Abstract
Immunotherapy is coming to the fore as a viable anti-cancer treatment modality, even in poorly immunogenic cancers such as glioblastoma (GBM). Accumulating evidence suggests that the central nervous system may not be impervious to tumor-specific immune cells and could be an adequate substrate for immunologic anti-cancer therapies. Recent advances in antigen-specific cancer vaccines and checkpoint blockade in GBM provide promise for future immunotherapy in glioma. As anti-GBM immunotherapeutics enter clinical trials, it is important to understand the interactions, if any, between immune-based treatment modalities and the current standard of care for GBM involving chemoradiation and steroid therapy. Current data suggests that chemoradiation may not preclude the success of immunotherapeutics, as their effects may be synergistic. The future of therapy for GBM lies in the power of combination modalities, involving immunotherapy and the current standard of care.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immunotherapy is rapidly becoming one of the pillars of anti-cancer therapy. While previously abandoned as an implausible cancer therapeutic, recent advances in the understanding of immune checkpoint molecules and cancer-specific antigens has propelled immunotherapeutics toward becoming robust anti-cancer agents. Immunotherapy has come to the fore of anti-cancer therapy with the approval in 2010 of sipuleucel-T, the first antigen-specific vaccine for castration-resistant prostate cancer, and in 2011 the approval of Ipilimumab, the first checkpoint inhibitor for advanced melanoma [1, 2]. While a number of cancers have been amenable to immunotherapeutic approaches—melanoma for its immunogenicity, prostate for its prevalence of characterized and targetable antigens—Glioblastoma (GBM) has not received similar clinical successes, likely for its poor immunogenicity, few characterized cancer antigens, and for its location in the immunologically distinct CNS.
The most prevalent brain tumor, GBM is associated with a dismal median overall survival of 1–2 years and a 5-year survival rate of <10 % [3–5]. Temozolomide, surgical resection and radiation comprise the current standard of care for GBM, but even standard therapy has only modestly improved overall survival [6]. Nevertheless, preclinical studies suggest that immunotherapies elicit significant anti-tumor responses in GBM, and that the CNS may not be an immunologically exclusive entity. Moreover, current immunotherapies may have the potential to work synergistically with the standard of care for GBM, providing promise for improvements in overall survival beyond the current standard. Immunotherapeutic agents have begun to alter the landscape of anti-tumor therapy for a number of cancer types, and GBM is no exception.
Principles of immunotherapy
The role of the immune system in the control and progression of tumor growth has been well described [7]. The adaptive immune response selectively destroys tumor cells displaying unique cancer antigens, initially protecting the body from aberrant cancer growth. Highly immunogenic tumors will arise in the absence of such effector mechanisms, as in Rag2-deficient mice that lack T, B, and Natural Killer (NK) cells [8]. While protective at the initiation of tumor cell proliferation, the selective pressure placed by immune effector cells on tumor growth results in the outgrowth of cancer cells that do not express immunogenic molecules [7, 9]. This process of immune-mediated editing of the tumor microenvironment has been described in three phases: elimination of immunogenic tumor cells by adaptive and innate immune responses, equilibrium between tumor cell growth and anti-tumor immunity, and escape of poorly immunogenic cancer cells that proliferate and outpace immune-mediated anti-tumor activity [9]. The basis of anti-cancer immunotherapy is to counteract immune evasion of cancer cells by priming, augmenting, or disinhibiting the effector immune response in the tumor microenvironment. Immunotherapeutic modalities may be divided into three major approaches: cytokine-based, passive therapy, an active immunotherapy.
Cytokine therapy
Cytokines are broad activators of immune function that stimulate anti-tumor effector cell proliferation. The earliest studies in immunotherapy tested intra-peritoneal injections of interferon in mice injected with Ehrlich ascites tumor cells; inhibition of tumor growth and increased survival were directly correlated to interferon therapy administered after tumor inoculation [10]. At a basic level, cytokines were understood to be potent regulators of immune activity, and the observation that their ability to stimulate anti-tumor immune function began decades of study in their use as anti-cancer agents [11]. Gamma-chain cytokines are the most widely studied class of cytokines, including interleukins IL-2, IL-7, IL-15, and IL-21 [12]. Interleukin-7 is responsible for T cell development and up-regulation on effector T-cells destined to become memory cells [13, 14]. Preclinical studies have shown increased anti-cancer immune-mediated cytotoxicity and IFN-γ expression in murine colon cancer treated with IL-7, while mouse sarcoma and glioma models have demonstrated tumor rejection mediated by CD8+ T-cells after IL-7 exposure [15–18]. A phase I study in renal cell carcinoma (RCC) treated with an IL-7-expressing tumor vaccine demonstrated increased IL-10 expression mediated by Th2 effector cells in responders, and increased circulating CD4+, CD8+ and CD3+ T-cells were observed in refractory solid cancers treated with IL-7 [19, 20]. Interleukin-15 is involved in CD8+ memory T-cell maintenance and NK cell maturation. While preclinical studies are limited to an increase in antigen-specific T-cells after culturing with IL-15 in melanoma and plasmacytoma, a number of phase I studies are ongoing in NSCLC, RCC, squamous head and neck cancer, and melanoma [21–23]. Produced by CD4+ T-cells, IL-21 possesses the dual effect of inhibiting regulatory T-cell (Treg) function while stimulating CD4+, CD8+ and NK cells [24–26]. Melanoma treated with IL-21 in a phase 2 study demonstrated a 22.5 % overall response rate, 12.4 month overall survival, and a progression free survival of 4.3 months [27]. Non-gamma chain cytokine IL-12 in combination with cytotoxic T-lymphocyte antigen-4 (CTLA-4) blockade has produced tumor regression in a syngeneic model of advanced glioma [28] (Fig. 1).
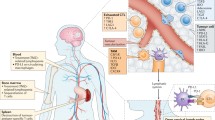
Immunologic anti-tumor strategies: T-cell stimulation occurs via cytokine binding to cognate receptors and cross presentation of vaccine-administered antigens by dendritic cells; monoclonal antibodies (mAb) to co-inhibitory checkpoint receptors such as programmed death-1 (PD-1) and cytotoxic T-lymphocyte antigen-4 (CTLA-4) block inhibitory interactions between the tumor cell and T-cell
While cytokine therapy is capable of inducing broad effector immune responses, this potentially anti-tumor effect is also responsible for widespread toxicity caused by systemic interleukin administration alone [12]. The value of cytokines may be as adjunctive therapies alongside immunotherapies such as adoptive T-cell transfer. Interleukin-2 is the only FDA-approved cytokine indicated in metastatic RCC for its ability to cause high rates of tumor regression [29]. While used successfully alone in the treatment of metastatic cancers, IL-2 garnered great utility for its ability to expand cancer-specific T-cells ex vivo and preserve the growth and number of adoptively transferred T-cells in vivo [30, 31]. Adoptively transferred autologous tumor-infiltrating T-cells administered alongside IL-2 in patients with melanoma resulted in 22 % complete tumor regression and a 5-year survival of 29 % [31].
Immune checkpoint blockade
Tumor-targeted antibody therapy encompasses antibodies that target tumor antigen to result in tumor cell death as well as antibodies that prevent tumor-mediated immune inhibition, also known as antibody-based immune checkpoint blockade. This section will focus on the mechanisms of immune checkpoints. Inhibitory immune checkpoints are responsible for down-modulating immune function to prevent autoimmunity in times of inflammation. Ligand-checkpoint receptor interactions may up- or down-regulate lymphocyte activity via a series of intracellular signaling cascades. Poorly immunogenic tumors have appropriated this machinery by expressing the ligands of inhibitory checkpoints and dampening the effector function of immune cells with the potential for anti-tumor activity. Programmed death-1 and CTLA-4 are two of such inhibitory immune checkpoints, among many others [32]. Parsa and colleagues have found that the loss of tumor suppressor PTEN in some GBM cells results in overexpression of B7 homolog 1 (B7-H1), also known as programmed death ligand-1 (PDL1). While an important finding, the actual expression profile of PDL1 in GBM is as yet unclear. Engagement of PD-1 with PDL1 in the periphery results in decreased activity of antigen-experienced T-cells and subsequent T-cell exhaustion and anergy. This system prevents autoimmunity in peripheral inflammatory responses, but can be utilized by PDL1-expressing tumors to depress cytotoxicity of lymphocytes in the tumor microenvironment. The combination of antibody mediated PD-1 blockade and stereotactic radiation in a murine intracranial glioma model has demonstrated significantly improved survival and tumor regression, suggesting synergism between PD-1 blockade and radiotherapy [33]. Cytotoxic T-lymphocyte antigen-4 (CTLA-4) is a co-inhibitory receptor that outcompetes stimulatory CD28 for binding to CD80 and CD86 [34, 35]. The inhibitory effects of CTLA-4 occur largely in naïve and resting T-cells, and act to down-regulate T-cell effector function and augment the inhibitory activity of Tregs. The combination of anti-CTLA-4 immunotherapy and radiation has induced tumor regression and prevention of metastases in a murine breast cancer model, and this effect was found to be mediated largely by CD8+ T-cells [36]. Combination checkpoint blockade has led to significant strides in anti-cancer therapy, with the combination of nivolumab (anti-PD-1, trade name Opdivo) and Ipilimumab (anti-CTLA-4, trade name Yervoy) resulting in a 53 % response rate with a manageable safety profile in patients with melanoma [37]. While anti-PD-1 and anti-CTLA-4 therapies are the closest to clinical use, a number of immune checkpoints, including TIM-3, GITR, and KIR, are in pre-clinical studies and may play a future role in clinical therapy.
Vaccine therapy
Active immunotherapy includes vaccine therapy targeted to tumor antigens. Cancer vaccines come in a number of forms, including dendritic cell (DC) vaccines that are loaded with tumor lysate, pulsed with specific MHC I derived tumor peptide, or injected directly into the tumor. While the clinical experience with anti-cancer vaccines overall has been disappointing, antigen-specific vaccines against GBM are entering clinical trials with initially promising results. One such DC vaccine for glioma involves the antigen epidermal growth factor receptor (EGFR), which is a transmembrane tyrosine kinase that is overexpressed in a structurally rearranged form in approximately 50 % of malignant gliomas and contributes to increased oncogenicity [38–40]. In human gliomas, the most common EGFR mutation is variant III (EGFRvIII); as such, EGFRvIII has become a major target of DC-based anti-glioma immunotherapy [41, 42]. A phase III clinical trial (ACT IV) is ongoing in newly diagnosed EGFRvIII positive GBM treated with CDX-110 (PEPvIII-KLH vaccine), a vaccine directed against a mutated segment of EGFRvIII (PEP-3). A phase II study (ReAct) is also studying the effects of CDX-110, this time in those with recurrent GBM that is refractory to bevacizumab. While checkpoint blockade and vaccine therapy have independently shown promise in pre-clinical and clinical models of GBM, it may be that the future of immunotherapeutics may be in combination regimens, involving current standard therapies and multiple types of immunotherapies targeting distinct anti-tumor immune mechanisms.
The current state of immunotherapeutics
The recent surge in discovery of unique cancer antigens and molecular modulators of immune function has catapulted immunotherapeutic agents such as anti-cancer vaccines and immune checkpoint inhibitors into clinical trials, with promising results (Table 1). The recent FDA approval of Pembrolizumab, the first approved anti-PD-1 therapy, has placed anti-PD-1 alongside anti-CTLA-4 (Ipilimumab) as the only immune checkpoints approved for use in humans. Immunotherapies are being actively investigated in a number of human cancers, including GBM, and may represent a paradigm shift in the treatment of disease resistant to standard therapies. Checkpoint inhibitors Ipilimumab and anti-PD-1 agents lambrolizumab and nivolumab have demonstrated improved survival and significant tumor regression in melanoma [43–45]. Even in poorly immunogenic tumors such as GBM, vaccines such as those targeted to heat shock protein-96 (HSP-96) are being pursued as safe and potentially efficacious treatments, with a median survival of 42.6 weeks in a phase I trial of HSP-96 [46]. A study of a tumor-associated antigen vaccine of DCs pulsed with multiple peptides resulted in median overall survival of 38.4 months [47], and a number of phase I and II trials involving tumor antigen associated vaccines are currently underway in GBM (NCT02078648, NCT01280552, NCT01920191). Currently, the most promising tumor specific antigen vaccine is rindopepimut, a synthetic peptide vaccine of the splice site of EGFRvIII, which is expressed in approximately 30 % of GBMs [39]. Phase I and II trials of rindopepimut have confirmed safety and elicited anti-tumor responses specific to EGFRvIII, albeit patients in these studies did not have high tumor burden or poor performance status [40, 42, 48]. A randomized controlled trial of rindopepimut is now underway in EGFRvIII positive, newly diagnosed GBM (NCT01480479). In addition to a number of ongoing studies of peptide vaccines in GBM, checkpoints are under active study in advanced glioma, such as the combination of Ipilimumab with nivolumab versus nivolumab alone [49]. Immunotherapies are no longer potential treatments limited to pre-clinical models and early phase clinical studies. The introduction of immune checkpoint inhibitors in the clinic has ushered in an era of immune-modulating therapeutics that may be used alone or alongside standard therapies to combat a wide range of cancers.
The CNS: immune-privileged or immune-discrete?
The previously prevailing notion that the CNS is an immunologically privileged site is now challenged by a number of pre-clinical and clinical observations. Survival of engrafted tissue in the CNS initially supported the idea that the CNS is not immunologically robust, but later observations of tumor-mediated breakdown of the blood–brain barrier (BBB), incursion of immune cells, and excursion of CNS antigens to cervical lymphatics recapitulated the CNS as an immunologically dynamic organ and not an immunologically distinct compartment [50–52]. Antigens or antigen-loaded APCs from the CNS may migrate to draining cervical lymph nodes via interstitial fluid of perivascular spaces or in the cerebrospinal fluid through the cribiform plate and to the nasal mucosa [53–55]. Moreover, there is clinical evidence for the role of immunosurveillance in CNS tumors—downregulation of HLA class I in GBM and low circulating CD4+ T-lymphocytes caused by standard therapy for GBM portend poorer survival [56, 57].
Supporting such clinical findings, preclinical data illuminates the interplay of peripherally activated T-cells with CNS targets. While T-cells have been found to enter CNS tissue at random, antigen-specific T-lymphocytes are retained within the CNS to induce inflammation [58]. Tumor antigen-specific T-cells may proliferate rapidly and home to tumor and draining lymph nodes if provided an appropriate immune stimulus, made possible by the up-regulation of genes responsible for cellular locomotion and chemokine adhesion [59, 60]. Finally, the CNS tumor microenvironment may foster further differentiation and enhanced effector function of antigen-specific T-cells, reflected by increased expression of interferon-γ and granzyme B [61].
Microglia are derived from the common myeloid progenitor, but are phenotypically distinct from macrophages and monocytes [62]. The phagocytic cells of the CNS, microglia are responsible for protecting the neuronal architecture from invading pathogens while minimizing inflammation damaging to the brain parenchyma. A homeostatic mechanism for repopulation of depleted microglia by circulating monocytes has been described, indicating an essential niche for the myeloid compartment in the CNS [63]. While the role of microglia in antigen presentation to adaptive immune cells is ill defined, the re-presentation of antigens to activated T-cells by microglia is crucial for the maintenance of antigen-specific T-cell populations in the CNS [62, 64]. Regarding the glioma microenvironment, microglia may facilitate tumor progression by releasing matrix metalloproteinases that enable tumor cell proliferation; microglia may also express immunosuppressive molecules such as programmed death ligand 1 (PD-L1), the ligand of PD-1 [65]. Nevertheless, inhibition of microglia-mediated immunosuppression in the tumor microenvironment may be an attractive target to enable an enhanced immune response.
Standard of care: barrier or facilitator of immunotherapeutic regimens?
Those who receive immunotherapeutic agents will have likely received or may be concurrently receiving chemotherapy or radiation for their cancer; it is important to understand whether the combination of standard therapies with immunotherapeutic drugs will facilitate or inhibit the anti-tumor effects of immunotherapy. In particular, the standard of care for GBM is the chemotherapeutic agent temozolomide (TMZ) in combination with radiation therapy (RT), and steroids such as dexamethasone are often prescribed to reduce the symptoms associated with tumor-induced intracranial edema. Chemotherapy has traditionally been viewed as antithetical to the mechanism of immunotherapy—its generalized cytotoxic effects often result in severe lymphopenia, and cell death incurred by chemotherapy occurs via a non-inflammatory apoptotic pathway [66, 67]. These assumptions have been challenged by findings that demonstrate a potentially synergistic effect of TMZ and EGFRvIII vaccine administration in GBM, provided that TMZ is administered outside of the period of effector T-cell proliferation [68]. Unexpectedly, these authors did not observe decline in overall CD4+ and CD8+ lymphocyte counts despite rounds of chemoradiation. The synergism between chemotherapy and immunotherapy may be a result of a decrease in circulating Tregs and consequent decline in their overall immunosuppressive effect [68, 69]. Nevertheless, it is important to emphasize that severe reductions in CD4 counts in individuals with high grade glioma treated with TMZ and radiation have been observed, and the increasing severity of CD4+ lymphopenia was associated with significantly poorer survival [57].
Similar to chemotherapy, RT has traditionally been considered an immunosuppressive agent as it targets rapidly dividing cells, such as T-lymphocytes [70]. Nevertheless, the consequences of such cytocidal effects may prove RT to be an effective adjuvant alongside immunotherapy. Tumor cells destroyed by ionizing radiation up-regulate a number of surface molecules and release cytokines that produce a pro-inflammatory environment, and the antigens released by apoptotic tumor cells provide an immunogenic substrate for anti-tumor inflammation [71–75]. Behaving as a vaccine, RT may prime an anti-tumor response by inducing expression of co-inhibitory molecules that may be subsequently blocked by immune checkpoints or other immunotherapeutics. Preclinical murine models of GBM have demonstrated synergism between PD-1 blockade and RT as well as between 4-1BB activation, CTLA-4 blockade and RT as evidenced by increased overall survival and significant tumor regression [33, 76]. Clinical studies in castration resistant prostate cancer have yielded favorable outcomes in a phase I/II clinical trial of anti-CTLA-4 therapy and radiotherapy, and phase III studies are now underway [77].
Dexamethasone therapy is routinely used in the symptomatic treatment of GBM to decrease vasogenic edema associated with tumor growth. Steroids have been known to cause immunosuppression, as one of their major functions is anti-inflammatory. Dexamethasone therapy in BALB/c mice has preferentially decreased CD4+ effector cell populations while preserving Tregs, possibly accounting for the anti-inflammatory effects of glucocorticoids [78]. If this is the case in the glioma tumor microenvironment, then dexamethasone therapy may abrogate the intended effects of immunotherapeutics, which commonly rely on up-regulation of the effector arms of the immune response and suppression of inhibitory forces such as Tregs. As yet, little is known about the effects of concomitant immunotherapy and dexamethasone in GBM.
Evidence regarding the potential pro-inflammatory role of chemotherapy and radiation in GBM and other tumor types provides promise for its use as a synergistic treatment modality alongside immunotherapeutics. While chemoradiation may facilitate immune-mediated anti-tumor activity, the anti-inflammatory effects of glucocorticoid therapy in GBM may prove to be counterproductive in immune-based treatment regimens. Additional studies are needed to examine the effects of dexamethasone on immunotherapy for GBM.
Future directions
Immunotherapy for GBM, in the form of immune checkpoint blockade and anti-tumor vaccines, is being actively studied in preclinical models and translated to clinical trials. Combination immunotherapies or treatment regimens involving both standard therapies and immunotherapies show promise as powerful anti-cancer therapies in GBM. A phase I clinical trial studying the effects of anti-PD-1 and anti-CTLA-4 combination therapy is currently recruiting for recurrent GBM (NCT02017717), and a number of studies of DC vaccines in recurrent and newly diagnosed GBM are also underway (NCT02010606, NCT02149225, NCT02049489, NCT01808820, NCT02078648). Preclinical successes in immunotherapy for GBM provide evidence that the brain is not an immunologically exclusive organ, but may participate dynamically with the immunologic periphery, providing opportunities for immune-based anticancer therapies. Combination approaches are promising future therapies for GBM, utilizing the anti-cancer and pro-inflammatory effects of chemoradiation in combination with multiple immunotherapeutic modalities.
References
Drake CG (2010) Prostate cancer as a model for tumour immunotherapy. Nat Rev Immunol 10:580–593. doi:10.1038/nri2817
Lipson EJ, Drake CG (2011) Ipilimumab: an anti-CTLA-4 antibody for metastatic melanoma. Clin Cancer Res 17:6958–6962. doi:10.1158/1078-0432.ccr-11-1595
Buckner JC (2003) Factors influencing survival in high-grade gliomas. Semin Oncol 30:10–14
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. doi:10.1056/NEJMoa043330
DeAngelis LM (2001) Brain tumors. N Engl J Med 344:114–123. doi:10.1056/nejm200101113440207
Nagasawa DT, Chow F, Yew A, Kim W, Cremer N, Yang I (2012) Temozolomide and other potential agents for the treatment of glioblastoma multiforme. Neurosurg Clin N Am 23:307–322. doi:10.1016/j.nec.2012.01.007
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3:991–998. doi:10.1038/ni1102-991
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science (New York, NY) 331:1565–1570. doi:10.1126/science.1203486
Dunn GP, Old LJ, Schreiber RD (2004) The three Es of cancer immunoediting. Annu Rev Immunol 22:329–360. doi:10.1146/annurev.immunol.22.012703.104803
Gresser I, Bourali C (1970) Antitumor effects of interferon preparations in mice. J Natl Cancer Inst 45:365–376
Gresser I, Boqali C, Chouroulinkov I, Fontaine-Brouty-Byé D, Thomas MT (1970) Treatment of Neoplasia in mice with interferon preparations. Ann N Y Acad Sci 173:694–707
Fewkes NM, Mackall CL (2010) Novel gamma-chain cytokines as candidate immune modulators in immune therapies for cancer. Cancer J (Sudbury, Mass) 16:392–398. doi:10.1097/PPO.0b013e3181eacbc4
Mazzucchelli R, Durum SK (2007) Interleukin-7 receptor expression: intelligent design. Nat Rev Immunol 7:144–154. doi:10.1038/nri2023
Kaech SM, Tan JT, Wherry EJ, Konieczny BT, Surh CD, Ahmed R (2003) Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat Immunol 4:1191–1198. doi:10.1038/ni1009
Murphy WJ, Back TC, Conlon KC, Komschlies KL, Ortaldo JR, Sayers TJ, Wiltrout RH, Longo DL (1993) Antitumor effects of interleukin-7 and adoptive immunotherapy on human colon carcinoma xenografts. J Clin Investig 92:1918–1924. doi:10.1172/jci116785
Jicha DL, Mule JJ, Rosenberg SA (1991) Interleukin 7 generates antitumor cytotoxic T lymphocytes against murine sarcomas with efficacy in cellular adoptive immunotherapy. J Exp Med 174:1511–1515
Aoki T, Tashiro K, Miyatake S, Kinashi T, Nakano T, Oda Y, Kikuchi H, Honjo T (1992) Expression of murine interleukin 7 in a murine glioma cell line results in reduced tumorigenicity in vivo. Proc Natl Acad Sci USA 89:3850–3854
Fritzell S, Eberstal S, Sanden E, Visse E, Darabi A, Siesjo P (2013) IFNgamma in combination with IL-7 enhances immunotherapy in two rat glioma models. J Neuroimmunol 258:91–95. doi:10.1016/j.jneuroim.2013.02.017
Westermann J, Florcken A, Willimsky G, van Lessen A, Kopp J, Takvorian A, Johrens K, Lukowsky A, Schonemann C, Sawitzki B, Pohla H, Frank R, Dorken B, Schendel DJ, Blankenstein T, Pezzutto A (2011) Allogeneic gene-modified tumor cells (RCC-26/IL-7/CD80) as a vaccine in patients with metastatic renal cell cancer: a clinical phase-I study. Gene Ther 18:354–363. doi:10.1038/gt.2010.143
Sportes C, Babb RR, Krumlauf MC, Hakim FT, Steinberg SM, Chow CK, Brown MR, Fleisher TA, Noel P, Maric I, Stetler-Stevenson M, Engel J, Buffet R, Morre M, Amato RJ, Pecora A, Mackall CL, Gress RE (2010) Phase I study of recombinant human interleukin-7 administration in subjects with refractory malignancy. Clin Cancer Res 16:727–735. doi:10.1158/1078-0432.ccr-09-1303
Klebanoff CA, Finkelstein SE, Surman DR, Lichtman MK, Gattinoni L, Theoret MR, Grewal N, Spiess PJ, Antony PA, Palmer DC, Tagaya Y, Rosenberg SA, Waldmann TA, Restifo NP (2004) IL-15 enhances the in vivo antitumor activity of tumor-reactive CD8+ T cells. Proc Natl Acad Sci USA 101:1969–1974. doi:10.1073/pnas.0307298101
Brentjens RJ, Latouche JB, Santos E, Marti F, Gong MC, Lyddane C, King PD, Larson S, Weiss M, Riviere I, Sadelain M (2003) Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat Med 9:279–286. doi:10.1038/nm827
Anichini A, Scarito A, Molla A, Parmiani G, Mortarini R (2003) Differentiation of CD8+ T cells from tumor-invaded and tumor-free lymph nodes of melanoma patients: role of common gamma-chain cytokines. J Immunol (Baltimore, Md: 1950) 171:2134–2141
Korn T, Bettelli E, Gao W, Awasthi A, Jager A, Strom TB, Oukka M, Kuchroo VK (2007) IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature 448:484–487. doi:10.1038/nature05970
Nurieva R, Yang XO, Martinez G, Zhang Y, Panopoulos AD, Ma L, Schluns K, Tian Q, Watowich SS, Jetten AM, Dong C (2007) Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 448:480–483. doi:10.1038/nature05969
Bucher C, Koch L, Vogtenhuber C, Goren E, Munger M, Panoskaltsis-Mortari A, Sivakumar P, Blazar BR (2009) IL-21 blockade reduces graft-versus-host disease mortality by supporting inducible T regulatory cell generation. Blood 114:5375–5384. doi:10.1182/blood-2009-05-221135
Petrella TM, Tozer R, Belanger K, Savage KJ, Wong R, Smylie M, Kamel-Reid S, Tron V, Chen BE, Hunder NN, Hagerman L, Walsh W, Eisenhauer EA (2012) Interleukin-21 has activity in patients with metastatic melanoma: a phase II study. J Clin Oncol 30:3396–3401. doi:10.1200/jco.2011.40.0655
Vom Berg J, Vrohlings M, Haller S, Haimovici A, Kulig P, Sledzinska A, Weller M, Becher B (2013) Intratumoral IL-12 combined with CTLA-4 blockade elicits T cell-mediated glioma rejection. J Exp Med 210:2803–2811. doi:10.1084/jem.20130678
Fyfe G, Fisher RI, Rosenberg SA, Sznol M, Parkinson DR, Louie AC (1995) Results of treatment of 255 patients with metastatic renal cell carcinoma who received high-dose recombinant interleukin-2 therapy. J Clin Oncol 13:688–696
Rosenberg SA (2014) IL-2: the first effective immunotherapy for human cancer. J Immunol (Baltimore, Md: 1950) 192:5451–5458. doi:10.4049/jimmunol.1490019
Rosenberg SA, Yang JC, Sherry RM, Kammula US, Hughes MS, Phan GQ, Citrin DE, Restifo NP, Robbins PF, Wunderlich JR, Morton KE, Laurencot CM, Steinberg SM, White DE, Dudley ME (2011) Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res 17:4550–4557. doi:10.1158/1078-0432.ccr-11-0116
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Zeng J, See AP, Phallen J, Jackson CM, Belcaid Z, Ruzevick J, Durham N, Meyer C, Harris TJ, Albesiano E, Pradilla G, Ford E, Wong J, Hammers HJ, Mathios D, Tyler B, Brem H, Tran PT, Pardoll D, Drake CG, Lim M (2013) Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int J Radiat Oncol Biol Phys 86:343–349. doi:10.1016/j.ijrobp.2012.12.025
Freeman GJ, Gribben JG, Boussiotis VA, Ng JW, Restivo VA Jr, Lombard LA, Gray GS, Nadler LM (1993) Cloning of B7-2: a CTLA-4 counter-receptor that costimulates human T cell proliferation. Science (New York, NY) 262:909–911
Linsley PS, Brady W, Urnes M, Grosmaire LS, Damle NK, Ledbetter JA (1991) CTLA-4 is a second receptor for the B cell activation antigen B7. J Exp Med 174:561–569
Demaria S, Kawashima N, Yang AM, Devitt ML, Babb JS, Allison JP, Formenti SC (2005) Immune-mediated inhibition of metastases after treatment with local radiation and CTLA-4 blockade in a mouse model of breast cancer. Clin Cancer Res 11:728–734
Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K, Burke MM, Caldwell A, Kronenberg SA, Agunwamba BU, Zhang X, Lowy I, Inzunza HD, Feely W, Horak CE, Hong Q, Korman AJ, Wigginton JM, Gupta A, Sznol M (2013) Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med 369:122–133. doi:10.1056/NEJMoa1302369
Wong AJ, Bigner SH, Bigner DD, Kinzler KW, Hamilton SR, Vogelstein B (1987) Increased expression of the epidermal growth factor receptor gene in malignant gliomas is invariably associated with gene amplification. Proc Natl Acad Sci USA 84:6899–6903
Wong AJ, Ruppert JM, Bigner SH, Grzeschik CH, Humphrey PA, Bigner DS, Vogelstein B (1992) Structural alterations of the epidermal growth factor receptor gene in human gliomas. Proc Natl Acad Sci USA 89:2965–2969
Heimberger AB, Crotty LE, Archer GE, Hess KR, Wikstrand CJ, Friedman AH, Friedman HS, Bigner DD, Sampson JH (2003) Epidermal growth factor receptor VIII peptide vaccination is efficacious against established intracerebral tumors. Clin Cancer Res 9:4247–4254
Wikstrand CJ, Hale LP, Batra SK, Hill ML, Humphrey PA, Kurpad SN, McLendon RE, Moscatello D, Pegram CN, Reist CJ et al (1995) Monoclonal antibodies against EGFRvIII are tumor specific and react with breast and lung carcinomas and malignant gliomas. Cancer Res 55:3140–3148
Sampson JH, Archer GE, Mitchell DA, Heimberger AB, Herndon JE 2nd, Lally-Goss D, McGehee-Norman S, Paolino A, Reardon DA, Friedman AH, Friedman HS, Bigner DD (2009) An epidermal growth factor receptor variant III-targeted vaccine is safe and immunogenic in patients with glioblastoma multiforme. Mol Cancer Ther 8:2773–2779. doi:10.1158/1535-7163.mct-09-0124
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723. doi:10.1056/NEJMoa1003466
Robert C, Thomas L, Bondarenko I, O’Day S, Webber J, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ, Davidson N, Richards J, Maio M, Hauschild A, Miller WH Jr, Gascon P, Lotem M, Harmankaya K, Ibrahim R, Francis S, Chen TT, Humphrey R, Hoos A, Wolchok JD (2011) Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med 364:2517–2526. doi:10.1056/NEJMoa1104621
Hamid O, Robert C, Daud A, Hodi FS, Hwu WJ, Kefford R, Wolchok JD, Hersey P, Joseph RW, Weber JS, Dronca R, Gangadhar TC, Patnaik A, Zarour H, Joshua AM, Gergich K, Elassaiss-Schaap J, Algazi A, Mateus C, Boasberg P, Tumeh PC, Chmielowski B, Ebbinghaus SW, Li XN, Kang SP, Ribas A (2013) Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 369:134–144. doi:10.1056/NEJMoa1305133
Bloch O, Crane CA, Fuks Y, Kaur R, Aghi MK, Berger MS, Butowski NA, Chang SM, Clarke JL, McDermott MW, Prados MD, Sloan AE, Bruce JN, Parsa AT (2014) Heat-shock protein peptide complex-96 vaccination for recurrent glioblastoma: a phase II, single-arm trial. Neuro-oncology 16:274–279. doi:10.1093/neuonc/not203
Phuphanich S, Wheeler CJ, Rudnick JD, Mazer M, Wang H, Nuno MA, Richardson JE, Fan X, Ji J, Chu RM, Bender JG, Hawkins ES, Patil CG, Black KL, Yu JS (2013) Phase I trial of a multi-epitope-pulsed dendritic cell vaccine for patients with newly diagnosed glioblastoma. Cancer Immunol Immunother 62:125–135. doi:10.1007/s00262-012-1319-0
Swartz AM, Li QJ, Sampson JH (2014) Rindopepimut: a promising immunotherapeutic for the treatment of glioblastoma multiforme. Immunotherapy 6:679–690. doi:10.2217/imt.14.21
A Randomized Study of Nivolumab Versus Bevacizumab and a Safety Study of Nivolumab or Nivolumab Combined With Ipilimumab in Adult Subjects With Recurrent Glioblastoma (GBM) (CheckMate 143). clinicaltrials.gov
Medawar PB (1948) Immunity to homologous grafted skin; the fate of skin homografts transplanted to the brain, to subcutaneous tissue, and to the anterior chamber of the eye. Br J Exp Pathol 29:58–69
Davies DC (2002) Blood-brain barrier breakdown in septic encephalopathy and brain tumours. J Anat 200:639–646
Rascher G, Fischmann A, Kroger S, Duffner F, Grote EH, Wolburg H (2002) Extracellular matrix and the blood-brain barrier in glioblastoma multiforme: spatial segregation of tenascin and agrin. Acta Neuropathol 104:85–91. doi:10.1007/s00401-002-0524-x
van Zwam M, Huizinga R, Melief MJ, Wierenga-Wolf AF, van Meurs M, Voerman JS, Biber KP, Boddeke HW, Hopken UE, Meisel C, Meisel A, Bechmann I, Hintzen RQ, t Hart BA, Amor S, Laman JD, Boven LA (2009) Brain antigens in functionally distinct antigen-presenting cell populations in cervical lymph nodes in MS and EAE. J Mol Med (Berlin, Germany) 87:273–286. doi:10.1007/s00109-008-0421-4
Goldmann J, Kwidzinski E, Brandt C, Mahlo J, Richter D, Bechmann I (2006) T cells traffic from brain to cervical lymph nodes via the cribroid plate and the nasal mucosa. J Leukoc Biol 80:797–801. doi:10.1189/jlb.0306176
Cserr HF, Harling-Berg CJ, Knopf PM (1992) Drainage of brain extracellular fluid into blood and deep cervical lymph and its immunological significance. Brain Pathol (Zurich, Switzerland) 2:269–276
Yeung JT, Hamilton RL, Ohnishi K, Ikeura M, Potter DM, Nikiforova MN, Ferrone S, Jakacki RI, Pollack IF, Okada H (2013) LOH in the HLA class I region at 6p21 is associated with shorter survival in newly diagnosed adult glioblastoma. Clin Cancer Res 19:1816–1826. doi:10.1158/1078-0432.ccr-12-2861
Grossman SA, Ye X, Lesser G, Sloan A, Carraway H, Desideri S, Piantadosi S (2011) Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin Cancer Res 17:5473–5480. doi:10.1158/1078-0432.ccr-11-0774
Hickey WF, Hsu BL, Kimura H (1991) T-lymphocyte entry into the central nervous system. J Neurosci Res 28:254–260. doi:10.1002/jnr.490280213
Prins RM, Shu CJ, Radu CG, Vo DD, Khan-Farooqi H, Soto H, Yang MY, Lin MS, Shelly S, Witte ON, Ribas A, Liau LM (2008) Anti-tumor activity and trafficking of self, tumor-specific T cells against tumors located in the brain. Cancer Immunol Immunother 57:1279–1289. doi:10.1007/s00262-008-0461-1
Odoardi F, Sie C, Streyl K, Ulaganathan VK, Schlager C, Lodygin D, Heckelsmiller K, Nietfeld W, Ellwart J, Klinkert WE, Lottaz C, Nosov M, Brinkmann V, Spang R, Lehrach H, Vingron M, Wekerle H, Flugel-Koch C, Flugel A (2012) T cells become licensed in the lung to enter the central nervous system. Nature 488:675–679. doi:10.1038/nature11337
Masson F, Calzascia T, Di Berardino-Besson W, de Tribolet N, Dietrich PY, Walker PR (2007) Brain microenvironment promotes the final functional maturation of tumor-specific effector CD8+ T cells. J Immunol (Baltimore, Md: 1950) 179:845–853
Ransohoff RM, Cardona AE (2010) The myeloid cells of the central nervous system parenchyma. Nature 468:253–262. doi:10.1038/nature09615
Varvel NH, Grathwohl SA, Baumann F, Liebig C, Bosch A, Brawek B, Thal DR, Charo IF, Heppner FL, Aguzzi A, Garaschuk O, Ransohoff RM, Jucker M (2012) Microglial repopulation model reveals a robust homeostatic process for replacing CNS myeloid cells. Proc Natl Acad Sci USA 109:18150–18155. doi:10.1073/pnas.1210150109
Carson MJ, Sutcliffe JG, Campbell IL (1999) Microglia stimulate naive T-cell differentiation without stimulating T-cell proliferation. J Neurosci Res 55:127–134
Yang I, Han SJ, Kaur G, Crane C, Parsa AT (2010) The role of microglia in central nervous system immunity and glioma immunology. J Clin Neurosci 17:6–10. doi:10.1016/j.jocn.2009.05.006
Okada H, Mak TW (2004) Pathways of apoptotic and non-apoptotic death in tumour cells. Nat Rev Cancer 4:592–603. doi:10.1038/nrc1412
Lake RA, Robinson BW (2005) Immunotherapy and chemotherapy–a practical partnership. Nat Rev Cancer 5:397–405. doi:10.1038/nrc1613
Heimberger AB, Sun W, Hussain SF, Dey M, Crutcher L, Aldape K, Gilbert M, Hassenbusch SJ, Sawaya R, Schmittling B, Archer GE, Mitchell DA, Bigner DD, Sampson JH (2008) Immunological responses in a patient with glioblastoma multiforme treated with sequential courses of temozolomide and immunotherapy: case study. Neuro-oncology 10:98–103. doi:10.1215/15228517-2007-046
Banissi C, Ghiringhelli F, Chen L, Carpentier AF (2009) Treg depletion with a low-dose metronomic temozolomide regimen in a rat glioma model. Cancer Immunol Immunother 58:1627–1634. doi:10.1007/s00262-009-0671-1
Gough MJ, Crittenden MR (2009) Combination approaches to immunotherapy: the radiotherapy example. Immunotherapy 1:1025–1037. doi:10.2217/imt.09.64
Tesniere A, Apetoh L, Ghiringhelli F, Joza N, Panaretakis T, Kepp O, Schlemmer F, Zitvogel L, Kroemer G (2008) Immunogenic cancer cell death: a key-lock paradigm. Curr Opin Immunol 20:504–511. doi:10.1016/j.coi.2008.05.007
Skoberne M, Beignon AS, Bhardwaj N (2004) Danger signals: a time and space continuum. Trends Mol Med 10:251–257. doi:10.1016/j.molmed.2004.04.001
Gasser S, Orsulic S, Brown EJ, Raulet DH (2005) The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature 436:1186–1190. doi:10.1038/nature03884
Larsson M, Fonteneau JF, Bhardwaj N (2001) Dendritic cells resurrect antigens from dead cells. Trends Immunol 22:141–148
Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E, Saulnier P, Yang H, Amigorena S, Ryffel B, Barrat FJ, Saftig P, Levi F, Lidereau R, Nogues C, Mira JP, Chompret A, Joulin V, Clavel-Chapelon F, Bourhis J, Andre F, Delaloge S, Tursz T, Kroemer G, Zitvogel L (2007) Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 13:1050–1059. doi:10.1038/nm1622
Belcaid Z, Phallen JA, Zeng J, See AP, Mathios D, Gottschalk C, Nicholas S, Kellett M, Ruzevick J, Jackson C, Albesiano E, Durham NM, Ye X, Tran PT, Tyler B, Wong JW, Brem H, Pardoll DM, Drake CG, Lim M (2014) Focal radiation therapy combined with 4-1BB activation and CTLA-4 blockade yields long-term survival and a protective antigen-specific memory response in a murine glioma model. PLoS One 9:e101764. doi:10.1371/journal.pone.0101764
Slovin SF, Higano CS, Hamid O, Tejwani S, Harzstark A, Alumkal JJ, Scher HI, Chin K, Gagnier P, McHenry MB, Beer TM (2013) Ipilimumab alone or in combination with radiotherapy in metastatic castration-resistant prostate cancer: results from an open-label, multicenter phase I/II study. Ann Oncol 24:1813–1821. doi:10.1093/annonc/mdt107
Chen X, Murakami T, Oppenheim JJ, Howard OM (2004) Differential response of murine CD4+ CD25+ and CD4+ CD25- T cells to dexamethasone-induced cell death. Eur J Immunol 34:859–869. doi:10.1002/eji.200324506
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patel, M.A., Pardoll, D.M. Concepts of immunotherapy for glioma. J Neurooncol 123, 323–330 (2015). https://doi.org/10.1007/s11060-015-1810-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1810-5