Abstract
Brain metastases are major complications of common cancers. Tumor type and proneness to the CNS are thought to define the number and size of brain metastases. It is not known if intrinsic vascular factors can also have an effect. Restricted perfusion due to cerebral small vessel disease is frequent in elderly patients and causes white matter lesions (WML). The aim of this analysis was to evaluate a possible negative effect of WML and patient age on the number and size of brain metastases (BM) of different tumor entities. Pre-therapeutic 3 T brain magnetic resonance imaging (MRI) of 200 patients with BM were analyzed. Location, size and number of BM (NoM) were determined. T2 hyperintensive WML were scored according to Fazekas-Score (grade I–III). Patients with WML grade 1 (NoM: 5.59; p = 0.009) and grade 2 (NoM: 3.68; p = 0.002) had significantly less BM than patients without WML (NoM: 6.99). This effect was present in subgroups of different tumors: NSCLC (p = 0.05), other tumors than NSCLC (p = 0.048). Age (≤65 or >65 years) was positively correlated with the degree of WML but not with number (pNoM = 0.832) or mean diameter (pmDM = 0.662) of brain metastases. While patient age did not appear to be relevant, increasing WML were associated with lower number of brain metastases in different tumor types.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerebral metastases are severe complications in patients with cancer. They occur in up to 40 % of patients with solid tumors [1]. According to diagnosis specific graded prognostic assessment (GPA), the number of brain metastases has prognostic impact in several tumors (lung cancer, melanoma, renal cell cancer) [2]. In addition, the number of metastases (NoM) is crucial in determining treatment, i.e. local or whole brain therapy [1, 3, 4]. Interestingly, factors that determine metastasis number are poorly described. It is more or less taken for granted that systemic tumor control is the major determinant, combined with CNS-proneness of tumor cells [5–7]. The extent of tissue perfusion has been shown to influence the distribution of brain metastases, i.e. most metastases develop in well perfused areas (e.g. the border of white and grey matter) [8] while less perfused areas (e.g. deep white matter) appear virtually protected from metastases [9, 10]. Whether vascular integrity or fragility can influence the NoM is not well investigated. The aim of this analysis was to evaluate a possible negative effect of cerebral small vessel disease (indicated by white matter lesions) and the patient age on the number and size of brain metastases.
Material and methods
Charts of all patients with the diagnosis “brain metastasis or brain metastases” that presented at the University Hospital Leipzig from October 2004—January 2015 were screened. Patients with different tumor entities at the time of initial diagnosis of BM were included in this study. 200 consecutive patients were included that fulfilled the following criteria: (a) complete and available patient chart with history and age and (b) 3 T cerebral MRI with 3D dataset of T1-weighted sequences after contrast agent injection (slice thickness of 1–1.5 mm) and T2/FLAIR sequence pre-treatment available. Patients were divided according to age and white matter lesion grade (WML 0–III).
Grading of white matter lesions
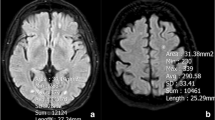
Initially, white matter hyperintensities in axial T2/FLAIR sequences of cerebral MRI were graded based on the Fazekas score [11]. In the manuscript, the term “T2-/FLAIR white matter hyperintensities” is replaced by the shorter synonymous term “white matter lesions” (WML). Degree of WML was defined as follows: no WML (grade 0), punctate WML (grade 1), beginning confluence of WML (grade 2) and large confluent areas (grade 3). Representative examples of WML grades are shown in Fig. 1.
MRI analysis of location, number and size of metastases
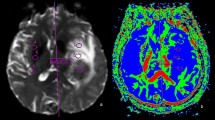
Secondly, the NoM was determined on axial 3D T1-weighted sequences of cerebral MRI, and the maximum axial diameter of each individual metastasis was measured. Metastases situated in deep white matter were termed white matter metastasis (WMM) as these might have a different vascular background than metastases at the border of grey and white matter [12]. The per-patient mean diameter of all metastases (mDM) was determined.
Age
Patients were split into two groups: (1) ≤65 years and (2) >65 years. In a second step, patients were subgrouped in seven groups: (1) >30–≤40 years, (2) >40–≤50 years, (3) >50–≤60 years, (4) >60–≤70 years, (5) >70–≤80 years, (6) >80–≤90 years, (7) 90–≤100 years.
Statistical analysis
IBM SPSS V22.0 was used for statistical analysis. At the beginning, the Kolmogorov–Smirnov test was used for testing normal distribution [13]. The data (NoM, mDM) was not normally distributed (p = 0.001). Therefore, statistical evaluation was adjusted to Mann–Whitney-U-Test (Ngroup = 2) or multiple Independent-Samples Kruskal–Wallis-Test (Ngroup > 2) described as p-value (p) [14]. Statistical significance was accepted at p < 0.05. Univariate (UVA) and multivariate data analyses (MVA) were combined. Multivariate analyses allowed an evaluation of more than one statistical variable [15]. By using Pearson’s Chi square test of Independence (pχ2), number and diameter of BM were analyzed on correlation with different other variables (e.g. age, WML grade, WMM). Figures were made with IBM SPSS V22.0 and Microsoft office version 2015.
BAB, SN performed MRI analysis under supervision of CS (7 years experience in neuroimaging). KH and SP collected clinical data. BAB, SN and CS performed statistic analysis and wrote the article with valuable input of CG, KTH and RDK.
Before initiation, the study design was evaluated and officially accepted by the local ethics committee.
Results
Patient characteristics
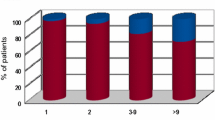
Patients were predominantly male (60.5 %, n = 121) and had a median age of 64.5 years with a range from 32 to 91 years. 47.5 % (n = 95) were older than 65 years. In patients ≤65 years, median age was 57 years (nmale = 56, nfemale = 49), while median age in patients >65 years was 72 years (nmale = 65, nfemale = 30) (Fig. 2a, b). The population comprised patients with non-small-cell lung cancer (NSCLC) (n = 89), small-cell lung cancer (SCLC) (n = 14), breast cancer (BC) (n = 20), malignant melanoma (MM) (n = 30), renal cell carcinoma (RCC) (n = 17), colorectal cancer (CRC) (n = 10) and patients with other tumors (OT) (n = 20). Patients with RCC were significantly older (73 years) than patients with BC (58 years, p = 0.001), MM (61 years, p = 0.009) and NSCLC (63 years, p = 0.005) or other tumors (62.7 years, p = 0.04). 31 of 200 patients (15.5 %) had a singular cerebral metastasis, 42 patients (21 %) had two metastases, 91 patients (45.5 %) 3–9 metastases and 36 patients had >9 metastases.
Patient groups and age-dependency of white matter hyperintensities. a Distribution of patients ≤65 years and older than 65 years. b Distribution of patients according to decades of age. c Patient age and different WML grades. d Correlation plot of patient age and WML grades. Data is displayed in boxplots (Box interquartile range (IQR), horizontal line median, whiskers max 1.5× IQR, N number of patients)
Frequency of white matter lesions
Overall, 88 (44 %) patients had no white matter lesions (WML0), while 74 (37 %) showed punctate foci (WML1), 37 patients (18.5 %) had confluent foci (WML2) and only in one case were (0.5 %) large confluent areas observed (WML3). WML were more frequent and severe in NSCLC, RCC and CRC (all median WML1) compared to MM, BC and SCLC (all median WML0).
Correlation of age and white matter lesions
A significant positive correlation between age and WML was identified by Pearson’s Chi square test of Independence (pχ2 = 0.001; r = 0.422). This was confirmed in Kruskallis–Wallis-Test of independent samples. There was a tendency of linear relation between age and grade of WML (Fig. 2).
White matter lesions and number and size of brain metastases
Across all tumor types the NoM significantly differed between WML grades (pNoM = 0.013). Grade 1 (Mean NoM = 5.59, Median = 3, p = 0.009) and grade 2 (Mean NoM = 3.68, Median = 3, p = 0.002) had significant less BMthan WML grade 0 (Mean NoM = 6.99, Median = 5), (Fig. 3a, b). Pearson’s Chi square test revealed significant negative correlation of NoM with increasing WML grade (pχ2 = 0.002, r = − 0.22). In patients with 1–2 or 3–9 brain metastases, the median WML was one, in patients with >9 metastases median WML was 0 (pχ2 = 0.009, r = − 0.18).
Influence of WML on mean number (a–c) and diameter (d) of BM. a Mean number of metastases according to WML grade. b Mean number of metastases according to WML grade in patients ≤65 and >65 years. c Mean number of metastases in patients with NSCLC and other tumors than NSCLC according to WML grade. d Mean diameter of metastases in mm according to WML grade. Data is displayed in boxplots (Box interquartile range (IQR), horizontal line median, whiskers max 1.5× IQR, N number of patients)
Referring to tumor type, patients with NSCLC and WML Grade 1 (Mean NoM = 6.79, Median = 4, p = 0.038) and grade 2 (Mean NoM = 2.88, Median = 2, p = 0.013) had significantly less BM than grade 0 (Mean NoM = 6.59, Median = 5). In the group of patients with tumors other than NSCLC (SCLC + MM + BC + RC + CRC + OT, N = 111 patients) the same observation of decreasing NoM with increasing WML was made (p = 0.05), (Table 1; Fig. 3c).
In contrast to NoM, mean diameter of BM was not different between all grades of WML (pmDM = 0.806) (Fig. 3d).
WML and white matter metastases
The number of patients with white matter metastases was low (N = 19, total number of WMM: 19). No significant correlation of white matter metastases with WML grade was seen (pχ2 = 0.67, r = −0.03).
Influence of age on number and size of brain metastases
Significant effects of age on number and size of cerebral metastases were not found, neither between higher/lower aged group (pNoM = 0.832; pmDM = 0.662) nor between the 10 years subgroups (pNoM = 0.968; pmDM = 0.848). The amount of whiter matter metastasis (WMM) was not affected by age (p = 0.3); no correlation was identified (pχ2 = 0.457).
Discussion
WML are signs of small vessel disease of the brain. Small vessel disease of the brain is characterized by histopathological changes, such as loss of structure in arteriolar walls, narrowing of the vessel lumen and thickening of the vessel walls [16–19]. WML are common in patients with vascular risk factors like arterial hypertension [19–21] and are associated with strokes, dementia [21, 22] and intracerebral bleeding (ICB) [16, 23, 24]. Furthermore, after stroke or ICB poor outcomes are reported in presence of WML [24]. Small vessel disease leads to reduced perfusion and chronic ischemia in deep brain structures [16, 19, 25]. Perfusion is essential for the process of brain metastasation [26]. Accordingly, a protective effect of small vessel disease against BM would be coherent. In our analysis, patients with WML had less BM than patients without WML. This effect was independent from patient age, correlated with the degree of WML, and was observed across different tumors. This implies that not only tumor type and proneness to CNS [5–7, 26] define the number of brain metastasis, but also the integrity of blood vessels.
Mazzone et al. [27] found the first indication of this relationship when they compared groups of patients with and without brain metastasis of NSCLC and compared the degree of WML between these groups. With the recent analysis of a differently composed patient cohort (various cancers, all patients with brain metastases) we can verify these findings by the use of more precise thin-sliced (1–1.5 mm) 3 T MRI with 3D T1 dataset. Further, we can extend an inverse relationship between WML-grade and BM from NSCLC to other tumor types. This appears relevant as different tumor types may utilize different forms of vasculogenesis (vascular cooption or angiogenesis [28, 29]). For example, the effects of WML may have only been present in NSCLC and not in other cancers. From our results, WML appear to reduce the development of BM in different tumors, however, our data is limited by small sample sizes in less frequent tumors (e.g. CRC, RCC).
Tumor entity dependent studies with larger patient cohorts are necessary to examine this suggestion for all tumor types. Most likely, cancers with proneness to the CNS (NSCLC, SCLC, MM) that rely on vascular cooption [30] can be influenced by WML. Whether the same applies to more angiogenic tumors (e.g. RCC [30, 31]), remains unclear. Additionally, it is not clear which step of brain metastasis—seeding or vasculogenesis [24, 26]—is impeded by WML.
Interestingly, deep white matter metastasis was not affected by WML. This could be due to small vessel disease inhibiting the cooption of existing blood vessels rather than prevention of neo-angiogenesis. This remains a hypothesis, as the total number of deep white matter metastases in our dataset was too low for appropriate statistical analysis.
Clinical studies have demonstrated that an alteration of vascular architecture occurring in long-standing diabetes mellitus patients could be a protective factor against metastases from lung cancer [32]. The mechanism of WML-induced modification of cerebral blood flow autoregulation, resulting from arterial hypertension and vessel wall thickening, has previously been suspected [33–35]. The relationship between single vascular risk factors, WML and brain metastasis in different tumor entities will be an interesting future field of research.
A surprising fact is the very small proportion of patients with heavy WML in our patient cohort. This could point to a nearly exclusive effect of very severe WML on brain metastasis. Regarding therapy, patients with severe WML appear not to be good candidates for WBRT, as their risk for development of multiple metastases appears naturally reduced; benefits of whole brain treatment might be negligible. Coherent with this notion is the lack of a survival benefit from WBRT compared to stereotactic radiotherapy in elderly patients with few metastases [3–5]. On the contrary, chronic ischemia of small vessel disease might be exacerbated by WBRT induced angiopathy [1, 36]. However, to fully answer this question, prospective data with different treatment arms is desirable.
In conclusion, there is increasing evidence for a negative correlation between small vessel disease and the number of brain metastases. WML may serve as a predictor of number and distribution of BM independent of tumor type. In future studies of this relationship quantitative MRI-measurements of brain perfusion e.g. like in [37] should be performed. Whether the knowledge about the negative impact of WML on BM may influence the treatment of BM remains to be answered in the future.
References
Soffietti R, Ruda R, Mutani R (2002) Management of brain metastases. J Neurol 249(10):1357–1369
Sperduto PW, Kased N, Roberge D, Xu Z, Shanley R, Luo X, Sneed PK, Chao ST, Weil RJ, Suh J, Bhatt A, Jensen AW, Brown PD, Shih HA, Kirkpatrick J, Gaspar LE, Fiveash JB, Chiang V, Knisely JP, Sperduto CM, Lin N, Mehta M (2011) Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol 30(4):419–425
Nussbaum ES, Djalilian HR, Cho KH, Hall WA (1996) Brain metastases. Histology, multiplicity, surgery, and survival. Cancer 78(8):1781–1788
Patil CG, Pricola K, Garg SK, Bryant A, Black KL (2010) Whole brain radiation therapy (WBRT) alone versus WBRT and radiosurgery for the treatment of brain metastases. Cochrane Database Syst Rev (6):CD006121
Ellis TL, Neal MT, Chan MD (2012) The role of surgery, radiosurgery and whole brain radiation therapy in the management of patients with metastatic brain tumors. Int J Surg Oncol 2012:952345
Carbonell WS, Ansorge O, Sibson N, Muschel R (2009) The vascular basement membrane as “soil” in brain metastasis. PLoS One 4(6):e5857
Donnem T, Hu J, Ferguson M, Adighibe O, Snell C, Harris AL, Gatter KC, Pezzella F (2013) Vessel co-option in primary human tumors and metastases: an obstacle to effective anti-angiogenic treatment? Cancer Med 2(4):427–436
Ge Y, Grossman RI, Babb JS, Rabin ML, Mannon LJ, Kolson DL (2002) Age-related total gray matter and white matter changes in normal adult brain. Part I: volumetric MR imaging analysis. AJNR Am J Neuroradiol 8:1327–1333
Klein B, Kuschinsky W, Schrock H, Vetterlein F (1986) Interdependency of local capillary density, blood flow, and metabolism in rat brains. Am J Physiol 6:1333–1340
Ellenbogen RG, Abdulrauf SI, Sekhar N (2012) Principles of neurological surgery. Saunders/Elsevier, Philadelphia
Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA (1987) MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol 149(2):351–356
Seidel C, Hambsch P, Hering K, Bresch A, Rohde S, Kortmann RD, Gaudino C (2015) Analysis of frequency of deep white matter metastasis on cerebral MRI. J Neurooncol 123(1):135–139
Ebermann E (2016) Kolmogorov-Smirnov-Test mit SPSS. https://www.univie.ac.at/ksa/elearning/cp/quantitative/quantitative-62.html. Accessed 15 May 2016
Bühl A (2014) SPSS 22: einführung in die moderne datenanalyse (pearson studium—scientific tools), 14th edn. Pearson Deutschland GmbH, Hallbergmoos, Germany
Johnson, RA, Wichern, Dean W (2007) Applied multivariate statistical analysis, 6th edn. Pearson Education Limited, Essex
Pantoni L (2010) Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol 9:689–701
Gouw AA, Seewann A, van der Flier WM, Barkhof F, Rozemuller AM, Scheltens P, Geurts JJ (2011) Heterogeneity of small vessel disease: a systematic review of MRI and histopathology correlations. J Neurol Neurosurg Psychiatr 82:126–135
Brown WR, Thore CR (2011) Review: cerebral microvascular pathology in ageing and neurodegeneration. Neuropathol Appl Neurobiol 37(1):56–74
Okroglic S, Widmann CN, Urbach H, Scheltens P, Heneka MT (2013) Clinical symptoms and risk factors in cerebral microangiopathy patients. PLoS One 8(2):e53455
Spolveri S, Baruffi MC, Cappelletti C, Semerano F, Rossi S, Pracucci G, Inzitari D (1998) Vascular risk factors linked to multiple lacunar infarcts. Cerebrovasc Dis 8(3):152–157
Brown WR, Moody DM, Thore CR, Anstrom JA, Challa VR (2009) Microvascular changes in the white mater in dementia. J Neurol Sci 283(1–2):28–31
Breteler MM, van Swieten JC, Bots ML, Grobbee DE, Claus JJ, van den Hout JH, van Harskamp F, Tanghe HL, de Jong PT, van Gijn J et al (1994) Cerebral white matter lesions, vascular risk factors, and cognitive function in a population-based study: The Rotterdam Study. Neurology 44:1246–1252
Yoo DH, Song SW, Yun TJ, Kim, Lee SH, Kim JH, Sohn CH, Park SH, Park CK, Kim IH, Choi SH (2015) MR imaging evaluation of intracerebral hemorrhages and T2 hyperintense white matter lesions appearing after radiation therapy in adult patients with primary brain tumors. PLoS One 10(8):e0136795
Sato S, Delcourt C, Heeley E, Arima H, Zhang S, Al-Shahi Salman R, Stapf C, Woo D, Flaherty ML, Vagal A, Levi C, Davies L, Wang J, Robinson T, Lavados PM, Lindley RI, Chalmers J, Anderson CS, INTERACT2 Investigators (2016) Significance of cerebral small-vessel disease in acute intracerebral hemorrhage. Stroke 47(3):7017
Pantoni L, Garcia JH (1997) Cognitive impairment and cellular/vascular changes in the cerebral white matter. Ann NY Acad Sci 826:92–102
Gavrilovic IT, Posner JB (2005) Brain metastases: epidemiology and pathophysiology. J Neurooncol 75(1):5–14
Mazzone PJ, Marchi N, Fazio V, Taylor JM, Masaryk T, Bury L, Mekhail T, Janigro D (2009) Small vessel ischemic disease of the brain and brain metastases in lung cancer patients. PLoS One 4(9):e7242
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Carmeliet P, Rakesh KJ (2011) Molecular mechanisms and clinical application of angiogenesis. Nature 473(7347):298–307
Berghoff AS, Ilhan-Mutlu A, Dinhof C, Magerle M, Hackl M, Widhalm G, Hainfellner JA, Dieckmann K, Pichler J, Hutterer M, Melchardt T, Bartsch R, Zielinski CC, Birner P, Preusser M (2015) Differential role of angiogenesis and tumor cell proliferation in brain metastases according to primary tumor type: analysis of 639 cases. Neuropathol Appl Neurobiol 41(2):e41–e55
Donnem T, Hu J, Ferguson M, Adighibe O, Snell C, Harris AL et al (2013) Vessel co-option in primary human tumors and metastases: an obstacle to effective anti-angiogenic treatment? Cancer Med 2(4):427–436
Hanbali A, Al-Khasawneh K, Cole-Johnson C, Divine G, Ali H (2007) Protective effect of diabetes against metastasis in patients with non-small cell lung cancer. Arch Intern Med 167(5):513
Quattrocchi CC, Errante Y, Mallio CA, Carideo L, Scarciolla L, Santini D, Tonini G, Zobel BB (2014) Inverse spatial distribution of brain metastases and white matter hyperintensities in advanced lung and non-lung cancer patients. J Neurooncol 120(2):321–330
Bakker SL, de Leeuw FE, de Groot JC, Hofman A, Koudstaal PJ, Breteler MM (1999) Cerebral vasomotor reactivity and cerebral white matter lesions in the elderly. Neurology 52:578–583
Kozera GM, Dubaniewicz M, Zdrojewski T, Madej-Dmochowska A, Mielczarek M, Wojczal J, Chwojnicki K, Swierblewska E, Schminke U, Wyrzykowski B, Nyka WM, SOPKARD Study Group (2010) Cerebral vaso- motor reactivity and extent of white matter lesions in middle-aged men with arterial hypertension: a pilot study. Am J Hypertens 23:1198–1203
Burns TC, Awad AJ, Li MD, Grant GA (2016) Radiation-induced brain injury: low-hanging fruit for neuroregeneration. Neurosurg Focus 40(5):E3
Nasel C, Boubela R, Kalcher K, Moser E (2016) Normalised time-to-peak-distribution curves correlate with cerebral white matter hyperintensities—Could this improve early diagnosis? J Cereb Blood Flow Metab. [Epub ahead of print]
Acknowledgments
B.A.B was recipient of a merit-based doctoral scholarship from the Hans-Böckler Foundation funded by the Federal Ministry of Education and Research—Germany (BMBF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Benjamin-Andreas Berk and Sandra Nagel are co-first authors.
Rights and permissions
About this article
Cite this article
Berk, BA., Nagel, S., Hering, K. et al. White matter lesions reduce number of brain metastases in different cancers: a high-resolution MRI study. J Neurooncol 130, 203–209 (2016). https://doi.org/10.1007/s11060-016-2235-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2235-5