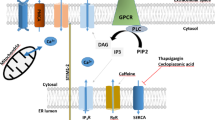
In neurons, the endoplasmic reticulum (ER) modulates elevations of the cytosolic free Ca2+ concentration ([Ca2+] ) in response to extracellular stimulation by accumulating Ca2+ via the sarcoplasmic/endoplasmic reticulum Ca2+-ATPases (SERCAs) or by releasing Ca2+ via the ER Ca2+ channels. SERCA inhibitors are often used as a tool for investigating the contribution from ER Ca2+ transport to the [Ca2+] dynamics. The respective effects on Ca2+ responses in different neurons are characterized by a large diversity. However, the factors that determine this diversity have not been completely understood. Using a simple two-compartment model of the Ca2+ dynamics, we showed that changes in the density and relative contribution of ryanodine receptors and SERCA pumps, stimulation conditions, and dye concentration are sufficient to reproduce either smoothly graded, or non-linearly graded, or all-or-non ER Ca2+ release and many effects of SERCA inhibitors on the Ca2+ transients observed in different neurons. Here, we have redefined main modes of the net ER Ca2+ transport (net Ca2+ uptake, non-regenerative Ca2+ release, and regenerative Ca2+ release) and showed that these modes are completely determined by the interplay between Ca2+ fluxes and the Ca2+ buffering rate. Our simulations demonstrate that low-gain models of Ca2+-induced calcium release (CICR) do not require any counteracting termination mechanism for the release termination. The simulations also suggested that Ca2+ transients in some neurons may be modestly amplified by regenerative CICR, which is, nonetheless, graded and self-limiting due to CICR termination mechanisms, such as store depletion and/or Ca2+-dependent inactivation of ryanodine receptors. However, a spatially homogeneous model fails to reproduce both smoothly graded and high- gain CICR.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
O. H. Petersen, M. Michalak, and A. Verkhratsky, “Calcium signalling: past, present and future,” Cell Calcium, 38, No. 3−4, 161–169 (2005); doi: https://doi.org/10.1016/j.ceca.2005.06.023.
A. Verkhratsky, “Physiology and pathophysiology of the calcium store in the endoplasmic reticulum of neurons,” Physiol. Rev., 85, No. 1, 201–279 (2005); doi: https://doi.org/10.1152/physrev.00004.2004.
D. D. Friel and R. W. Tsien, “A caffeine- and ryano- dine-sensitive Ca2+ store in bullfrog sympathetic neurones modulates effects of Ca2+ entry on [Ca2+] i.,” J. Physiol., 450, 217–246 (1992); doi: https://doi.org/10.1113/jphysiol.1992.sp019125.
N. Solovyova, N. Veselovsky, E. C. Toescu, and A. Ver- khratsky, “ Ca2+ dynamics in the lumen of the endoplasmic reticulum in sensory neurons: direct visualization of Ca2+-induced Ca2+ release triggered by physiological Ca2+ entry,” EMBO J., 21, No. 4, 622−630 (2002); doi: https://doi.org/10.1093/emboj/21.4.622.
A. S. Cohen, K. A. Moore, R. Bangalore, et al., “Ca2+- induced Ca2+ release mediates Ca2+ transients evoked by single action potentials in rabbit vagal afferent neurones,” J. Physiol., 499, Pt. 2, 315−328 (1997); doi: https://doi.org/10.1113/jphysiol.1997.sp021929.
Y. M. Usachev and S. A. Thayer, “All-or-none Ca2+ release from intracellular stores triggered by Ca2+ influx through voltage-gated Ca2+ channels in rat sensory neurons,” J. Neurosci., 17, No. 19, 7404−7414 (1997); doi: https://doi.org/10.1523/JNEUROSCI.17-19-07404.1997.
L. Fierro, R. DiPolo, and L. Llano, “Intracellular calcium clearance in Purkinje cell somata from rat cerebellar slices,” J. Physiol. 510, Pt. 2, 499−512 (1998); doi: https://doi.org/10.1111/j.1469-7793.1998.499bk.x.
B. L. Sabatini, T. G. Oertner, and K. Svoboda, “The life cycle of Ca2+ ions in dendritic spines,” Neuron, 33, No. 3, 439–452 (2002); doi: https://doi.org/10.1016/s0896-6273(02)00573-1.
T. Akita and K. Kuba, “Functional triads consisting of ryanodine receptors, Ca2+ channels, and Ca2+-activated K+ channels in bullfrog sympathetic neurons. Plastic modulation of action potential,” J. Gen. Physiol., 116, No. 5, 697−720 (2000); doi: https://doi.org/10.1085/jgp.116.5.697.
D. Gall, F. Prestori, E. Sola, et al., “Intracellular calcium regulation by burst discharge determines bidirectional long-term synaptic plasticity at the cerebellum input stage,” J. Neurosci., 25, No. 19, 4813–4822 (2005); doi: https://doi.org/10.1523/JNEUROSCI.0410-05.2005.
S. Alford, B. G. Frenguelli, J. G. Schofield, and G. L. Collingridge, “Characterization of Ca2+ signals induced in hippocampal CA1 neurons by the synaptic activation of NMDA receptors,” J. Physiol., 469, 693– 716 (1993); doi: https://doi.org/10.1113/jphysiol.1993.sp019838.
Y. Liang, L. L. Yuan, D. Johnston, and R. Gray, “Calcium signaling at single mossy fiber presynaptic terminals in the rat hippocampus,” J. Neurophysiol., 87, No. 2, 1132−1137 (2002); doi: https://doi.org/10.1152/jn.00661.2001.
T. Szikra and D. Križaj, “Intracellular organelles and calcium homeostasis in rods and cones,” Vis. Neurosci., 24, No. 5, 733−743 (2007); doi: https://doi.org/10.1017/S0952523807070587.
V. M. Sandler and J. G. Barbara, “Calcium-induced calcium release contributes to action-potential evoked calcium transients in hippocampal CA1 pyramidal neurons,” J. Neurosci., 19, No. 11, 4325–4336 (1999); doi: https://doi.org/10.1523/JNEUROSCI.19-11-04325.1999.
F. Helmchen, K. Imoto, and B. Sakmann, “Ca2+ buffering and action potential-evoked Ca2+ signaling in dendrites of pyramidal neurons,” Biophys. J., 70, No. 2, 1069–1081 (1996); doi: https://doi.org/10.1016/S0006-3495(96)79653-4.
D. W. Tank, W. G. Regehr, and K. R. Delaney, “A quantitative analysis of presynaptic calcium dynamics that contribute to short-term enhancement,” J. Neurosci., 15, No. 12, 7940–7952 (1995); doi: https://doi.org/10.1523/JNEUROSCI.15-12-07940.1995.
M. A. Albrecht, S. L. Colegrove, J. Hongpaisan, et al., “Multiple modes of calcium-induced calcium release in sympathetic neurons I: attenuation of endoplasmic reticulum Ca2+ accumulation at low [Ca2+] during weak depolarization,” J. Gen. Physiol., 118, No. 1, 83−100 (2001); doi: https://doi.org/10.1085/jgp.118.1.83.
M. A. Albrecht, S. L. Colegrove, and D. D. Friel, “Dif- ferential regulation of ER Ca2+ uptake and release rates accounts for multiple modes of Ca2+-induced Ca2+ release,” J. Gen. Physiol., 119, No. 3, 211–233 (2002); doi: https://doi.org/10.1085/jgp.20028484.
S. D. Brenowitz and W. G. Regehr, “Reliability and heterogeneity of calcium signaling at single pre- synaptic boutons of cerebellar granule cells,” J. Neurosci., 27, No. 30, 7888–7898 (2007); doi: https://doi.org/10.1523/JNEUROSCI.1064-07.2007.
S. L. Colegrove, M. A. Albrecht, and D. D. Friel, “Quantitative analysis of mitochondrial Ca2+ uptake and release pathways in sympathetic neurons. Reconstruction of the recovery after depolarization-evoked [Ca2+] elevations,” J. Gen. Physiol., 115, No. 3, 371–388 (2000); doi: https://doi.org/10.1085/jgp.115.3.371.
T. Xu, M. Naraghi, H. Kang, and E Neher, “Kinetic studies of Ca2+ binding and Ca2+ clearance in the cytosol of adrenal chromaffin cells,” Biophys. J., 73, No. 1, 532–545 (1997); doi: https://doi.org/10.1016/S0006-3495(97)78091-3.
M.-H. Kim, N. Korogod, R. Schneggenburger, et al. “Interplay between Na+/Ca2+ exchangers and mito- chondria in Ca2+ clearance at the calyx of Held,” J. Neurosci., 25, No. 26, 6057−6065 (2005); doi: https://doi.org/10.1523/JNEUROSCI.0454-05.2005.
M. Patterson, J. Sneyd, and D. D. Friel, “Depolarization- induced calcium responses in sympathetic neurons: relative contributions from Ca2+ entry, extrusion, ER/mitochondrial Ca2+ uptake and release, and Ca2+ buffering,” J. Gen. Physiol., 129, No. 1, 29−56 (2007); doi: https://doi.org/10.1085/jgp.200609660.
E. D’Angelo, T. Nieus, A. Maffei, et al. “Theta- frequency bursting and resonance in cerebellar granule cells: experimental evidence and modeling of a slow K+-dependent mechanism,” J. Neurosci., 21 , No. 3 , 759 – 770 ( 2001 ) ; doi: https://doi.org/10.1523/JNEUROSCI.21-03-00759.2001.
J. Liou, M. Fivaz, T. Inoue, and T. Meyer, “Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after Ca2+ store depletion,” Proc. Natl. Acad. Sci. USA, 104, No. 22, 9301−9306 (2007); doi: https://doi.org/10.1073/pnas.0702866104.
M. Maravall, Z. F. Mainen, B. L. Sabatini, and K Svo- boda, “Estimating intracellular calcium concentrations and buffering without wavelength rationing,” Biophys. J., 78, No. 5, 2655−2667 (2000); doi: https://doi.org/10.1016/S0006-3495(00)76809-3.
T. Akita and K. Kuba, “Ca2+-dependent inactivation of Ca2+-induced Ca2+ release in bullfrog sympathetic neurons,” J. Physiol., 586, 3365−3384 (2008); doi: https://doi.org/10.1113/jphysiol.2008.153833.
M. L. Hines and N. T. Carnevale, “The NEURON simulation environment,” Neural Comput., 9, No. 6, 1179–1209 (1997); doi: https://doi.org/10.1162/neco.1997.9.6.1179.
F. Helmchen and D. W. Tank, “A single-compartment model of calcium dynamics in nerve terminals and dendrites,” Cold Spring Harb. Protoc., 2015, No. 2, 155−167 (2015); doi: https://doi.org/10.1101/pdb.top085910.
J. Klingauf and E. Neher, “Modeling buffered Ca2+ diffusion near the membrane: implications for secretion in neuroendocrine cells,” Biophys. J. 72, No. 2, Pt. 1, 674–690 (1997); doi: https://doi.org/10.1016/s0006-3495(97)78704-6.
S. P. Robertson, J. D. Johnson, and J. D. Potter, “The time course of Ca2+ exchange with calmodulin, troponin, parvalbumin, and myosin in response to transient increases in Ca2+,” Biophys. J., 34, No. 3, 559–569 (1981); doi: https://doi.org/10.1016/S0006-3495(81)84868-0.
M. Naraghi and E. Neher, “Linearized buffered Ca2+ diffusion in microdomains and its implications for calculation of [Ca2+] at the mouth of a calcium channel,” J. Neurosci., 17, No. 18, 6961–6973 (1997); doi: https://doi.org/10.1523/JNEUROSCI.17-18-06961.1997.
M.T. Alonso, M. J. Barrero, E. Carnicero, et al., “Functional measurements of [Ca2+] in the endoplasmic reticulum using a herpes virus to deliver targeted aequorin,” Cell Calcium, 24, No. 2, 87−96 (1998); doi: https://doi.org/10.1016/s0143-4160(98)90076-8.
A. Miyawaki, J. Lopis, R. Heim, et al., “Fluorescent indicators for Ca2+ based on green fluorescent proteins and calmodulin”, Nature, 388, No. 6645, 882−887 (1997); doi: https://doi.org/10.1038/42264.
K. H. Krause and M. Michalak, “Calreticulin,” Cell, 88, No. 4, 439–443 (1997); doi: https://doi.org/10.1016/s0092-8674(00)81884-x.
H. Mogami, J. Gardner, O. V. Gerasimenko, et al., “Calcium binding capacity of the cytosol and endo- plasmic reticulum of mouse pancreatic acinar cells”, J. Physiol., 518, Pt. 2, 463–467 (1999); doi: https://doi.org/10.1111/j.1469-7793.1999.0463p.x.
M. M. Wu, M Grabe, S. Adams, et al., “Mechanisms of pH regulation and the regulated secretory pathway,” J. Biol. Chem., 276, No. 35, 33027–33035 (2001); doi: https://doi.org/10.1074/jbc.M103917200.
E. De Schutter and P. Smolen, “Calcium dynamics in large neuronal models”, in: Methods in Neuronal Modeling: from Ions to Networks, C. Koch and I. Segev (Eds.), 2nd ed., MIT Press, Cambridge (1998), pp. 211−250.
M. D. Stern, “Theory of excitation–contraction coupling in cardiac muscle,” Biophys J., 63, No. 2, 497−517 (1992); doi: https://doi.org/10.1016/S0006-3495(92)81615-6.
E. Eggermann and P. Jonas, “How the ‘slow’ Ca2+ buffer parvalbumin affects transmitter release in nanodomain- coupling regimes,” Nat. Neurosci., 15, No. 1, 20–22 (2011); doi: https://doi.org/10.1038/nn.3002.
C. M. Hackney, S. Mahendrasingam, A. Penn, and R. Fettiplace, “The concentrations of calcium buffering proteins in mammalian cochlear hair cells”, J. Neurosci., 25, No. 34, 7867–7875 (2005); doi: https://doi.org/10.1523/JNEUROSCI.1196-05.2005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saftenku, E. Modes of Operation of the Endoplasmic Reticulum Ca2+ Transport Systems in Neurons: Insights from the Compartmental Models. Neurophysiology 54, 2–13 (2022). https://doi.org/10.1007/s11062-023-09930-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-023-09930-6