairment of language and communication skills with restricted/repetitive behaviors. The plasma agmatine levels in the ASD patients have been found to be remarkably lower than the non-ASD patients which suggests the involvement of agmatine to the ASD pathogenesis [165]. In the valproic acid animal model of autism agmatine ameliorated the ASD like symptoms by modulating the over-excitability of the neural circuit via inhibiting the over activation of the ERK1/2 signaling in the prefrontal cortex and hippocampus [166]. On the other hand, the plasma level of agmatine in the patients with schizophrenia found to be increased and in the rodent schizophrenia model agmatine, at a dose of (160 mg/kg i.p.), disrupted the paradigm of prepulse inhibition (PPI) of the acoustic startle reflex [167, 168]. Kotagale et al., suggested that agmatine alone can not induce schizophrenia like effects in rodents rather significant schizophrenic catalepsy can be found when agmatine (80 mg/kg i.p.) is injected into 5-HT1A receptor antagonist pretreated rodents [169]. However, agmatine was found to rescue the negative and cognitive schizophrenic symptoms but not the sensorimotor gating in MK-801-induced rat model of schizophrenia and pretreatment of a low dose of agmatine (20 mg/kg i.p.) could rescue the psychotomimetic drug phencyclidine (PCP) induced PPI deficit [170]. In the OCD rodents agmatine can effectively reduces the compulsive like behaviours which might be related to NO in brain [171, 172]. The agmatine level in the postmortem brain of the individuals with/without major depressive disorder (MDD) and suicide found to be lower than the normal individuals [173]. In the stress induced and CREB-regulated transcription coactivator 1 (CRTC1) knockout rodent models of depression agmatine showed marked anti-depressive and anti-anxiolytic effects through oxidative homeostasis pathway which may also involve serum CORT and BDNF levels [131, 174, 175].
Conclusion
The presence of agmatine in mammals was first noted in 1994 after its initial discovery by Nobel Laureate Albrecht Kossel in 1910. In mammals, it was identified as a CDS while searching for the endogenous ligand for I-1. A wide range of studies have since been performed and have shown that agmatine possesses most of the characteristics of an endogenous neurotransmitter and can bind to a variety of receptors or ion channels and modulate their functions (Fig. 4). Previous studies showed that, by modulating receptors or ion channels, agmatine exerted protective effects in many chronic and acute neurological diseases such as ischemic stroke, traumatic injury, AD, PD, schizophrenia, anxiety, depression, autism, and addiction. In addition to neuroprotection against neurological diseases, agmatine has been reported to function as an anti-diabetic, anti-hypertensive, nephron-protective, and gastro-protective drug. Our review provides a brief summary of the diseases related to the nervous system, and receptors and ion channels acted upon by this primary amine. This review will provide the basic information to the researchers studying the functional mechanism of agmatine on those receptors and ion channels which are not clearly understood yet. However, to perfect the use of agmatine as a choice pharmacological agent, a wide range of future studies are needed in order to determine its ligand binding and functional mechanisms related to the receptors and ion channels.
Abbreviations
- CNS:
-
Central nervous system
- PNS:
-
Peripheral nervous system
- AD:
-
Alzheimer’s disease
- PD:
-
Parkinson’s disease
- HD:
-
Huntington’s disease
- ADC:
-
Arginine decarboxylase
- ROS:
-
Reactive oxygen species
- NF-κB:
-
Nuclear factor kappa B
- TBI:
-
Traumatic brain injury
- NO:
-
Nitric oxide
- SCI:
-
Spinal cord injury
- BMP:
-
Bone morphogenetic protein
- Aβ:
-
Amyloid-beta
- Nrf2:
-
Nuclear factor (erythroid derived 2)-like 2
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- MES:
-
Maximal electroshock seizures
- PTZ:
-
Pentylenetetrazole
- NMDAR:
-
N-Methyl-d-aspartate receptor
- AMPAR:
-
α-Amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor
- NOS:
-
Nitric oxide synthase
- KAR:
-
Kainite receptor
- GPCR:
-
G protein-coupled receptor
- CDS:
-
Clonidine displacing substance
- AChR:
-
Acetylcholine
- mAChR:
-
Muscarinic acetylcholine receptor
- nAChR:
-
Nicotinic acetylcholine receptor
- 5-HT:
-
5-Hydroxytryptamine
- VDCC:
-
Voltage-dependent calcium channel
- LVA:
-
Low-voltage activated
- HVA:
-
High-voltage activated
- ENaC/DEG:
-
Epithelial Na+ channel/degenerin
- ASIC:
-
Acid-sensing ion channel
- EL/IL:
-
Extracellular/intracellular loops
References
Kumar P, Kumar D, Jha SK, Jha NK, Ambasta RK (2016) Ion channels in neurological disorders. Adv Protein Chem Struct Biol 103:97–136
Bano D, Zanetti F, Mende Y, Nicotera P (2011) Neurodegenerative processes in Huntington’s disease. Cell Death Dis 2:e228
Kumar VS, Gopalakrishnan A, Naziroglu M, Rajanikant GK (2014) Calcium ion-the key player in cerebral ischemia. Curr Med Chem 21:2065–2075
Shirwany NA, Payette D, Xie J, Guo Q (2007) The amyloid beta ion channel hypothesis of Alzheimer’s disease. Neuropsychiatry Dis Treat 3:597–612
Weilinger NL, Maslieieva V, Bialecki J, Sridharan SS, Tang PL, Thompson RJ (2013) Ionotropic receptors and ion channels in ischemic neuronal death and dysfunction. Acta Pharmacol Sin 34:39–48
Lim D, Fedrizzi L, Tartari M, Zuccato C, Cattaneo E, Brini M, Carafoli E (2008) Calcium homeostasis and mitochondrial dysfunction in striatal neurons of Huntington disease. J Biol Chem 283:5780–5789
Vitvitsky VM, Garg SK, Keep RF, Albin RL, Banerjee R (2012) Na+ and K+ ion imbalances in Alzheimer’s disease. Biochim Biophys Acta 1822:1671–1681
Hubner CA, Jentsch TJ (2002) Ion channel diseases. Hum Mol Genet 11:2435–2445
Gorbatyuk OS, Milner TA, Wang G, Regunathan S, Reis DJ (2001) Localization of agmatine in vasopressin and oxytocin neurons of the rat hypothalamic paraventricular and supraoptic nuclei. Exp Neurol 171:235–245
Reis DJ, Regunathan S (2000) Is agmatine a novel neurotransmitter in brain? Trends Pharmacol Sci 21:187–193
Kossel A (1911) Über das agmatin. Z Physiol Chem 66:257–261
Li G, Regunathan S, Barrow CJ, Eshraghi J, Cooper R, Reis DJ (1994) Agmatine: an endogenous clonidine-displacing substance in the brain. Science 263:966–969
Li G, Regunathan S, Reis DJ (1995) Agmatine is synthesized by a mitochondrial arginine decarboxylase in rat brain. Ann N Y Acad Sci 763:325–329
Lortie MJ, Novotny WF, Peterson OW, Vallon V, Malvey K, Mendonca M, Satriano J, Insel P, Thomson SC, Blantz RC (1996) Agmatine, a bioactive metabolite of arginine. Production, degradation, and functional effects in the kidney of the rat. J Clin Investig 97:413–420
Otake K, Ruggiero DA, Regunathan S, Wang H, Milner TA, Reis DJ (1998) Regional localization of agmatine in the rat brain: an immunocytochemical study. Brain Res 787:1–14
Peters D, Berger J, Langnaese K, Derst C, Madai VI, Krauss M, Fischer KD, Veh RW, Laube G (2013) Arginase and arginine decarboxylase—where do the putative gate keepers of polyamine synthesis reside in rat brain? PLoS ONE 8:e66735
Askalany AR, Yamakura T, Petrenko AB, Kohno T, Sakimura K, Baba H (2005) Effect of agmatine on heteromeric N-methyl-D-aspartate receptor channels. Neurosci Res 52:387–392
Feng Y, Piletz JE, Leblanc MH (2002) Agmatine suppresses nitric oxide production and attenuates hypoxic-ischemic brain injury in neonatal rats. Pediatr Res 52:606–611
Kim JH, Lee YW, Park YM, Park KA, Park SH, Lee WT, Lee JE (2011) Agmatine-reduced collagen scar area accompanied with surface righting reflex recovery after complete transection spinal cord injury. Spine 36:2130–2138
Kuo JR, Lo CJ, Chang CP, Lin KC, Lin MT, Chio CC (2011) Agmatine-promoted angiogenesis, neurogenesis, and inhibition of gliosis-reduced traumatic brain injury in rats. J Trauma 71:E87–E93
Laube G, Bernstein HG (2017) Agmatine: multifunctional arginine metabolite and magic bullet in clinical neuroscience? Biochem J 474:2619–2640
Kim JH, Kim JY, Jung JY, Lee YW, Lee WT, Huh SK, Lee JE (2017) Endogenous agmatine induced by ischemic preconditioning regulates ischemic tolerance following cerebral ischemia. Exp Neurobiol 26:380–389
Mun CH, Lee WT, Park KA, Lee JE (2010) Regulation of endothelial nitric oxide synthase by agmatine after transient global cerebral ischemia in rat brain. Anat Cell Biol 43:230–240
Piletz JE, Aricioglu F, Cheng JT, Fairbanks CA, Gilad VH, Haenisch B, Halaris A, Hong S, Lee JE, Li J, Liu P, Molderings GJ, Rodrigues AL, Satriano J, Seong GJ, Wilcox G, Wu N, Gilad GM (2013) Agmatine: clinical applications after 100 years in translation. Drug Discov Today 18:880–893
Seo SK, Yang W, Park YM, Lee WT, Park KA, Lee JE (2013) Overexpression of human arginine decarboxylase rescues human mesenchymal stem cells against H2O2 toxicity through cell survival protein activation. J Korean Med Sci 28:366–373
Uzbay TI (2012) The pharmacological importance of agmatine in the brain. Neurosci Biobehav Rev 36:502–519
Mayeur C, Veuillet G, Michaud M, Raul F, Blottiere HM, Blachier F (2005) Effects of agmatine accumulation in human colon carcinoma cells on polyamine metabolism, DNA synthesis and the cell cycle. Biochim Biophys Acta 1745:111–123
Wang JF, Su RB, Wu N, Xu B, Lu XQ, Liu Y, Li J (2005) Inhibitory effect of agmatine on proliferation of tumor cells by modulation of polyamine metabolism. Acta Pharmacol Sin 26:616–622
Marx M, Trittenwein G, Aufricht C, Hoeger H, Lubec B (1995) Agmatine and spermidine reduce collagen accumulation in kidneys of diabetic db/db mice. Nephron 69:155–158
Marc RE, Kalloniatis M, Jones BW (2005) Excitation mapping with the organic cation AGB2+. Vis Res 45:3454–3468
Benitez J, Garcia D, Romero N, Gonzalez A, Martinez-Oyanedel J, Figueroa M, Salas M, Lopez V, Garcia-Robles M, Dodd PR, Schenk G, Carvajal N, Uribe E (2018) Metabolic strategies for the degradation of the neuromodulator agmatine in mammals. Metabolism 81:35–44
Cai SX (2006) Glycine/NMDA receptor antagonists as potential CNS therapeutic agents: ACEA-1021 and related compounds. Curr Top Med Chem 6:651–662
Missale C, Fiorentini C, Busi C, Collo G, Spano PF (2006) The NMDA/D1 receptor complex as a new target in drug development. Curr Top Med Chem 6:801–808
Traynelis SF, Wollmuth LP, McBain CJ, Menniti FS, Vance KM, Ogden KK, Hansen KB, Yuan H, Myers SJ, Dingledine R (2010) Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev 62:405–496
Brittain MK, Brustovetsky T, Sheets PL, Brittain JM, Khanna R, Cummins TR, Brustovetsky N (2012) Delayed calcium dysregulation in neurons requires both the NMDA receptor and the reverse Na+/Ca2+ exchanger. Neurobiol Dis 46:109–117
Huang Z, Huang PL, Panahian N, Dalkara T, Fishman MC, Moskowitz MA (1994) Effects of cerebral ischemia in mice deficient in neuronal nitric oxide synthase. Science 265:1883–1885
Kaindl AM, Degos V, Peineau S, Gouadon E, Chhor V, Loron G, Le Charpentier T, Josserand J, Ali C, Vivien D, Collingridge GL, Lombet A, Issa L, Rene F, Loeffler JP, Kavelaars A, Verney C, Mantz J, Gressens P (2012) Activation of microglial N-methyl-D-aspartate receptors triggers inflammation and neuronal cell death in the developing and mature brain. Ann Neurol 72:536–549
Ahn SK, Hong S, Park YM, Lee WT, Park KA, Lee JE (2011) Effects of agmatine on hypoxic microglia and activity of nitric oxide synthase. Brain Res 1373:48–54
Wang WP, Iyo AH, Miguel-Hidalgo J, Regunathan S, Zhu MY (2006) Agmatine protects against cell damage induced by NMDA and glutamate in cultured hippocampal neurons. Brain Res 1084:210–216
Costa VV, Del Sarto JL, Rocha RF, Silva FR, Doria JG, Olmo IG, Marques RE, Queiroz-Junior CM, Foureaux G, Araujo JMS, Cramer A, Real A, Ribeiro LS, Sardi SI, Ferreira AJ, Machado FS, de Oliveira AC, Teixeira AL, Nakaya HI, Souza DG, Ribeiro FM, Teixeira MM (2017) N-Methyl-d-aspartate (NMDA) receptor blockade prevents neuronal death induced by Zika virus infection. MBio 8:e00350
Zhu MY, Piletz JE, Halaris A, Regunathan S (2003) Effect of agmatine against cell death induced by NMDA and glutamate in neurons and PC12 cells. Cell Mol Neurobiol 23:865–872
Fairbanks CA, Schreiber KL, Brewer KL, Yu CG, Stone LS, Kitto KF, Nguyen HO, Grocholski BM, Shoeman DW, Kehl LJ, Regunathan S, Reis DJ, Yezierski RP, Wilcox GL (2000) Agmatine reverses pain induced by inflammation, neuropathy, and spinal cord injury. Proc Natl Acad Sci USA 97:10584–10589
Hardingham GE, Bading H (2010) Synaptic versus extrasynaptic NMDA receptor signalling: implications for neurodegenerative disorders. Nat Rev Neurosci 11:682–696
Moretti M, Matheus FC, de Oliveira PA, Neis VB, Ben J, Walz R, Rodrigues AL, Prediger RD (2014) Role of agmatine in neurodegenerative diseases and epilepsy. Front Biosci 6:341–359
Regunathan S, Piletz JE (2003) Regulation of inducible nitric oxide synthase and agmatine synthesis in macrophages and astrocytes. Ann N Y Acad Sci 1009:20–29
Jacob AL, Weinberg RJ (2015) The organization of AMPA receptor subunits at the postsynaptic membrane. Hippocampus 25:798–812
Kumar J, Schuck P, Mayer ML (2011) Structure and assembly mechanism for heteromeric kainate receptors. Neuron 71:319–331
Liu SJ, Zukin RS (2007) Ca2+-permeable AMPA receptors in synaptic plasticity and neuronal death. Trends Neurosci 30:126–134
Sun HY, Bartley AF, Dobrunz LE (2009) Calcium-permeable presynaptic kainate receptors involved in excitatory short-term facilitation onto somatostatin interneurons during natural stimulus patterns. J Neurophysiol 101:1043–1055
Anzai T, Tsuzuki K, Yamada N, Hayashi T, Iwakuma M, Inada K, Kameyama K, Hoka S, Saji M (2003) Overexpression of Ca2+-permeable AMPA receptor promotes delayed cell death of hippocampal CA1 neurons following transient forebrain ischemia. Neurosci Res 46:41–51
Liu S, Lau L, Wei J, Zhu D, Zou S, Sun HS, Fu Y, Liu F, Lu Y (2004) Expression of Ca2+-permeable AMPA receptor channels primes cell death in transient forebrain ischemia. Neuron 43:43–55
Darstein M, Petralia RS, Swanson GT, Wenthold RJ, Heinemann SF (2003) Distribution of kainate receptor subunits at hippocampal mossy fiber synapses. J Neurosci 23:8013–8019
Lowry ER, Kruyer A, Norris EH, Cederroth CR, Strickland S (2013) The GluK4 kainate receptor subunit regulates memory, mood, and excitotoxic neurodegeneration. Neuroscience 235:215–225
Lee JY, Kim JH, Palmiter RD, Koh JY (2003) Zinc released from metallothionein-iii may contribute to hippocampal CA1 and thalamic neuronal death following acute brain injury. Exp Neurol 184:337–347
Kwak S, Weiss JH (2006) Calcium-permeable AMPA channels in neurodegenerative disease and ischemia. Curr Opin Neurobiol 16:281–287
Neis VB, Moretti M, Bettio LE, Ribeiro CM, Rosa PB, Goncalves FM, Lopes MW, Leal RB, Rodrigues AL (2016) Agmatine produces antidepressant-like effects by activating AMPA receptors and mTOR signaling. Eur Neuropsychopharmacol 26:959–971
Buerkle H, Yaksh TL (1998) Pharmacological evidence for different alpha 2-adrenergic receptor sites mediating analgesia and sedation in the rat. Br J Anaesth 81:208–215
Henderson R, Baldwin JM, Ceska TA, Zemlin F, Beckmann E, Downing KH (1990) Model for the structure of bacteriorhodopsin based on high-resolution electron cryo-microscopy. J Mol Biol 213:899–929
Salminen T, Varis M, Nyronen T, Pihlavisto M, Hoffren AM, Lonnberg T, Marjamaki A, Frang H, Savola JM, Scheinin M, Johnson MS (1999) Three-dimensional models of α2A-adrenergic receptor complexes provide a structural explanation for ligand binding. J Biol Chem 274:23405–23413
Boxwalla M, Matwyshyn G, Puppala BL, Andurkar SV, Gulati A (2010) Involvement of imidazoline and opioid receptors in the enhancement of clonidine-induced analgesia by sulfisoxazole. Can J Physiol Pharmacol 88:541–552
Han Z, Xiao MJ, Shao B, Zheng RY, Yang GY, Jin K (2009) Attenuation of ischemia-induced rat brain injury by 2-(-2-benzofuranyl)-2-imidazoline, a high selectivity ligand for imidazoline I2 receptors. Neurol Res 31:390–395
Li JX, Zhang Y (2011) Imidazoline I2 receptors: target for new analgesics? Eur J Pharmacol 658:49–56
Atlas D, Burstein Y (1984) Isolation and partial purification of a clonidine-displacing endogenous brain substance. Eur J Biochem 144:287–293
Coupry I, Atlas D, Podevin RA, Uzielli I, Parini A (1990) Imidazoline-guanidinium receptive site in renal proximal tubule: asymmetric distribution, regulation by cations and interaction with an endogenous clonidine displacing substance. J Pharmacol Exp Ther 252:293–299
Zonnenchein R, Diamant S, Atlas D (1990) Imidazoline receptors in rat liver cells: a novel receptor or a subtype of α2-adrenoceptors? Eur J Pharmacol 190:203–215
Aricioglu F, Korcegez E, Bozkurt A, Ozyalcin S (2003) Effect of agmatine on acute and mononeuropathic pain. Ann N Y Acad Sci 1009:106–115
Aricioglu F, Paul IA, Regunathan S (2004) Agmatine reduces only peripheral-related behavioral signs, not the central signs, of morphine withdrawal in nNOS deficient transgenic mice. Neurosci Lett 354:153–157
Dixit MP, Thakre PP, Pannase AS, Aglawe MM, Taksande BG, Kotagale NR (2014) Imidazoline binding sites mediates anticompulsive-like effect of agmatine in marble-burying behavior in mice. Eur J Pharmacol 732:26–31
Demehri S, Homayoun H, Honar H, Riazi K, Vafaie K, Roushanzamir F, Dehpour AR (2003) Agmatine exerts anticonvulsant effect in mice: modulation by α2-adrenoceptors and nitric oxide. Neuropharmacology 45:534–542
Shelkar GP, Gakare SG, Chakraborty S, Dravid SM, Ugale RR (2016) Interactions of nitric oxide with α2-adrenoceptors within the locus coeruleus underlie the facilitation of inhibitory avoidance memory by agmatine. Br J Pharmacol 173:2589–2599
Zomkowski AD, Hammes L, Lin J, Calixto JB, Santos AR, Rodrigues AL (2002) Agmatine produces antidepressant-like effects in two models of depression in mice. Neuroreport 13:387–391
Li F, Wu N, Su RB, Liu Y, Lu XQ, Li J (2009) Comparison of agmatine with moxonidine and rilmenidine in morphine dependence in vitro: role of imidazoline I1 receptors. Eur J Pharmacol 612:1–8
Taksande BG, Kotagale NR, Patel MR, Shelkar GP, Ugale RR, Chopde CT (2010) Agmatine, an endogenous imidazoline receptor ligand modulates ethanol anxiolysis and withdrawal anxiety in rats. Eur J Pharmacol 637:89–101
Taksande BG, Kotagale NR, Tripathi SJ, Ugale RR, Chopde CT (2009) Antidepressant like effect of selective serotonin reuptake inhibitors involve modulation of imidazoline receptors by agmatine. Neuropharmacology 57:415–424
Giusepponi ME, Cifani C, Micioni Di Bonaventura MV, Mattioli L, Hudson A, Diamanti E, Del Bello F, Giannella M, Mammoli V, Paoletti CD, Piergentili A, Pigini M, Quaglia W (2016) Combined interactions with I1-, I2-imidazoline binding sites and α2-adrenoceptors to manage opioid addiction. ACS Med Chem Lett 7:956–961
Fredriksson R, Lagerstrom MC, Lundin LG, Schioth HB (2003) The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol 63:1256–1272
Kruse AC, Kobilka BK, Gautam D, Sexton PM, Christopoulos A, Wess J (2014) Muscarinic acetylcholine receptors: novel opportunities for drug development. Nat Rev Drug Discov 13:549–560
Wess J, Eglen RM, Gautam D (2007) Muscarinic acetylcholine receptors: mutant mice provide new insights for drug development. Nat Rev Drug Discov 6:721–733
Eglen RM (2012) Overview of muscarinic receptor subtypes. Handb Exp Pharmacol 2012:3–28
Conn PJ, Jones CK, Lindsley CW (2009) Subtype-selective allosteric modulators of muscarinic receptors for the treatment of CNS disorders. Trends Pharmacol Sci 30:148–155
Liao DL, Hong CJ, Chen HM, Chen YE, Lee SM, Chang CY, Chen H, Tsai SJ (2003) Association of muscarinic M1 receptor genetic polymorphisms with psychiatric symptoms and cognitive function in schizophrenic patients. Neuropsychobiology 48:72–76
Peretto I, Petrillo P, Imbimbo BP (2009) Medicinal chemistry and therapeutic potential of muscarinic M3 antagonists. Med Res Rev 29:867–902
Albuquerque EX, Pereira EF, Alkondon M, Rogers SW (2009) Mammalian nicotinic acetylcholine receptors: from structure to function. Physiol Rev 89:73–120
Zhang L, Xiong W (2009) Modulation of the Cys-loop ligand-gated ion channels by fatty acid and cannabinoids. Vitam Horm 81:315–335
Dani JA, Bertrand D (2007) Nicotinic acetylcholine receptors and nicotinic cholinergic mechanisms of the central nervous system. Annu Rev Pharmacol Toxicol 47:699–729
Rode F, Munro G, Holst D, Nielsen EO, Troelsen KB, Timmermann DB, Ronn LC, Grunnet M (2012) Positive allosteric modulation of α4β2 nAChR agonist induced behaviour. Brain Res 1458:67–75
Li M, Lester HA (2001) Ion channel diseases of the central nervous system. CNS Drug Rev 7:214–240
Picciotto MR, Caldarone BJ, Brunzell DH, Zachariou V, Stevens TR, King SL (2001) Neuronal nicotinic acetylcholine receptor subunit knockout mice: physiological and behavioral phenotypes and possible clinical implications. Pharmacol Ther 92:89–108
Wootten D, Christopoulos A, Sexton PM (2013) Emerging paradigms in GPCR allostery: implications for drug discovery. Nat Rev Drug Discov 12:630–644
Loring RH (1990) Agmatine acts as an antagonist of neuronal nicotinic receptors. Br J Pharmacol 99:207–211
Aricioglu-Kartal F, Uzbay IT (1997) Inhibitory effect of agmatine on naloxone-precipitated abstinence syndrome in morphine dependent rats. Life Sci 61:1775–1781
Kotagale NR, Taksande BG, Gahane AY, Ugale RR, Chopde CT (2010) Repeated agmatine treatment attenuates nicotine sensitization in mice: modulation by α2-adrenoceptors. Behav Brain Res 213:161–174
Morgan AD, Campbell UC, Fons RD, Carroll ME (2002) Effects of agmatine on the escalation of intravenous cocaine and fentanyl self-administration in rats. Pharmacol Biochem Behav 72:873–880
Su RB, Wang WP, Lu XQ, Wu N, Liu ZM, Li J (2009) Agmatine blocks acquisition and re-acquisition of intravenous morphine self-administration in rats. Pharmacol Biochem Behav 92:676–682
Uzbay IT, Yesilyurt O, Celik T, Ergun H, Isimer A (2000) Effects of agmatine on ethanol withdrawal syndrome in rats. Behav Brain Res 107:153–159
Yesilyurt O, Uzbay IT (2001) Agmatine potentiates the analgesic effect of morphine by an α2-adrenoceptor-mediated mechanism in mice. Neuropsychopharmacology 25:98–103
Kotagale NR, Walke S, Shelkar GP, Kokare DM, Umekar MJ, Taksande BG (2014) Agmatine attenuates nicotine induced conditioned place preference in mice through modulation of neuropeptide Y system. Behav Brain Res 262:118–124
Taksande BG, Kotagale NR, Gawande DY, Bharne AP, Chopde CT, Kokare DM (2014) Neuropeptide Y in the central nucleus of amygdala regulates the anxiolytic effect of agmatine in rats. Eur Neuropsychopharmacol 24:955–963
Moosavi M, Khales GY, Abbasi L, Zarifkar A, Rastegar K (2012) Agmatine protects against scopolamine-induced water maze performance impairment and hippocampal ERK and Akt inactivation. Neuropharmacology 62:2018–2023
Tsien RW, Ellinor PT, Horne WA (1991) Molecular diversity of voltage-dependent Ca2+ channels. Trends Pharmacol Sci 12:349–354
Nowycky MC, Fox AP, Tsien RW (1985) Three types of neuronal calcium channel with different calcium agonist sensitivity. Nature 316:440–443
Scott RH, Pearson HA, Dolphin AC (1991) Aspects of vertebrate neuronal voltage-activated calcium currents and their regulation. Prog Neurobiol 36:485–520
Catterall WA, Perez-Reyes E, Snutch TP, Striessnig J (2005) International union of pharmacology. XLVIII. Nomenclature and structure-function relationships of voltage-gated calcium channels. Pharmacol Rev 57:411–425
Catterall WA (2010) Ion channel voltage sensors: structure, function, and pathophysiology. Neuron 67:915–928
Anekonda TS, Quinn JF, Harris C, Frahler K, Wadsworth TL, Woltjer RL (2011) L-type voltage-gated calcium channel blockade with isradipine as a therapeutic strategy for Alzheimer’s disease. Neurobiol Dis 41:62–70
Ostacher MJ, Iosifescu DV, Hay A, Blumenthal SR, Sklar P, Perlis RH (2014) Pilot investigation of isradipine in the treatment of bipolar depression motivated by genome-wide association. Bipolar Disord 16:199–203
Ziyatdinova S, Gurevicius K, Kutchiashvili N, Bolkvadze T, Nissinen J, Tanila H, Pitkanen A (2011) Spontaneous epileptiform discharges in a mouse model of Alzheimer’s disease are suppressed by antiepileptic drugs that block sodium channels. Epilepsy Res 94:75–85
Weng XC, Gai XD, Zheng JQ, Li J (2003) Agmatine blocked voltage-gated calcium channel in cultured rat hippocampal neurons. Acta Pharmacol Sin 24:746–750
Zheng JQ, Weng XC, Gai XD, Li J, Xiao WB (2004) Mechanism underlying blockade of voltage-gated calcium channels by agmatine in cultured rat hippocampal neurons. Acta Pharmacol Sin 25:281–285
Wang G, Gorbatyuk OS, Dayanithi G, Ouyang W, Wang J, Milner TA, Regunathan S, Reis DJ (2002) Evidence for endogenous agmatine in hypothalamo-neurohypophysial tract and its modulation on vasopressin release and Ca2+ channels. Brain Res 932:25–36
Kim YH, Jeong JH, Ahn DS, Chung S (2016) Agmatine suppresses peripheral sympathetic tone by inhibiting N-type Ca2+ channel activity via imidazoline I2 receptor activation. Biochem Biophys Res Commun 477:406–412
Chu XP, Papasian CJ, Wang JQ, Xiong ZG (2011) Modulation of acid-sensing ion channels: molecular mechanisms and therapeutic potential. Int J Physiol Pathophysiol Pharmacol 3:288–309
Sherwood TW, Frey EN, Askwith CC (2012) Structure and activity of the acid-sensing ion channels. Am J Physiol Cell Physiol 303:C699–C710
Gonzales EB, Kawate T, Gouaux E (2009) Pore architecture and ion sites in acid-sensing ion channels and P2X receptors. Nature 460:599–604
Benson CJ, Xie J, Wemmie JA, Price MP, Henss JM, Welsh MJ, Snyder PM (2002) Heteromultimers of DEG/ENaC subunits form H+-gated channels in mouse sensory neurons. Proc Natl Acad Sci USA 99:2338–2343
Bohlen CJ, Chesler AT, Sharif-Naeini R, Medzihradszky KF, Zhou S, King D, Sanchez EE, Burlingame AL, Basbaum AI, Julius D (2011) A heteromeric Texas coral snake toxin targets acid-sensing ion channels to produce pain. Nature 479:410–414
Yu Y, Chen Z, Li WG, Cao H, Feng EG, Yu F, Liu H, Jiang H, Xu TL (2010) A nonproton ligand sensor in the acid-sensing ion channel. Neuron 68:61–72
Hu R, Duan B, Wang D, Yu Y, Li W, Luo H, Lu P, Lin J, Zhu G, Wan Q, Feng H (2011) Role of acid-sensing ion channel 1a in the secondary damage of traumatic spinal cord injury. Ann Surg 254:353–362
Pignataro G, Simon RP, Xiong ZG (2007) Prolonged activation of ASIC1a and the time window for neuroprotection in cerebral ischaemia. Brain 130:151–158
Wemmie JA, Chen J, Askwith CC, Hruska-Hageman AM, Price MP, Nolan BC, Yoder PG, Lamani E, Hoshi T, Freeman JH Jr, Welsh MJ (2002) The acid-activated ion channel ASIC contributes to synaptic plasticity, learning, and memory. Neuron 34:463–477
Li WG, Yu Y, Zhang ZD, Cao H, Xu TL (2010) ASIC3 channels integrate agmatine and multiple inflammatory signals through the nonproton ligand sensing domain. Mol Pain 6:88
Peroutka SJ, Howell TA (1994) The molecular evolution of G protein-coupled receptors: focus on 5-hydroxytryptamine receptors. Neuropharmacology 33:319–324
Hoyer D, Clarke DE, Fozard JR, Hartig PR, Martin GR, Mylecharane EJ, Saxena PR, Humphrey PP (1994) International union of pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol Rev 46:157–203
McCorvy JD, Roth BL (2015) Structure and function of serotonin G protein-coupled receptors. Pharmacol Ther 150:129–142
Wang C, Jiang Y, Ma J, Wu H, Wacker D, Katritch V, Han GW, Liu W, Huang XP, Vardy E, McCorvy JD, Gao X, Zhou XE, Melcher K, Zhang C, Bai F, Yang H, Yang L, Jiang H, Roth BL, Cherezov V, Stevens RC, Xu HE (2013) Structural basis for molecular recognition at serotonin receptors. Science 340:610–614
Booij L, Tremblay RE, Szyf M, Benkelfat C (2015) Genetic and early environmental influences on the serotonin system: consequences for brain development and risk for psychopathology. J Psychiatry Neurosci 40:5–18
Li YF, Gong ZH, Cao JB, Wang HL, Luo ZP, Li J (2003) Antidepressant-like effect of agmatine and its possible mechanism. Eur J Pharmacol 469:81–88
Zomkowski ADE, Rosa AO, Lin J, Santos AR, Calixto JB, Rodrigues ALS (2004) Evidence for serotonin receptor subtypes involvement in agmatine antidepressant like-effect in the mouse forced swimming test. Brain Res 1023:253–263
Molderings GJ, Schmidt K, Bönisch H (1996) Inhibition of 5-HT3 receptor function by imidazolines in mouse neuroblastoma cells: potential involvement of σ2 binding sites. Naunyn-Schmiedeberg’s Arch Pharmacol 354:245–252
Shopsin B (2013) The clinical antidepressant effect of exogenous agmatine is not reversed by parachlorophenylalanine: a pilot study. Acta Neuropsychiatry 25:113–118
Freitas AE, Egea J, Buendia I, Navarro E, Rada P, Cuadrado A, Rodrigues AL, Lopez MG (2015) Agmatine induces Nrf2 and protects against corticosterone effects in hippocampal neuronal cell line. Mol Neurobiol 51:1504–1519
Gilad GM, Salame K, Rabey JM, Gilad VH (1996) Agmatine treatment is neuroprotective in rodent brain injury models. Life Sci 58:PL 41–46
Gilad GM, Gilad VH (2000) Accelerated functional recovery and neuroprotection by agmatine after spinal cord ischemia in rats. Neurosci Lett 296:97–100
Halaris A, Plietz J (2007) Agmatine: metabolic pathway and spectrum of activity in brain. CNS Drugs 21:885–900
Yang XC, Reis DJ (1999) Agmatine selectively blocks the N-methyl-D-aspartate subclass of glutamate receptor channels in rat hippocampal neurons. J Pharmacol Exp Ther 288:544–549
Yu CG, Marcillo AE, Fairbanks CA, Wilcox GL, Yezierski RP (2000) Agmatine improves locomotor function and reduces tissue damage following spinal cord injury. Neuroreport 11:3203–3207
Kriz J, Lalancette-Hebert M (2009) Inflammation, plasticity and real-time imaging after cerebral ischemia. Acta Neuropathol 117:497–509
Kim JH, Yenari MA, Giffard RG, Cho SW, Park KA, Lee JE (2004) Agmatine reduces infarct area in a mouse model of transient focal cerebral ischemia and protects cultured neurons from ischemia-like injury. Exp Neurol 189:122–130
Lee WT, Hong S, Yoon SH, Kim JH, Park KA, Seong GJ, Lee JE (2009) Neuroprotective effects of agmatine on oxygen-glucose deprived primary-cultured astrocytes and nuclear translocation of nuclear factor-kappa B. Brain Res 1281:64–70
Kim DJ, Kim DI, Lee SK, Suh SH, Lee YJ, Kim J, Chung TS, Lee JE (2006) Protective effect of agmatine on a reperfusion model after transient cerebral ischemia: temporal evolution on perfusion MR imaging and histopathologic findings. AJNR Am J Neuroradiol 27:780–785
Kim JH, Lee YW, Park KA, Lee WT, Lee JE (2010) Agmatine attenuates brain edema through reducing the expression of aquaporin-1 after cerebral ischemia. J Cereb Blood Flow Metab 30:943–949
Yang MZ, Mun CH, Choi YJ, Baik JH, Park KA, Lee WT, Lee JE (2007) Agmatine inhibits matrix metalloproteinase-9 via endothelial nitric oxide synthase in cerebral endothelial cells. Neurol Res 29:749–754
Kim JY, Lee YW, Kim JH, Lee WT, Park KA, Lee JE (2015) Agmatine attenuates brain edema and apoptotic cell death after traumatic brain injury. J Korean Med Sci 30:943–952
Kuo JR, Lo CJ, Chio CC, Chang CP, Lin MT (2007) Resuscitation from experimental traumatic brain injury by agmatine therapy. Resuscitation 75:506–514
Goracke-Postle CJ, Nguyen HO, Stone LS, Fairbanks CA (2006) Release of tritiated agmatine from spinal synaptosomes. Neuroreport 17:13–17
Park YM, Han SH, Seo SK, Park KA, Lee WT, Lee JE (2015) Restorative benefits of transplanting human mesenchymal stromal cells overexpressing arginine decarboxylase genes after spinal cord injury. Cytotherapy 17:25–37
Park YM, Lee WT, Bokara KK, Seo SK, Park SH, Kim JH, Yenari MA, Park KA, Lee JE (2013) The multifaceted effects of agmatine on functional recovery after spinal cord injury through Modulations of BMP-2/4/7 expressions in neurons and glial cells. PLoS ONE 8:e53911
Kang S, Kim CH, Jung H, Kim E, Song HT, Lee JE (2017) Agmatine ameliorates type 2 diabetes induced-Alzheimer’s disease-like alterations in high-fat diet-fed mice via reactivation of blunted insulin signalling. Neuropharmacology 113:467–479
Arteni NS, Lavinsky D, Rodrigues AL, Frison VB, Netto CA (2002) Agmatine facilitates memory of an inhibitory avoidance task in adult rats. Neurobiol Learn Mem 78:465–469
Liu P, Bergin DH (2009) Differential effects of i.c.v. microinfusion of agmatine on spatial working and reference memory in the rat. Neuroscience 159:951–961
Lu W, Dong HJ, Gong ZH, Su RB, Li J (2010) Agmatine inhibits morphine-induced memory impairment in the mouse step-down inhibitory avoidance task. Pharmacol Biochem Behav 97:256–261
Moosavi M, Zarifkar AH, Farbood Y, Dianat M, Sarkaki A, Ghasemi R (2014) Agmatine protects against intracerebroventricular streptozotocin-induced water maze memory deficit, hippocampal apoptosis and Akt/GSK3β signaling disruption. Eur J Pharmacol 736:107–114
Song J, Hur BE, Bokara KK, Yang W, Cho HJ, Park KA, Lee WT, Lee KM, Lee JE (2014) Agmatine improves cognitive dysfunction and prevents cell death in a streptozotocin-induced Alzheimer rat model. Yonsei Med J 55:689–699
Bergin DH, Liu P (2010) Agmatine protects against β-amyloid25-35-induced memory impairments in the rat. Neuroscience 169:794–811
Condello S, Calabro E, Caccamo D, Curro M, Ferlazzo N, Satriano J, Magazu S, Ientile R (2012) Protective effects of agmatine in rotenone-induced damage of human SH-SY5Y neuroblastoma cells: Fourier transform infrared spectroscopy analysis in a model of Parkinson’s disease. Amino Acids 42:775–781
Condello S, Curro M, Ferlazzo N, Caccamo D, Satriano J, Ientile R (2011) Agmatine effects on mitochondrial membrane potential and NF-kappaB activation protect against rotenone-induced cell damage in human neuronal-like SH-SY5Y cells. J Neurochem 116:67–75
Gilad GM, Gilad VH, Finberg JP, Rabey JM (2005) Neurochemical evidence for agmatine modulation of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxicity. Neurochem Res 30:713–719
Bence AK, Worthen DR, Stables JP, Crooks PA (2003) An in vivo evaluation of the antiseizure activity and acute neurotoxicity of agmatine. Pharmacol Biochem Behav 74:771–775
Luszczki JJ, Czernecki R, Wojtal K, Borowicz KK, Czuczwar SJ (2008) Agmatine enhances the anticonvulsant action of phenobarbital and valproate in the mouse maximal electroshock seizure model. J Neural Transm 115:1485–1494
Bahremand A, Ziai P, Khodadad TK, Payandemehr B, Rahimian R, Ghasemi A, Ghasemi M, Hedayat T, Dehpour AR (2010) Agmatine enhances the anticonvulsant effect of lithium chloride on pentylenetetrazole-induced seizures in mice: involvement of L-arginine/nitric oxide pathway. Epilepsy Behav 18:186–192
Riazi K, Honar H, Homayoun H, Rashidi N, Kiani S, Ebrahimkhani MR, Noorian AR, Ghaffari K, Jannati A, Dehpour AR (2005) The synergistic anticonvulsant effect of agmatine and morphine: possible role of alpha 2-adrenoceptors. Epilepsy Res 65:33–40
Moezi L, Shafaroodi H, Hojati A, Dehpour AR (2011) The interaction of melatonin and agmatine on pentylenetetrazole-induced seizure threshold in mice. Epilepsy Behav 22:200–206
Su RB, Wei XL, Zheng JQ, Liu Y, Lu XQ, Li J (2004) Anticonvulsive effect of agmatine in mice. Pharmacol Biochem Behav 77:345–349
Xu H, Ou F, Wang P, Naren M, Tu D, Zheng R (2014) High dosage of agmatine alleviates pentylenetetrazole-induced chronic seizures in rats possibly by exerting an anticonvulsive effect. Exp Ther Med 8:73–78
Esnafoglu E, Irende I (2018) Decreased plasma agmatine levels in autistic subjects. J Neural Transm 125:735–740
Kim JW, Seung H, Kim KC, Gonzales ELT, Oh HA, Yang SM, Ko MJ, Han SH, Banerjee S, Shin CY (2017) Agmatine rescues autistic behaviors in the valproic acid-induced animal model of autism. Neuropharmacology 113:71–81
Uzbay T, Goktalay G, Kayir H, Eker SS, Sarandol A, Oral S, Buyukuysal L, Ulusoy G, Kirli S (2013) Increased plasma agmatine levels in patients with schizophrenia. J Psychiatr Res 47:1054–1060
Uzbay T, Kayir H, Goktalay G, Yildirim M (2010) Agmatine disrupts prepulse inhibition of acoustic startle reflex in rats. J Psychopharmacol 24:923–929
Kotagale NR, Taksande BG, Wadhwani PJ, Palhade MW, Mendhi SM, Gawande DY, Hadole PN, Chopde CT (2012) Psychopharmacological study of agmatine in behavioral tests of schizophrenia in rodents. Pharmacol Biochem Behav 100:398–403
Palsson E, Fejgin K, Wass C, Klamer D (2008) Agmatine attenuates the disruptive effects of phencyclidine on prepulse inhibition. Eur J Pharmacol 590:212–216
Gawali NB, Chowdhury AA, Kothavade PS, Bulani VD, Nagmoti DM, Juvekar AR (2016) Involvement of nitric oxide in anticompulsive-like effect of agmatine on marble-burying behaviour in mice. Eur J Pharmacol 770:165–171
Patel VMSFR (2014) Agmatine ameliorates social isolation induced obsessive-compulsive behavior in mice. J Glob Trends Pharm Sci 5:2048–2051
Chen GG, Almeida D, Fiori L, Turecki G (2018) Evidence of reduced agmatine concentrations in the cerebral cortex of suicides. Int J Neuropsychopharmacol 21:895–900
Gawali NB, Bulani VD, Gursahani MS, Deshpande PS, Kothavade PS, Juvekar AR (2017) Agmatine attenuates chronic unpredictable mild stress-induced anxiety, depression-like behaviours and cognitive impairment by modulating nitrergic signalling pathway. Brain Res 1663:66–77
Meylan EM, Breuillaud L, Seredenina T, Magistretti PJ, Halfon O, Luthi-Carter R, Cardinaux JR (2016) Involvement of the agmatinergic system in the depressive-like phenotype of the Crtc1 knockout mouse model of depression. Transl Psychiatry 6:e852
Acknowledgements
This study was supported by National Research Foundation of Korea (NRF) grant funded by the Korea Government (MSIP) (2017R1A2B2005350).
Funding
This study was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea Government (MSIP) (2017R1A2B2005350).
Author information
Authors and Affiliations
Contributions
JEL provided concept, design and overall supervision of this study. SB, JYK1 contributed in the writing and drawing. JYK2, JHK and JEL participated in the discussion and revision. All authors approved and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barua, S., Kim, J.Y., Kim, J.Y. et al. Therapeutic Effect of Agmatine on Neurological Disease: Focus on Ion Channels and Receptors. Neurochem Res 44, 735–750 (2019). https://doi.org/10.1007/s11064-018-02712-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-018-02712-1